Abstract
Infertility is an increasing global health concern. Growing evidence suggests that systemic inflammatory conditions, including periodontal disease, may contribute to impaired reproductive outcomes. This narrative review highlights recent conceptual advances linking periodontal disease with male and female infertility, focusing on biological mechanisms (i.e., microbial translocation, chronic inflammation, immune dysregulation, oxidative stress, and epigenetic modifications). The review critically examines available studies focusing on scientific quality, design, and clinical relevance. In females, periodontal disease has been associated with idiopathic infertility and polycystic ovary syndrome. These conditions are characterized by immune dysregulation and low-grade systemic inflammation. In males, impaired semen parameters and idiopathic infertility have been linked to poor periodontal status. Despite growing interest, existing studies are largely associative and limited by methodological heterogeneity, insufficient control for confounders, and a lack of standardized outcome measures. This review proposes a framework for improved future research strategies addressing these shortcomings to clarify causality and therapeutic potential.
Introduction
Infertility, defined as the inability to conceive after 12 months or more of regular unprotected intercourse, affects 48.5 to 72.4 million couples worldwide (Mascarenhas et al 2012). Infertility may result from reproductive disorders or remain idiopathic. Beyond physiological impacts, infertility carries profound social and psychological consequences, including ostracism and divorce, and more subtle forms of stigma, such as isolation and mental health disorders.
Its etiology is complex, influenced by biological, lifestyle, and environmental factors. While some causes are preventable, many require medical interventions. Recently, the possible influence of systemic inflammation on reproductive outcomes has drawn attention, with oral health as the key research focus. Observational evidence suggests associations between oral microbiota composition, immune system interactions, and reproductive health outcomes (Ludovichetti et al 2021), although causal relationships remain incompletely understood. Periodontal disease, albeit an oral condition, demonstrates significant systemic effects. It amplifies extraoral inflammation, potentially modifying menstrual cycles and embryo implantation in women while affecting sperm quality and quantity in men.
This review critically explores recent findings (Table 1) and emerging hypotheses connecting periodontitis to infertility in both sexes (Fig. 1), focusing on shared pathophysiological pathways and future research directions. Risk of bias was evaluated for included studies using the ROBINS-I tool (Appendix Tables 1-6).
Summary of Discussed Studies Investigating the Association between Periodontal Disease and Infertility-Related Conditions.
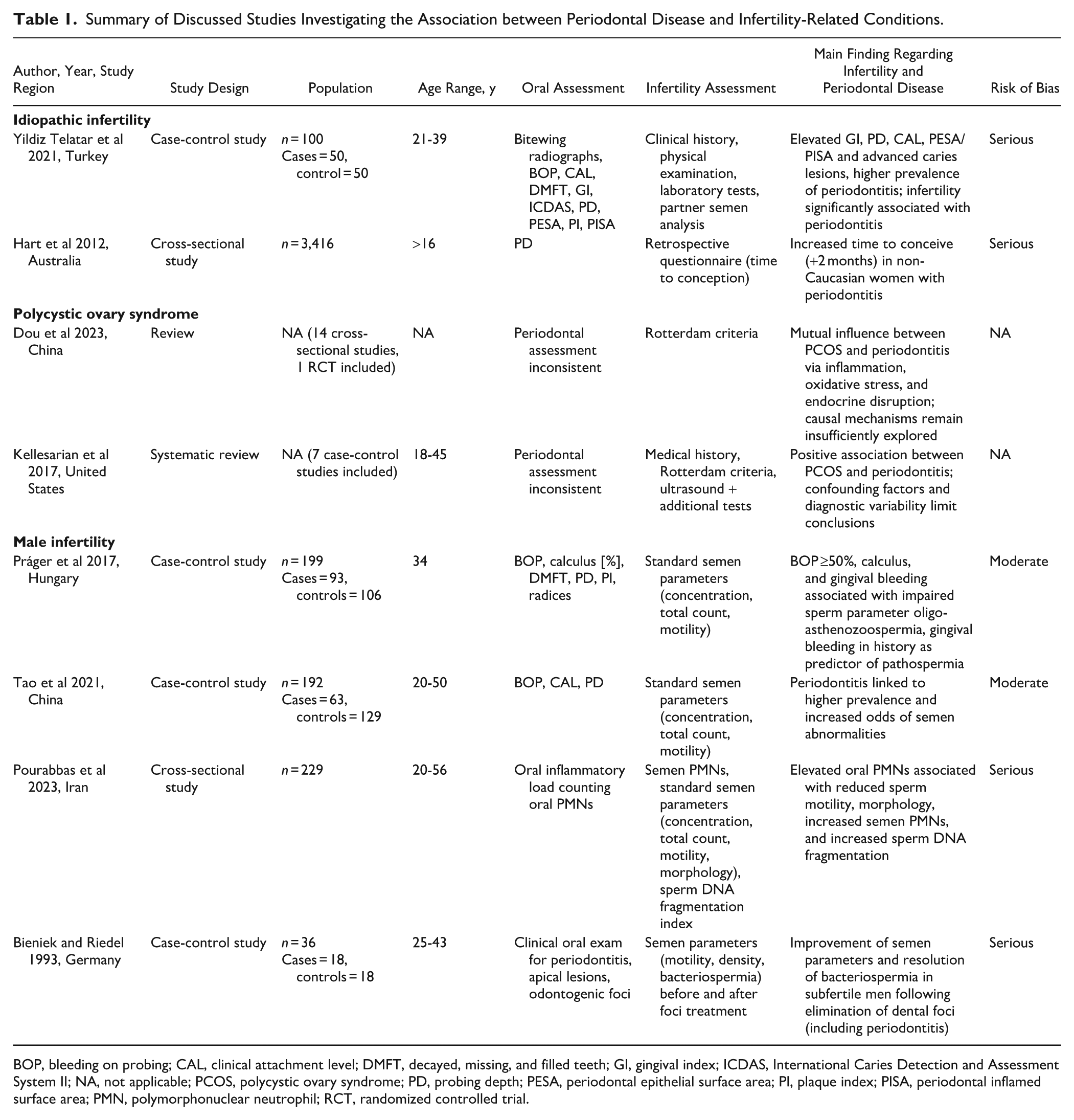
BOP, bleeding on probing; CAL, clinical attachment level; DMFT, decayed, missing, and filled teeth; GI, gingival index; ICDAS, International Caries Detection and Assessment System II; NA, not applicable; PCOS, polycystic ovary syndrome; PD, probing depth; PESA, periodontal epithelial surface area; PI, plaque index; PISA, periodontal inflamed surface area; PMN, polymorphonuclear neutrophil; RCT, randomized controlled trial.
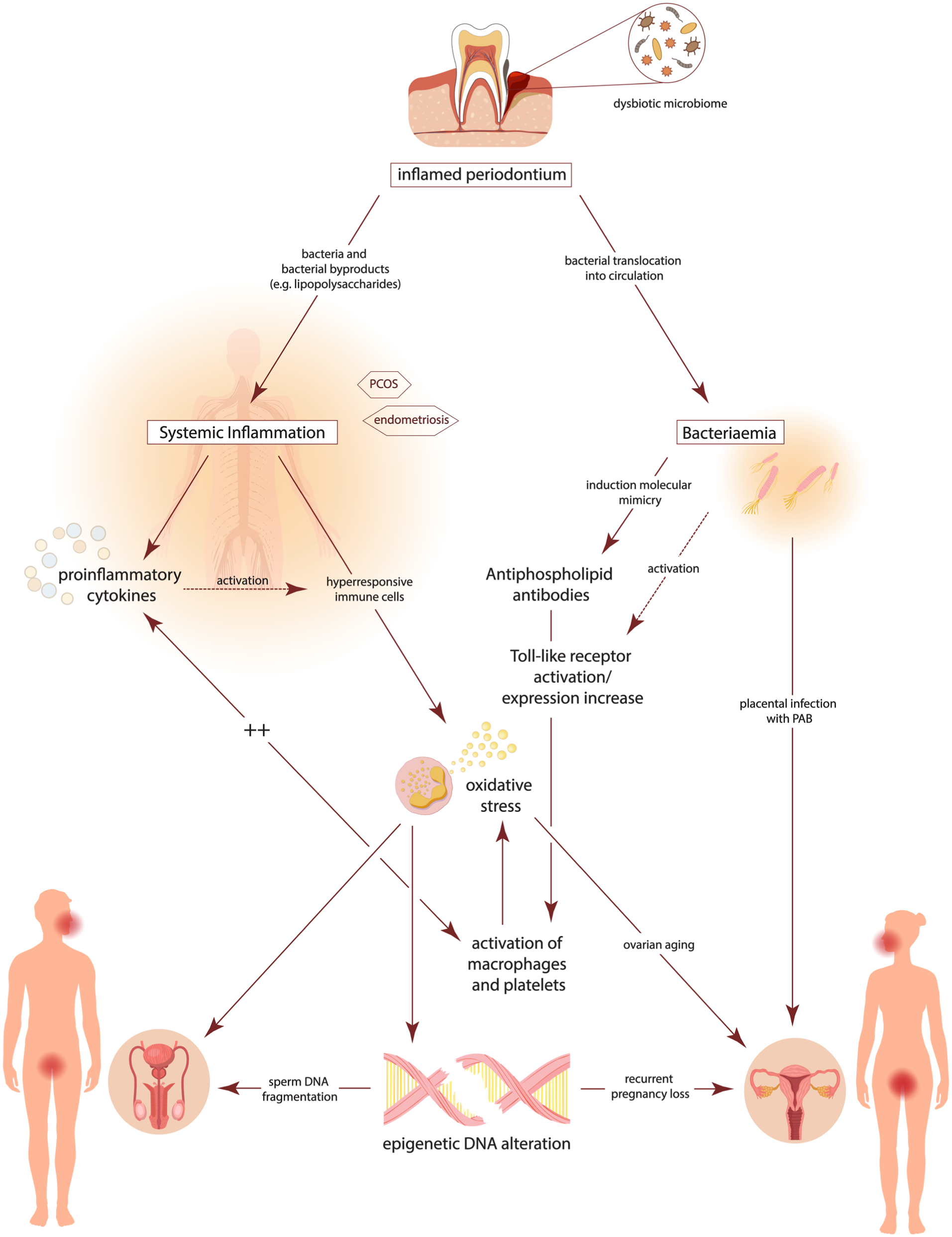
Proposed pathophysiological link between periodontal disease and reproductive disorders. The diagram outlines potential pathways connecting periodontal disease to reproductive dysfunction. Key mechanisms include systemic inflammation, bacterial translocation, oxidative stress, and immune activation, which may contribute to ovarian aging, sperm DNA fragmentation, polycystic ovary syndrome, endometriosis, and pregnancy loss. DNA, deoxyribonucleic acid; PAB, periodontitis-associated bacteria; PCOS, Polycystic ovary syndrome; ++, mutually reinforcing.
Recent Discoveries and Clinical Evidence
The periodontitis-pregnancy relationship has been extensively investigated, with observational studies demonstrating moderate associations with low birth weight and preterm birth, although interventional evidence remains inconclusive (Machado et al 2023). In contrast, periodontal effects on fertility and conception remain substantially underexplored.
Female Reproductive Disorders and Periodontal Health
Female reproductive disorders are commonly classified based on affected system, underlying causes, or clinical presentation. Main causes include idiopathic infertility, endometriosis and polycystic ovary syndrome. Notably, this review is not covering etiology and pathogenesis in detail but focuses on the association with periodontal disease.
Idiopathic Infertility
Idiopathic infertility in women accounts for approximately 30% of infertility cases (Romualdi et al 2023) and has been previously associated with poorer periodontal health (Yildiz Telatar et al 2021). An extended time to conceive (TTC) by 2 months among non-Caucasian women with periodontitis was observed in a randomized controlled multicenter trial (Hart et al 2012). However, limitations include retrospective TTC assessment via questionnaire and mid-pregnancy periodontal diagnosis, which may not reflect the pre-pregnancy periodontal status due to pregnancy-associated changes in the immune system, hormone levels, and oral microbiome composition (Saadaoui et al 2021). These limitations are particularly important when investigating the effects of periodontitis on TTC. Additionally, the study does not account for key confounding factors (Appendix Table 2).
Polycystic Ovary Syndrome
Polycystic ovary syndrome (PCOS) is a hormonal disorder characterized by irregular menstrual cycles, excess androgen levels, polycystic ovaries, and chronic low-grade systemic inflammation, with the latter also known to be caused by periodontitis (Dou et al 2023). Periodontitis is proposed to contribute to PCOS pathogenesis through the modulation of immunoinflammatory responses, specifically by altering proinflammatory and anti-inflammatory signaling pathways and promoting oxidative damage (Dou et al 2023). Although PCOS appears to be the most extensively analyzed condition of female infertility in relation to periodontitis, its multifactorial nature limits conclusions to positive associative findings (Kellesarian et al 2017) rather than establishing clear causal relationships. Small sample sizes, heterogeneous and inconsistently applied PCOS diagnostic criteria, and accompanying metabolic disorders such as hyperglycemia, insulin resistance, and undiagnosed higher central fat levels affecting systemic inflammation limit data validity.
Male Reproductive Disorders and Periodontal Health
Male infertility accounts for almost 50% of all infertility cases and is associated with insufficient sperm production (oligozoospermia), impaired motility (asthenozoospermia), or abnormal morphology (teratozoospermia) (Imamovic Kumalic and Pinter 2014).
Studies have reported a positive correlation between reduced semen quality and periodontal inflammation (Práger et al 2017; Tao et al 2021; Pourabbas et al 2023). This association is supported by proposed biological mechanisms linking oral neutrophil phenotypes to sperm quality (Pourabbas et al 2023), while improvements in semen characteristics following treatment of periodontitis, chronic apical osteitis, and advanced odontogenic cysts have been observed, suggesting a microbial or inflammatory link (Bieniek and Riedel 1993). Notably, existing studies in this field (Mortazavi et al 2025) suffer from small sample sizes, cross-sectional study designs, and insufficient statistical analyses, including inadequate confounder adjustment, which limits generalizability and leaves space for speculative interpretation.
Potential Mechanisms and Biological Plausibility
Chronic Inflammation and Immune Dysregulation
Several chronic inflammatory conditions are associated with fertility impairment, providing insight into systemic immune pathways potentially involved (Ashonibare et al 2024). Periodontitis is a chronic inflammatory disease driven by a dysbiotic shift in the subgingival microbial community and dysregulated host immune-inflammatory responses. Beyond systemic effects of bacterial dissemination and locally expressed inflammatory mediators, the immune response associated with periodontitis might also imprint immune function at non-oral sites.
Specifically, inflammatory bowel disease (IBD) has been associated with higher infertility risk, particularly when inflammation is poorly controlled (Zapounidou et al 2025). Periodontitis and IBD share common pathogenic features – dysbiotic microbiota, immune dysregulation, and chronic inflammation in genetically susceptible hosts – evidenced by elevated periodontitis prevalence in patients with IBD and spontaneous periodontal disease in Crohn’s disease animal models (Zhang et al 2021).
The mechanistic intersection of both diseases with infertility centers on dysbiotic microbiota-driven TH17/Treg imbalance across oral, intestinal, and reproductive tissues (Zhang et al 2021; Ashonibare et al 2024). Systemically circulating TH17 cells generated during oral inflammation can migrate to distant tissues expressing appropriate chemokine ligands. During periodontitis, oral pathobiont-reactive TH17 cells expand through lymphatics to the intestine, where orally derived bacteria (particularly Klebsiella and Enterobacter species), which ectopically colonize from the oral cavity (Sedgley and Samaranayake 1994), promote inflammasome-mediated IL-1β secretion and subsequent TH1/TH17 cell expansion (Atarashi et al 2017; Kitamoto et al 2020). These orally primed pathogenic TH17 cells demonstrate tissue-specific homing capacity, thereby amplifying local inflammation (Hajishengallis and Chavakis 2021). TH17 cells expressing chemokine receptors are abundant in female reproductive tissues, with elevated TH17 and IL-17A in endometriosis, suggesting roles in establishing reproductive inflammatory foci (Delbandi et al 2025).
This paradigm extends to reproductive dysfunction, where pathobiont-driven TH17/Treg imbalance mediates fertility impairment (Ashonibare et al 2024). The TH17/Treg dysregulation of periodontitis-induced intestinal inflammation (Jia et al 2024) constitutes the immunological signature of reproductive failure, where excessive TH17 activity compromises endometrial receptivity and spermatogenesis through proinflammatory cytokines that abrogate immune tolerance. Women with active IBD show significantly reduced anti-Müllerian hormone levels compared to those in remission (Sun et al 2022). The proposed mechanism involves proinflammatory factors released during intestinal inflammation that penetrate the bowel wall, triggering peritoneal inflammation extending to the ovaries, accelerating follicular depletion (Xiang et al 2023).
The chronic systemic inflammatory status imposed by both conditions generates oxidative stress through excessive proinflammatory cytokine-mediated reactive oxygen species production, resulting in pathological damage to reproductive tissues, including meiotic arrest in oocytes, granulosa cell apoptosis, corpus luteum dysfunction, and accelerated ovarian aging that potentially triggers chromosomal non-disjunction and impairs embryo developmental competence (Agarwal et al 2012; Shi et al 2023).
Oxidative modification of cytoskeletal proteins maintaining sperm shape and motility leads to loss of motility and abnormal morphology, while protein oxidation affects enzymes regulating sperm metabolism and energy production (Wang et al 2025). At the neuroendocrine level, proinflammatory cytokines from the IL-1 family inhibit the hypothalamic-pituitary-gonadal axis by affecting gonadotropin-releasing hormone secretion from the hypothalamus, which subsequently regulates luteinizing hormone and follicle-stimulating hormone release. This hormonal disturbance leads to ovulation failure in females and compromises testicular steroidogenesis and spermatogenesis in males (Ignatiuk et al 2025). These findings demonstrate that periodontitis-associated bacteria (PAB) recapitulate IBD pathomechanisms with established fertility-impairing roles, providing biological plausibility for oral-to-reproductive effects.
Microbial Translocation and Microbiome Dysbiosis
Alterations in microbial composition have been linked to female infertility-related conditions such as PCOS. Affected women demonstrate significant gut dysbiosis characterized by reduced α-diversity correlating with hyperandrogenemia and elevated testosterone levels (Zou et al 2023), altered bacterial composition (including increased Escherichia-Shigella, Bacteroides vulgatus, and Prevotella copri, alongside decreased Faecalibacterium, Alistipes, and Lachnospiraceae) (Xia et al 2025), and metabolic dysfunction with depleted short-chain fatty acids, dysregulated bile acid metabolism, and elevated branched-chain amino acids (Qi et al 2019; Xia et al 2025). These alterations promote PCOS pathogenesis through lipopolysaccharide-mediated chronic inflammation via Toll-like receptor 4 activation, compromised intestinal barrier integrity, impaired insulin signaling, and altered steroid hormone metabolism (Gautam et al 2024). Regarding pregnancy, placental microbiome existence in healthy pregnancies remains highly controversial. While early molecular studies suggested placental colonization with oral bacteria (Pelzer et al 2017), the largest rigorously controlled study to date failed to detect a placental microbiome in healthy term deliveries, concluding that detected bacterial signals primarily represent reagent contamination or acquisition during labor (de Goffau et al 2019). This conclusion is supported by expert consensus emphasizing that up to 36% of bacterial genera reported in earlier placental microbiome studies correspond to known laboratory contaminants (Kennedy et al 2023). Nevertheless, case reports and animal models provide evidence that PAB can translocate to the placenta during pathological states.
The relationship between oral dysbiosis and reproductive tract colonization is particularly evident in endometriosis, demonstrating elevated levels of oral-origin bacteria (Dialister and Streptococcus) in the uterine endometrium of patients with endometriosis and repeated implantation failure (Marcickiewicz et al 2025). PAB, such as Fusobacterium nucleatum and Porphyromonas gingivalis, may translocate from the oral cavity to the pelvic peritoneum and induce local inflammation via activating peritoneal macrophages and IL-1 synthesis.
While semen was long considered sterile, next-generation sequencing revealed that semen harbors a rich microbiome with >104 bacteria per milliliter (Farahani et al 2021). The seminal microbiome originates from seminal vesicles, prostate, testes, and epididymis, each possessing a unique microbiome altered by hygiene, circumcision, sexual practices, and diet (Lundy et al 2021). In healthy men, the urogenital microbiome is dominated by Lactobacillus, Prevotella, Streptococcus, Corynebacterium, Staphylococcus, and Enterococcus, although considerable interindividual variation exists in species composition throughout the urogenital tract. Notably, Prevotella, a taxon frequently recognized as PAB, accounts for 49% of anaerobic bacterial isolates in semen (Kwaśniewski and Karpiński 2025). Lactobacillus appears to protect sperm parameters, whereas Ureaplasma urealyticum, Enterococcus faecalis, Mycoplasma hominis, and Prevotella negatively impact semen parameters (Farahani et al 2021).
Infertile men demonstrated increased seminal α-diversity and distinct β-diversity compared to fertile controls, characterized by increased Aerococcus and decreased Collinsella (Lundy et al 2021). Prevotella abundance was inversely associated with sperm concentration, while Pseudomonas showed a direct association with sperm motility. Seminal microbiome dysbiosis has been linked to upregulation of the S-adenosyl-L-methionine cycle, implicating microbial contributions to DNA methylation, oxidative stress, and polyamine synthesis, which are key processes in spermatogenesis (Lundy et al 2021). Disruption through the microbiome-mediated S-adenosyl-L-methionine cycle may result in aberrant epigenetic programming, compromising sperm quality and fertilization potential. Moreover, PAB can influence gut dysbiosis and intestinal barrier function in IBD through multiple mechanisms: disrupting tight junction proteins (downregulating ZO-1, occludin, and claudin), degrading the mucus layer, inducing ferroptosis in colonic epithelial cells, and triggering proinflammatory cytokine release (IL-1β, IL-17, TNF-α) (Kitamoto et al 2020; Hajishengallis and Chavakis 2021). This intestinal dysbiosis operates through the gut-testis axis, whereby microbial metabolites (short-chain fatty acids, lipopolysaccharides) and inflammatory mediators translocate systemically to compromise blood-testis barrier integrity, suppress the hypothalamic-pituitary-gonadal axis, induce oxidative stress, and alter testicular metabolite profiles, thereby impairing spermatogenesis (Wang and Xie 2022). Specific gut bacteria (i.e., Clostridium scindens and Ruminococcus gnavus) residing in the distal colon and cecum possess the enzymatic capacity to convert glucocorticoids secreted via bile into the intestinal lumen into androgens through side-chain cleavage, directly contributing to circulating testosterone pools. Additionally, these colonic microbiota deglucuronidate conjugated testosterone excreted by the liver into the bile, facilitating reabsorption and maintaining systemic androgen levels (Colldén et al 2019; Ashonibare et al 2024).
Furthermore, disruption of gut microbiota by oral bacteria may affect the integrity of the blood-testis barrier, a specialized structure formed by tight junctions, adherens junctions, gap junctions, and ectoplasmic specializations between adjacent Sertoli cells that creates an immunologically privileged adluminal compartment separating developing meiotic and postmeiotic germ cells from the immune system. Gut dysbiosis-derived lipopolysaccharides translocate systemically through compromised intestinal barriers and trigger downregulation of tight junction proteins (occludin, claudin-11, ZO-1), compromising blood-testis barrier integrity and exposing developing spermatocytes to immune attack and inflammatory damage, ultimately impairing spermatogenesis (Hajishengallis and Chavakis 2021; Guo et al 2024). Germ-free mice exhibit delayed blood-testis barrier development, with downregulation of cell adhesion molecules, resulting in impaired spermatogenesis and reduced fertility (Al-Asmakh et al 2014; Ashonibare et al 2024). PAB (Porphyromonas gingivalis and Prevotella intermedia) and gut microbiota dysbiosis additionally trigger lipopolysaccharide-mediated low-grade inflammation, inducing insulin resistance through inhibition of insulin signaling pathways (Hajishengallis and Chavakis 2021). Resultant hyperinsulinemia suppresses hepatic sex hormone-binding globulin synthesis; increases circulating free testosterone and endogenous testosterone production, ultimately impairing spermatogenesis; and activates negative feedback that suppresses luteinizing hormone and follicle-stimulating hormone in women (Zou et al 2023; Ashonibare et al 2024; Xia et al 2025).
Challenges in Current Evidence and Future Research Directions
A major limitation of available studies examining the relationship between periodontal disease and infertility is methodological heterogeneity, as well as general shortcomings in study design and data analysis. Many studies suffer from small, selective sample sizes, lack of age stratification, nonstandardized periodontitis and infertility diagnoses, and inadequate control of lifestyle and metabolic confounders (Appendix Tables 1-6). Most associations are descriptive, lacking mechanistic validation or longitudinal follow-up.
Future research should particularly consider proper age stratification, as it is a well-established factor influencing female fertility and a significant risk factor for the onset and progression of periodontitis. Inclusion of older individuals, who naturally experience decreased fertility and higher periodontitis susceptibility, introduces a potential confounding variable that would compromise the validity of findings.
As many studies focus on rather unspecific indicators of periodontitis, future studies should use standard diagnostic criteria (i.e., probing pocket depths and clinical attachment levels) to ensure adequate periodontitis diagnosis and mitigate potential misclassification biases. Given growing evidence linking periodontal disease with impaired reproductive outcomes, the dental research community is positioned to conduct longitudinal studies evaluating the impact of periodontl interventions within fertility care frameworks. Investigating whether periodontal treatment prior to conception improves clinical outcomes, such as TTC, embryo implantation success, or live birth rates, would provide key evidence to support integrated care models. These efforts should be complemented by interprofessional collaboration between dental and reproductive health providers. Together, these strategies represent a critical step toward ultimately enhancing patient care and developing evidence-based, interdisciplinary guidelines serving both dental and fertility patients.
Conclusions
Periodontitis might represent a previously underestimated contributor to infertility and fertility treatment outcomes. While current evidence remains largely associative, derived from studies that insufficiently address confounding factors, plausible biological mechanisms provide a compelling rationale for further investigation. High-quality, methodically grounded studies are needed to define the therapeutic potential and clinical implications of addressing periodontitis in the reproductive health context. In the absence of robust data, it is crucial to avoid creating premature expectations regarding the therapeutic potential of periodontal treatment for infertility management. Further research is strongly encouraged to delineate mechanisms through which periodontitis may impair fertility and to identify the most effective interventions for mitigating these effects.
Author Contributions
C. Wetzel, N. Werner, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; C.V. Bumm, J. Becker, F. Schwendicke, M. Folwaczny, contributed to analysis and interpretation, critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345261416502 – Supplemental material for Infertility and Periodontitis: Are We Connecting the Right Dots?
Supplemental material, sj-docx-1-jdr-10.1177_00220345261416502 for Infertility and Periodontitis: Are We Connecting the Right Dots? by C. Wetzel, C.V. Bumm, J. Becker, F. Schwendicke, M. Folwaczny and N. Werner in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability
No datasets were generated or analyzed for this review. Data availability is therefore not applicable.
A supplemental appendix to this article is available online.
