Abstract
Keywords
Introduction
Testosterone replacement therapy (TRT) is increasingly medicalised, framed as a controlled intervention for managing testosterone deficiency while also entangled in broader discourses of pharmaceuticalisation (Dunn et al., 2021), anti-doping (Gibbs et al., 2024; Piatkowski, Gibbs, & Dunn, 2024), and illicit drug control (Piatkowski, Gibbs, et al., 2024; Underwood et al., 2021). Despite TRT’s legitimacy within biomedicine, many men face restrictive prescribing guidelines (Handelsman, 2013, 2020, 2021), and medical scepticism (Cox et al., 2024; Piatkowski, Ayurzana et al., 2025), leading them to seek testosterone through informal markets and peer networks (Henning & Andreasson, 2022; Turnock et al., 2023). Concerningly, alternate (non-medical) procurement pathways are known to be associated with a heightened risk of harms (Piatkowski et al., 2025a, 2025b; Turnock et al., 2023). As such, this study examines how men navigate access to testosterone within a landscape shaped by biomedical regulation, risk governance, and harm reduction gaps. Using qualitative interviews with men (all of whom identified as male) consuming TRT within Australia, we explore how structural barriers shape testosterone access and usage practices. Drawing on biomedicalisation theory (Clarke et al., 2003) and using a peer lens (Piatkowski, Seear, et al., 2024), we argue that TRT must be understood beyond the (non)medical dichotomy, recognising the need for regulated safe supply and peer-driven harm reduction. While a variety of safe supply models have been implemented for other substances (Ferguson et al., 2022; Kalicum et al., 2024), they remain underdeveloped for testosterone, leading men into fragmented, sometimes risky markets (Piatkowski et al., 2023). We advocate for pragmatic harm reduction strategies involving relational peer-led care, alongside Australian drug policy reform, to better align with the realities of using anabolic-androgenic steroids (AAS).
Background
AAS, including testosterone, have long been central to biomedical, cultural, and regulatory debates, shaping how masculinity, ageing, and health are understood and managed (Hoberman & Yesalis, 1995). While initially developed for treating hypogonadism (Bhasin et al., 2018), TRT has expanded beyond strict clinical necessity to include broader applications such as ageing-related decline, vitality enhancement, and body optimisation (Underwood et al., 2021). TRT fundamentally differs from broader AAS use—which is commonly targeted towards image and performance enhancement and can increase testosterone beyond high-natural levels—instead aiming to medically restore testosterone to healthy natural levels (Dunn et al., 2021; Piatkowski, Turnock, et al., 2024). Despite this medicalisation, access to testosterone remains highly regulated, creating barriers that shape how men navigate TRT (Handelsman, 2021). Testosterone’s regulatory status in Australia is complex and varies between national and state-level frameworks. Under the national Poisons Standard, testosterone is generally classified as a Schedule 4 medication—indicating that it is available only by prescription (Handelsman, 2020). However, in Queensland and New South Wales, AAS (including testosterone and its derivative dihydrotestosterone) are regulated under state legislation as Schedule 1 substances—carrying more severe legal ramifications in response to possession, distribution, and non-medical use (Piatkowski, Gibbs, & Dunn, 2024). This more restrictive classification in some states reflects a heightened control and politicised stance over AAS (van de Ven & Fomiatti, 2025). Such regulatory discrepancies underscore the challenges faced by men seeking safe and reliable TRT, as they must navigate not only stringent medical gatekeeping practices but also variable legal frameworks.
These regulatory structures position formal medical access as the ‘legitimate’ route while simultaneously criminalising or discouraging alternative pathways, despite evidence that many men procure testosterone outside the healthcare system due to medical gatekeeping, stigma, and restrictive prescribing guidelines (Fomiatti et al., 2019). As a result, informal peer networks and underground markets have emerged as parallel sources of access, often filling gaps left by rigid regulatory frameworks. Research on testosterone use has often centred on a dichotomy of either medical TRT or illicit AAS use, creating a division that fails to capture the complex realities of self-directed use. Recent studies have challenged this dichotomy, emphasising the fluidity between therapeutic and enhancement uses, and elucidating the ways in which men negotiate medical authority, self-experimentation, and embodied expertise (Dunn et al., 2021; James & Wynn, 2022; Underwood et al., 2021). These perspectives highlight the need for harm reduction strategies that extend beyond conventional models of risk, acknowledging that many consumers are not merely patients but active participants in managing their own health.
Empirical research has increasingly documented the challenges men face when seeking TRT, including inconsistent clinical practices around diagnostic thresholds, medical scepticism regarding treatment motives, and constraints on ongoing monitoring and care (Barbonetti et al., 2020; Cox et al., 2024; McVeigh & Bates, 2022; Underwood et al., 2021). Such experiences can leave men feeling delegitimised and disillusioned within clinical settings, contributing to disengagement from care and occasional transitions into informal supply pathways (Cox et al., 2024; Piatkowski, Ayurzana, et al., 2025). Studies from Australia and similar contexts further highlight that prescriber reluctance, stigma linked to performance enhancement, and financial burden shape treatment continuity and healthcare-seeking behaviour (Cox et al., 2024; Dunn et al., 2021; Piatkowski, Kill, et al., 2025). While these findings demonstrate the complex interplay of clinical, social, and regulatory factors affecting TRT access, most research continues to dichotomise testosterone use as either medically legitimate or illicit (Handelsman, 2020; Paoli & Cox, 2024). These empirical gaps indicate a need to understand how men actively navigate and negotiate TRT access across multiple, intersecting pathways.
Rather than positioning men who use testosterone as passive recipients of medical care or deviant consumers of illicit substances, we foreground their agency in navigating, contesting, and reconfiguring access pathways. This perspective aligns with participatory and peer-led frameworks that recognise people who use AAS as active agents in their health decisions (Nourse, Moore, & Fraser, 2024; Piatkowski et al., 2024), often drawing on experiential and situated knowledge (Fraser, Moore, et al., 2020), and peer networks to manage risks and optimise benefits. Our study builds on this scholarship by examining how men engage with testosterone across multiple access points, from formal prescriptions to underground markets, and how these practices intersect with broader regulatory, medical, and social structures.
Approach
The concept of biomedicalisation provides the core lens for our study by situating personal narratives of TRT within the larger transformation of health and body management in contemporary society. As articulated by Clarke and Shim (2010) and Clarke et al. (2023), biomedicalisation extends beyond the traditional framework of medicalisation by capturing how naturally occurring phenomena, such as testosterone production, are redefined as tightly controlled pharmaceutical commodities. This redefinition carries promises of safety and standardised care while simultaneously enacting significant barriers to access. According to Clarke et al. (2023), biomedicalisation is characterised by several key processes: the expansion of medical interventions into realms of enhancement rather than mere disease treatment; an increased emphasis on surveillance and risk management; the integration of technoscientific innovations; and the commodification of biomedical knowledge and practice. These processes are evident in the context of TRT. For instance, pharmaceutical marketing, clinical guidelines, and strict regulatory frameworks construct a normative pathway where low testosterone is pathologised, and a highly standardised solution is provided; however, available only through formal, costly, and bureaucratic channels (Nourse, Moore, & Fraser, 2024).
In this light, testosterone becomes a tool for self-surveillance and self-discipline, aligning an individual’s bodily function with contemporary ideals of vitality, youth, and masculinity (Nourse, Moore, & Fraser, 2024). However, while the regulated biomedical regime promotes uniform standards of care, it simultaneously produces inequities. Those who are excluded by rigid protocols or who face insurmountable financial barriers become expert negotiators of their own care, accumulating a form of peer-led biomedical literacy that challenges formal medical authority (Henning & Andreasson, 2022; Turnock et al., 2023). This theoretical approach compels us to view TRT not solely as a clinical intervention, but as a locus where health, identity, and embodied expertise are co-constructed (Fomiatti et al., 2019, 2020; Fraser, Fomiatti, et al., 2020). The controlled distribution of testosterone through regulated channels, as well as the parallel emergence of underground supply networks (Piatkowski, Kill, et al., 2025), illustrates the paradox of biomedicalisation. While regulated pathways promise safety through standardisation, they also compel many people to develop alternative strategies that blend formal and informal practices.
Despite increasing clinical and regulatory attention to TRT, limited research has examined how men prescribed testosterone actively navigate access across the intersecting domains of formal healthcare and informal peer-driven markets. This gap restricts our understanding of how TRT is lived and managed in practice, particularly when clinical pathways prove inaccessible or misaligned with men’s needs. By centring the lived expertise and navigation strategies men employ to obtain and maintain treatment, this study provides essential insight into how current governance frameworks shape health outcomes and care experiences. In doing so, our analysis generates new knowledge to strengthen harm-reduction and policy responses that more accurately reflect the realities of TRT access and support safer, more inclusive models of care.
Methods
Sampling and Recruitment
This study was exploratory and drew on a larger research project that investigated the use of AAS. Data were gathered from nine individuals who identified as men (M age = 38.86, SD = 2.85), all aged 18 or older and currently using testosterone, recruited through purposive sampling. A total of 14 interviews were conducted, as five participants completed a second interview for deeper insight. The study was approved by the Griffith University Human Research Ethics Community (approval number: 2023/784).
Second interviews were conducted selectively to follow- up on notations, questions, and emergent insights with some participants rather than offered automatically to all participants. These follow-up interviews were requested when the first interview raised complex or under-developed topics (e.g., experiences of navigating multiple healthcare providers, combining prescribed and underground compounds) that warranted further elaboration. This approach aligns with iterative qualitative design principles (Piatkowski, Kill, et al., 2025), where data collection and analysis proceed concurrently, and later interviews build on analytic needs rather than strict participant availability. Interviewing ceased once analytic sufficiency was reached, with later interviews yielding no substantially new insights. Recruitment was also limited by the challenges of engaging a relatively hidden population of men prescribed TRT in Australia.
Recruitment was conducted via the authors’ personal and professional networks, including social media promotion, word of mouth, and snowball sampling. Recruitment was voluntary, with participants opting in by contacting the senior author, who was involved in a separate harm reduction initiative offering anonymous laboratory analysis of AAS products. This testing program was entirely independent of the present qualitative study, and no biological samples or drug products were collected as part of this research. Some participants became aware of the study through prior engagement in this separate community service, but participation was voluntary and in no way conditional upon access to testing.
To maximise anonymity and encourage participation from a population navigating significant stigma and legal risk, demographic data collection was intentionally minimised. Consistent with best practice for research involving potentially criminalised behaviours, only core eligibility information (age, gender identification, current testosterone use) was gathered. Pilot discussions indicated that collecting more detailed demographic variables (e.g., socioeconomic background, training history, comorbidities) reduced willingness to participate. Therefore, demographic breadth was intentionally limited to prioritise participant safety and engagement, though this necessarily constrains the extent to which the sample can be contextually profiled.
Data Collection
Informed consent was obtained verbally prior to conducting semi-structured interviews with participants about their experiences. Interviews were conducted via Microsoft Teams, each lasting between 36 and 72 minutes (Mean = 56.79 minutes, SD = 9.82 minutes). Audio was recorded locally rather than stored on cloud-based servers to minimise third-party data access. Automatic transcription was generated using Microsoft’s built-in transcription tool, and transcripts were manually checked for accuracy. Immediately following verification, all audio files were deleted from the recording platform.
To protect participants’ confidentiality, given that illicit AAS possession carries legal risks in parts of Australia (Piatkowski et al., 2024), pseudonyms were assigned at transcription, identifying information was removed, and data were stored on an encrypted university drive accessible only to the research team. No links between participants’ identities and their transcripts were retained.
The interviews included demographic questions and in-depth, open-ended questions related to their use of TRT, health-related outcomes, and the broader sociocultural context of their experiences. Example questions include: Could you please share some information about your experience with steroids and information about the compound and dosage? How was your experience attaining the prescription? And How do access to healthcare services and social stigma around AAS use affect your ability to practice safer use? The interviews were recorded, transcribed automatically, and reviewed for accuracy by TP. Transcripts were then imported into NVivo (QSR, v12) for analysis.
Data Analysis
The data were analysed using a narrative analytic approach that foregrounds participants' lived-living experiences with TRT. Importantly, participants reported varied motivations for testosterone use. While some described clinically indicated treatment for low testosterone, others framed their use around goals such as wellbeing, physique, or performance enhancement. We therefore treated motivations as fluid and shifting rather than as fixed categories, as this heterogeneity offered essential insight into how structural barriers differently shape access needs and risk profiles. This approach enabled an in-depth exploration of how people construct, negotiate, and make sense of their experiences within a complex healthcare landscape. Our analysis was iterative and reflexive, drawing on iterative categorisation (Neale, 2016), a systematic technique for analysing qualitative data developed by Jo Neale. This approach involved multiple readings of the transcripts, open coding, and the development of thematic categories that emerged directly from the data. In this process, we closely attended to both the content of participants’ accounts (e.g., descriptions of physical and mental wellbeing, technical details of dosing) and the ways in which these narratives were structured (e.g., metaphors such as “jumping through hoops”). The initial phase followed Neale’s iterative categorisation method, beginning with line-by-line coding of the transcripts (Neale, 2016). This stage identified a range of concepts—including “gatekeeping,” “stigma,” “barriers,” “wellbeing,” and “self-regulation”—that were central to participants’ accounts. As coding progressed, these concepts began to coalesce into three overarching theme-categories: Jumping through hoops: Negotiating institutional gatekeeping, Transforming wellbeing, and Topping up: Dosage and self-regulation.
After establishing these broader themes, interpretative work was done to link the participants’ narratives to broader cultural and biomedical discourses (Neale, 2021). For example, the critiques of regulatory frameworks that, while designed for safety, inadvertently restrict access. Similarly, narratives of transformative wellbeing were interpreted as contestations of biomedical advice that fails to fully address the complex interplay between mental and physical health. Finally, detailed accounts of dosage and self-regulation were analysed to underscore how participants not only adhere to, but also actively negotiate biomedical standards, reflecting a form of peer-led biomedical literacy. This process ensured that both the content and structure of the narratives were central to our understanding of the lived-living experience of TRT, highlighting the tension between personal benefit and systemic barriers.
This negotiation is not without risk, as participants also highlight the dangers of unregulated products—a concern that resonates with broader debates on illicit AAS markets in Australia and globally. Reflections on navigating similar choices underscore a critical point: expertise is often cultivated outside formal institutions, and this lived expertise can offer valuable insights into the safe and effective use of TRT. Having encountered the complexities of both pharmaceutical-grade testosterone—often diverted through formal channels—and underground sources when medical access proved elusive, our research perspective incorporates a peer-informed lens that enriches and grounds the analysis. This reflexive positioning allowed us to appreciate the nuances of risk, safety, and personal agency from an insider’s perspective while also critically interrogating the systems that produce and regulate these experiences. In doing so, our theoretical framework is not abstract or removed from practice; rather, it is rooted in the everyday realities of men whose bodies, health, and identities are shaped by both clinical protocols and informal practices. This peer-informed lens acknowledges both the promise and the pitfalls of current biomedical systems. By interweaving detailed participant accounts with critical theoretical insights, we offer a nuanced portrayal of TRT use, one that acknowledges participants’ reported experiences of personal transformation while critically interrogating the structural forces that shape access and safety. We adopt a neutral and non-advocacy stance regarding testosterone and other AAS use. Our aim is to accurately represent participants’ lived-living experiences, including both perceived benefits and harms, without promoting or discouraging any specific health decisions. This reflexive positioning acknowledges the legitimacy of participants’ embodied perspectives while maintaining critical attention to the structural and regulatory contexts that shape access, safety, and wellbeing.
The following section presents the three overarching theme categories: Jumping through Hoops: Negotiating Institutional Gatekeeping Transforming Wellbeing, and, Topping up: Dosage and Self-Regulation.
Findings
Demographic Information and Pseudonyms of Participants (N = 9)
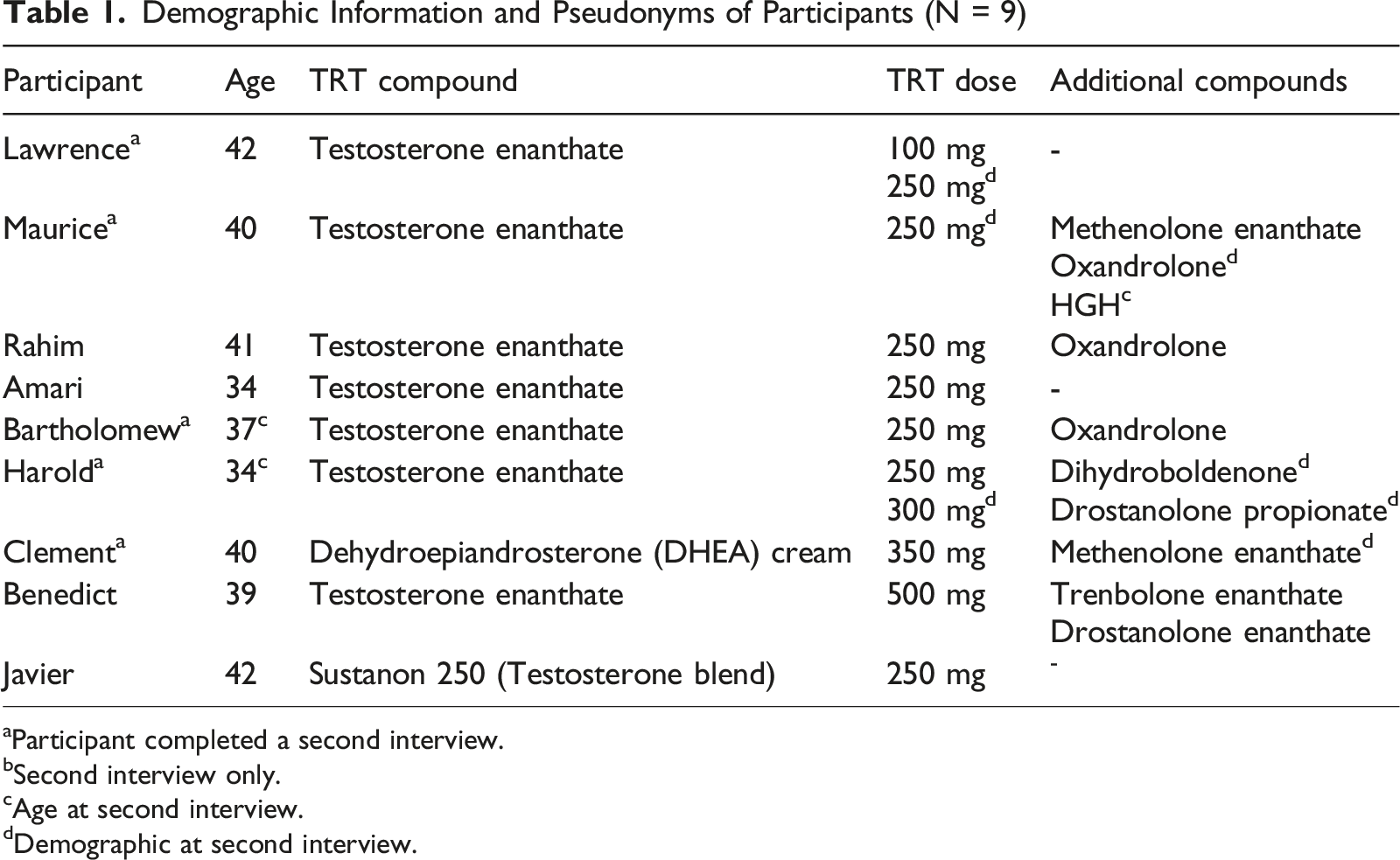
aParticipant completed a second interview.
bSecond interview only.
cAge at second interview.
dDemographic at second interview.
Jumping through Hoops: Negotiating Institutional Gatekeeping
This theme describes how structural, financial, and stigma-related barriers within formal healthcare systems restricted access to TRT, prompting some participants to pursue informal alternatives. It encompassed two sub-patterns: (1) structural barriers to accessing TRT (e.g., cost, wait-times, prescriber reluctance), and (2) stigma and delegitimisation within healthcare encounters. Participants’ narratives frequently foregrounded the cumbersome and often stigmatising processes associated with accessing testosterone. These accounts revealed not only personal frustration but also a broader critique of a medical system that enforced strict gatekeeping. In doing so, participants constructed their experiences as battles against outdated, bureaucratic practices that fail to recognise the nuanced benefits of TRT. For example, Amari, 34, described his experience succinctly: I’m currently on TRT from a doctor. Testosterone enanthate. Like one millilitre (ml) every 10 days, something like that. Yeah. it's hard to find the right, right doctors willing to help you. You just need to a doctor who's open enough to and understand it outside the taboo. Testosterone could actually be helpful for individuals, and a lot of doctors are quite old school about it. Because obviously [there is] a lot of new knowledge around testosterone and its benefits for men.
This narrative immediately establishes the notion that within the biomedical community, outdated perceptions continue to be perceived to hinder access. His account reflects both a personal struggle for a receptive provider, and a broader cultural critique of entrenched biomedical norms for people seeking testosterone through regulated pathways. Rahim, 41, added further detail: It's like you have to do like doctors, doctors meet appointment taking time off work, then go see your endo [endocrinologist]. Endo then wants us to get a blood test. You have to go back to the endo another time, and you can't just do this week after week. It's a long and protected process. Then you only get a script of maybe five or six repeats. So yeah, to do it legitimately, you can get it done, but it's hard. It's very hard, very long and expensive. Much easier just to buy underground from a guy at the gym.
Rahim lays out the step-by-step hurdles that many people face, exposing how the regulated process, while intended to ensure safety, can become prohibitively complex. The explicit contrast with the underground route not only hints at a pragmatic workaround but also signals a loss of trust in the regulated system, revealing a dissonance between ideal biomedical practices and real-world access. Clement, 40, added: I think I told you the last place I was with for the TRT stuff, same deal, had to jump through all the hoops. And he basically just again told me (pause) the stigma. So it was like. And then once you have that experience, you kind of go, oh, fuck, nobody's willing to help me or you feel embarrassed to then go and chat with somebody. Like, you know, what avenues do I have here? And they [medical professionals] actually don't know most of them. Which is kind of scary.
Clement’s account reinforces the hoops are not just procedural but are imbued with stigma. His frustration and disillusionment that arises when medical professionals reinforce negative stereotypes. The narrative highlights how repeated exposure to dismissive attitudes can lead to feelings of isolation and uncertainty about available treatment avenues, further complicating access to safe and regulated TRT. These accounts reveal not only personal inconvenience but also a broader sociopolitical dynamic in which clinical gatekeeping regulates who is deemed a “legitimate” recipient of testosterone, reinforcing normative assumptions about masculinity, ageing, and acceptable bodies. Bartholomew, 37, provided a financial and procedural perspective: I think the initial consult [is] around the $300 mark [AUD] and then the blood test […] then the blood test results get sent to the clinic and then the clinic goes “Yep, sweet.” And then they either compound the testosterone enanthate or they send you prescription testosterone. So if you can get it on PBS [Pharmaceutical Benefits Scheme], it's about $11 a ml. On a private script, you're sort of anywhere from $25 to $40 a ml. So, if you don't get it on PBS, you're paying three or four times [more].
Here, Bartholomew’s narrative links the structural barriers to significant financial hurdles for some people. These hoops are not only administrative, but they also translate into real monetary barriers for people which can create inequitable access to what many participants view as a life-enhancing treatment. Harold, 34, added: Yep, so I'm 34 male. At the moment I'm not doing a lot. I'm on pharmaceutical as in like prescribed. At 250 milligrams (mg) testosterone enanthate per week. Yeah it’s on PBS, I think it end up about $30 for three ml. So 10 bucks a week. Which is probably like a little bit more expensive than black market. Yeah. It's also stronger. 250 mg of [pharmacy name] is very different from 250 mg from the guy at the gym.
This represents a cost–benefit analysis that many people who use androgens are forced to consider. Yet, emphasis on quality reinforces the trust in the biomedical system’s ability to deliver safe, standardised doses despite its hurdles. This is an important consideration in light of the dangers of the unregulated AAS market both in Australia and globally. Clement also engaged in this analysis and stated: It's not much more expensive, to be honest than going underground, I mean. So actually, for three months for this it cost $85. But the testing process cost nearly $1000 to get to that stage. And then, but underground, I mean, you can get anything from, you know, $10 to $220 depending. But it can be anything from just canola oil to some backyard chemical mix of whatever.
This highlights the duality of accounts, and the technical rationale for people’s choices when deciding between efficacy and accessibility. The substantial testing costs underscore how safety measures and regulatory requirements can become prohibitive, pushing some toward less reliable underground options despite the inherent risks.
Together, these accounts highlight how biomedical regulation constructs legitimacy through complex gatekeeping, while simultaneously producing conditions that can push men toward underground markets.
Transforming Wellbeing
This theme captures participants’ accounts of TRT as crucial to improving energy, mood, and daily functioning, reflecting a perceived mismatch between lived-experience benefits and medical scepticism. It included two sub-patterns: (1) perceived improvements in physical and mental wellbeing, and (2) the role of TRT in sustaining everyday functioning and identity. Beyond the structural hurdles, participants recounted transformative narratives of improved physical and mental wellbeing, positioning TRT as a critical intervention in their health journeys. Harold, 34, described his personal transformation: I was very, very overweight and had a lot of depression […] obsessed with trying to, like, get a better physique and just no matter how much like I reduce food or like trained like, I just wasn’t getting anywhere. So I've kind of gone if I'm doing the process and it doesn't work, then there must be another variable out. Got blood test, found out my testosterone was absolutely in the dirt. And I went and saw my GP and he's like, “I think you need to be on something - but I can't do it”. So he refused to prescribe it. So I went OK, cool. And I had a very long talk with my wife and we went, this is health issues, you know. We don't agree with [the] medical professional. So I'm going to start using it.”
This narrative captures both physical struggles (e.g., obesity, lack of progress despite effort) and mental health challenges (e.g., depression), which are perceived to benefit from TRT. Importantly, decisions to override traditional medical advice reveal a strong sense of agency and a peer-informed trust in his own lived-living knowledge—challenging biomedical protocols. Javier, 42, added a focus on mental health: I’ve done a lot of work post discharge referring to mental health. I suffer very bad with thoughts of self-harm and all that sort of thing, and did a lot of work with that […] I got myself to the point of even in my darkest moments I had slipped into… darkest period of my life ever, like when I had these low test [testosterone] scores.
This linkage between low testosterone and mental health challenges is not new but reinforces the argument that TRT can serve as a holistic intervention. This narrative is crucial in juxtaposing clinical skepticism with the everyday reality that many men face. This everyday support and assistance chimed with Bartholomew’s narrative: I don't train as often as I used to because. I've been studying full time for the last four years and I finish at the end of this year so. That's so it's [TRT] not really used for training anymore. It's just used to function as an adult, yeah.
Bartholomew, 37, redefines the stereotypical role of TRT, from enhancing athletic performance to sustaining daily functioning. His narrative underscores that for many, TRT is not about achieving a superhuman physique but rather about maintaining energy, focus, and overall wellbeing. Across participants’ accounts, maintaining energy, mood, sexual function, and everyday capacity was consistently framed as “getting back to normal” rather than pursuing extraordinary performance—demonstrating that health-focused enhancement was central to how men understood the purpose of TRT. Javier, 42, also summarised the everyday benefits: I feel like, yeah, this [TRT] can really be a stepping stone and a tool in our fight against mental health and all that sort of thing.
This focus on sustained functionality speaks to a qualitative improvement in daily life, supporting both mental and physical health. Taken together, these accounts demonstrate the need for a patient-centred understanding of testosterone efficacy, one that values lived-living experience as well as clinical benchmarks. They also emphasise wellbeing restoration and self-determination, challenging narrow biomedical framings of what constitutes a “legitimate” health outcome.
Topping up: Dosage and Self-Regulation
This theme illustrates how men actively adapt prescribed regimens to optimise outcomes, often blending formal and informal practices based on peer knowledge and embodied experience. It involved two sub-patterns: (1) refining prescribed regimens based on embodied experience, and (2) integrating informal compounds when clinical access was insufficient. A further dimension of participants’ experiences involved the technical management of TRT regimens. These accounts reveal a sophisticated negotiation between prescribed protocols and personal adaptations. Lawrence, 42, explained the clinical rationale: I guess some people start, they say it's a TRT dose, but a TRT dose of testosterone would typically be about 1 mg per kilogram (kg) of body weight. So, if that's the case a male my size, say 100 kg, a starting dose for him would be anywhere from 100 mg to say, 125 for a genuine TRT dosage.
Lawrence continued with a critical distinction: But there's a bit of a misconception people will say they're going on TRT, but they're just doing steroids. Like they're pushing the limits going from 1. 1.5 up most people as a first cycle would start about 250 mg a week and go upwards from there.”
This differentiates between legitimate TRT use and the use of AAS. This commentary underscores a key tension: while TRT is framed as a regulated, safe intervention, there exists a blurred boundary around safe supply and dosage. In this light, Rahim, 41, offered his personal regimen: So I'm currently on 1.1 ml. So basically 250 mg per ml. So one dose of testosterone enanthate from the doctor on prescription he was prescribing me one dose every 10 days to keep my levels up. I'm dosing once every seven days. On top of that, I've bought an underground version of Anavar [oxandrolone], which supposedly is a 20 mg tablet. I'm going to say it's probably around about 10 or 12 mgs and I'm using one to two of those a day. So weekly I would be using 250 mgs of testosterone enanthate and I would be using 140 to 150 mgs of Anavar in tablet form.
The addition of the underground oxandrolone reveals how consumers actively blend formal and informal practices. People with lived-living expertise often are placed in the position of managing their own care, reflecting a form of peer-led biomedical literacy that challenges the notion of passive patienthood. However, Clement acknowledges the unregulated nature of the market regarding compounds outside of the prescription. This does not come without dangers. Clement, 40, in his second interview, provided another technical perspective: I did a very slight bulk. I'm on TRT regularly. So that's just from a compounding pharmacy through a script that comes as a cream. So basically, I mean I'm pretty good because my bloods are very even, very nice I guess. So if your kidney function and the enzymes start to rise significantly […] they remove it from you, and then they're pretty good because they've got a full fertility programme as well.”
Clement’s account underscores how biomedical monitoring (e.g., regular blood tests) reassures him of the safety and effectiveness of his testosterone usage. Considered collectively, these accounts highlight both sophisticated self-management and exposure to risk in under-regulated supply environments, demonstrating the dual nature of peer-led biomedical expertise. Furthermore, they delineate that health monitoring practices are not rigidly enforced outside of formal medically supervised testosterone and other AAS use. As such, members of the AAS use community, scholars, policymakers, and practitioners have a responsibility to shape the illicit and licit use of testosterone in Australia.
Discussion
Our findings demonstrate the contested terrain of TRT within contemporary biomedical regimes, highlighting how men navigate, negotiate, and, at times, resist the structural constraints imposed by medicalised testosterone access to achieve their desired health outcomes. TRT is not merely a clinical intervention, but a sociopolitical process through which bodies are disciplined, regulated, and commodified, as evident in men’s accounts of stigma during clinical encounters, having to “justify” need, and feeling gatekept by prescribing thresholds and procedural hurdles. Yet, as our participants’ narratives demonstrate, these biomedicalised pathways are neither stable nor universally accessible. For example, while some men obtained testosterone through medical professionals (e.g., general practitioner, endocrinologist), others reported medical professionals refused to prescribe it—despite acknowledging their low testosterone levels and the benefits TRT could provide. As such, some men seeking TRT must engage in strategic negotiations that blur the boundaries between formal healthcare systems, peer networks, and underground markets.
A key tension in our data is the contradiction inherent in biomedicalised testosterone governance: while formal TRT pathways are framed as the gold standard for safety and efficacy, their restrictive nature paradoxically drives many men toward informal routes of procurement (Dunn et al., 2021; Handelsman, 2021; Underwood et al., 2021). For many men in our study, the promise of optimisation is undermined by regulatory hurdles, stringent diagnostic criteria, and financial barriers. In this way, TRT becomes emblematic of a broader phenomenon wherein biomedical regimes construct risk while simultaneously limiting access to standardised care. Participants’ experiences underscore how biomedical gatekeeping practices, such as rigid diagnostic thresholds for hypogonadism, serve not only as medical criteria but also as sociopolitical instruments. By determining who is deemed a ‘legitimate’ recipient of TRT and who is not, these practices regulate masculinity, moral worthiness, and compliance with normative bodily ideals. Denial of access, or the threat of criminalisation, functions to police the boundaries between health and enhancement, reinforcing a hierarchy in which some men’s embodied needs are authorised and others are delegitimised.
In addition, it is important to acknowledge that participants’ decisions to initiate or continue TRT were grounded in their perceived health needs, which may not always align with biomedical diagnostic criteria. Our analysis does not evaluate the clinical appropriateness of testosterone for managing symptoms such as low mood or fatigue, nor does it suggest individuals should bypass medical guidance. Rather, we examine how experiences of delegitimisation and unmet care needs shape decision-making in practice. This distinction is central to a harm-reduction lens, understanding why people self-manage enables health and policy responses that reduce risk, rather than assuming that individuals who fall outside biomedical thresholds have no legitimate concerns.
Furthermore, beyond merely responding to biomedical constraints, men engaging with TRT outside formal channels cultivate their own forms of embodied expertise, thereby constructing alternative biomedical knowledge systems. Rose (2007a) emphasises the increasing responsibilisation of individuals in managing their health, a dynamic that our findings support. Many of our participants actively curated their own knowledge through peer communities, online forums, and experiential learning, positioning themselves as informed decision-makers rather than passive patients. This distributed biomedicalisation (Rose, 2007)—the emergence of biomedical knowledge beyond institutional confines—demonstrates how expertise is collectively constructed in ways that challenge the traditional patient-doctor hierarchy echoing Fraser’s work on peer-led care (Fraser, Fomiatti, et al., 2020; Fraser, Moore, et al., 2020). Peer networks in our study functioned as crucial infrastructures of safety and legitimacy, enabling harm-reduction practices such as self-monitoring hormone levels and adjusting dosages responsively to lived experience rather than rigid clinical guidelines.
However, self-directed TRT is not a risk-free or ideologically “liberating” alternative. Participants described uncertainties around product quality, dosage accuracy, and the absence of medical monitoring when turning to informal suppliers or modifying prescriptions. Our analysis does not endorse bypassing clinical oversight nor presume that all self-identified needs warrant intervention. Instead, we highlight how delegitimisation and unmet care needs shape decisions in practice, and how governance structures can unintentionally heighten harm by restricting access to regulated treatment. A balanced harm-reduction approach therefore requires acknowledging the value of lived expertise while ensuring evidence-based monitoring and safer supply pathways remain accessible.
TRT’s biomedicalisation is also deeply entangled with shifting ideals of bodily optimisation. These experiences were shaped not only by regulatory access constraints but also by participants’ differing goals for testosterone use, resulting in distinct navigation strategies within the same system. Our findings align with the work of scholars such as Conrad (2013), who highlight the pharmaceuticalisation of everyday life as a process that reconfigures social norms. Testosterone, historically associated with virility and strength, has become a molecular fix, promising the restoration of lost vitality and productivity. Yet, our participants’ narratives complicate this view: rather than uncritically adopting TRT, many men engaged in critical reflections about its role in their lives. For some, TRT was framed as a necessary intervention to maintain a sense of selfhood disrupted by declining hormone levels, aligning with dominant biomedical scripts (Dunn et al., 2021; Underwood et al., 2021). For others, it was positioned as an act of resistance, an assertion of bodily autonomy in the face of restrictive healthcare systems. Importantly, our findings challenge the notion that TRT use outside biomedical frameworks is purely driven by enhancement motives. Instead, they reveal how men negotiate TRT through complex affective and social considerations, informed as much by care and wellbeing as by performance and optimisation.
Our study underscores the need to reconfigure harm reduction strategies for testosterone use, particularly by addressing the limitations of existing regulatory frameworks. Current public health models often construct a rigid dichotomy between ‘legitimate’ medical prescriptions and ‘illegitimate’ self-directed use, failing to account for the (non)medical complexities of how men actually access and manage testosterone. This binary framing not only obscures the structural barriers that limit access to pharmaceutical-grade testosterone through formal healthcare channels but also risks masking how these very barriers push people toward unregulated markets — such as underground suppliers and peer-sourced products — where product inconsistency, contamination, and absence of clinical monitoring heighten harm (Magnolini et al., 2022). A more pragmatic, process-oriented model of care is needed, acknowledging both the necessity of safe supply, and the realities of self-directed use.
A comparative policy lens clarifies why these barriers produce different harm landscapes across jurisdictions. In Australia, testosterone and other AAS are classified as controlled substances under the Poisons Standard, with illegality extending to possession, importation, and non-prescribed use (Handelsman, 2021; James & Wynn, 2022). Penalties vary significantly by state—from minor offences to 2–25-year prison terms for possession deemed “supply-related” (James & Wynn, 2022). In states such as Queensland and New South Wales, even simple possession can result in criminal prosecution, generating deterrence not just from illicit supply, but from seeking healthcare altogether (Piatkowski, Kill, et al., 2025).
In contrast, the United Kingdom (UK) adopts a partial-decriminalisation model: AAS are classified as Class C under the Misuse of Drugs Act (1971), where manufacture and distribution are criminalised, but personal possession for use is legal (McVeigh & Begley, 2017; Mullen et al., 2020). This distinction has important practical consequences: because individuals can legally possess both diverted pharmaceutical-grade testosterone and underground products without fear of arrest, informal peer-regulated markets operate in a far more open environment (McVeigh & Begley, 2017). Rather than being driven entirely underground, these markets can circulate a wider range of products—including diverted prescription esters—while enabling open discussion about quality, dosing, and risk-reduction practices (McVeigh & Begley, 2017; Mullen et al., 2020). The reduced threat of criminalisation increases honesty within user networks, facilitates access to harm-reduction advice, and creates a market less constrained by fear or punitive enforcement. Thus, informal networks in the UK frequently function as a de facto safe supply mechanism, helping to maintain product quality and facilitating engagement with harm-reduction advice (McVeigh & Begley, 2017).
By contrast, Australia’s stricter criminalisation pushes acquisition further underground, increasing exposure to counterfeit products, product mislabelling, and absence of clinical monitoring (Piatkowski et al., 2024). Policy reform in Australia must therefore recognise that criminalising possession does not prevent use, it redistributes harm. A shift toward models that consider decriminalisation and enable more options for clinical health monitoring would align regulatory intent with on-the-ground realities of testosterone use.
This study has several limitations that inform how findings should be interpreted. Our sample consisted of a small and demographically homogenous group of cis men prescribed testosterone, recruited through purposive methods. As such, the results offer context-specific insights rather than generalisable conclusions about all testosterone users. While testosterone is also a core medication for trans men, their healthcare pathways, legal contexts, and gatekeeping experiences differ substantially from those of cis men (Fraser, Moore, et al., 2020; Moravek, 2018). As trans men were not included in the present sample, we do not draw conclusions about their experiences here. Furthermore, because participants were recruited from a hidden population and anonymity was a key condition for engagement, only minimal demographic and contextual information was collected. Beyond age and TRT use patterns, we did not gather detailed socioeconomic data (e.g., employment/study status, income), ethnicity, training history, or other contextual characteristics that may shape testosterone access and usage. This decision supported participant safety and trust but limits the depth of sample characterisation we can provide. Taking these points together, future research including more diverse populations, particularly trans men and others who rely on testosterone for differing purposes, will be essential for developing harm-reduction and policy responses that are inclusive of the full spectrum of testosterone use.
Our analysis of TRT highlights the paradoxes and negotiations that define its biomedicalisation. While medicalised testosterone governance purports to offer standardised, evidence-based care, it also enacts exclusions that drive many men toward alternative strategies. By centring the lived-living experiences of those engaged in TRT, we illuminate the ways in which biomedical authority is contested, adapted, and, at times, subverted through peer-based expertise and self-directed care. Notably, our interviews revealed that some of the men who self-managed their testosterone use, did not demonstrate legitimate biomedical expertise, but rather engaged in trial and error, thus raising concerns about harm exposure. As such, future harm reduction efforts must move beyond conventional frameworks that position self-directed testosterone use as inherently deviant or dangerous. Instead, a more nuanced, participatory approach is required, one that recognises the legitimacy of peer knowledge and seeks to create more inclusive, responsive pathways for those navigating TRT outside formal biomedical structures. In doing so, we not only challenge dominant narratives of risk and legitimacy but also contribute to a broader reimagining of care in the context of contemporary biomedicalisation.
Footnotes
Acknowledgments
The authors wish to thank the participants for generously sharing their time and experiences. We also acknowledge the broader peer and harm reduction networks that supported recruitment and provided valuable insights throughout the research process.
Ethical Considerations
This study was approved by the Griffith University Human Research Ethics Committee (Approval Number: Approval: 2023/784).
Consent to Participate
All participants provided informed verbal consent prior to participation, including consent to record and transcribe interviews.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Correction (January 2026):
This article has been updated with minor style corrections since its original publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Due to the sensitive and identifiable nature of the qualitative interview data, the datasets generated and/or analysed during the current study are not publicly available. De-identified excerpts may be made available upon reasonable request to the corresponding author, subject to ethical approval.
