Abstract
The purpose of this study was to determine the average treatment effect (ATE) of tooth extraction before radiotherapy on the risk of osteoradionecrosis (ORN) in patients with head and neck cancer (HNC) and to estimate the conditional average treatment effect (CATE) to characterize heterogeneous treatment effects (HTE). In this retrospective cohort study of adults with HNC treated with curative radiotherapy from 2011 to 2018 with ongoing follow-up, the intervention was the extraction of at least 1 tooth before radiotherapy, and the outcome was ORN (ClinRad grade ≥1). Twenty sociodemographic and clinical covariates were recorded. A causal survival forest targeting restricted mean survival time (RMST) was trained with 100 repetitions of 5-fold cross-validation. Calibration used out-of-fold augmented inverse probability weighting (AIPW) scores as pseudo-outcomes, and treatment prioritization was assessed using the rank-weighted average treatment effect (RATE). A best linear projection regression identified covariates with direct associations to the predicted benefit. Among 2,466 patients, 810 underwent preradiotherapy extraction, and 183 developed ORN during the follow-up. The ATE was not significant at −0.26 mo (95% confidence interval [CI]: −0.68 to 0.17). However, the CATE distributions revealed substantial HTE, with patients in the lowest quartile (Q1) experiencing harm and those in the highest quartile (Q4) deriving benefit. Calibration against AIPW scores confirmed median RMST differences of −1.47 mo in Q1 and +0.94 mo in Q4. The RATE was positive at 1.10 (95% CI: 0.04 to 2.15). Best linear projection identified Eastern Cooperative Oncology Group performance status 1 to 4 versus 0 (β = 2.68; 95% CI: 0.19 to 5.17) and periodontal grade III–IV versus 0 (β = 4.33; 95% CI: 1.05 to 7.60) as independent modifiers of benefit. Radiation dose had a nonlinear association with treatment effect. If preradiotherapy tooth extraction was applied across all eligible patients, it would not alter the overall risk of ORN. However, individualized treatment effects varied, with some patients benefiting and others harmed, underscoring the importance of targeted interventions.
Keywords
Introduction
Osteoradionecrosis (ORN) of the jaws is a severe, late complication of radiotherapy for head and neck cancer (HNC), characterized by exposed, nonhealing bone that can lead to pain, infection, and pathological fracture (Chronopoulos et al 2018; Topkan et al 2023). Risk factors span tumor-related variables (e.g., site, stage), treatment parameters (e.g., radiation dose, concurrent chemotherapy), and patient-related factors (e.g., poor oral hygiene, dental insurance, tobacco use) (Nabil and Samman 2012; Chronopoulos et al 2018; Topkan et al 2023). One potentially modifiable factor is tooth extraction before radiotherapy, a strategy recommended to reduce the risk of ORN by removing teeth that are likely to require invasive interventions after radiotherapy, thereby minimizing posttreatment ORN risk. However, evidence from 3 recent systematic reviews remains conflicting, with 1 concluding that preradiotherapy extractions have not been shown to reduce ORN incidence (Balermpas et al 2022). Another review found that tooth extraction before radiotherapy may represent a risk factor for ORN development (Lajolo et al 2021), and a third reported substantial heterogeneity and inconsistent results, noting that while some studies linked extraction timing to ORN risk, others emphasized the role of additional contributing factors (Beaumont et al 2021).
In recent years, causal machine learning (ML) methods for uncovering heterogeneous treatment effects (HTE) have gained popularity in clinical research. HTE refers to the variation in treatment effect across individuals or subgroups, recognizing that a given intervention may benefit or harm some patients more than others (Wager and Athey 2018). By directly targeting the conditional average treatment effect (CATE), also referred to as the individualized treatment effect, which is defined as the expected difference in outcome under treatment versus control for patients sharing a common covariate profile, causal ML approaches operate within a counterfactual framework that addresses “what if” questions and overcome key limitations of traditional subgroup analyses (Wager and Athey 2018). These methods address the risk of spurious subgroup findings from multiple testing via data-driven discovery with cross-validation; flexibly discover complex, higher-order nonparametric interactions without prespecified hypotheses or interaction terms (Sverdrup et al 2025); and can mitigate false-negative findings seen in narrow, single-variable subgroup comparisons by modeling the interplay of multiple factors (Desai et al 2024).
Preradiotherapy tooth extraction is a clinician-selected intervention; accordingly, the target estimand should support prospective allocation, that is, who should be offered extraction, rather than merely describe historical choices. The average treatment effect (ATE) denotes the covariate-adjusted mean difference in outcomes that would arise in a target population if every eligible patient were assigned to treatment rather than control (Imbens 2004). The objective of this study is to develop and validate a causal ML approach using time-to-event data to (1) estimate the ATE of preradiotherapy tooth extraction on subsequent ORN risk, (2) calculate CATE and assess HTE in a cohort of patients with HNC, and (3) identify subgroups most likely to benefit from or be harmed by extraction through a fully data-driven approach.
Methods
Reporting Guideline
This study followed the guidelines of Strengthening the Reporting of Observational Studies in Epidemiology (STROBE; Von Elm et al 2007), Predictive Approaches to Treatment effect Heterogeneity (PATH; Kent et al 2020), and Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis with Artificial Intelligence (TRIPOD+AI; Collins, Moons, et al 2024).
Data and Study Population
A retrospective cohort study was conducted using time-to-event data derived from a prospectively maintained institutional database. Consecutive adults (≥18 y) with HNC who received definitive or postoperative intensity‑modulated radiotherapy (≥45 Gy) at Princess Margaret Cancer Center (2011 to 2018) and who underwent a pretreatment dental evaluation were included. Patients were excluded if they had prior head and neck irradiation, were edentulous, had primary tracheal cancer, or had early-stage laryngeal cancer (T1–2, N0, M0). Radiation and dental oncology records were merged to ascertain ORN outcomes. Follow‑up examinations were scheduled every 3 mo for the first 2 y, every 4 to 6 mo during years 3 through 5, and annually thereafter, with unscheduled dental assessments for extractions or suspected ORN. Mandibular dose–volume histogram parameters were extracted from the Pinnacle treatment planning system (Philips Healthcare). A flow diagram and detailed dataset description have been published previously (Moharrami et al 2025 (Watson 2024)).
Estimand and Framework
Under the potential outcome framework (Rosenbaum and Rubin 1983), each patient i has 2 latent survival times, Ti(1) under treatment and Ti(0) under control. The estimand of interest is CATE, defined as τ(x) = E[Ti(1) − Ti(0)|Xi = x], representing the expected difference in survival time for individuals with covariate profile Xi = x. A doubly robust estimating equation that combines inverse probability weights for treatment and censoring with an outcome regression adjustment was used, such that consistency of τ(x) estimates is maintained if either the weighting component or the outcome model is correctly specified (Cui et al 2023). Three assumptions were assumed: (1) consistency, requiring that the observed survival time equals the potential survival time corresponding to the treatment actually received; (2) exchangeability, assuming that treatment assignment is independent of the potential outcomes given measured covariates; and (3) overlap or positivity, requiring the probability of receiving treatment, that is, the propensity score, e(x) = P(Wi = 1|Xi = x), to satisfy 0 < e(x) < 1 (Rosenbaum and Rubin 1983).
Treatments, Outcomes, and Covariates
The intervention of interest was the extraction of at least 1 tooth following the initial dental evaluation and before the initiation of radiotherapy, performed in accordance with institutional clinical practice guidelines for dental management of HNC patients (Watson et al 2021). The outcome was the development of ORN, which was assessed using the ClinRad system (0 to 4) (Watson et al 2024), and subsequently dichotomized: patients with a ClinRad score of ≥1 were classified as ORN positive, while those with a score of 0, or those with no radiographic or clinical signs, were considered ORN negative. In addition, to perform a sensitivity analysis, a secondary ORN outcome of ClinRad ≥2 was defined. A comprehensive set of 20 covariates was documented before the commencement of radiotherapy, including age, sex, Eastern Cooperative Oncology Group (ECOG) performance status, alcohol consumption history, smoking pack-years index, periodontal condition, decay–missing–filled surfaces (DMFS-160) (Watson et al 2020), number of teeth before planned preradiotherapy extractions, individual income, type of dental insurance, primary tumor site, histological diagnosis, human papillomavirus status, overall TNM stage (American Joint Committee on Cancer/International Union Against Cancer, 7th edition), primary treatment modality, use of concurrent chemotherapy, total prescribed radiotherapy dose, radiotherapy duration in days, number of radiotherapy fractions, and the planned mandibular D10cc (Gy), a proxy for local dose defined as the minimum dose received by the most irradiated 10 cm³ of the mandible.
Preprocessing and Missing Data Imputation
Histological diagnoses were grouped as either squamous cell carcinoma (SCC) or non-SCC. The overall TNM stage was binarized into stage I to II versus III to IV. ECOG performance status was categorized as fully active (ECOG = 0) versus any limitation in activity or self-care (ECOG = 1 to 4). For covariates with less than 5% random missingness, primarily smoking and drinking history, imputation was performed using the missForest algorithm (Stekhoven and Bühlmann 2012).
Model Development and Validation
Details of model development and validation are provided in Appendix 1. In summary, we used the grf package (https://grf-labs.github.io/grf) to implement a causal survival forest (CSF) using the R programming language (Cui et al 2023), to estimate the CATE on restricted mean survival time (RMST) at a 60-mo horizon. Clinically, the RMST represents the average time a patient remains free of ORN within the specified follow-up window. A 60-mo horizon was selected to align with the standard 5-y clinical surveillance period used at our center. Furthermore, this window encompasses the primary risk period for ORN, as the density and distribution of events in our dataset showed. This approach aligns with epidemiological evidence indicating that most ORN cases manifest within the first few years following radiotherapy (Chronopoulos et al 2018).
Model assumptions were rigorously checked. The positivity assumption (overlap) was assessed by examining the distribution of propensity scores to ensure common support across treated and untreated groups. The exchangeability assumption (no unmeasured confounding) for observed covariates was evaluated by calculating standardized mean differences (SMDs) before and after weighting. An absolute SMD of less than 10% was considered indicative of adequate covariate balance (Austin 2009).
Model development followed a repeated stratified cross-validation framework consisting of 100 repetitions of 5-fold cross-validation, yielding 500 development–test splits. To prevent data leakage, preprocessing steps were performed independently within each fold. In each split, we trained a CSF model and computed out-of-fold augmented inverse probability weighting pseudo-outcomes (AIPW scores) and CATEs, which were estimated across all 500 cross-validation iterations, and the values were averaged for each patient. Patients with CATE above zero were considered likely to benefit from tooth extraction, and those with CATE below zero were considered harmed. Patients were then grouped into quartiles based on their CATE scores, and baseline covariates were compared across quartiles. The ranking ability was measured by the rank-weighted average treatment effect (RATE; corresponding to the area under the targeting operator characteristic [TOC] curve) (Yadlowsky et al 2025), which captures incremental RMST gain when treating top-ranked patients. Calibration was evaluated by stratifying patients in each individual split into quartiles of split-specific predicted CATE and comparing mean predicted versus AIPW (observed) scores, visualized using scatter and box plots (Desai et al 2024). Finally, we fit a best-linear-projection regression on the full-sample model to quantify each baseline covariate’s linear association with predicted CATE (Cui et al 2023).
Results
The cohort comprised 2,466 patients, with 183 ORN events observed during follow-up. The median time to develop ORN from the end of radiotherapy was 22.0 mo (range: 1.0 to 114.0 mo). The median age of all HNC patients was 60 y (range: 18 to 96 y), with males comprising 73.0% of the study population. Of the total cohort, 810 patients underwent at least 1 tooth extraction before radiotherapy. Preradiotherapy extraction was observed in 63 patients (34.4%) who developed ORN and in 747 patients (32.7%) who did not. Based on the subsample of patients who underwent at least 1 extraction, the time elapsed between the last extraction and the start of RT ranged from 0 to 169 d (median: 8 d), and the number of teeth extracted varied from 1 to 28 (median: 2). Also, 58.4% had unilateral extractions, while 41.6% had bilateral extractions. Considering the entire patient sample, the total number of teeth before extraction ranged from 1 to 32 (median: 25), and the count after extraction ranged from 0 to 32 (median: 24). Furthermore, among the patients who subsequently developed ORN, 77.5% had posterior teeth extracted, 12.5% had both anterior and posterior extractions, and 2.5% had only anterior extractions.
Estimated propensity scores ranged from 0.11 to 0.59 (mean = 0.32; median = 0.35). As shown in Figure 1, patients who did not undergo preradiotherapy extraction predominated at the lower end of the score distribution, while treated patients were concentrated toward the higher end, yet there remained substantial overlap across the midrange, demonstrating that all patients had a nonzero likelihood of receiving or not receiving treatment. The exchangeability assumption was supported by the effective balancing of covariates; as detailed in Appendix 2, the unadjusted cohort exhibited substantial imbalances. However, after weighting, the SMDs for all sociodemographic and clinical covariates were reduced to less than 5%, confirming that the weighting procedure successfully balanced the treatment and control groups.
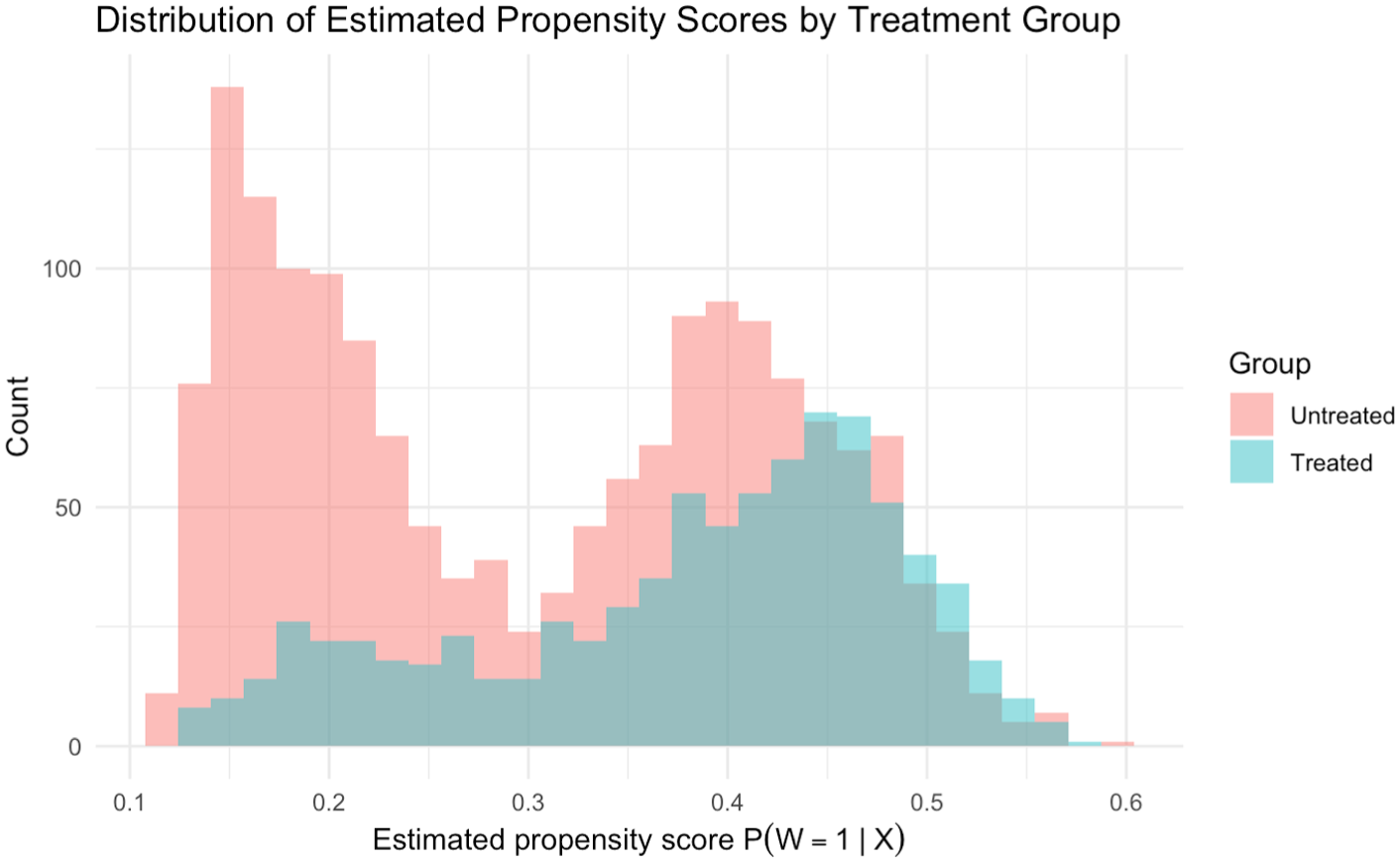
Overlap of estimated propensity score distributions by treatment group, preradiotherapy extraction versus no extraction.
The average ATE estimated over the whole sample was −0.26 (95% CI: −0.68 to 0.17). Figure 2A displays the distribution of predicted CATEs by quartile. Patients in Q2 and Q3 are tightly clustered around zero, indicating minimal treatment effect. In contrast, those in the lowest quartile (Q1), who appear to be harmed by the treatment, show predicted RMST differences of −10 mo or more; conversely, patients in the highest quartile (Q4), who benefit from the treatment, exhibit predicted differences of +10 mo or more.
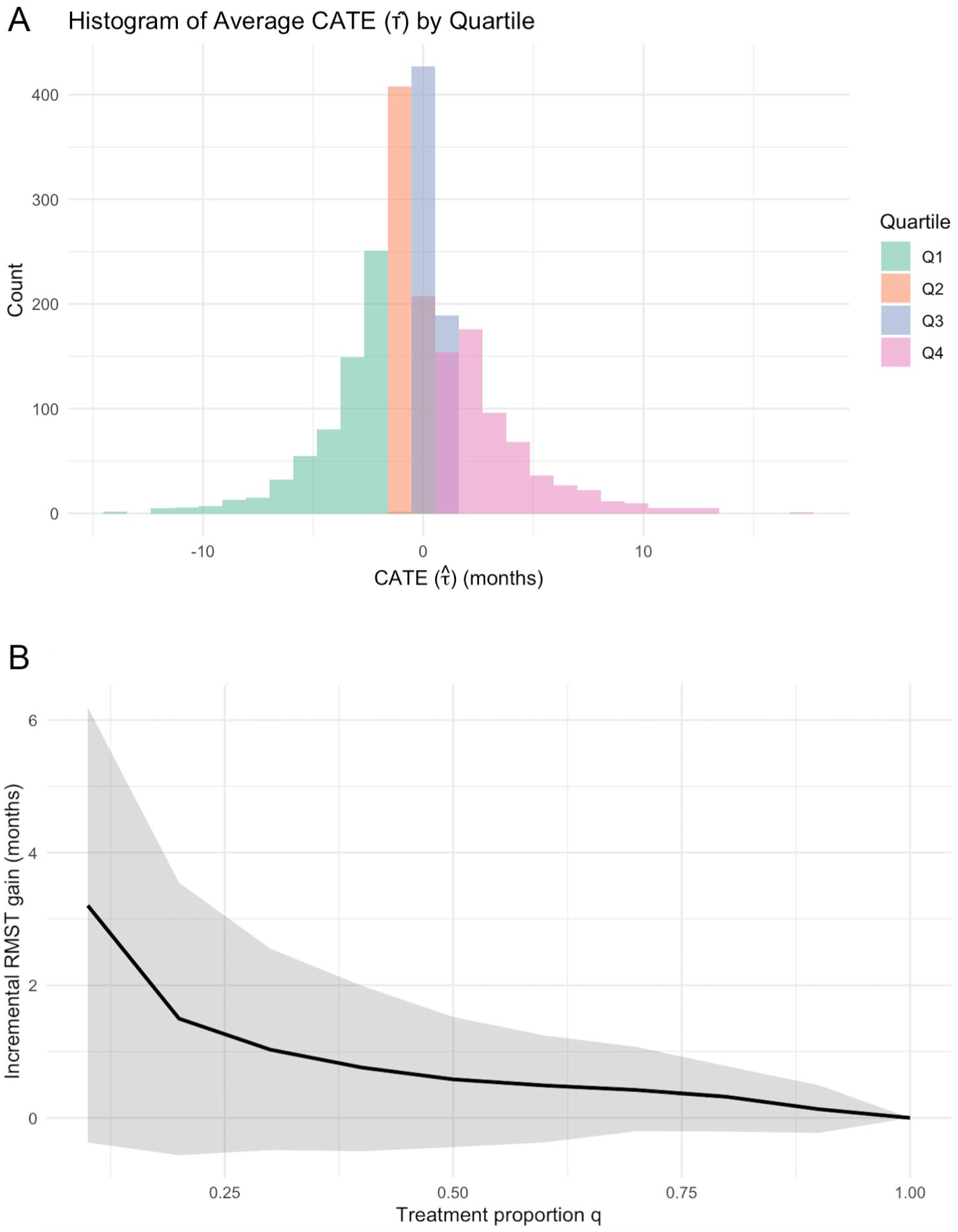
(
Table 1 summarizes the distribution of baseline covariates across predicted CATE quartiles, revealing distinct linear and nonlinear patterns. Linearly, patients in the higher benefit quartiles were more likely to exhibit impaired performance status (ECOG ≥1), have severe periodontitis (grade III to IV), have advanced-stage disease (TNM stage III to IV), have higher income, and receive concurrent chemotherapy; however, the best-linear-projection analysis of the full CSF model identified only ECOG performance status (β = 2.68; 95% CI: 0.19 to 5.17) and periodontal disease (grade III to IV: β = 4.33; 95% CI: 1.05 to 7.60; grade I to II: β = 3.30; 95% CI: 0.07 to 6.53) as having significant linear associations with predicted benefit (Table 2). Concurrently, the data exhibited strong nonlinear associations, most prominently for mandibular dose (D10cc), which followed a U-shaped distribution with higher doses in both the highest-harm (Q1) and highest-benefit (Q4) quartiles compared with a considerable drop in the middle quartiles (Q2 and Q3), a nonlinear pattern similarly observed for histology and primary tumor site.
Baseline Characteristics by Quartile of Predicted Conditional Average Treatment Effect (CATE) on Restricted Mean Survival Time (Preradiotherapy Tooth Extraction vs No Extraction).
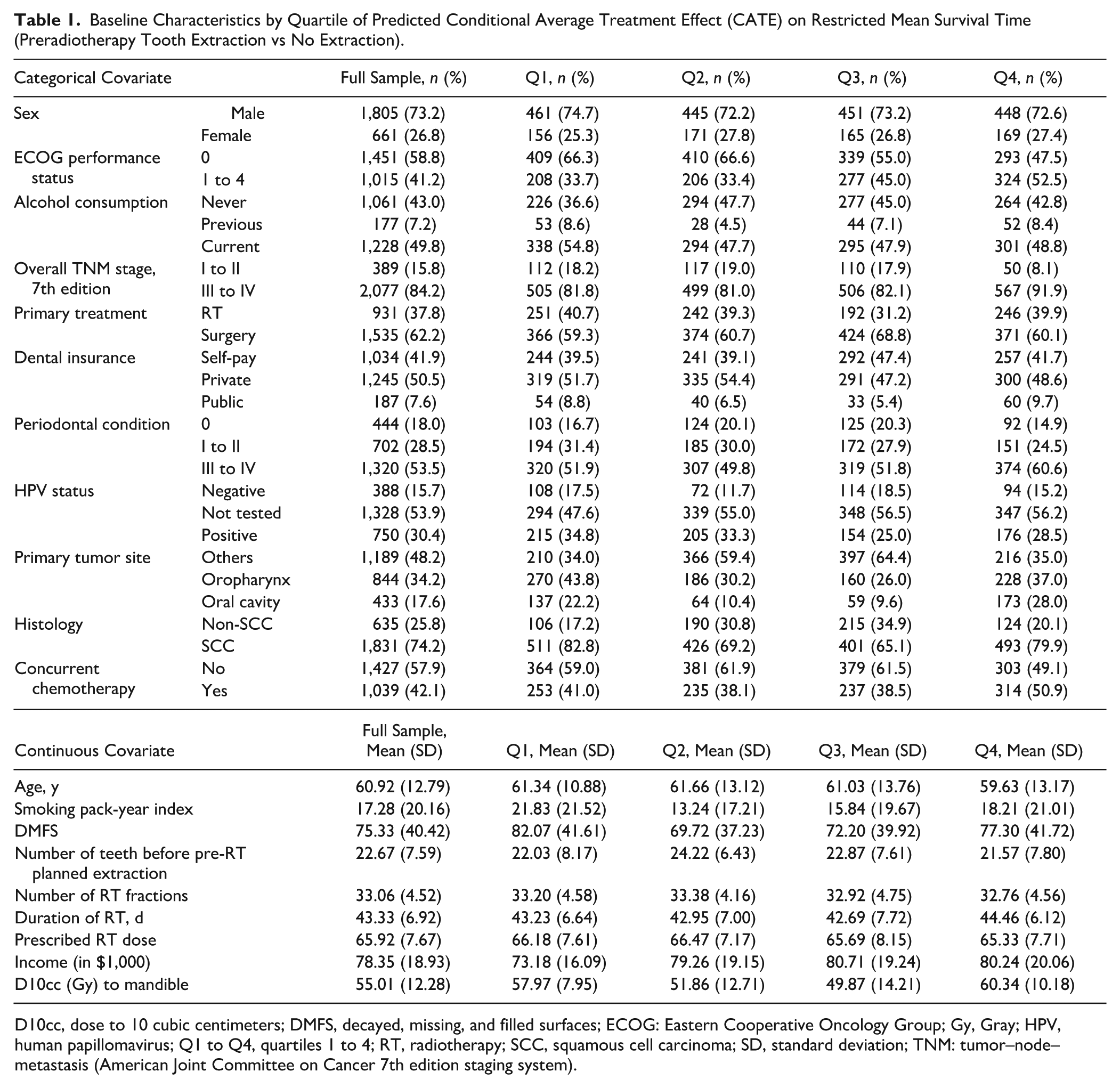
D10cc, dose to 10 cubic centimeters; DMFS, decayed, missing, and filled surfaces; ECOG: Eastern Cooperative Oncology Group; Gy, Gray; HPV, human papillomavirus; Q1 to Q4, quartiles 1 to 4; RT, radiotherapy; SCC, squamous cell carcinoma; SD, standard deviation; TNM: tumor–node–metastasis (American Joint Committee on Cancer 7th edition staging system).
Best Linear Projection of Baseline Covariates of Predicted Conditional Average Treatment Effect.
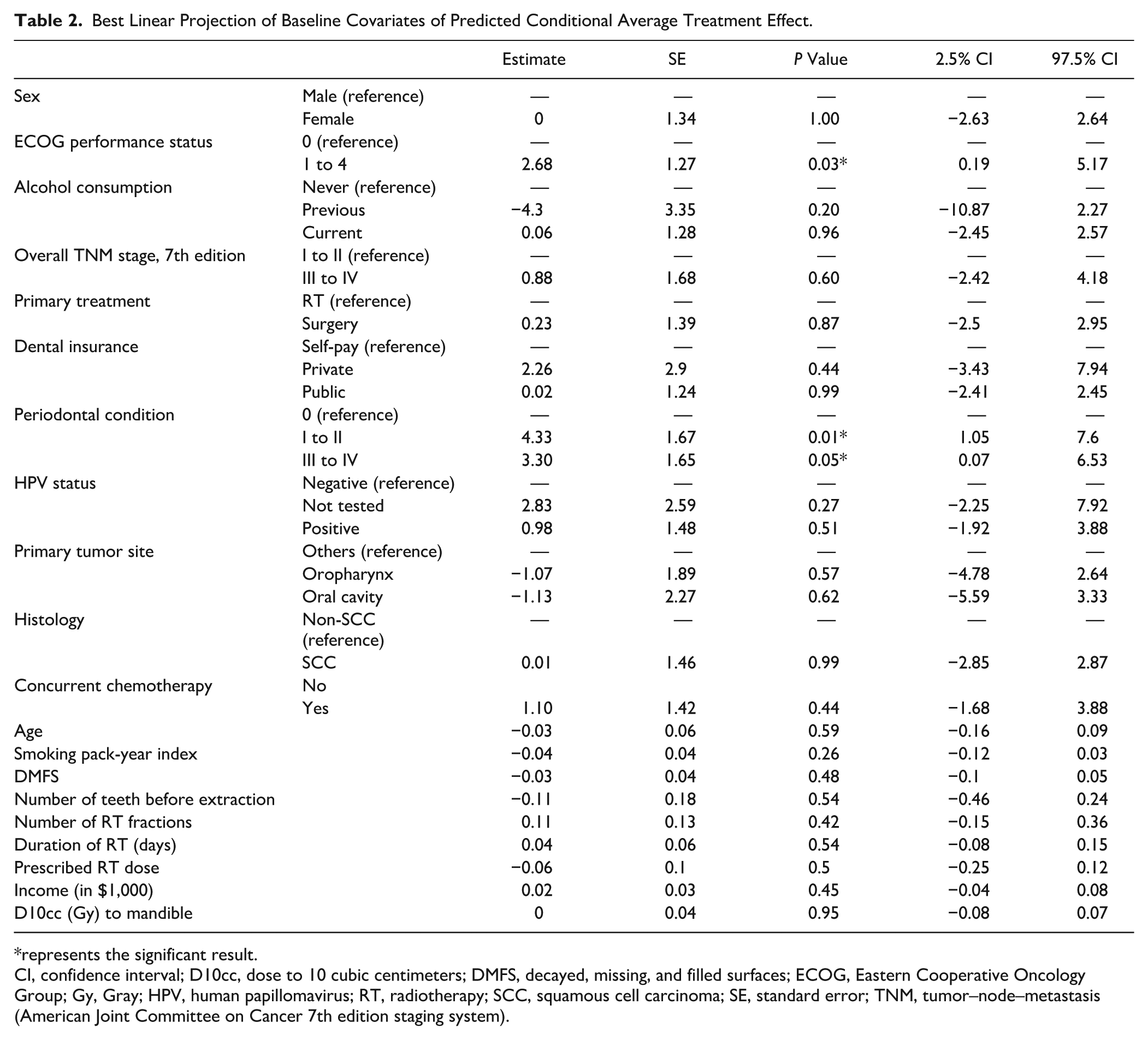
represents the significant result. CI, confidence interval; D10cc, dose to 10 cubic centimeters; DMFS, decayed, missing, and filled surfaces; ECOG, Eastern Cooperative Oncology Group; Gy, Gray; HPV, human papillomavirus; RT, radiotherapy; SCC, squamous cell carcinoma; SE, standard error; TNM, tumor–node–metastasis (American Joint Committee on Cancer 7th edition staging system).
The model’s ability to rank patients by predicted benefit was positive, as reflected in a RATE of 1.10 mo (95 % CI: 0.04 to 2.15), reflecting the average additional survival gained by treating patients in descending order of predicted CATE. Clinically, this indicates that a prioritized approach, in which preradiotherapy extractions are targeted toward patients identified by the model as high benefit, yields a larger reduction in ORN risk than treating patients indiscriminately. As illustrated in Figure 2B, the averaged TOC curve climbs sharply for small treatment proportions when treating the top 25% of patients sorted by predicted CATE and then decreases monotonically toward 0 at q = 1.0, when the entire cohort is treated.
In Figure 3A, each dot summarizes 1 split–quartile pair: for that split’s quartile, the mean CATE predicted by a CSF model is plotted against the mean AIPW pseudo-outcome (ie, the observed effect on RMST). With 4 quartiles per split across 500 splits, the scatter comprises 2,000 points. The companion boxplot (Fig 3B) shows, for each predicted-benefit quartile, the distribution across splits of the within-quartile mean AIPW values. Medians (IQR) are −1.47 mo (–2.05 to −0.81) in Q1, −0.24 mo (–0.90 to +0.31) in Q2, −0.30 mo (–0.82 to +0.13) in Q3, and +0.94 mo (+0.32 to +1.67) in Q4, indicating a clear increase in observed effect across predicted-benefit quartiles.
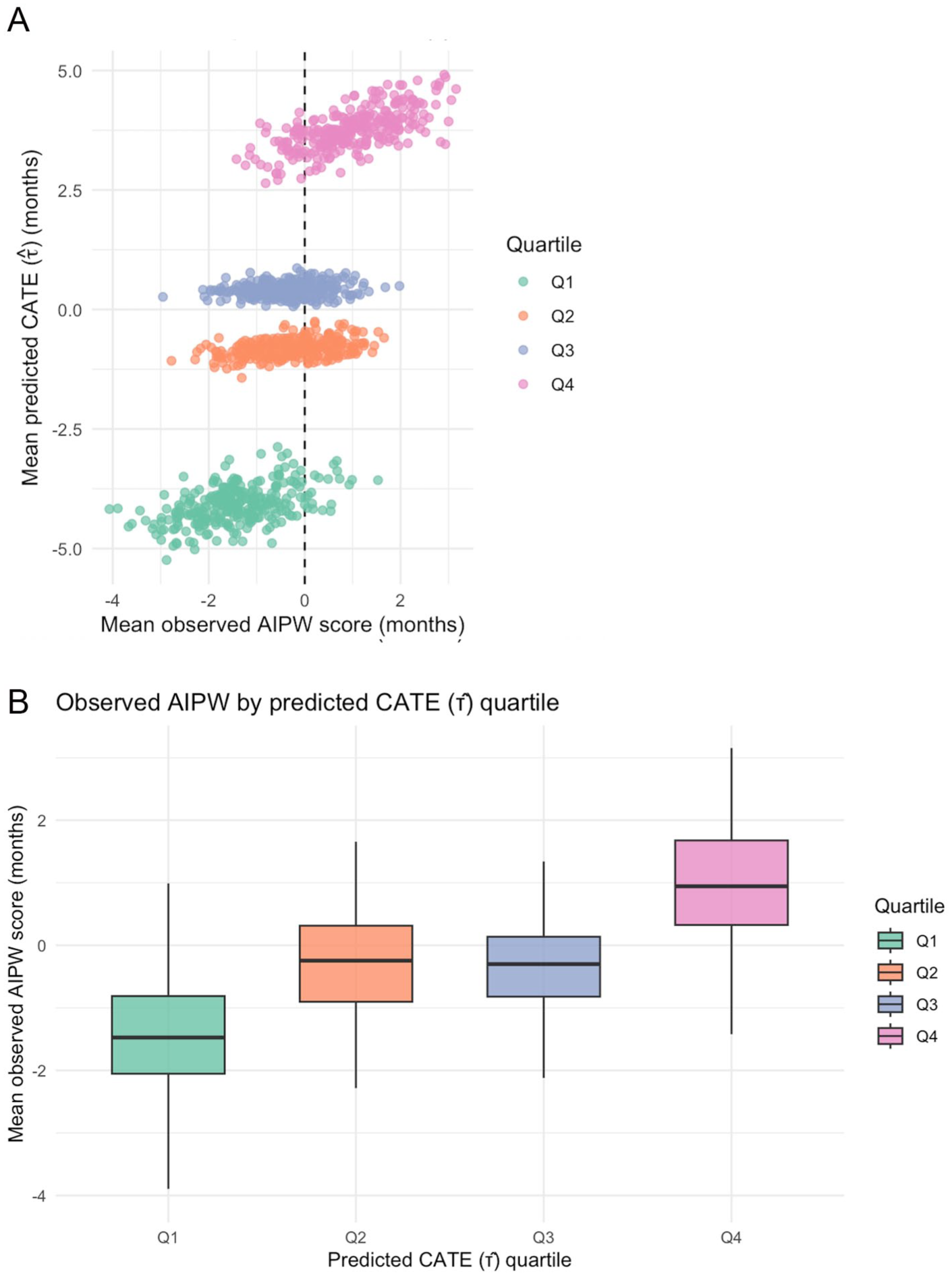
(
A sensitivity analysis was conducted using the stricter outcome definition of ORN (ClinRad grade ≥2). The ATE was not significant but skewed further toward the negative range at −0.41 mo (95% CI: −0.86 to 0.07). The CATE distributions revealed underlying heterogeneity, albeit less pronounced than in the primary analysis. Calibration against observed AIPW scores showed median RMST differences (IQR) of −1.62 mo (−2.15 to −1.05) in Q1 and +0.35 mo (−0.12 to +0.85) in Q4. The RATE was positive at 0.91 (95% CI: −0.06 to 1.90). In the best linear projection, ECOG performance status 1 to 4 (β = 2.25; 95% CI: 0.05 to 4.45) and periodontal disease grade III to IV (β = 3.64; 95% CI: 0.51 to 6.77) remained significant modifiers of benefit, whereas periodontal disease grade I to II (β = 2.70; 95% CI: −0.50 to 5.89) was not statistically significant.
Discussion
This study showed that if all eligible patients had undergone preradiotherapy tooth extraction, the average risk of ORN in the overall HNC population would not have meaningfully differed, as reflected by the ATE. However, substantial HTE was observed, suggesting that certain patients may either benefit from or be harmed by the intervention. The fully data-driven CSF model, integrating sociodemographic, clinical, and dosimetric covariates, successfully captured individualized treatment effects through nonlinear relationships and higher-order interactions. The best linear projection then indicated that, with all other covariates held constant, periodontal condition and ECOG performance status alone could influence the effect of tooth extraction on the risk of ORN. The trained model was well-calibrated and demonstrated reliable performance in ranking patients for receiving preradiotherapy extraction treatment.
The estimated treatment effect based on RMST must be interpreted in the context of the low overall incidence of ORN, which was 7.4%. Because these differences are calculated across a population in which the majority remains event free, the resulting values dilute the true magnitude of the effect for the susceptible minority. For instance, in the highest predicted benefit quartile (Q4), the observed median gain of +0.94 mo implies an approximate 8-mo delay in ORN onset for the ~11.7% of patients susceptible to the condition in that quartile. Similarly, the −1.47-mo median loss observed in the highest harm quartile (Q1) suggests an acceleration of nearly 2 y for the ~6.3% of patients in that quartile who are at risk of ORN. Figure 2A demonstrates this heterogeneity, in which the distributions of individual predictions for the highest benefit and harm groups exhibit tails extending to ±10 mo or more. Thus, these modest population-level shifts actually represent clinically substantial changes for high-risk profile patients.
The distribution of baseline covariates across CATE quartiles suggested linear trends for several factors. However, the best-linear-projection model identified only ECOG performance status and periodontal disease as having statistically significant, independent linear associations with the benefit of preradiotherapy extraction. These findings align with recent clinical guidelines, which identify periodontally compromised teeth as being at higher risk of failure following radiotherapy and therefore recommend prophylactic extraction (Watson et al 2021; Lalla et al 2023), particularly given that poor periodontal health further impairs bone healing (Schuurhuis et al 2018). Similarly, the benefit observed in patients with compromised ECOG performance status likely reflects limited self-care ability and reduced adherence to daily oral hygiene (Santibañez-Bedolla et al 2023); by performing extractions upfront, these patients may avoid later extractions postradiotherapy that could precipitate ORN.
Conversely, the analysis also revealed distinct nonlinear clinical profiles, particularly regarding radiation dose. The relationship between mandibular D10cc and treatment effect appeared U-shaped; both the highest-harm (Q1) and highest-benefit (Q4) quartiles exhibited mean doses (58 and 60 Gy, respectively) aligning with high-risk thresholds previously identified as critical for ORN development (Kubota et al 2021; Moharrami et al 2025). This suggests that while high radiation dose acts as a primary modifier increasing mandibular vulnerability, the divergence in outcomes likely stems from interactions with other covariates. In Q4, the combination of high dose with severe periodontal disease, compromised performance status, advanced tumor stage, and concurrent chemotherapy likely necessitates preradiotherapy extraction as vital source control to prevent catastrophic posttreatment extractions in an immunosuppressed environment. In contrast, for Q1, the interaction between high dose and the synergistic microvascular impairment from peak smoking exposure and alcohol use likely compromises immediate healing, converting the preventative extraction itself into a precipitating factor for ORN (Owosho et al 2017; Chronopoulos et al 2018). This underscores the value of the CSF model; while linear modifiers can serve as concise clinical flags for initial screening, they might fail to capture the full complexity of risk. Therefore, a tiered workflow should use linear clinical flags for first-line stratification, reserving full CSF predictions for high-dose patients in whom infection prevention must be balanced against healing capacity.
The results of this study should be interpreted considering several limitations. First, the analysis relied on mandibular D10cc rather than socket-specific dosimetry. Although local radiation dose pathophysiologically precipitates tissue failure, an extraction-site dose is counterfactually undefined for control patients who underwent no extractions and unavailable in standard clinical datasets. Consequently, D10cc was used as a robust, definable surrogate for the mandible’s overall susceptibility to ORN. In addition, data on other potential confounders, such as the use of antiresorptive medications (eg, bisphosphonates), were unavailable, which may limit the ability to distinguish between radiation-induced necrosis and medication-related osteonecrosis of the jaw (Peterson et al 2024). Furthermore, we excluded variables related to socket healing, such as extraction time and number, because they are logically undefined for the nonextraction control group. Incorporating these factors would introduce posttreatment bias by conditioning on mediators rather than baseline covariates. Accordingly, our estimates should be interpreted as the average effect of a composite extraction strategy.
Finally, while independent external evaluation across other settings may be possible in the future, once comparable time-to-event datasets became available, we used a “targeted validation” (Sperrin et al 2022), estimating model performance within our intended use population and institute. This was achieved using repeated resampling-based internal validation to fully exploit our representative ORN cohort and correct for in-sample optimism without the inefficiencies of data splitting (Collins, Dhiman, et al 2024). Moreover, since the workflow operates on routine tabular clinical and dosimetric data and leverages open-source implementations, it is computationally efficient and cost-effective to retrain; other institutions can readily implement the same pipeline to fit and validate site-specific models using locally available data.
Conclusion
This study showed that offering preradiotherapy tooth extraction to all eligible HNC patients would not meaningfully change the average risk of ORN. However, substantial HTE was revealed, with some individuals experiencing harm and others benefiting from preradiotherapy tooth extraction. The CSF model reliably generated individualized treatment-effect predictions by capturing complex interactions and nonlinearities, while linear analysis specifically identified impaired performance status and moderate-to-severe periodontitis as the independent subgroups most likely to benefit. These findings support a tiered decision-making strategy wherein these clinical flags serve as a first-line screening for prophylactic extraction, reserving full model predictions for high-dose patients to address complex risks by balancing infection control against healing capacity.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345261424748 – Supplemental material for Estimating the Individualized Effect of Tooth Extraction before Radiotherapy on Osteoradionecrosis Using Causal Machine Learning
Supplemental material, sj-docx-1-jdr-10.1177_00220345261424748 for Estimating the Individualized Effect of Tooth Extraction before Radiotherapy on Osteoradionecrosis Using Causal Machine Learning by M. Moharrami, E. Watson, S. Singhal, S.H. Huang, C. Yao, A. Hosni, C. Quinonez and M. Glogauer in Journal of Dental Research
Footnotes
Author Contributions
M. Moharrami, contributed to data conception and design, acquisition, analysis, and interpretation, drafted the manuscript; E. Watson, contributed to data study design, acquisition and interpretation, critically revised the manuscript; S. Singhal, C. Quinonez, contributed to data design, interpretation, critically revised the manuscript; S.H. Huang, C. Yao, contributed to data acquisition and interpretation, critically revised manuscript; A. Hosni, contributed to data acquisition and analysis, critically revised the manuscript; M. Glogauer, contributed to data conception and design, acquisition and interpretation, critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Canada Graduate Scholarships-Doctoral (CGS-D) from the Canadian Institute of Health Research (CIHR), reference No. 187502.
Data Availability Statement
The individual-level clinical data for this study are not publicly available due to patient privacy and confidentiality obligations. However, where applicable, the numerical data underlying the figures are available in the online supplementary material associated with this article.
A supplemental appendix to this article is available online.
