Abstract
Periodontitis is a complex, multifactorial inflammatory condition characterized by progressive destruction of the periodontal supporting structures. It profoundly affects oral health, esthetics, and masticatory function and is increasingly recognized as a contributing risk factor for systemic disorders. Deep periodontal pockets establish a severely hypoxic microenvironment induced by periodontal pathogens, increased oxygen consumption of infiltrated inflammatory cells, and accompanying periodontal vascular changes. The cellular response to hypoxia is centrally regulated by hypoxia-inducible factor 1 alpha (HIF-1α), which is also recognized as a critical factor driving the progression of periodontal tissue destruction. In vivo and in vitro studies have shown the upregulation of HIF-1α in both animal periodontitis models and clinical samples from individuals with periodontitis, where its expression correlates positively with deteriorating clinical periodontal parameters. Experimental and clinical studies using mouse conditional knockout models, selective small-molecule inhibitors, and human-derived materials have demonstrated clear causal roles for hypoxia-driven HIF-1α signaling in the progression of periodontitis. This conclusion is supported by mechanistic evidence demonstrating that HIF-1α induces aberrant neovascularization, enhances osteoclastogenesis leading to subsequent alveolar bone resorption, and promotes M1 macrophage polarization along with proinflammatory cytokine production. Hypoxia, via HIF-1α, synergistically acts with periodontal pathogens to amplify periodontal inflammation and oxidative stress, driving persistent extracellular matrix destruction in periodontal tissues. This critical review summarizes recent findings, using in vitro and in vivo approaches using animal and human-derived materials, on the role of hypoxia, primarily through the HIF-1α pathway, in periodontitis pathogenesis as well as the potential of hypoxia-based strategies and targeted modulation of HIF-1α signaling through HIF-1α stabilizers for managing periodontitis and promoting periodontal regeneration, while highlighting the existing gaps in our understanding and the limitations of current research, which can serve as a foundation for guiding future studies in this area.
Keywords
Introduction
Periodontitis, a chronic inflammatory disease of complex etiology, leads to the gradual destruction of tooth-supporting structures (Nascimento et al 2024). The primary clinical features of periodontitis include loss of periodontal attachment, periodontal pocket formation, and alveolar bone resorption (Nascimento et al 2024). These changes can culminate in tooth mobility and eventual tooth loss, accompanied by esthetic concerns and impaired masticatory function, ultimately diminishing quality of life (Nascimento et al 2024). Recently, the global burden of severe periodontitis was estimated to exceed 1 billion cases, with a prevalence rate of ~12.5% (Nascimento et al 2024). Periodontitis worsens the severity and progression of systemic inflammatory disorders and is associated with a significantly increased risk of cardiovascular disease and mortality in older adults (Bengtsson et al 2021).
Periodontitis is characterized by dynamic interactions among bacterial pathogens, host immune and inflammatory responses, and other various risk factors, such as systemic diseases associated with immunologic dysregulation or metabolic disorders and smoking (Bozyel et al 2025; Okano et al 2025). These interactions result in hypoxia, oxidative stress, microvascular changes, and persistent inflammation, collectively promoting irreversible destruction of periodontal tissues and progressive alveolar bone resorption (Zheng et al 2025). The hypoxic microenvironment in periodontitis is evidenced by significantly reduced gingival baseline oxygen saturation compared with healthy individuals, and diminished oxygen availability in periodontal tissues has been shown to accelerate disease progression (Mayr et al 2024). This implication of hypoxia in the pathogenesis of periodontitis is also supported by the evidence demonstrating the upregulation of hypoxia-inducible factor-1α (HIF-1α), a key transcriptional regulator that mediates cellular adaptation to low oxygen tension (Lee et al 2020; Zheng et al 2025), in both in vivo models and patients with periodontitis (Ebersole et al 2021; Wang et al 2022; Kzar and Abbas 2025; Taşdemir et al 2025), suggesting its potential as a biological marker for disease activity and progression. Thus, this critical review aims to elucidate the contribution of hypoxia/HIF-1α signaling to periodontitis pathogenesis while highlighting its context-dependent roles in periodontal homeostasis. It further synthesizes current preclinical evidence on hypoxic preconditioning and HIF-1α modulation as potential therapeutic strategies for periodontal repair and identifies key knowledge gaps limiting mechanistic insight and clinical translation. In this review, literature was identified through searches of PubMed using combinations of keywords including “HIF-1α,” “hypoxia,” “periodontitis,” “gingiva,” “periodontal ligament,” “bone resorption,” and “regeneration.” Priority was given to primary experimental, translational, and clinical studies published within the last 10 to 15 y. Studies were selected based on relevance to hypoxia-responsive mechanisms in periodontal biology and disease and clinical or translational implications. Earlier seminal studies were included where necessary to provide fundamental or conceptual context.
Structure, Regulation, and Physiological Roles of HIF-1α in Periodontal Health
HIF is a heterodimeric transcriptional factor composed of an oxygen-labile, inducibly expressed α-subunit and a stable, constitutively expressed β-subunit (Lee et al 2020). Mammalian cells express 3 HIFα isoforms: HIF-1α, HIF-2α, and the less extensively studied HIF-3α. These isoforms share a conserved structure comprising an N-terminal basic helix–loop–helix and PAS domains for DNA binding and dimerization, a central oxygen-dependent degradation domain, and C-terminal transactivation domains that regulate hypoxia-responsive gene transcription and vary among HIFα isoforms (Wenger 2000) (Fig 1). Although HIF-1α is ubiquitously expressed in all cell types, HIF-2α and HIF-3α exhibit tissue-specific expression; however, data on their levels in oral tissues remain limited (Wenger 2000; Bozyel et al 2025). In normoxia, the HIFα subunit is hydroxylated at specific proline residues by prolyl hydroxylase domain (PHD) enzymes, whose activity depends on oxygen, 2-oxoglutarate (α-ketoglutarate), and iron (Yu et al 2001). Following hydroxylation, HIFα is targeted by the von Hippel–Lindau (VHL) tumor suppressor protein (pVHL) complex for ubiquitylation and proteasomal degradation (Lee et al 2020). Under hypoxia, reduced PHD activity stabilizes HIFα, allowing its nuclear translocation, dimerization with HIFβ, and recruitment of the transcriptional coactivators p300/CBP to form an active complex that binds to the hypoxia-response elements in target gene promoters, thereby driving the expression of genes that mediate adaptive responses to hypoxia, including tissue homeostasis and repair, angiogenesis, proliferation, metabolism, and extracellular matrix (ECM) remodeling (Lee et al 2020) (Fig 1). For example, in healthy gingiva, localized HIF-1α stabilization in sulcular and oral epithelium supports cell survival through metabolic adaptation within the physiological oxygen gradients, and its positive correlation with vascular endothelial growth factor (VEGF) expression suggests a physiological role in maintaining tissue homeostasis (Ng et al 2011; Vasconcelos et al 2016). In addition, HIF-1α plays protective roles in promoting angiogenesis and tissue repair essential for tooth extraction socket healing and in supporting postimplantation survival and the angiogenic capacity of stem cells from human exfoliated deciduous teeth (Han et al 2021; Linawati et al 2024). Furthermore, under compressive orthodontic strain or mildly excessive occlusal forces, HIF-1α stabilization in the periodontal ligaments (PDLs) driven by vascular compression-induced hypoxia and mechanotransduction is considered to maintain tissue homeostasis and promote angiogenesis essential for physiological periodontal remodeling (Ullrich et al 2019; Takedachi et al 2022). However, HIF-1α appears to play a context-dependent role in periodontal tissues, supporting physiological hypoxia-responsive processes essential for periodontal homeostasis and wound healing while acting as a potential contributor to periodontitis pathogenesis when chronically activated and further induced by metabolic alterations and inflammatory signals (Su et al 2020).
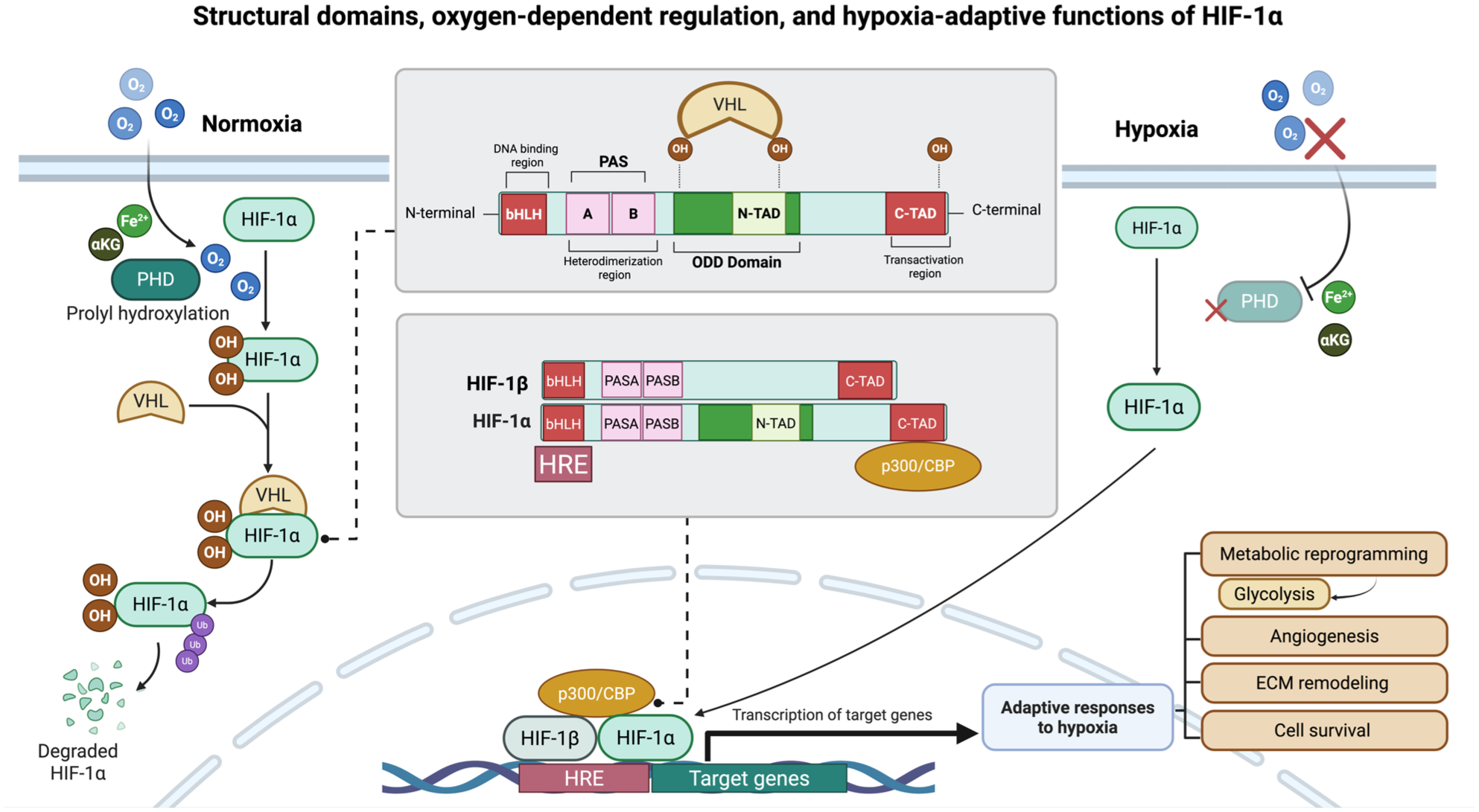
Structural domains, oxygen-dependent regulation, and hypoxia-adaptive functions of HIF-1α. Hypoxia-inducible factor (HIF) is a heterodimeric transcription factor composed of an oxygen-labile α-subunit and a constitutively expressed β-subunit. HIF-1α contains an N-terminal basic helix–loop–helix (bHLH) for DNA binding and PAS (PAS-A and PAS-b) domains required and dimerization with HIF-1β, a central oxygen-dependent degradation domain (ODDD), and 2 transactivation domains: an N-terminal transactivation domain (N-TAD), which overlaps with the ODDD and contributes to hypoxia-induced transcriptional activity, and a C-terminal transactivation domain (C-TAD), which mediates recruitment of transcriptional coactivators such as p300/CBP. Under normoxic conditions, HIF-1α is hydroxylated at specific proline residues by prolyl hydroxylase domain (PHD) enzymes in an oxygen-, Fe2+-, and α-ketoglutarate (αKG)–dependent manner, promoting recognition by the von Hippel–Lindau (VHL) E3 ubiquitin ligase complex and subsequent proteasomal degradation. Under hypoxic conditions, reduced PHD activity stabilizes HIF-1α, allowing its nuclear translocation, heterodimerization with HIF-1β, and recruitment of p300/CBP via the C-TAD. The resulting active HIF-1 complex binds the hypoxia-response element (HRE) in target gene promoters, inducing transcriptional programs that mediate cellular adaptation to hypoxia, including angiogenesis, metabolic reprogramming, cell survival, and extracellular matrix remodeling to maintain tissue homeostasis. Figure created in https://BioRender.com.
Periodontitis Tissues Are Characterized by Hypoxia and Exhibit Elevated HIF-1α Levels
Gingival tissues from patients with periodontitis exhibit significantly reduced baseline oxygen saturation compared with healthy tissues (Mayr et al 2024). Specifically, oxygen tension at the base of untreated periodontal pockets, which ranged from 5 to 27 mm, was reported to be as low as 13.3 mm Hg (≈1.8% O2) in early studies (Loesche et al 1983). Consistent with this hypoxic microenvironment, HIF-1α, the central transcriptional regulator of cellular responses to hypoxia, shows markedly elevated expression in mRNA and protein levels in the periodontal tissues of patients with periodontitis compared with healthy controls (Ng et al 2011; Xiao et al 2020; Zhang et al 2025). HIF-1α expression has also been linked in many studies to disease severity, with higher levels observed in advanced compared with moderate chronic periodontitis (Shi et al 2015). Furthermore, microarray analysis of gingival samples from an in vivo periodontitis model showed early and sustained upregulation of HIF-1α pathway–related genes that correlated with periodontitis-associated bacterial pathogenic complexes, highlighting a reciprocal interaction between microbial dysbiosis and hypoxia that drives disease progression (Ebersole et al 2021).
HIF-1α as a Potential Diagnostic Biomarker in Periodontitis
HIF-1α has shown strong diagnostic potential as a biomarker for periodontitis (Afacan et al 2019; Taşdemir et al 2025). Elevated salivary HIF-1α levels reported in systemically healthy, nonsmoking patients with stage III periodontitis showed a positive correlation with clinical parameters of disease severity, including gingival index, plaque index, bleeding on probing, probing depth, and clinical attachment level (Taşdemir et al 2025). At the gingival crevicular fluid (GCF) level, elevated HIF-1α expression has been reported in patients with chronic or generalized aggressive periodontitis compared with gingivitis or healthy controls and was correlated with site-specific clinical periodontal parameters and other hypoxia- and inflammation-related biomarkers (Afacan et al 2019). Recently, increased HIF-1α levels in GCF from patients with stage III grade B/C periodontitis were found to positively correlate with other hypoxia-responsive factors, including HIF-2α, HIF-3α, and VEGF (Bozyel et al 2025). However, although elevated HIF-1α expression in GCF has been confirmed in cohorts including different clinical stages and grades of periodontitis, the possible stage-dependent variation in HIF-1α levels has not been systematically evaluated (Ning et al 2025). Despite existing evidence implicating HIF-1α in periodontitis pathogenesis, limited disease stratification and longitudinal evaluation across disease stages highlight the need for stage-stratified, longitudinal clinical studies with careful control of systemic confounders to define temporal HIF-1α dynamics and validate its potential as a diagnostic and disease-monitoring biomarker.
HIF-1α and Aberrant Angiogenesis in Periodontitis
Aberrant angiogenesis constitutes a pathological manifestation of periodontitis, characterized by the uncontrolled formation of new blood vessels within inflamed tissues (Lin et al 2025). Despite reduced oxygen saturation, periodontitis-affected gingival tissues paradoxically exhibit increased blood flow compared with healthy tissues (Mayr et al 2024). This is likely attributable to hypoxia-induced angiogenesis and inflammation-driven vasodilation, increasing microvascular perfusion to support immune cell infiltration and the sustained release of proinflammatory mediators (Mayr et al 2024). However, the elevated metabolic and oxygen demands of infiltrating immune cells deplete available oxygen, indicating that the enhanced perfusion does not fully compensate for tissue hypoxia (Mayr et al 2024). Histologically, periodontitis tissues exhibit increased vascularization, evidenced by the elevated expression of the vascular marker CD31and the proangiogenic factors VEGF-A and Angiopoietin 1 (ANGPT1) (Lin et al 2025). The mechanism underlying this reactive vascularization in periodontitis may involve the development of a hypoxic microenvironment within periodontal pockets, largely driven by the metabolic activity of oral pathogens, which stabilizes HIF-1α that induces VEGF transcription, promoting pathological angiogenesis and bleeding tendency in inflamed sites (Kzar and Abbas 2025) (Fig 2). Consistent with this, elevated HIF-1α levels in gingival tissues from patients with chronic periodontitis have been associated with increased VEGF-A expression and positively correlated with VEGF-C levels, which increase with periodontitis severity as compared with healthy controls (Ng et al 2011; Shi et al 2015; Vasconcelos et al 2016).
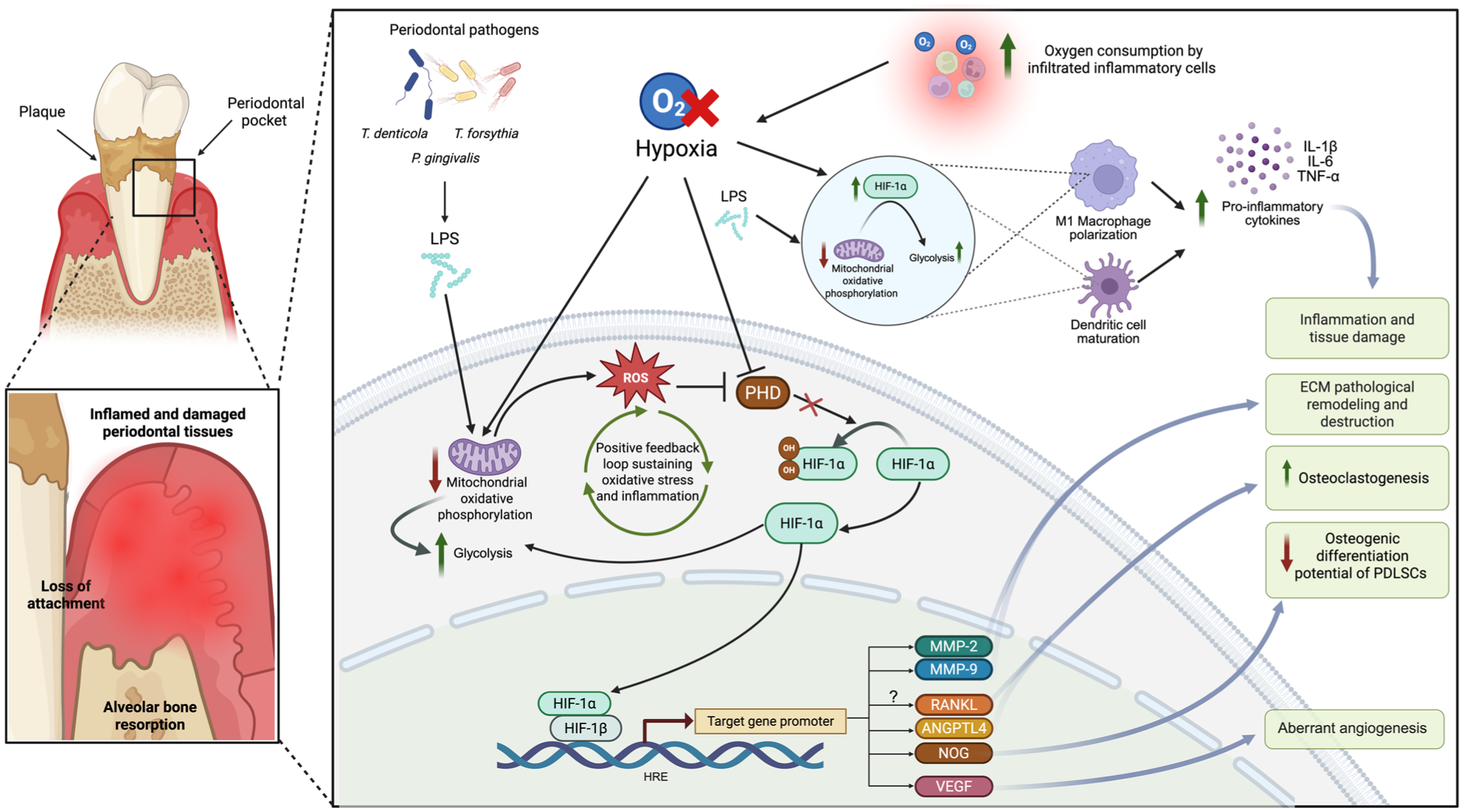
Molecular mechanisms underlying hypoxia-induced inflammation, aberrant angiogenesis, extracellular matrix (ECM) remodeling, and alveolar bone resorption in periodontitis. The hypoxic microenvironment is a characteristic of deep periodontal pockets. Low oxygen levels inhibit prolyl hydroxylase domain (PHD) enzymes, resulting in stabilization and nuclear translocation of hypoxia-inducible factor 1 alpha (HIF-1α), which activates the transcription of genes contributing to periodontitis pathogenesis. These include vascular endothelial growth factor (VEGF), promoting aberrant angiogenesis, and matrix metalloproteinases MMP-2 and MMP-9, which facilitate ECM degradation. HIF-1α also enhances osteoclastogenesis by upregulating angiopoietin-like protein 4 (ANGPTL4) and NOG (Noggin), the latter associated with reduced osteogenic differentiation of periodontal ligament stem cells. Although HIF-1α has been implicated in regulating RANKL expression under hypoxia, its direct role in this pathway in the context of periodontitis remains unconfirmed, as indicated by the question mark in the figure. In addition, HIF-1α promotes a metabolic shift from oxidative phosphorylation to glycolysis and increases reactive oxygen species (ROS), which further inhibit prolyl hydroxylase domain (PHD) activity and reinforce HIF-1α stabilization, generating a positive feedback loop that sustains oxidative stress and inflammation. Lipopolysaccharides (LPS) from periodontal pathogens can also stabilize HIF-1α through glycolysis induction and succinate-driven ROS production, promoting M1 macrophage polarization and dendritic cell maturation, together increasing interleukin (IL)–6, IL-1β, and tumor necrosis factor–α production and driving inflammatory ECM destruction and alveolar bone resorption. Figure created in https://BioRender.com.
The enhanced proangiogenic capacity observed in periodontitis has been shown to be largely mediated by periodontal ligament stem cells (PDLSCs; Zhang et al 2021). For instance, PDLSCs isolated from patients with stage 3 periodontitis exhibit upregulation of ubiquitin C-terminal hydrolase L1 (UCHL1), a deubiquitinating enzyme regulating protein stability that was shown to promote PDLSC-driven angiogenesis by increasing VEGF-A and ANGPT1 secretion through HIF-1α–dependent activation of Yes-associated protein (YAP) signaling (Lin et al 2025). In vivo, a murine model of periodontitis showed elevated HIF-1α and YAP expression associated with upregulated VEGF-A, ANGPT1, and CD31, all of which were attenuated by UCHL1 inhibition (Lin et al 2025). Although these studies implicate HIF-1α signaling in periodontitis-associated angiogenesis, they rely largely on correlative human data and indirect pathway modulation, such as UCHL1 inhibition, underscoring the need for direct, cell-specific manipulation of HIF-1α in periodontitis models to establish causality and define its mechanistic role in this process.
Potential Contribution of HIF-1α to Periodontal ECM Destruction
Periodontitis is characterized by the irreversible degradation of the ECM, a key structural component of the periodontium and surrounding alveolar bone (Ning et al 2025). This inflammatory destruction is triggered by a dysbiotic oral microbiota and mediated by excessive host-derived proteinases, leading to periodontal attachment loss and alveolar bone resorption (Ning et al 2025). Stimulation of PDLSCs in vitro with the bacterial endotoxin lipopolysaccharide (LPS) under hypoxia increased the expression of HIF-1α, ECM-degrading matrix metalloproteinases (MMPs) MMP-2 and MMP-9, and inflammatory cytokines, whereas siRNA-mediated inhibition of HIF-1α attenuated these responses (Ning et al 2025). These findings are further supported clinically by elevated HIF-1α levels in the GCF of patients with periodontitis, which are positively correlated with MMP-2, MMP-9, and the proinflammatory cytokines interleukin-6 (IL-6), IL-1β, and tumor necrosis factor–α (TNF-α), highlighting the role of HIF-1α in MMP-mediated ECM degradation and inflammatory responses in periodontitis (Ning et al 2025) (Fig 2). At the tissue level, studies on gingival tissues from periodontitis patients reported increased MMP-8 and reduced collagen-related enzymes alongside divergent patterns of HIF-1α expression; a study showed elevated HIF-1α levels in stage III grade B lesions (Karatas et al 2020), whereas another reported reduced HIF-1α expression in stage III grade B/C lesions, with the lowest levels observed in the most severe grade C (Taskan et al 2019), whereas no significant change was observed in an earlier study that compared chronic periodontitis with healthy controls and lacked stratification by disease stage and severity (Balci Yuce et al 2019). Together, limited disease stratification and insufficient integration of clinical parameters across studies hinder cross-study comparison and interpretation of how HIF-1α signaling evolves during periodontitis progression, while methodological differences, including biomarkers in GCF versus tissue-level histological analyses, provide complementary but nonequivalent insights into HIF-1α activity in ECM destruction. Addressing these gaps through clinical stage-stratified analyses and targeted HIF-1α inhibition in relevant in vivo models will be essential to define its temporal dynamics and potential stage-dependent roles in periodontitis-associated ECM destruction, which may guide strategies to modulate HIF-1α activity to balance ECM degradation and regeneration.
Role of HIF-1α in Periodontitis-Induced Alveolar Bone Resorption
Hypoxia influences alveolar bone homeostasis, as demonstrated in periodontitis-free rat models in which unilateral nasal obstruction–induced hypoxia enhanced osteoclastogenesis and inflammatory responses, leading to a reduction in mandibular bone mineral density and destruction of alveolar bone (Kim et al 2022; Xu et al 2023). A hypoxic microenvironment is commonly observed in osteolytic diseases such as periodontitis, in which progressive alveolar bone resorption underlies disease progression (Yu et al 2020; Qi et al 2023). Hypoxia was shown to exacerbate alveolar bone resorption in a periodontitis rat model by promoting osteoclast activity and the expression of the inflammatory mediator prostaglandin E2 (Terrizzi et al 2021). With regard to the contribution of HIF-1α to this process, chemical stabilization of HIF-1α using the hypoxia mimetic cobalt chloride (CoCl2) enhanced the osteoclastogenesis of RAW264.7 macrophages via upregulation of angiopoietin-like protein 4 (ANGPTL4), while osteoclast-specific deletion of HIF-1α in a periodontitis mouse model markedly reduced osteoclast numbers, ANGPTL4 expression in periodontitis tissue, and alveolar bone resorption compared with controls (Qi et al 2023). Furthermore, hypoxia suppresses the osteogenic differentiation capacity of PDLSCs in vitro via HIF-1α–dependent upregulation of NOG (Fig 2), encoding Noggin, a bone morphogenetic protein antagonist, consistent with observations in the alveolar bone of rat hypoxia models (Pan et al 2022); notably, the in vitro inhibition of HIF-1α in LPS-stimulated PDLSCs under hypoxia rescues their osteogenic differentiation (Ning et al 2025). In addition, in vitro hypoxia was shown to stabilize HIF-1α together with increased expression of the osteoclastogenic marker receptor activator of nuclear factor-κB ligand (RANKL) in PDL cells (Yu et al 2015; Hase et al 2025) and enhanced RANKL-induced osteoclast differentiation and resorptive activity in bone marrow–derived macrophages (Yu et al 2020); these findings align with in vivo evidence of increased HIF-1α and RANKL expression in alveolar bone in a rat model of nasal obstruction–induced hypoxia (Kim et al 2022). Although not examined in periodontitis models, these findings suggest that HIF-1α may regulate RANKL-mediated osteoclast differentiation; however, the mechanistic role of HIF-1α in controlling RANKL expression remains incompletely defined and warrants investigation using cell-specific HIF-1α inhibition in in vitro and in vivo periodontitis models.
Role of HIF-1α in Modulating Host Inflammatory Responses in Periodontitis
HIF-1α and Proinflammatory M1 Macrophage Polarization
Increased HIF-1α expression has been reported in inflamed periodontal tissues, in ligature-induced periodontitis in an in vivo model, and in LPS-stimulated macrophages and periodontal cells (Ng et al 2011; Zeng et al 2024). Stabilized HIF-1α drives a metabolic shift in macrophages toward glycolysis, driving M1 proinflammatory macrophage polarization and inducing the production of inflammatory mediators such as IL-1β in vitro (Zeng et al 2024; Zhang et al 2025) (Fig 2). Consistent with this mechanism, suppression of HIF-1α–mediated glycolysis pathways using phloretin reduced M1 polarization in LPS-stimulated macrophages and attenuated periodontal inflammation and alveolar bone loss in a ligature-induced periodontitis model (Zeng et al 2024). In addition, the ability of human gingival mesenchymal stem cell–derived exosomes to induce metabolic reprogramming toward an anti-inflammatory M2 macrophage phenotype was inhibited by the stabilization of HIF-1α using dimethyloxallyl glycine (DMOG) in LPS-stimulated macrophages (Wang et al 2025). Together with patient-derived evidence, single-cell RNA sequencing analysis demonstrated significant upregulation of the HIF-1α pathway and proinflammatory M1-associated genes, including NLR family pyrin domain–containing protein 3 (NLRP3) and IL-1β, in gingival macrophages from patients with periodontitis compared with healthy individuals (Zhang et al 2025). However, definitive evidence for the role of HIF-1α–mediated metabolic reprogramming in driving the polarization of periodontitis-associated proinflammatory macrophages will require macrophage-specific deletion of HIF-1α in in vivo periodontitis models that could be integrated with spatial transcriptomics to resolve the tissue-level microenvironmental cues and cell–cell interactions that govern HIF-1α–dependent regulatory networks.
Role of HIF-1α in Periodontal Pathogen-Induced Inflammatory Response
Periodontitis is significantly associated with the “red complex” of gram-negative anaerobes, including Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia, with P. gingivalis recognized as the keystone pathogen driving its onset and progression (Mineoka et al 2008). Exposure to P. gingivalis induces a metabolic shift in vitro in PDLSCs from oxidative phosphorylation to glycolysis, resulting in intracellular succinate accumulation and elevated reactive oxygen species (ROS) (Su et al 2020) (Fig 2). These changes inhibit PHD2 activity, leading to stabilization of HIF-1α and upregulation of proinflammatory mediators such as IL-6 and MCP-1, an effect that is significantly attenuated by the pharmacologic inhibition of HIF-1α with 2-methoxyestradiol (Su et al 2020). A subsequent in vitro study on human gingival fibroblasts (HGFs) showed that hypoxia together with P. gingivalis–LPS synergistically stabilized HIF-1α and promoted the production of IL-1β and the activation of the NLRP3 inflammasome complex, suggesting that hypoxia in periodontal pockets may amplify LPS-driven inflammatory signaling in periodontitis (Yang et al 2021). However, neither hypoxia nor P. gingivalis–LPS alone was sufficient to activate the NLRP3 inflammasome (Yang et al 2021). Rather, hypoxia is required for P. gingivalis–LPS–induced NLRP3 expression, caspase-1 activation, and subsequent IL-1β production and maturation in vitro in HGFs, consistent with observations of increased HIF-1α, NLRP3, and IL-1β expression in gingival tissues from patients with chronic periodontitis compared with healthy controls (Cheng et al 2017). However, the precise mechanistic role of HIF-1α in regulating this inflammatory response remains to be clarified through cell-specific HIF-1α inhibition and validations in relevant in vivo periodontitis models.
Furthermore, HIF-1α regulates the glycolytic metabolism and maturation of dendritic cells (DCs), key mediators in periodontal inflammation and alveolar bone resorption (Wu et al 2023) (Fig 2). In vitro, the suppression of HIF-1α expression in LPS-stimulated DCs by butyrate, an anti-inflammatory microbial metabolite, significantly reduced glycolytic activity and impaired DC maturation, accompanied by reduced production of the proinflammatory cytokines TNF-α, IL-1β, and IL-12 (Wu et al 2023). These findings are consistent with reduced maturation of DCs, attenuated inflammation, and alleviated alveolar bone resorption in butyrate-treated ligature-induced periodontitis mice (Wu et al 2023). Conversely, overexpression of HIF-1α via lentiviral transfection restored glycolysis and proinflammatory cytokine release despite butyrate treatment, suggesting that HIF-1α drives LPS-induced inflammatory response of DCs (Wu et al 2023).
Moreover, a recent bioinformatic analysis of patient-derived periodontitis microarray datasets identified differentially expressed neutrophil extracellular trap (NET)–related genes and proposed a 7-gene diagnostic signature that includes HIF1A with strong predictive performance (Yu et al 2025). Validation in a P. gingivalis–induced periodontitis mouse model confirmed marked HIF-1α upregulation alongside increased NET formation, suggesting a potential role for HIF-1α in hypoxia-driven neutrophil activation (Yu et al 2025). However, validation on patient periodontal samples and experimental investigations are still needed to determine whether HIF-1α directly induces NET formation and whether neutrophil-specific inhibition of HIF-1α can attenuate periodontal inflammation in vivo.
Role of HIF-1α in Oxidative Stress–Induced Periodontal Inflammatory Responses
Periodontitis is characterized by hypoxia and excessive oxidative stress, which drive sustained inflammation and tissue destruction (Zheng et al 2025). In hypoxia, impaired electron transfer within the mitochondrial respiratory chain reduces oxidative phosphorylation and increases reliance on glycolysis, leading to excessive ROS generation, which amplifies oxidative stress and inflammation in part through HIF-1α signaling (Fig 2) (Wu et al 2020; Zheng et al 2025). P. gingivalis has been shown to induce a similar response in vitro in PDLSCs, promoting ROS accumulation, which stabilizes HIF-1α and amplifies downstream inflammatory signaling, an effect further intensified under hypoxia (Su et al 2020). This relationship appears to be reciprocal, forming a positive feedback loop in which HIF-1α may promote further ROS production, thereby sustaining oxidative stress–driven inflammation in periodontitis (Zhang et al 2025). Supporting this, in vitro studies showed that stabilization of HIF-1α with CoCl2 increased intracellular and mitochondrial ROS and triggered apoptosis in PDL cells; importantly, siRNA-mediated HIF-1α silencing reversed these effects (Gao et al 2021), while suppression of HIF-1α in LPS-stimulated macrophages reduced glycolytic metabolism, ROS generation, and M1 macrophage polarization (Zhang et al 2025). These observations suggest that HIF-1α may regulate ROS-mediated inflammatory responses and propose its contribution to periodontal tissue damage. Furthermore, in vitro ROS accumulation in CoCl2-treated PDL cells induced additional inflammatory pathways, including the NLRP3 inflammasome, thioredoxin-interacting protein (TXNIP), and IL-1β, effects reversed by ROS inhibition, while activation of the same inflammatory mediators was also reported in periodontal tissues from a rat model of unilateral nasal obstruction–induced hypoxia (Zhu et al 2022). Although the role of HIF-1α in ROS-driven periodontal inflammatory responses was not directly assessed in the periodontitis models in these studies, its potential involvement is still supported by evidence showing that both hypoxia and CoCl2 stabilize HIF-1α, together with prior findings linking HIF-1α to ROS-dependent inflammatory signaling in periodontal cells (Zhu et al 2022). In human periodontitis tissues, the increased expression of the ROS-generating enzyme NADPH oxidase 4 (NOX4) has been reported, and in vitro studies further show that NOX4-dependent ROS production is amplified in PDL cells exposed to hypoxia and/or P. gingivalis–LPS (Gölz et al 2014). Given the limited in vivo and clinical evidence defining the mechanistic role of the HIF-1α–ROS axis in periodontitis, these findings underscore the need for future studies using cell-specific HIF-1α inhibition in relevant in vivo periodontitis models and longitudinal human sampling across different stages of periodontitis to examine correlations between HIF-1α–ROS signaling and periodontal clinical parameters such as pocket depth and bone resorption.
Hypoxia as a Link between Systemic Disease–Induced Periodontitis and the Progression of Systemic Diseases
Systemic autoimmune disorders and chronic inflammatory diseases have been associated with the development and progression of periodontitis (Okano et al 2025). Hypoxia, mediated by HIF-1α, is linked to diseases, particularly those associated with microvasculopathy, and contributes to the destruction of both soft and hard tissues, as observed in conditions such as diabetes, rheumatoid arthritis, and systemic sclerosis (Taskan et al 2019). For instance, HIF-1α has been identified as a biomarker, reflecting microvascular changes in systemic sclerosis (Maciejewska et al 2023). In addition, periodontitis has also been shown to exacerbate the severity and progression of systemic autoimmune diseases (Okano et al 2025). Evidence further indicates that hypoxia, through the HIF-1α pathway, enhances NLRP3 inflammasome activation in response to the periodontitis-related pathogen P. gingivalis, contributing to the progression of multiple sclerosis in vivo (Okano et al 2025). However, despite the association between these inflammatory autoimmune diseases and vasculopathy-induced hypoxia (Maciejewska et al 2023), there are a few, if any, clinical studies examining the role of hypoxia through HIF-1α in mediating periodontitis linked to systemic disorders, highlighting an urgent need for future research.
Controlled Modulation through Hypoxic Preconditioning and Pharmacologic HIF-1α Stabilization May Promote Periodontal Tissue Repair
The effects of hypoxia through HIF-1α signaling in periodontal health are highly context dependent (Su et al 2020). Chronic hypoxia and persistent HIF-1α activation, combined with metabolic alterations and inflammatory signals, contribute to periodontitis pathogenesis. Yet, acute hypoxic responses, mediated by transient HIF-1α expression, are essential for tissue homeostasis, angiogenesis, and reparative processes during wound healing, including posttooth extraction socket healing (Linawati et al 2024) (Fig 3). Therefore, the biological outcome of HIF-1α activation is strongly influenced by timing, duration, cellular context, and microenvironmental conditions, suggesting that controlled modulation rather than complete inhibition may be therapeutically advantageous, as strategies such as hypoxic preconditioning or pharmacologic hypoxia mimetics have been studied for their potential to alleviate periodontitis and enhance regeneration (Yu et al 2016; Chen et al 2025) (Fig 4). For instance, short-term hypoxic preconditioning improved PDLSCs’ function in vitro and enhanced periodontal regeneration in vivo (Yu et al 2016), extracellular vesicles derived from hypoxia-preconditioned bone marrow stem cells improved peri-implant osseointegration in a diabetic rat model (Cao et al 2025), and delivery of hypoxia-preconditioned deciduous dental pulp stem cells within HIF-1α–loaded biomaterials attenuated periodontitis and promoted bone regeneration in vivo (Chen et al 2025). Furthermore, pharmacologic stabilization of HIF-1α using the hypoxia mimetic CoCl2 promoted alveolar bone defect healing in vivo, and transplanted CoCl2-pretreated human umbilical cord mesenchymal stem cells reduced inflammation and osteoclastogenesis in a peri-implantitis model (Fan et al 2024; Kuntjoro et al 2025). Similarly, deferoxamine, an iron-chelating HIF-1α stabilizer, and the hypoxia mimetic DMOG alleviated periodontitis and alveolar bone loss when delivered in vivo (Zhu et al 2024). Interestingly, other approaches, such as in vivo delivery of recombinant human HIF-1α protein to extraction sockets or distraction gaps, have been shown to enhance new bone formation (Jiang et al 2016; Lim et al 2018). Although these approaches show promising potential, more extensive preclinical investigations are required before translation to human applications, including the integration of human-derived models and clinically relevant disease settings to establish standardized translational protocols, define context-specific applications across different stages of periodontitis and patient conditions, and rigorously assess the safety of hypoxia-mimetic agents.
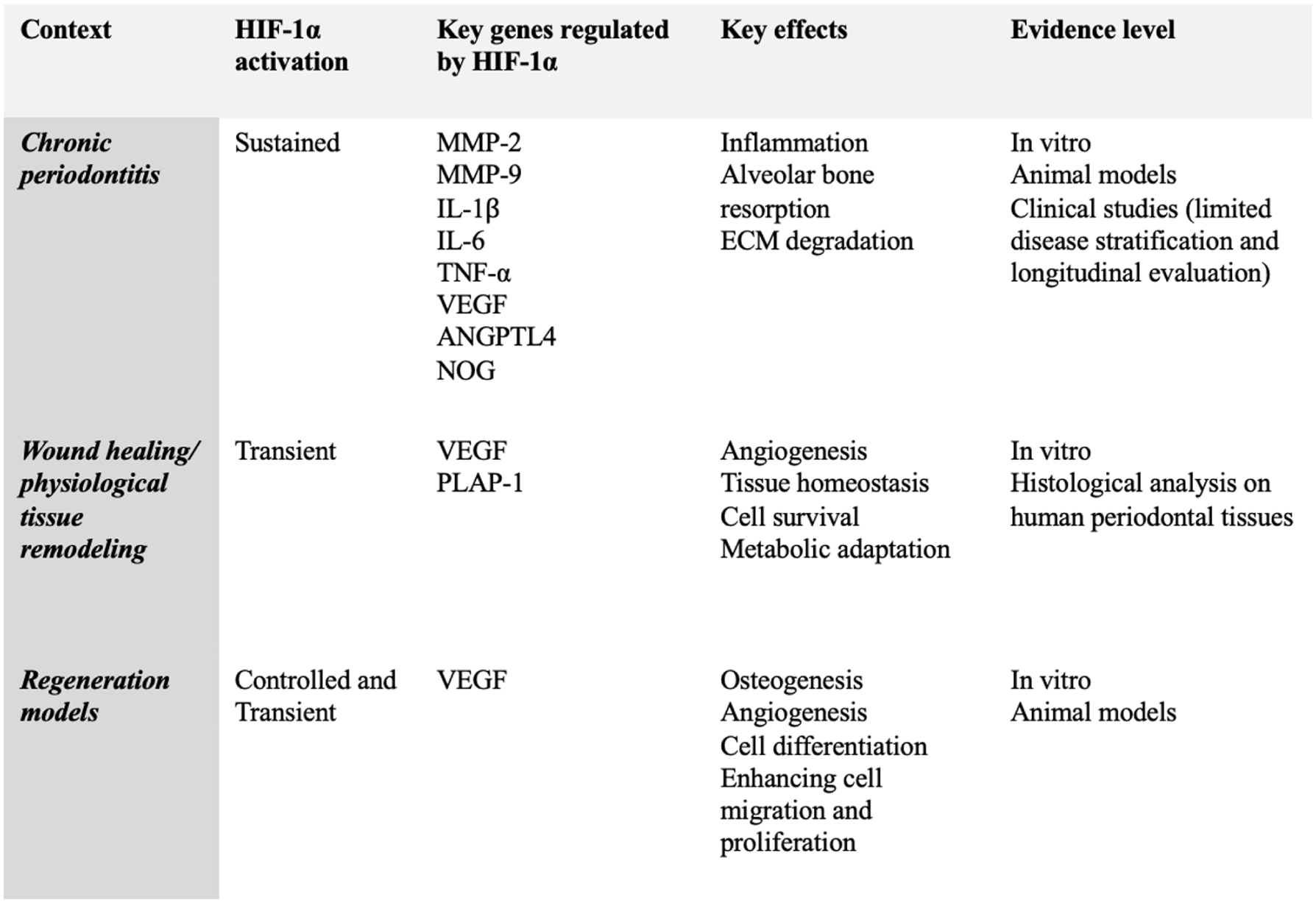
Context-dependent roles of hypoxia-inducible factor 1 alpha (HIF-1α) in periodontal tissues.
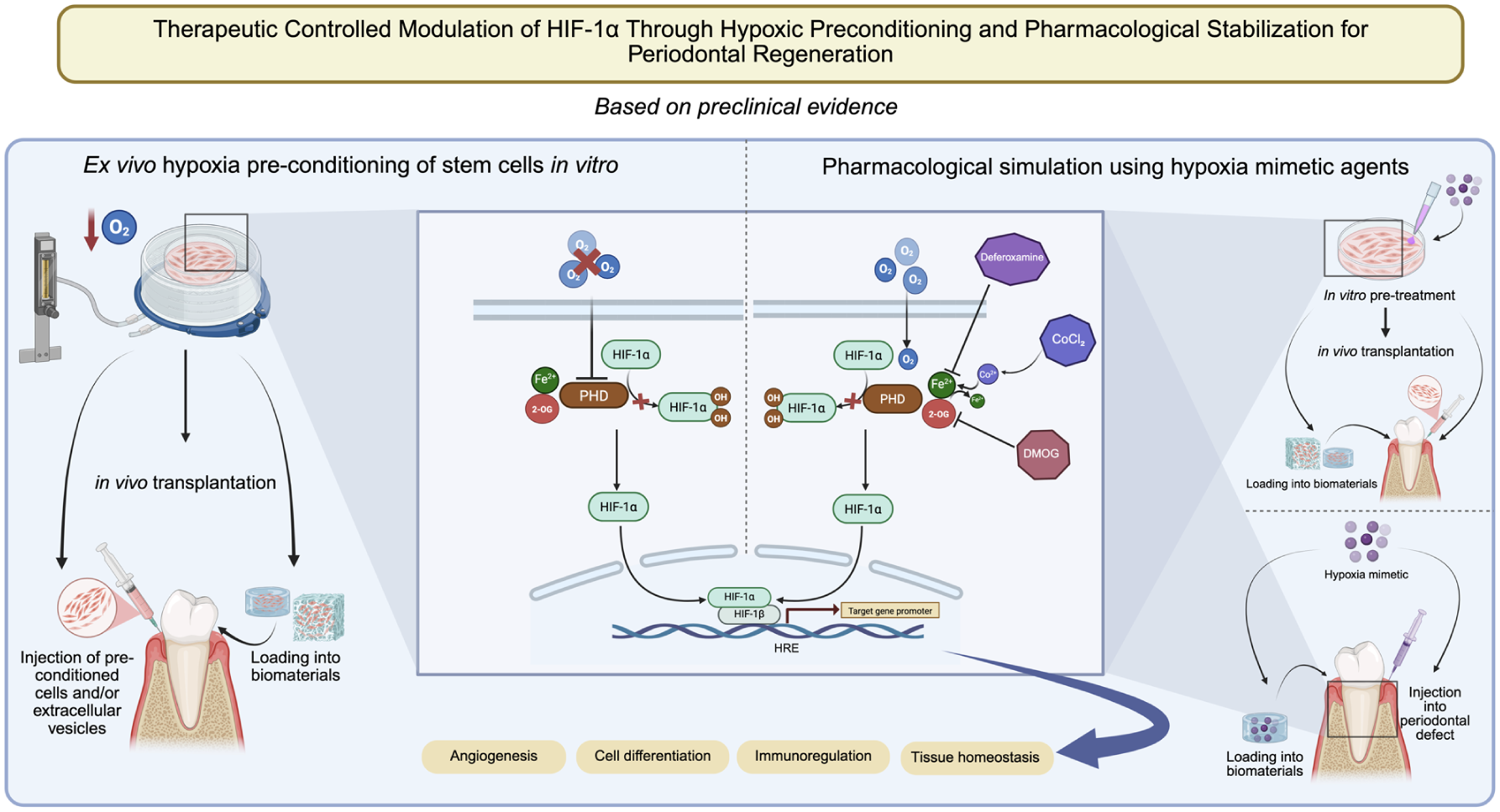
Hypoxia-based strategies for periodontal regeneration. These strategies include hypoxia preconditioning and pharmacologic simulation with hypoxia mimetics. Ex vivo hypoxia preconditioning is performed by exposing stem cells to low oxygen levels in vitro, after which preconditioned cells or their extracellular vesicles can be transplanted into periodontal defects or incorporated into biomaterials such as hydrogels and scaffolds. Hypoxia-mimetic agents that stabilize hypoxia-inducible factor 1 alpha (HIF-1α) by inhibiting prolyl hydroxylase domain (PHD) activity may be used to pretreat cells prior to transplantation, injected directly to defect sites, or loaded within biomaterial carriers. Both approaches act through HIF-1α signaling to maintain tissue homeostasis and enhance angiogenesis, cell differentiation, and immunomodulation essential for periodontal healing and regeneration. Figure created in https://BioRender.com.
Conclusion
Periodontitis is a chronic inflammatory disease characterized by the destruction of tooth-supporting structures, primarily driven by the interaction between pathogens and the host inflammatory response within a hypoxic microenvironment (Ng et al 2011). Hypoxia stabilizes HIF-1α protein, which plays a significant role in the disease’s pathogenesis by regulating inflammation, aberrant angiogenesis, ECM degradation, and alveolar bone resorption (Afacan et al 2019). However, rather than being inherently detrimental, cellular responses to hypoxia mediated by HIF-1α exert a context-dependent influence on periodontal health, serving as a key mediator of physiological responses to hypoxia that support tissue homeostasis, angiogenesis, wound healing, and periodontal remodeling while contributing to periodontitis progression and tissue destruction when activation is persistent (Su et al 2020). It should be noted that limited stratification by disease stage and longitudinal sampling in the available clinical studies, together with insufficient cell-specific modulation of HIF-1α in periodontitis models, complicates the interpretation of HIF-1α–associated mechanisms across disease progression. In addition, future in vivo and clinical studies could advance mechanistic understanding and support the development of targeted therapeutic strategies for individuals at high risk of periodontal destruction associated with systemic disorders characterized by microvasculopathy and hypoxia-induced HIF-1α signaling activation (Taskan et al 2019). Finally, controlled modulation of HIF-1α signaling through hypoxia preconditioning or hypoxia-mimetic strategies has shown promise in vivo and may offer new avenues for periodontal regeneration; however, extensive preclinical studies are still required to evaluate efficacy and safety and to establish translational, patient- and stage-specific protocols before human application.
Author Contributions
A. Fadl, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; A. Leask, contributed to conception and design, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A. Leask is supported by the Canadian Institutes of Health Research (MOP-183830). A. Fadl is the recipient of a Saskatchewan Innovation & Opportunity Doctoral Scholarship.
Data Availability
As a review article, no data were used or generated for this article.
