Abstract
Sexual dimorphism is found in gene expression and polarization of macrophages in mammals but remains unclear in the ontogeny of tissue-resident macrophages. Remarkable sex differences are present in salivary glands and risks for a related autoimmune disease, Sjögren’sdisease. Macrophages are the most abundant immune cells in healthy mouse salivary glands and essential for the maintenance of immune quiescence and tissue repair after radiation or inflammatory damages. Therefore, we compared the origins of macrophages in salivary glands between male and female mice using conditional Cx3cr1 and Ccr2 lineage-tracing approaches. We found that among salivary gland macrophages in adult mice, most are locally maintained and derived from yolk sac progenitors or perinatal monocytes in males, but much more are short-lived and continuously replenished by monocytes in females. In wild type C57BL/6 mice, female adult submandibular glands (SMGs) consistently contain more leukocytes, including classical monocytes expressing Ccr2, Ly6c, or Csf2rb and macrophages carrying these monocyte markers, as compared with male SMGs. Single-cell RNA sequencing and flow cytometry indicated that female SMG macrophages are more polarized and express several proinflammatory genes at higher levels. Meanwhile, female SMGs contain more innate lymphoid cells and T/NKT cells expressing Csf2 and other proinflammatory cytokines. The potential contributions of these sexual differences warrant further studies as they relate to Sjögren’s disease, the most female-dominant autoimmune disease, characterized by chronic inflammation in salivary and lacrimal glands. Also, these differences need be considered in developing macrophage-targeting therapies of dry mouth caused by autoimmunity or radiation.
Keywords
Introduction
In mammals, infectious diseases are more frequent and severe in males, whereas autoimmune diseases are more prevalent in females, indicating the sexual dimorphism in the immune system (Feng et al 2024). This dimorphism is mostly evident in macrophages in mice at the transcriptome level (Gal-Oz et al 2019). In mouse peritoneal and pleural cavities, female macrophages exhibit more M1-like activities than males (Scotland et al 2011). Conversely, macrophages in the heart or lungs of male mice are more prone to M1 polarization than females, which leads to the sex-related susceptibility to viral myocarditis and asthma (Li et al 2009; Melgert et al 2010). These paradoxical reports suggest that sex differences in macrophage polarization might be tissue specific. However, it remains unclear whether the ontogeny of macrophages in the same organ is comparable or different between the sexes.
In humans and mice, substantial sex differences have been noticed in biochemistry and the flow of saliva in salivary glands (SGs; Srivastava et al 2008; Mukaibo et al 2019). Moreover, Sjögren’s disease, a systemic autoimmune disease characterized by SG manifestations, occurs predominately in females over males in human (9 to 16:1) and mouse models (Brandt et al 2015; Gao et al 2020). Macrophages are the most abundant immune cells in steady-state mouse SGs (Lu et al 2022) and are essential for SG function through paracrine interactions with epithelial progenitors and endothelial cells (Zhao et al 2020). The study on the origins of adult SG resident macrophages is limited and not conclusive (McKendrick et al 2023), and it remains unclear whether such origins are comparable or different between the sexes.
Among the 3 pairs of major SGs, submandibular glands (SMGs) are the largest in mice. Therefore, we compared the origin and dynamics of SMG macrophages in male and female mice side-by-side with genetic lineage tracing, flow cytometry, and single-cell RNA sequencing (scRNA-seq). Our data indicate that among SG macrophages in adult mice, most are locally maintained and derived from perinatal progenitors in males, but much more are short-lived and continuously replenished by adult monocytes in females. Consistently, adult female mouse SMGs contain more classical monocytes, macrophages expressing monocyte markers, and polarized macrophages.
Materials and Methods
For all animal studies, we complied with the ARRIVE 2.0 guidelines, as outlined in the supplementary information. All details of materials and methods are also in the supplementary information.
Results
SMG Macrophages Derived from Yolk Sac Progenitors Are Long-lived in Both Sexes but Much More Abundant in Adult Males
Yolk sac macrophage progenitors are Cx3cr1+ and traffic to the embryo from embryonic day 8.5 (E8.5); therefore, we used heterozygous Cx3cr1-CreER;Ai9(RCL-tdT) mice induced with 4-hydroxy-tamoxifen (4-OHT) at E8.5 (Fig. 1A) to trace their progeny in SMGs as reported previously (Ensan et al 2016). This approach labeled most microglial cells derived from yolk sac progenitors but negligible monocyte-enriched splenocytes with tdTomato (tdT) in adult mice of both sexes as expected (Fig. S2A–B). In SMG sections from E18.5 mice, tdT+F4/80+ macrophages were detected by immunofluorescent staining in both sexes (Fig. 1B), indicating that yolk sac–derived macrophage progenitors migrate into SMGs. In flow cytometry, mouse SMG macrophages have been identified as F4/80+, F4/80+Ly-6G−Siglec-F−, or CD45+CD64+ cells and are mostly MERTK− (Zhao et al 2020; Lu et al 2022; McKendrick et al 2023). In the SMGs of postnatal day 30 (P30), P90, and P180 C57BL/6 mice, the percentages of macrophages identified by these 3 methods were comparable between the sexes, and almost all F4/80+ cells were Ly-6G−Siglec-F− (Fig. S1). Therefore, we mainly used F4/80 alone as the marker of SMG macrophages in the flow cytometry assays of our lineage-tracing mice expressing tdT. In SMG cells, the percentages of F4/80+ macrophages were comparable between the sexes at E18.5, P30, and P60 but lower in females at P90 and P150 (Figs. 1C–D and S2C). At E18.5, tdT+% in F4/80+ macrophages and tdT+Cx3cr1+% in CD45+ cells were comparable between males and females (Figs. 1E and S2D), indicating the similar initial labeling efficiency of this approach in SMGs between the sexes. At P60, the percentages of tdT+ cells in F4/80+ macrophages in male SMGs increased to ~35% and were maintained at P90 and P150. This index did not significantly change in female SMGs through P30 to P150 at ~15% and was much lower than males from P60 to P150 (Fig. 1E), as validated by P90 data based on other macrophage markers (Fig. S2E–F). These data indicated that macrophages derived from yolk sac progenitors are long-lived in SMGs of both sexes but much more abundant in adult males.
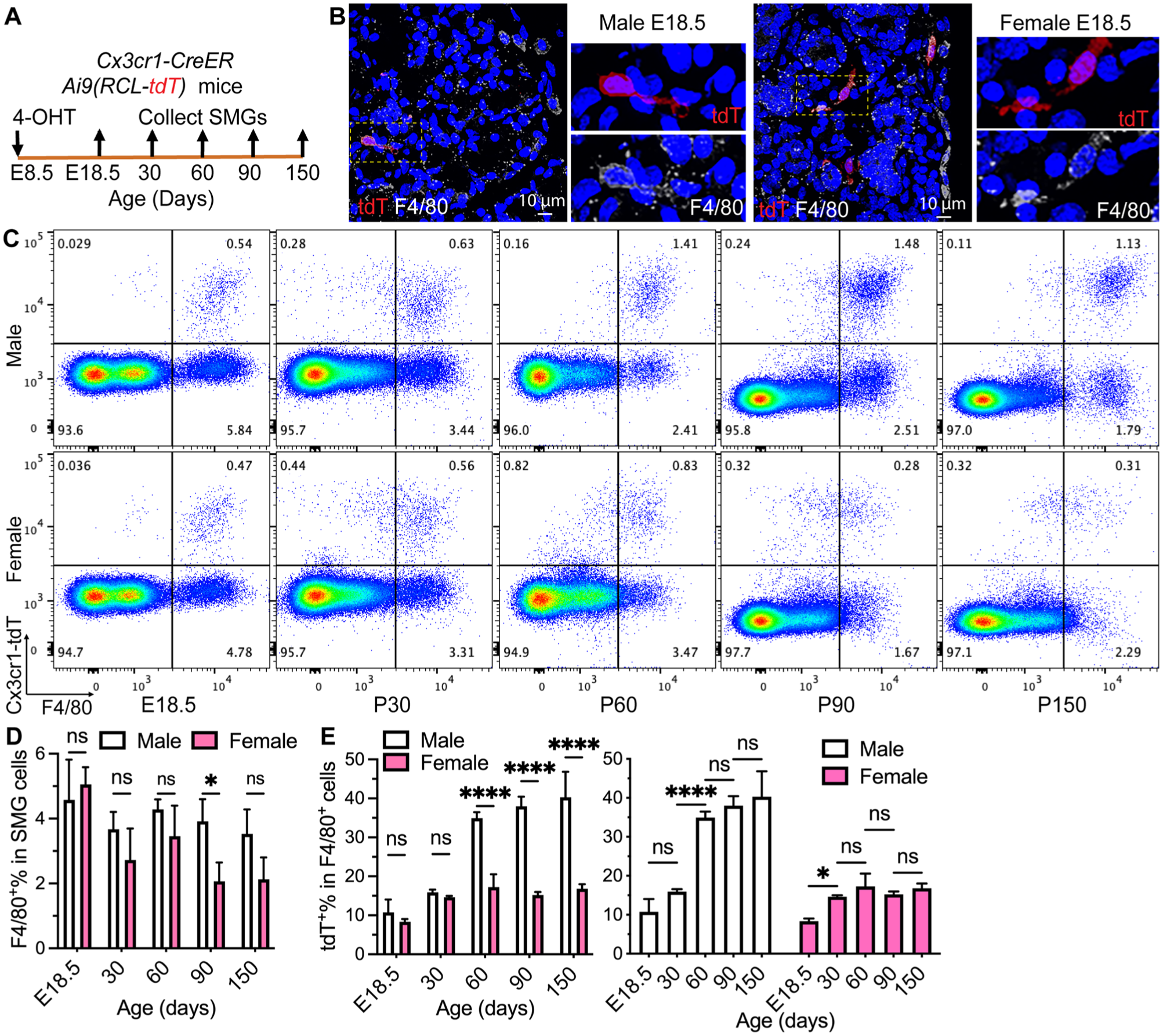
Lineage tracing of yolk sac progenitors in mouse SMG macrophages. (
Most SMG Macrophages Derived from Fetal Monocytes Are Short-lived in Both Sexes
Circulating fetal monocytes (FMs) are mostly Ccr2+ and give rise to adult tissue-resident macrophages in multiple organs (Hoeffel et al 2015). Therefore, we first used Ccr2-CreER;Ai9(RCL-tdT) mice induced with 4-OHT at E18.5 to trace the progeny of Ccr2+ FMs in adult SMGs. At P30, ~60% of F4/80+ macrophages in the SMGs of both sexes were labeled with Ccr2-CreER–mediated expression of tdT (Ccr2-tdT; Figs. 2A–D and S3A), indicating that Ccr2+ FMs are a dominant source of macrophages in juvenile SMGs. However, Ccr2-tdT+% in F4/80+ macrophages and all SMG cells decreased gradually in both sexes since P60 and to negligible levels at P90 and P150 (Figs. 2A–D and S3A–C), indicating that most SMG macrophages derived from Ccr2+ FMs are short-lived in both sexes.
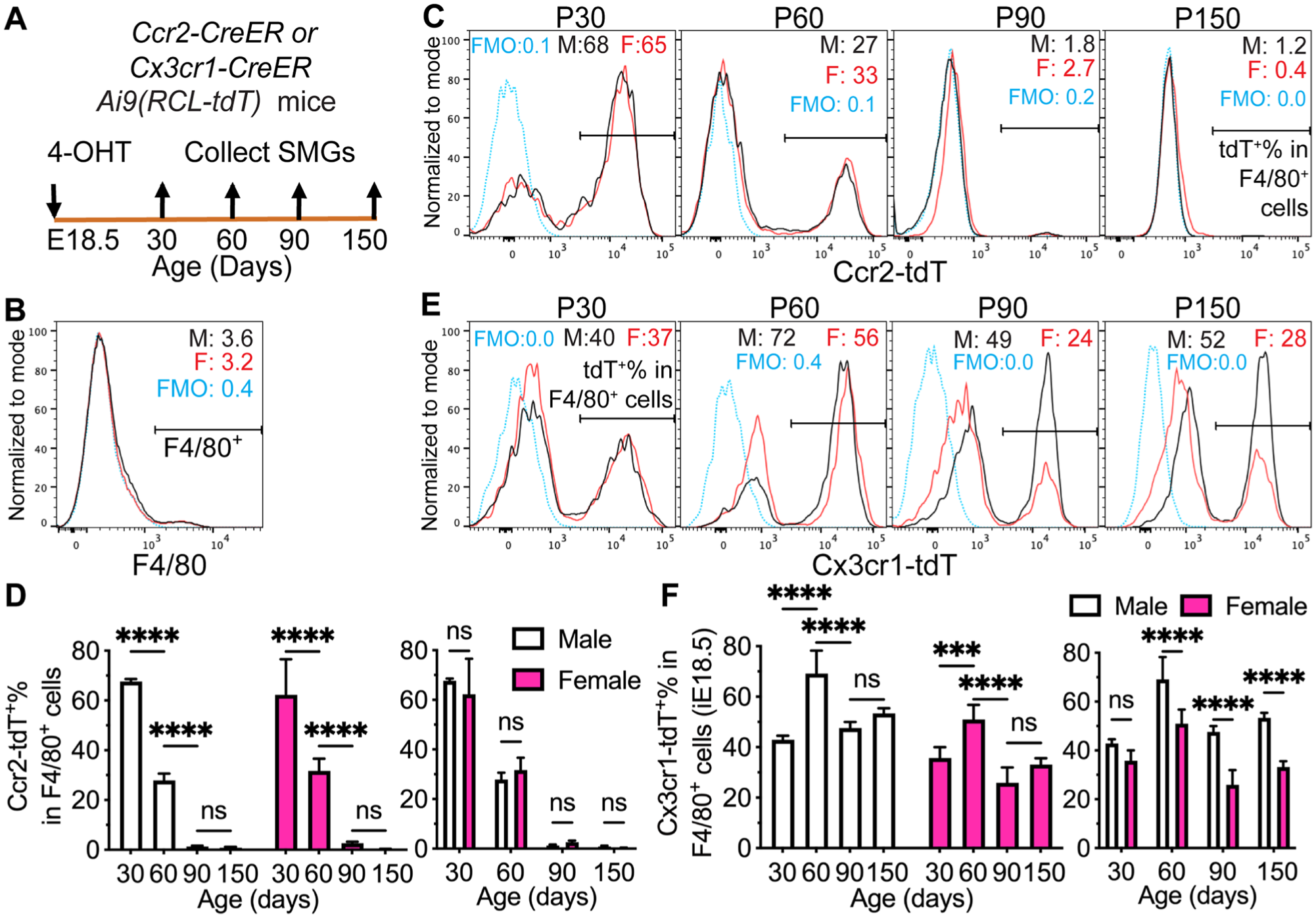
Lineage tracing of fetal progenitors in mouse SMG macrophages. (
Since Ccr2+ FMs decrease Ccr2 expression but increase Cx3cr1 expression upon tissue infiltration (Hoeffel et al 2015), we induced Cx3cr1-CreER;Ai9 mice with 4-OHT at E18.5 to trace the progeny of all Cx3cr1+ fetal cells, including those derived from FM and yolk sac in SMG macrophages. At P90, tdT was detected in most microglial cells but negligible in monocyte-enriched splenocytes without sex differences (Fig. S3D–E), validating the specificity of this approach in tracing fetal-derived long-lived macrophages. At P30, Cx3cr1-tdT+% and Cx3cr1-tdT+F4/80+% in SMG cells and Cx3cr1-tdT+% in F4/80+ macrophages are all comparable between males and females (Figs. 2E–F and S3F), indicating the similar initial labeling efficiency. Despite a transient increase of Cx3cr1-tdT+% in F4/80+ macrophages at P60 in both sexes, this index decreased to ~50% in males and ~25% in females at P90, was maintained at these levels at P150, and was significantly higher in males than females from P60 to P150 (Figs. 2E–F and S3F–H). Sex differences in the progeny of Cx3cr1+ fetal progenitors were similar to that of yolk sac–derived macrophages, but the latter did not decrease from P60 to P150 (Fig. 1E). These data confirmed that the progeny of FMs is dominant in the SMG macrophages of juvenile mice but mostly short-lived in both sexes.
SMG Macrophages Derived from Postnatal Monocytes Are Short-lived in Females but Long-lived in Males
Mouse postnatal monocytes (PMs) are Ccr2+ and contribute to adult tissue-resident macrophages in other organs (Ginhoux and Guilliams 2016). Since macrophages derived from Ccr2+ FMs were negligible in SMGs by P90 (Fig. 2A–D), we traced the PM progeny since P90 in Ccr2-CreER;Ai9 mice induced at P30. At P90, tdT was negligible in monocyte-enriched splenocytes without sex differences (Fig. S4A), validating the efficient washout of labeling in short-lived monocytes after 60 d. At P90, ~20% to 35% of SMG macrophages (F4/80+, CD45+F4/80+, CD45+F4/80+Ly-6G−Siglec-F−, or CD45+CD64+) were Ccr2-tdT+ without significant sex differences (Figs. 3A–D and S4B–J), indicating that Ccr2+ PMs initially contribute to SMG macrophages in both sexes at comparable levels. At P150 and P210, Ccr2-tdT+% in F4/80+ macrophages and SMG cells and Ccr2-tdT+F4/80+% in SMG cells decreased gradually in females to negligible levels but were maintained in males (Figs. 3A–D and S4B–D), indicating that SMG macrophages derived from Ccr2+ PMs are short-lived in females but long-lived in males.
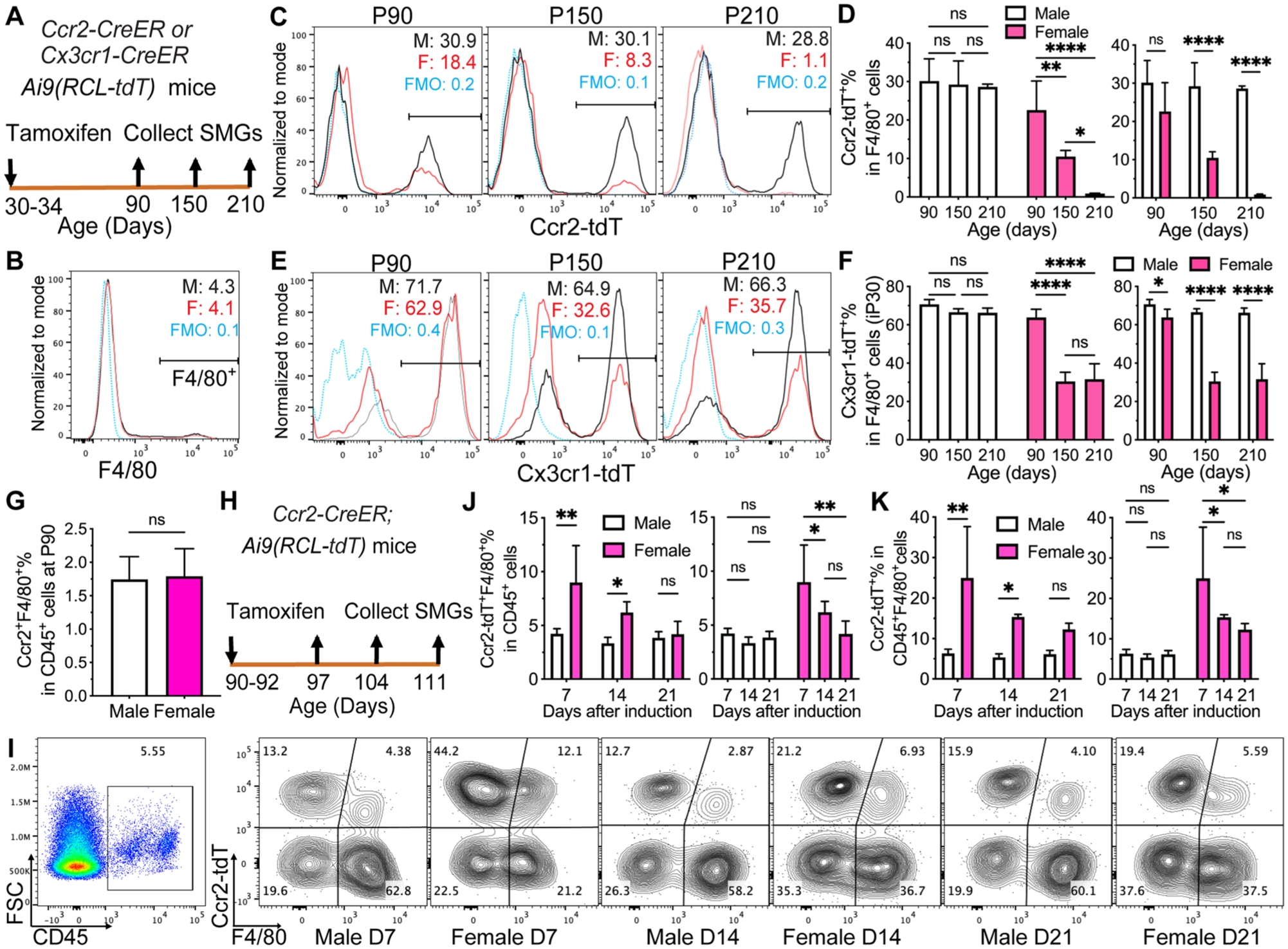
Lineage tracing of postnatal progenitors in mouse SMG macrophages. (
Since some macrophages derived from Ccr2+ PMs also lose Ccr2 expression and begin to express Cx3cr1 in other mouse organs (Dick et al 2022), we induced Cx3cr1-CreER;Ai9 mice at P30 to trace these PMs besides Cx3cr1+ macrophages derived from fetal progenitors. At P90, tdT was detected in most microglial cells but negligible in monocyte-enriched splenocytes without sex differences (Fig. S5A–B), validating the specificity of this approach in tracing long-lived macrophages. In SMGs, Cx3cr1-tdT+% in F4/80+ macrophages at P90, P150, and P210 was maintained without significant changes in males at ~70% but decreased greatly from ~60% to ~30% in females (Fig. 3E–F), which is consistent with data in Ccr2-CreER;Ai9 mice. In all 3 age groups, this index on the progeny of all perinatally established macrophages was significantly lower in females than males (Figs. 3E–F and S5F–H), which is consistent with sex differences of yolk sac– and fetal-derived macrophages (Figs. 1 and 2E–F).
In other organs of adult mice, CCR2+ macrophages are short-lived and constantly replenished by monocytes (Dick et al 2022). In P90 SMGs, the percentages of Ccr2+F4/80+ macrophages in CD45+ cells were very low and comparable between the sexes (Fig. 3G). Therefore, to estimate the progeny of adult classical monocytes, we induced P90 Ccr2-CreER;Ai9 mice for 3 d and examined the tdT+ progeny of Ccr2+ cells in SMGs at 7, 14, and 21 d later (Fig. 3H). At 7 d, tdT was detected in many monocyte-enriched splenocytes but negligible in microglia without sex differences (Fig. S6A–B), validating the specific labeling of monocytes and their progeny. In SMGs, the percentages of tdT+F4/80+ and tdT+F4/80− cells in CD45+ cells and tdT+% in CD45+F4/80+ macrophages were significantly higher in females than males at 7 and 14 d after induction but not 21 d, and these indexes significantly decreased at days 14 and 21 than day 7 in females but not males (Figs. 3I–K and S6C–D). Similar sex differences were observed with CD45+CD64+ as the macrophage marker (Fig. S6E–F). These data confirmed that macrophages derived from adult classical monocytes are much more abundant in female SMGs and their life spans are short.
These data collectively suggested that most adult SMG macrophages are established before adulthood and long-lived in males but not in females and much more adult female SMG macrophages are continually replenished by monocytes.
SMGs of Female Wild Type Mice Contain More Proinflammatory Classical Monocytes and Macrophages Recently Derived from Them Than Males
To validate these lineage-tracing data in wild type mice, we examined the expression of pan-immune cell marker CD45, macrophage marker F4/80, and multiple monocyte markers in the SMGs from male and female C57BL/6 mice at P30, P90, and P180 with flow cytometry. Ly6c and Ccr2 are widely used markers of proinflammatory classical monocytes in mice (Yona and Jung 2010), whereas Csf2rb is required for the inflammatory signature of Ccr2+Ly6c+ monocytes (Croxford et al 2015) and M1 polarization of macrophages (Castro-Dopico et al 2020). The expression of these markers in monocyte-derived macrophages is generally transient (Li et al 2022); moreover, F4/80+ cells expressing Ly6c or Ccr2 are considered macrophages derived from newly recruited monocytes, whereas macrophages expressing Csf2rb have higher proinflammatory potentials (Castro-Dopico et al 2020). In the SMGs of C57BL/6 mice, the percentages of CD45+ immune cells were comparable between the sexes at P30 but higher in females than males at P90 and P180 (Figs. 4A–B and S7A). In gated CD45+ cells, the percentages of F4/80+ macrophages were comparable between the sexes at P30 but significantly lower in females than males at P90 and P180 (Figs. 4C–D and S7B–D). Notably, F4/80 expression decreased greatly in some female SMGs (3 or 4 of 6) but not all at P90 and P180 than P30, and overall percentages of F4/80+ in female CD45+ SMG cells remained comparable between P30 and P90 and between P90 and P180 (Fig. 4D). Despite high variations of F4/80 expression, the percentages of Ly6c+F4/80−, Ccr2+F4/80−, and Csf2rb+F4/80− in CD45+ cells and Ly6c+, Ccr2+, and Csf2rb+ in CD45+F4/80+ cells in these adult female SMGs were consistently higher than in male SMGs (Figs. 4C–L and S7B–D). Moreover, in CD11b+CD45+ myeloid cells from P90 SMGs, CD64+Ly6c+ macrophages recently derived from monocytes were more abundant in females than males; however, unlike that reported in intestine myeloid cells (Tamoutounour et al 2012), the numbers of CD64+Ly6c+ cells were limited, and the expression of these 2 markers did not show the waterfall-shaped distribution in both sexes (Fig. S7E–F), supporting the coexistence of long- and short-lived macrophages in SMGs rather than the conventional monocyte waterfall model. At P30, some of these indexes were already significantly higher in females than males, whereas other indexes were only slightly higher, which is consistent with the significant sexual differences of yolk sac– or fetal-derived macrophages at P60 but not P30 (Figs. 1 and 2). Considering the much higher contribution of adult monocytes to SMG macrophages in females and the short life span of adult monocyte-derived macrophages (Fig. 3H–I), it is possible that the variable F4/80 expression in female SMGs is the consequence of fast macrophage turnover and variable monocyte differentiation. At P180, similar sex differences were observed by CD45 and CD64 for identifying macrophages as F4/80: female CD45+ SMG cells contained much fewer CD64+ macrophages and more Ly6c+CD64− monocytes, and female CD45+CD64+ macrophages contained much more Ly6c+ cells recently derived from monocytes than males (Fig. S7G–J). These data indicate that the SMGs of adult female C57BL/6 mice contain more leukocytes, classical monocytes, and macrophages recently derived from monocytes than males.
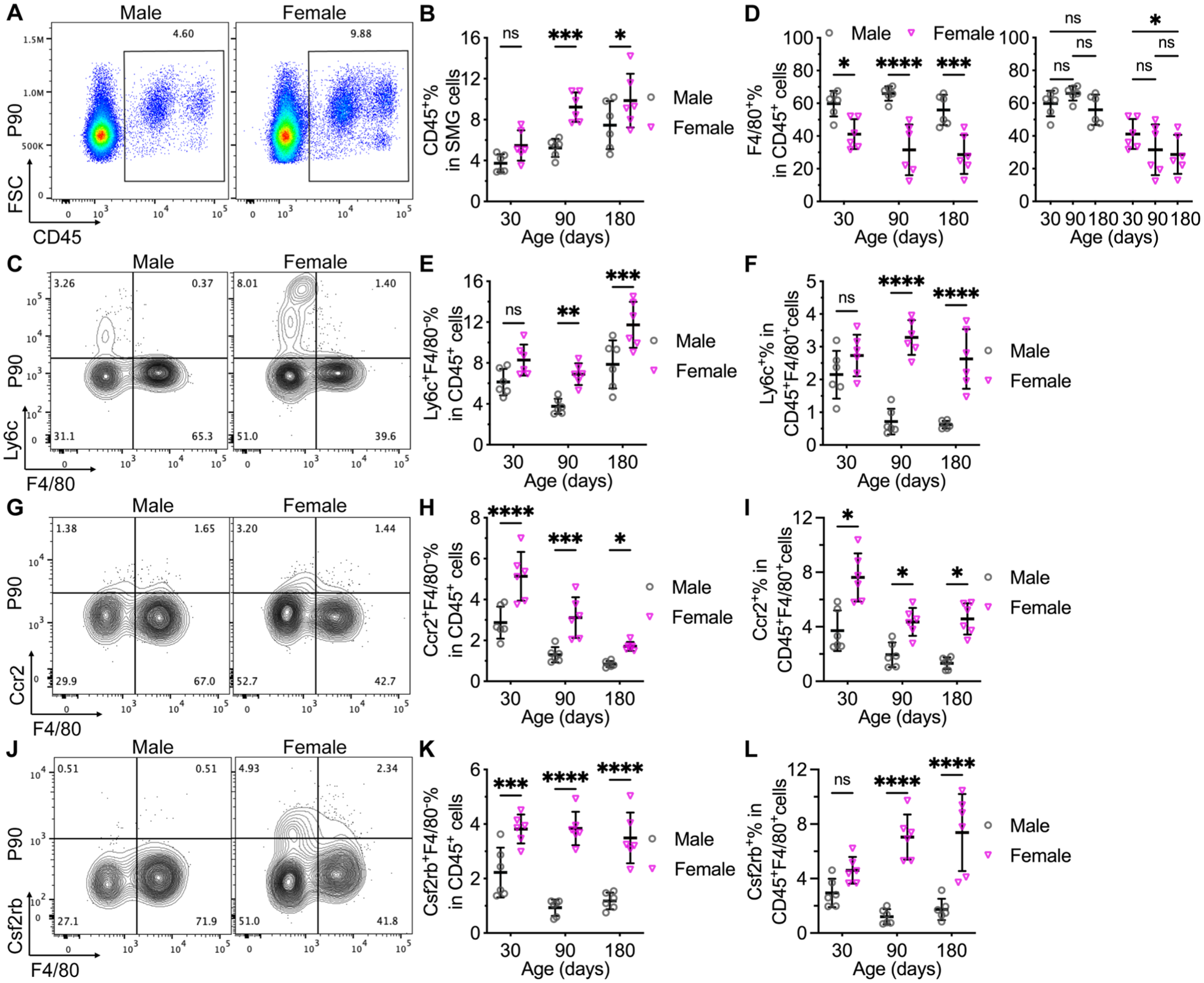
Sex differences in the percentages of immune cells, classical monocytes, and macrophages expressing monocyte markers in SMGs from wild type mice. SMGs were collected from wild type male and female C57BL/6 mice at postnatal day 30 (P30), P90, and P180 for flow cytometry. CD45+ immune cells were gated and quantified (
scRNA-seq Revealed Sexual Differences in Composition and Gene Expression of SMG Immune Cells
To determine the sexual dimorphism of SMG immune cells, we isolated CD45+ cells from the SMGs of P90 male and female C57BL/6 mice with magnetic cell sorting, performed scRNA-seq, and deposited the dataset to Gene Expression Omnibus as GSE298595. Our scRNA-seq data identified 1 macrophage cluster (Adgre hi Csf1r hi Mafb hi ) containing 7 subclusters; 4 myeloid/monocyte clusters (Fcgr1+Adgre1 low Csf1r low Mafb low ); and individual clusters of innate lymphoid cells (Ncr1+Ccr2+Ifng+), T cells (Cd3d+Ccr2 hi Csf2+), dendritic cells (Flt3+Csf2rb hi ), ductal epithelial cells (Krt8+Krt18+Prol1 hi Ptprc−), and endothelial cells (Flt1+Pecam1+Ptprc−; Figs. 5A–B and S8A). Ptprc/Cd45− ductal epithelial cells and endothelial cells were likely isolated due to the breakage and adhesion of macrophage protrusions and not counted as immune cells. The 4 myeloid/monocyte clusters include Ccr2+Csf2rb+Itgam hi Ly6c+ classical monocytes, Cd163+Cd206+ M2-like monocytes, Irf7+Isg15+ M1-like monocytes, and Birc5+Stmn1+ mitotic myeloid cells. The proportions of classical monocytes, innate lymphoid cells, and T cells among total immune cells were notably higher in female SMGs; those of total macrophages, M1-like monocytes, and M2-like monocytes were slightly higher in males; and those of mitotic myeloid cells and dendritic cells were comparable (Fig. 5C–D). Among macrophages, female SMGs contained more cells in Cd163+Cd206+ M2-like and Cd86hiTnf+ M1-like clusters and slightly fewer cells in nonpolarized M0 clusters (Fig. 5B and E). Notably, multiple phagocytosis-related genes, including Mertk, Itgam, Lyz2, and Pf4, were enriched in the cells of M2-like cluster, consistent with the report that phagocytosis promotes M2 polarization (A-Gonzalez et al 2017). A small subset of macrophages highly expressed acinar markers such as Prol1 and was termed epithelial-like macrophages. This macrophage subset was likely formed by the phagocytosis of apoptotic acinar cells, and more cells in epithelial-like macrophages and M2-like clusters of female than male SMGs suggests higher phagocytosis activities. The flow cytometry of P90 SMGs confirmed that the percentages of innate lymphoid cells and NKT cells in CD45+ immune cells were significantly higher in females than males, whereas that of T cells was slightly higher in females (Figs. 5F and S8D). In CD45+F4/80+ macrophages, the percentages of M1- and M2-like cells were significantly higher in females, and that of M0-like cells was higher in males (Figs. 5G and S8E), confirming stronger macrophage polarization in female SMGs. In our scRNA-seq data, the most female-biased gene in all macrophage clusters was Xist, a lncRNA transcribed only from the inactive X chromosome in females and promoting autoimmunity and M2 polarization (Pi et al 2022; Dou et al 2024; Figs. 5H–I and S8B–C). Among other female-biased genes, estrogen target genes Klk1 and Hspa1a/1b showed context-dependent pro- or anti-inflammatory activities (Yiu et al 2014); Fkbp5 and Lgals3 were proinflammatory (Dragomir et al 2012; Jantaruk et al 2017); and acini-abundant Scgb2b26 suggested higher phagocytosis (Fig. S8A). Among male-biased genes, Wfdc12 and Y-chromosome gene Uty showed anti-inflammatory activities (Glasgow et al 2015; Cunningham et al 2022); another Y-chromosome gene, Ddx3y, modulated macrophage activation and cytokine production (McGill et al 2025); and functions of 2310057J18Rik are largely unknown. These data indicated that healthy female adult SMGs contain more immune cells and polarized macrophages, which is related to the sex-biased expression of multiple immune modulatory genes in macrophages.
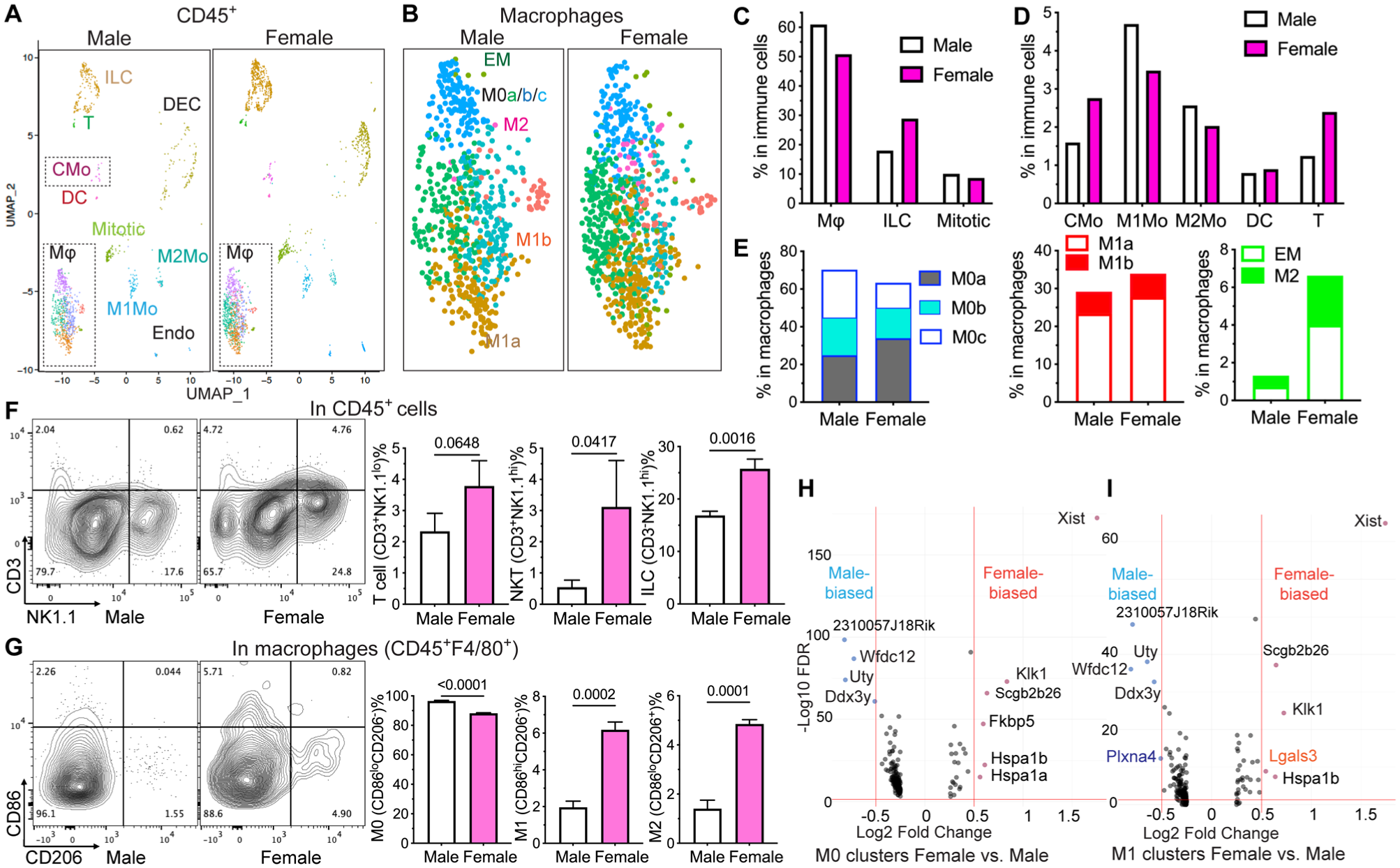
Sex differences in the composition of SMG immune cells and gene expression in SMG macrophages. (
Discussion
Our data showed that most SMG macrophages are long-lived in adult male mice and derived mainly from yolk sac progenitors and perinatal monocytes. Conversely, much more macrophages in adult female SMGs are short-lived and continuously replenished by monocytes. The percentages of macrophages in SMG leukocytes are consistently much lower in adult female C57BL/6 mice than males, whereas the percentages of leukocytes in all SMG cells and those of classical monocytes, innate lymphoid cells, and T cells in leukocytes are higher in adult female SMGs. Moreover, SMG macrophages in adult female mice appear more polarized and express higher levels of proinflammatory factors than in males. The macrophage ontogeny and percentages of leukocytes in SMGs are comparable between adolescent male and female mice, indicating that the sexual differences in adult SMG macrophages are mainly due to the maintenance, not recruitment, of macrophage progenitors before adulthood. In many steady-state adult mammal tissues, long-lived tissue-resident macrophages of perinatal origins coexist with short-lived adult monocyte-derived macrophages. Long-lived tissue-resident macrophages prevent autoimmunity by efferocytosis and support tissue functions via homeostatic crosstalk with specialized tissue cells, whereas short-lived monocyte-derived macrophages generally increase inflammation. Upon aging or pathologic inflammation, long-lived tissue-resident macrophages are replaced by short-lived monocyte-derived macrophages, which increases autoimmunity risks, compromises homeostatic crosstalk, and leads to tissue dysfunction (Lazarov et al 2023; Mass et al 2023). Moreover, the chronic low-grade inflammation may shorten the life span of macrophages (Viola and Boeckxstaens 2021). Therefore, the substantially greater contributions of adult monocytes to macrophages in female than male SMGs likely lead to stronger baseline inflammation, which may together contribute to the much higher risks of Sjögren’s disease in females in human and mouse models through linking innate and adaptive immunity (Sequi-Sabater and Beretta 2022; Baldini et al 2024).
Mechanisms underlying sex differences in SG macrophages need further exploration. Male rodent SGs contain unique granular convoluted ducts that are absent in human and female rodents, but our previous scRNA-seq data showed that granular convoluted duct cells are not a major source of macrophage growth factors (Zhao et al 2023), suggesting that these ducts are unlikely the cause of sexual dimorphism of SG macrophages. In adult C57BL/6 mice, genes related to immune activation are enriched in female SMGs (Mukaibo et al 2019). In humans, anti-inflammatory genes are enriched in the minor SGs of men (Michael et al 2011), whereas genes related to responses to wounding are enriched in the parotids of women (Srivastava et al 2008). Xist expressed only in females can activate TLR7 and affect macrophage polarization (Syrett et al 2019). Moreover, TLR4 signaling in macrophages is regulated negatively by male hormones but positively by female hormones (Rettew et al 2008, 2009). We confirmed that female SMG macrophages highly express Xist, estrogen target gene Klk1, and several proinflammatory genes. Considering the prevalence of Sjögren’s disease in women over men and the conserved Xist, X-chromosome genes, and immune modulatory genes regulated by sex hormones, it is likely that similar sexual differences in SG immune cells are present in human.
In summary, our data indicate that macrophages in the steady-state SMGs of adult mice are mostly long-lived and established before adulthood in males, whereas much more SMG macrophages in females are short-lived and continuously replenished by classical monocytes. These sexual differences need be considered in mouse models of diseases affecting SGs, whereas the clinical significance of these findings remains unclear and warrants further studies.
Author Contributions
Q. Zhao, contributed to design, data acquisition and analysis, drafted the manuscript; S. Pan, L. Zhang, contributed to data analysis and interpretation, critically revised the manuscript; J. Jaiswal, L.-T. Wang, E. Chang, contributed to data acquisition and analysis, critically revised the manuscript; A. Shahsavari, Y. Zhang, V. Yu, R. Zheng, T. Chen, contributed to data acquisition, critically revised the manuscript; F. Liu, contributed to conception and design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave their final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251411907 – Supplemental material for The Ontogeny of Mouse Salivary Gland Macrophages Is Distinct between Sexes
Supplemental material, sj-docx-1-jdr-10.1177_00220345251411907 for The Ontogeny of Mouse Salivary Gland Macrophages Is Distinct between Sexes by Q. Zhao, S. Pan, J. Jaiswal, L. Zhang, L.-T. Wang, E. Chang, A. Shahsavari, Y. Zhang, V. Yu, R. Zheng, T. Chen and F. Liu in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345251411907 – Supplemental material for The Ontogeny of Mouse Salivary Gland Macrophages Is Distinct between Sexes
Supplemental material, sj-pdf-2-jdr-10.1177_00220345251411907 for The Ontogeny of Mouse Salivary Gland Macrophages Is Distinct between Sexes by Q. Zhao, S. Pan, J. Jaiswal, L. Zhang, L.-T. Wang, E. Chang, A. Shahsavari, Y. Zhang, V. Yu, R. Zheng, T. Chen and F. Liu in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health/National Institute of Dental and Craniofacial Research (1R01DE031478 to F.L.).
Data Availability Statement
The single-cell RNA sequencing data are shared at Gene Expression Omnibus as GSE298595.
A supplemental appendix to this article is available online.
