Abstract
Diabetic periodontitis constitutes a serious and multifaceted public health issue, acting as a significant risk factor for various systemic diseases by exacerbating inflammatory responses and disrupting metabolic stability. Its high prevalence, severe periodontal tissue destruction, and complex pathological mechanisms present substantial therapeutic challenges. Conventional therapies frequently fail to target underlying molecular pathological mechanisms. Ferroptosis, an iron-dependent form of regulated cell death characterized by iron accumulation and lipid peroxidation, has been increasingly implicated in diabetic periodontitis. However, translating ferroptosis inhibition into effective therapy remains problematic. Current strategies, primarily relying on single-pathway inhibitors, exhibit insufficient effectiveness. To overcome this, we engineered a bimetallic ZIF-8 codelivery nanoplatform (FGZ nanoparticles [NPs]) for sustained release of gallium ions (Ga³+) and canonical ferroptosis inhibitor ferrostatin-1 (Fer-1), synergistically integrating the functions of the 2 drugs. FGZ NPs potently activated the Nrf2/HO-1 cytoprotective pathway, simultaneously reestablishing iron homeostasis and strengthening antioxidant capacity, resulting in effective dual-pathway ferroptosis inhibition. FGZ NPs demonstrated outstanding therapeutic outcomes, significantly promoting the regeneration of damaged periodontal tissues. This study validates ferroptosis as a promising target for diabetic periodontitis and introduces a novel strategy that surpasses the constraints of single-pathway ferroptosis inhibition, providing a new design framework for biomaterials targeting ferroptosis-related diseases.
This is a visual representation of the abstract.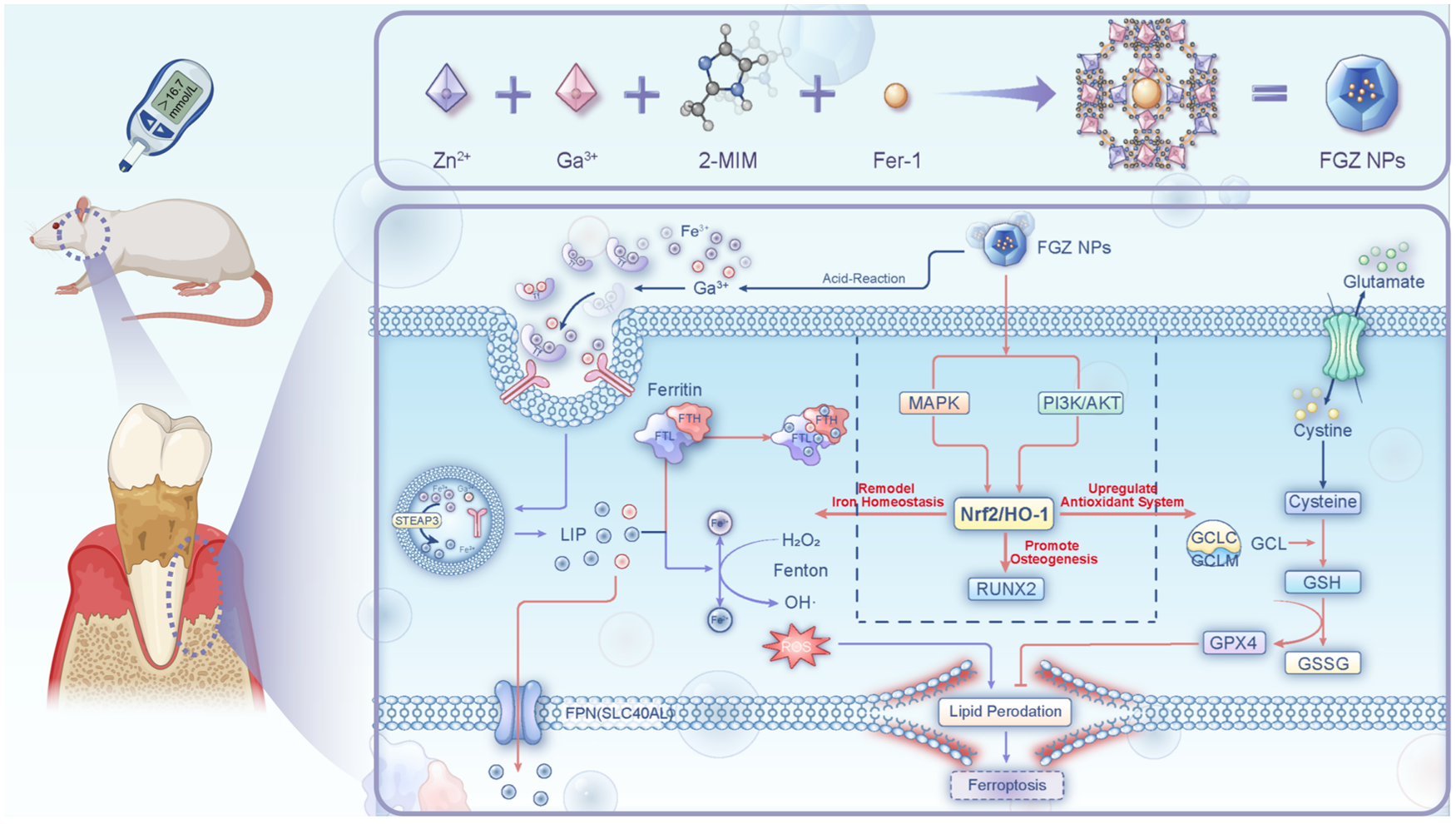
Get full access to this article
View all access options for this article.
