Abstract
Mastication is a vital function that relies on precise synchronization among multiple brainstem regions, known as being part of a central pattern generator (CPG). Movements can be triggered either by stimulating a sensory-motor region called the cortical masticatory area (CMA), well-documented in various species but not yet formally identified in mice, or by stimulating the orofacial sensory primary afferents, the fibers of which form the trigeminal tract (Vtr). However, it is unclear whether these different inputs activate distinct components of the CPG or converge on the same. This study aims to map the brainstem areas activated by cortical and sensory inputs using immunohistochemistry against the cellular activity markers, c-Fos and Ca2+ imaging, respectively. Optogenetic stimulation of the cortical masticatory area in awake, head-fixed mice reliably induced rhythmic jaw movements and increased c-Fos expression in multiple brainstem regions, with the strongest activation in the peritrigeminal area (PeriV). In contrast, in vitro electrical stimulation of the Vtr predominantly activated neurons and astrocytes in the main sensory nucleus (NVsnpr), the dorsal area of PeriV adjacent to it, and the parvocellular reticular nucleus (PCRt). The areas containing the highest numbers of activated cells differed clearly according to the type of inputs and overlapped only in the PCRt, ventral and slightly medial to the trigeminal motor nucleus and the most dorsomedial part of PeriV. These findings demonstrate that cortical and sensory inputs take part in distinct components of the brainstem masticatory circuitry, with PCRt emerging as a potential point of convergence and providing new insights into the components of the CPG of mastication.
Keywords
Introduction
Mastication, a vital function that involves preparing food for digestion, is a key component of feeding behavior that relies on sensory and cortical inputs to adapt the movements to the properties of the food bolus. Using decerebrate and paralyzed animals, Dellow and Lund (1971) were the first to demonstrate that, like other rhythmic movements such as breathing and locomotion, mastication relies on a specific neuronal network known as a central pattern generator (CPG) located in the brainstem (Dellow and Lund 1971; Feldman and Grillner 1983; Lund 1991; Falardeau et al 2023). Later, using an isolated brainstem preparation, Kogo et al (1996) and Tanaka et al (1999) suggested that the masticatory CPG was confined to the pontine area of the brainstem defined rostrocaudally by the rostral limits of the trigeminal (NVmt) and facial (NVII) motor nuclei and mediolaterally by the column of the last-order interneurons surrounding the NVmt and the trigeminal sensory complex formed at this level by the trigeminal main sensory nucleus (NVsnpr) and the rostral part of the spinal trigeminal nucleus (NVspo). The areas dorsal, lateral, and medial to the NVmt are named the supra- (SupV), inter- (IntV), and juxta-trigeminal (JuxtV) areas, respectively. The area ventral to the NVmt, which also extends caudally, is the parvocellular reticular formation (PCRt). We refer to the 4 areas surrounding the NVmt as the peritrigeminal area (PeriV). Besides a small proportion of SupV neurons, intrinsic bursting properties were found only in neurons of the dorsomedial part of NVsnpr (Sandler et al 1998; Bourque and Kolta 2001; Brocard et al 2006), which led to the proposal that it forms the rhythmogenic core of the masticatory CPG (Tsuboi et al 2003; Brocard et al 2006; Morquette et al 2015).
In in vivo animal models, rhythmic jaw movements (RJMs) resembling mastication can be elicited by sustained repetitive stimulation of a cortical area named the cortical masticatory area (CMA) or of the trigeminal sensory afferents (Dellow and Lund 1971), both of which project to components of the brainstem area thought to contain the masticatory CPG. The aim of this study was to examine whether these inputs engage different components or converge on the same areas to better understand the circuitry underlying mastication. The CMA is well defined in many species but still not formerly identified in mice; however, Mercer Lindsay et al (2019) pointed at its existence in a study mapping corticobulbar projections. In rabbits, c-Fos expression in trigeminal areas has been used to map neurons activated by the stimulation of the CMA (Athanassiadis et al 2005). Here, a similar approach was used to describe the distribution of neurons activated by stimulation of the CMA in mice but could not be used to map the distribution of cells activated by sensory inputs, since it is difficult to activate these specifically and strongly in vivo. Thus, we used Ca2+ imaging in in vitro slice preparations to map the distribution of cells responding to electrical stimulation of the portion of the Vth tract presumably containing the fibers of sensory afferents most needed for mastication (periodontal and muscle spindle afferents). Our results confirmed the existence of a circumscribed CMA in mice and revealed distinct patterns of cellular activation. CMA stimulation broadly increased neuronal activity across multiple brainstem regions, including the NVsnpr with pronounced activation primarily in JuxtV and PCRt. In contrast, sensory stimulation predominantly revealed cellular activation in NVsnpr, IntV, SupV, and PCRt. These observations suggest that PCRt may serve as a critical convergence hub for both cortical and sensory inputs.
Methods
All procedures were approved by the University of Montreal Animal Care Committee (#23-207) and followed Canadian Institutes of Health Research guidelines. Full methods are given in the appendix. The study complies with ARRIVE 2.0 guidelines.
Results
Optogenetic Stimulation of a Circumscribed Cortical Area Induces RJMs
Using whole-cell patch clamp recordings of layer 5 pyramidal neurons, we first examined in in vitro cortical slices from VGlut2-ChR2 mice, the effects of photostimulation to ensure that it efficiently activates pyramidal neurons. Figure 1A shows that blue light (440 nm; top trace), but not red light (561 nm; bottom trace) induced sustained firing in the recorded neurons (n = 5; N = 2). Then, as in rats, we identified 2 areas, 1 anterior (coordinates: 2.5 mm AP, 2.0 mm L, and 0.75 mm D relative to Bregma) in the motor cortex (Fig 1C) and 1 posterior (coordinates: 0 mm AP, 3.5 mm L, and 2.0 mm D relative to Bregma) in the granular and agranular insular posterior cortices (Fig 1C), for which optogenetic stimulation through implanted optic fibers (Fig 1B) in VGlut2-ChR2 mice elicited RJMs. We confirmed ChR2 expression and the location of optic fiber implantation by performing immunohistochemistry on the brain sections of the tested animals to reveal the green fluorescent protein (GFP)–stained cells (arrows) and their NeuN-stained nuclei (arrowheads) as well as the lesion (*) made by the optic fiber (Fig 1D). Optogenetic stimulation at 40 Hz (for 3 s) induced stereotypical RJMs, as shown in Figure 1E and F for the anterior and posterior areas, respectively. For the anterior area (A-area) stimulation, the movements began with stimulation and continued throughout its duration. Conversely, for the posterior area (P-area), movements sometimes persisted for 1 to 2 s after the end of the stimulation. We then assessed whether RJMs maintained a relatively stable frequency over the course of the 30-min stimulation period. The frequency of RJMs in mice stimulated in both the A-area and P-area combined decreased over time but was not statistically significant (start = 10.79 ± 0.59 Hz vs end = 8.85 ± 1.00 Hz, P = 0.2188 > 0.05, 2-tailed Wilcoxon matched-pairs signed rank test, n = 6; Fig 1G). Overall, we established a robust method to induce RJMs for the first time in awake and head-fixed mice using optogenetics, demonstrating consistent results over time.
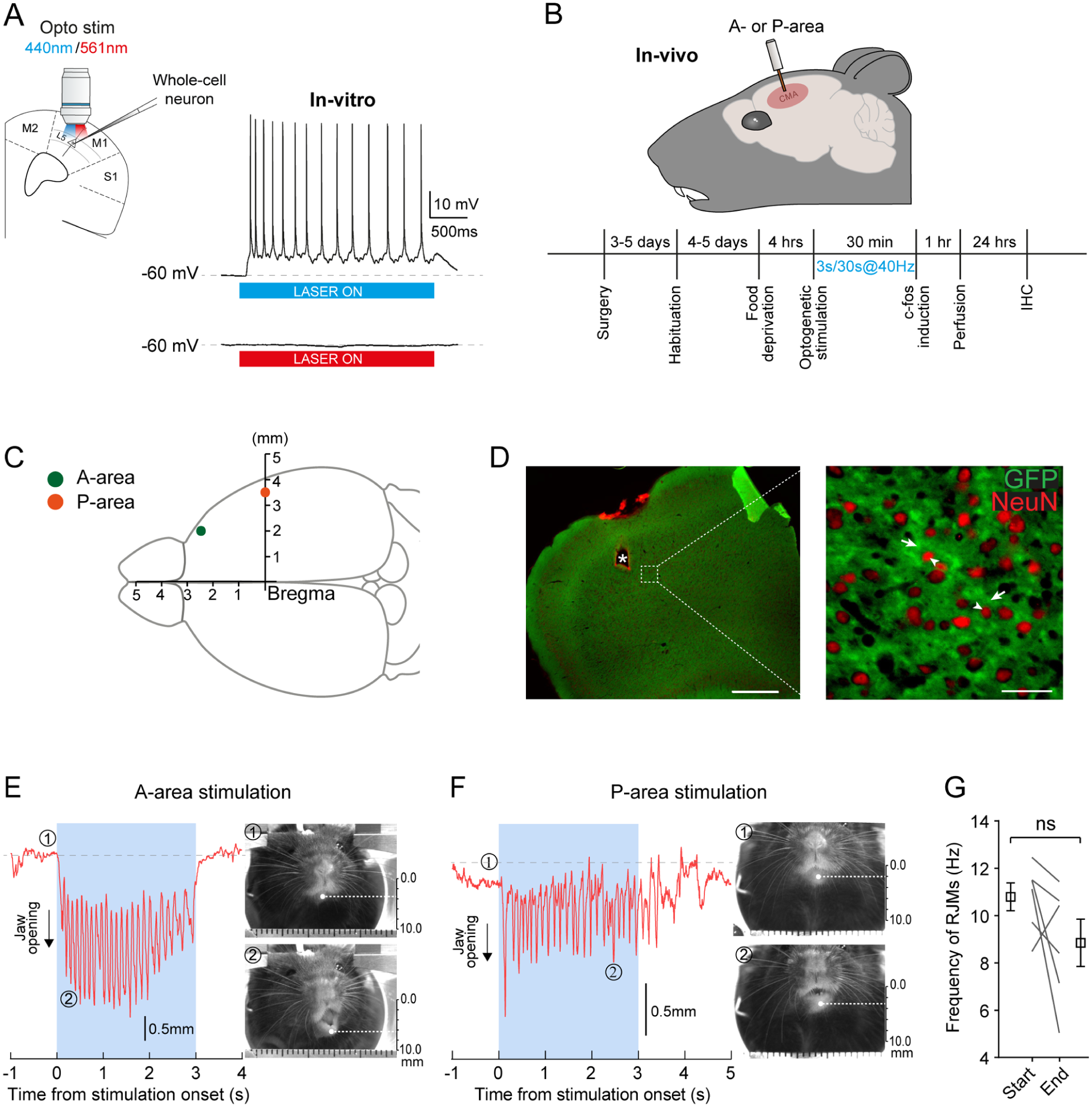
In vivo optogenetic stimulation of the cortical masticatory area (CMA) induces rhythmic jaw movements (RJMs). (
Expression of c-Fos in Brainstem Areas after Optogenetic Stimulation of the CMA
Brainstem c-Fos–stained cells were mapped, as described in the methods, for 6 mice that were subjected to optogenetic stimulation of the CMA (stimulated) and for 5 mice that were implanted but unstimulated (control). The paired interleaved scatter plot in Figure 2A displays data from both control (empty circles) and stimulated (blue circles) animals, illustrating the quantitative distribution of c-Fos staining among the brainstem nuclei and regions. The P values for all regions are presented in Appendix Table 1. Since projections from the CMA are predominantly contralateral, we investigated whether the position of the fibers influenced activation patterns. To this end, we compared the ipsilateral (purple) and contralateral (orange) sides to identify any significant differences. As shown in Appendix Figure 1A, no significant changes were observed in any of the regions analyzed. We then examined whether stimulation of the A-area led to changes in c-Fos–stained cell counts compared with stimulation of the P-area (Appendix Fig 1B, gray and mauve circles, respectively). Although the number of mice implanted in the P-area was limited, no differences were detected between the 2 groups across all regions. We then performed a density mapping of all brainstem sections to pinpoint the hotspot of c-Fos–stained cells. For analysis purposes, data for regions expanding on multiple sections (as shown in Fig 2B and C) were combined in the plots (Fig 2A and Appendix Fig 1A, B) and in Appendix Table 1. Overall, several regions showed an increase in c-Fos–stained cells in stimulated animals relatively to control, including the PCRt, JuxtV, SupV, IntV, PCRtA, IRt, NVmt, the trigeminal accessory motor nucleus (NVac), and the NVsnpr.
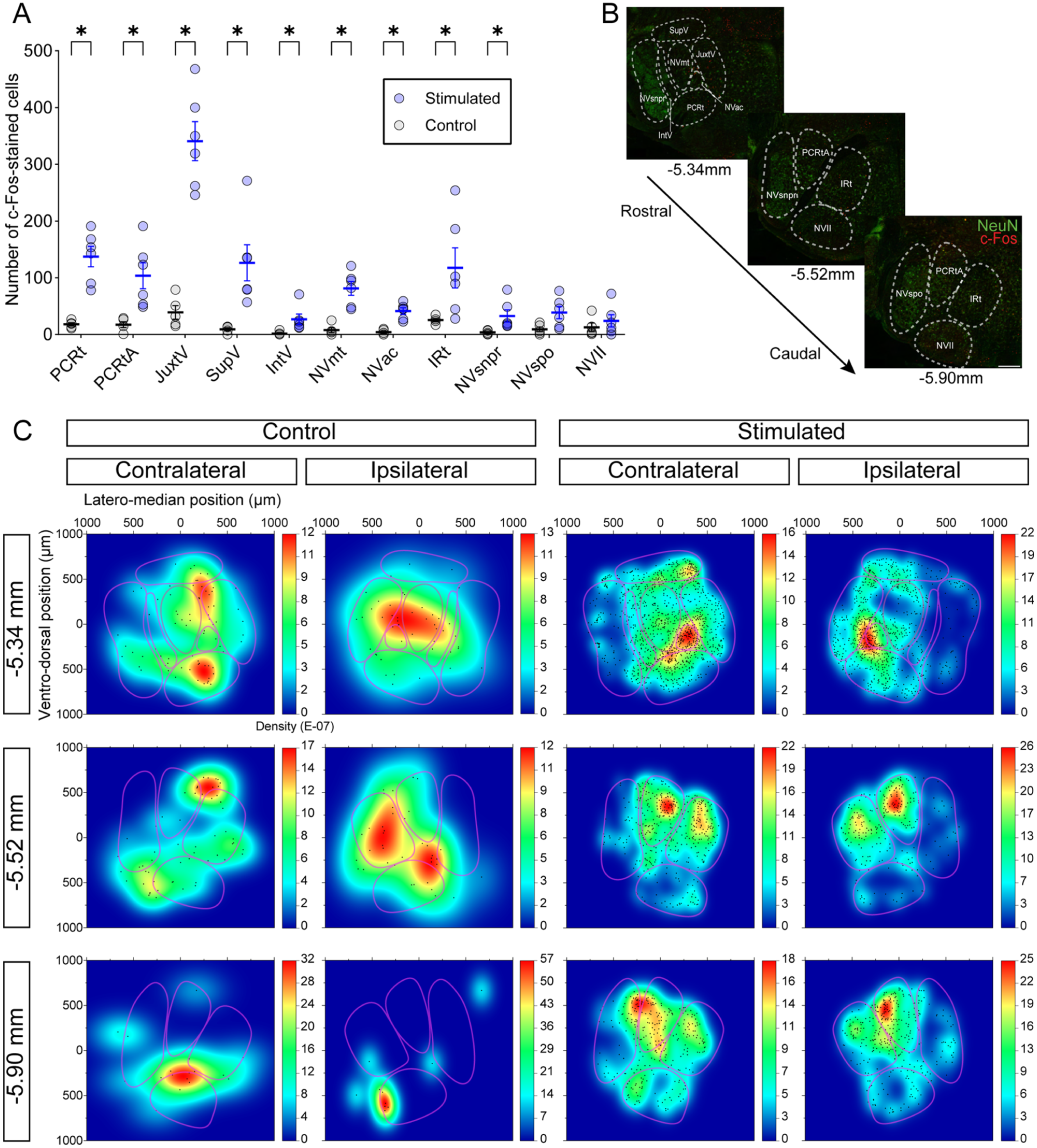
Distribution of c-Fos–stained cells in the brainstem induced by optogenetic stimulation of the cortical masticatory area. (
At the level of the NVmt (−5.34 mm relative to Bregma; Fig 2C, top), density maps on the contralateral side revealed that c-Fos–positive cells were primarily distributed in the PCRt, NVac, NVmt, and JuxtV. A similar pattern was observed ipsilaterally. Control experiments revealed clusters of c-Fos–positive cells localized partially in the PCRt, JuxtV, and NVmt on the contralateral side and partially around the JuxtV, NVac, and NVmt on the ipsilateral side.
Similar results were obtained at the level of the medial pons (−5.52 mm relative to Bregma; Fig 2C, middle) and close to the medulla (−5.90 mm relative to Bregma; Fig 2C, bottom). In both levels, c-Fos–stained cells were predominantly located in the dorsomedial part of the brainstem, with density hotspots centered around the PCRtA and IRt. Density maps from the control experiments at the medial pons showed sparse staining at the junction of the IRt and PCRtA on the contralateral side, while on the ipsilateral side, staining was primarily localized within the IRt. In the medulla, control results differed from the experimental condition, with staining hotspots observed in the PCRtA on the contralateral side at both examined locations.
Thus, optogenetic stimulation of the CMA led to a marked increase in c-Fos expression in several brainstem regions. These increases were more important for rostral areas analyzed, slightly higher on the contralateral side and highest in JuxtV. These results highlight a lateralized and region-specific activation pattern within the masticatory CPG circuitry.
In Vitro Stimulation of the Sensory Inputs Generates Repetitive Calcium Transients in Neurons and Astrocytes of the NVsnpr and PeriV
Based on the above results, we pursued experimentation using calcium imaging techniques in in vitro brainstem slices at the level of the NVmt, where the highest increases in c-Fos were seen, to map areas responding most strongly to the stimulation of sensory inputs conveyed by primary afferent axons forming the trigeminal tract (Vtr). This approach makes it possible to visualize both neuronal and astrocytic calcium responses to stimulation.
Expression of the calcium indicator GCaMP6f by brainstem neurons was confirmed in 2 lines of transgenic mice expressing it under the control of VGlut2 (Fig 3A) and Thy1 promoters (Fig 3B). To detect astrocytic responses simultaneously with neuronal responses, we added Fluo8L-AM, which is a calcium indicator primarily taken up by astrocytes when added to the bath, and SR-101 was also used to label astrocytes specifically (Fig 3C, middle). Neurons and astrocytes in the dorsal NVsnpr (Fig 3E, traces 1–2) and PeriV (Fig 3E, traces 3–4) responded to glutamate (500 µM, 30 s) applications, confirming the functionality of our 2 calcium indicators (GCaMP6f and Fluo8L).
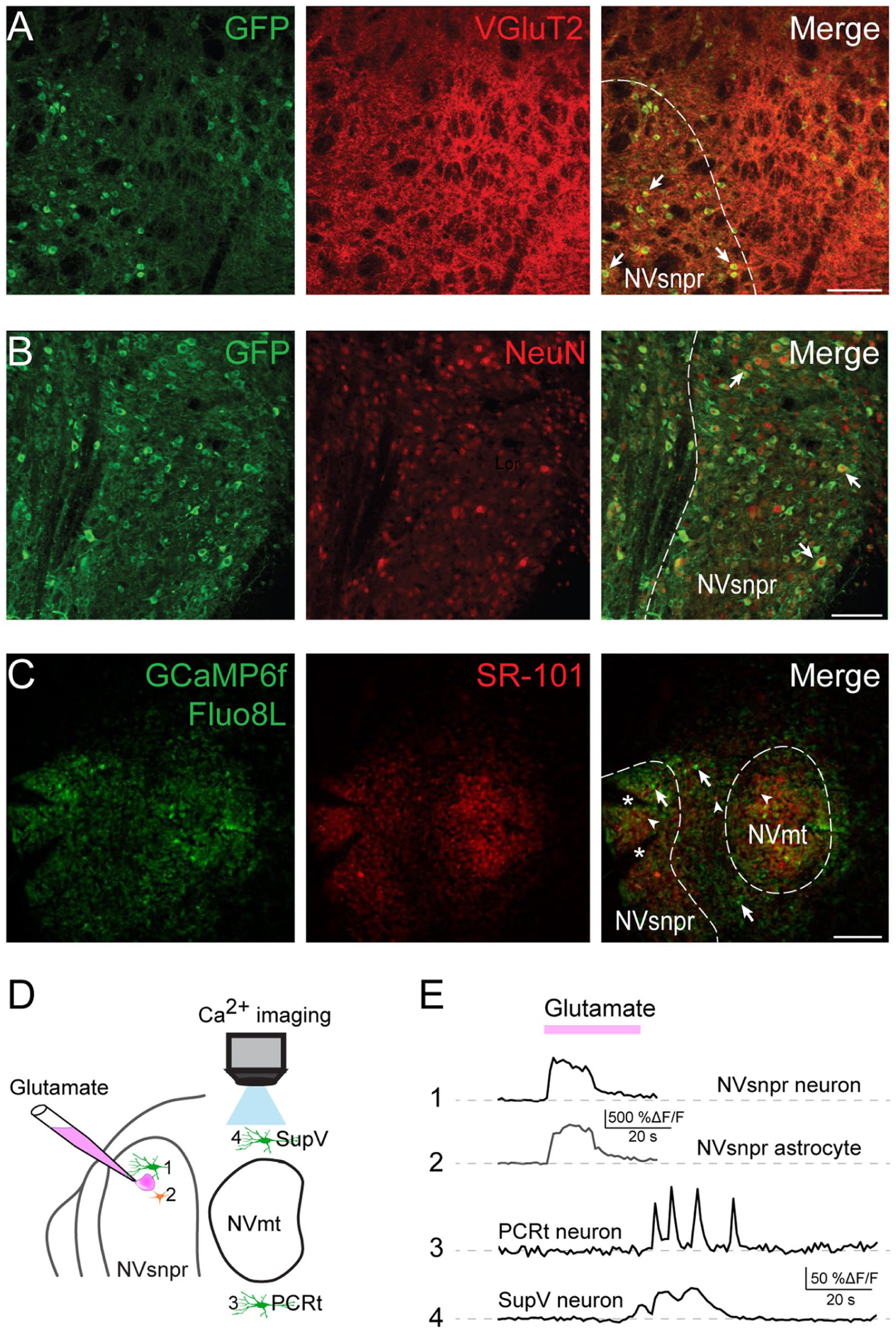
Validation of the in vitro model to assess brainstem Ca2+ imaging. The calcium indicator GCaMP6f is expressed under the control of the neuronal promoters Thy1 and VGlut2 in 2 separate strains of mice. (
Electrical stimulation (300 ms pulses, 40 Hz, 50–300 µA) of the dorsal part of the Vtr (Fig 4A) generated calcium responses in 180 cells (Fig 4B, 56 neurons [black empty circles] and 124 astrocytes [gray-filled circles], N = 14) in its vis-à-vis areas of NVsnpr and the different areas of PeriV known to mostly respond to periodontal and spindle afferents inputs. In the NVsnpr, most neuronal and astrocytic responses were concentrated in the dorsal half with only a few cells in the ventral part (Fig 4B–D). Electrical stimulation of the Vtr also generated neuronal calcium responses in NVmt and in the area dorsomedial to it in SupV and ventral to it in PCRt. Very few neurons were responsive in the JuxtV (n = 2), IntV (n = 3), and NVac (n = 1) (Fig 4C and E, black bars). Surprisingly, the distribution of activated astrocytes did not faithfully mirror that of neurons and presented some differences (Fig 4D and E, gray bars). Stimulation of Vtr activated more astrocytes than neurons in most areas of PeriV and dorsal NVsnpr with the highest densities found dorsally in SupV and NVsnpr. In contrast, the opposite was observed for PCRt (Fig 4D and E, gray bars). As illustrated in Figure 4F, 3 types of calcium responses were observed: single transients, repetitive transients, and sustained calcium elevations. The bar graph in Figure 4G illustrates the distribution of these responses across the 7 monitored nuclei for both neurons and astrocytes.
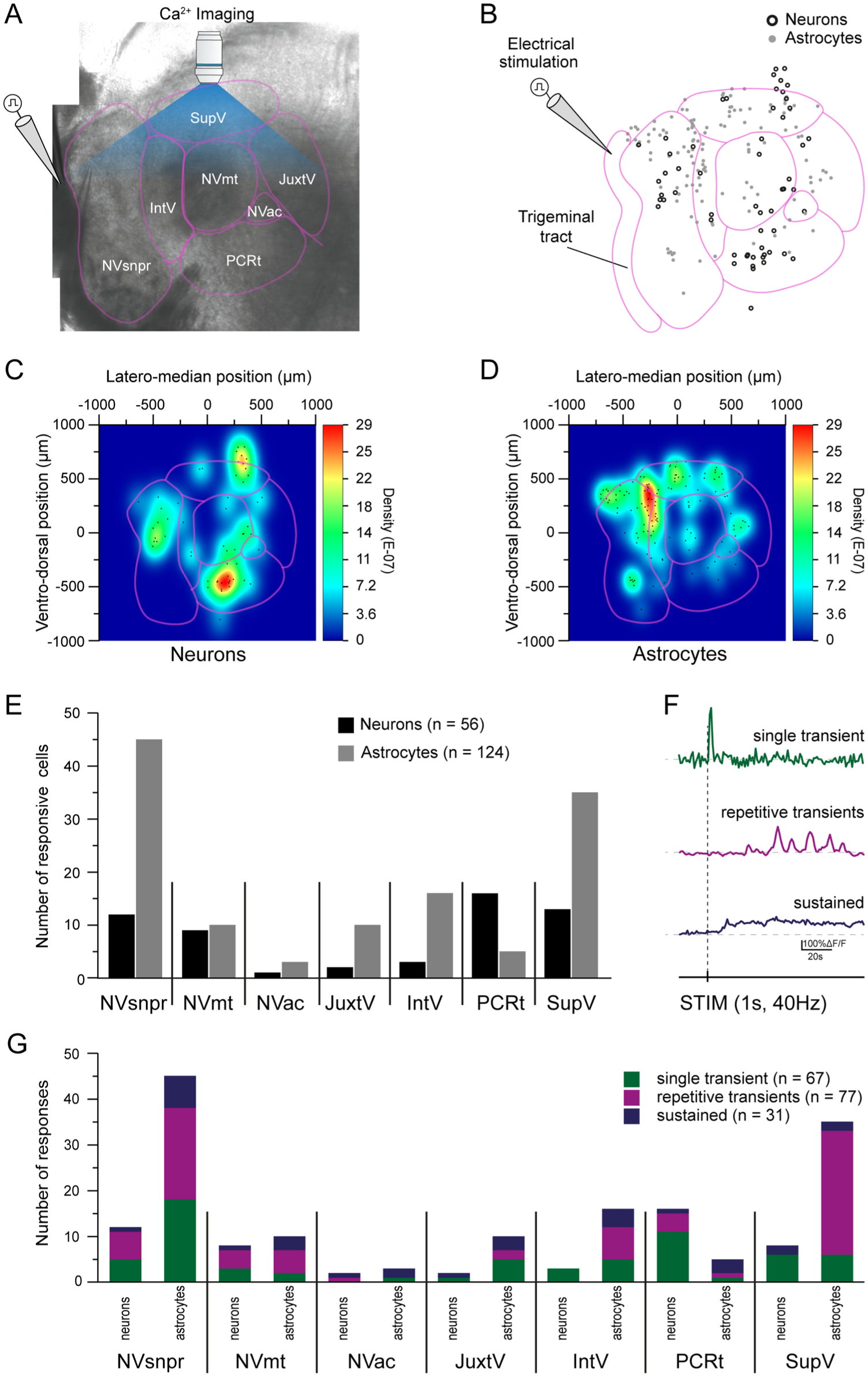
Electrical stimulation of the dorsal part of the trigeminal spinal tract generates responses in both neurons and astrocytes of the NVsnpr and PeriV. (
Since NVsnpr is known to act as a relay of sensory inputs to the thalamus and perhaps other targets, and because our previous work suggests that it may form the rhythmogenic core of the CPG, we examined the distribution of cells activated when rhythmic firing is induced in its neurons by local applications of the Ca2+ chelator, BAPTA (as we have done before in Morquette et al 2015; Slaoui Hasnaoui et al 2020; Ryczko et al 2021; Gaudel et al 2025) (Appendix Fig 2A). BAPTA applications in the dorsal NVsnpr elicited calcium responses in 40 neurons that were mostly concentrated in the PCRt, SupV, and the dorsal part of the NVsnpr (Appendix Fig 2B, black empty circles; Appendix Fig 2C and E, black bars; N = 28). Responsive astrocytes were mostly concentrated at the junction of the NVsnpr, SupV, and IntV and were sparsely distributed in the PCRt, NVmt, and JuxtV (Appendix Fig 2B, gray-filled circles; Appendix Fig 2D and E, gray bars; n = 63, N = 28). No neurons nor astrocytes showed signs of activation in the NVac. The same response patterns observed with the electrical stimulation of the Vtr were again identified in both neurons and astrocytes following local BAPTA application in the dorsal NVsnpr (Appendix Fig 2F). The bar graph in Appendix Figure 2G shows the distribution of these responses in the 7 monitored nuclei for neurons and astrocytes.
These results confirm that electrical stimulation of the Vtr and pharmacologic stimulation of NVsnpr selectively activate distinct populations of neurons and astrocytes across brainstem regions involved in sensorimotor integration. Notably, astrocytic responses were more prominent than neuronal responses in several areas, highlighting their differential spatial activation patterns and potential roles in trigeminal processing.
Discussion
Existence of the CMA in Mice
Optogenetic stimulation of glutamatergic neurons of 2 cortical areas elicited RJMs in head-fixed awake animals. As in rats (Zhang and Sasamoto 1990; Maeda et al 2014), movements evoked by the stimulation of the anterior area, at a similar location to what was found by Mercer Lindsay et al (2019) in the mice, were simpler and occurred at a higher frequency than those observed during natural mastication (as reported by Kobayashi et al 2002). In rats, stimulation of the P-area induces more complex, circular jaw motions with greater jaw opening (Zhang and Sasamoto 1990). Although this was sometimes the case here, further investigations are needed to compare the kinematics of the movements elicited by stimulation of the 2 areas. Although few mice were implanted in the P-area, no regional differences were observed between groups. Previous reports indicate that projections from the A- and P-areas converge in the SupV, IntV, dorsal NVsnpr, and reticular formation (Zhang and Sasamoto 1990), which may account for the observed similarities. A limitation of this study is that c-Fos expression could be influenced by jaw movement, as the animals were not paralyzed.
Comparison of Trigeminal Areas Activated by Cortical and Sensory Stimulations
Motoneuronal pools
Although no attempts were made to distinguish between motoneurons (MNs) or interneurons, which are also found in NVmt (Kolta 1997; McDavid et al 2006), a greater number of activated cells was found in nuclei (NVmt and NVac) containing MNs after either cortical or sensory tract stimulation, in agreement with Slaoui-Hasnaoui et al (2020) in the latter case, although this increase was modest. As previously reported for other MNs (Barajon et al 1992; Athanassiadis et al 2005; Dai et al 2005), the increase of c-Fos staining was modest, perhaps due to the potential absence of biochemical messengers regulating Fos activation as suggested by Dragunow and Faull (1989).
Reticular formation
In the reticular formation, all subdivisions of PeriV showed significant c-Fos elevation following cortically driven RJMs, with the greatest number of c-Fos–stained cells being in JuxtV. In rats, the A-area seems also mostly related to JuxtV (Yoshida et al 2009; Haque et al 2010), which contains jaw-opening pre-MNs (Chang et al 2009) and contributes an inhibitory input to the other subdivisions of PeriV, particularly to the SupV (Bourque and Kolta 2001). PCRt is also strongly activated during CMA stimulation and shares extensive interconnections with SupV as well as with other PeriV areas and NVsnpr and a portion of its glutamatergic neurons project to NVmt and PCRtA (Kajiwara et al 2022), which, as PCRt, contains last-order interneurons to jaw muscles in many species (Fay and Norgren 1997; Huard et al 1999; Kolta et al 2000). However, considering the lack of intrinsic bursting properties in this nucleus (Bourque and Kolta 2001), the rhythmic bursts reported in PCRt during jaw opening suggest that premotor neurons contained in this region relay the rhythm generated by other structures of the masticatory CPG to trigeminal MNs (Nozaki et al 1986). Our results suggest that PCRt may be a site of convergence for cortical and sensory inputs, but causal experiments are needed to confirm its role as an integration hub for mastication.
More caudally, the IRt shows less c-Fos staining than in its rostral part following the cortically induced RJMs. This area, known to be involved in different orofacial behaviors, including mastication (Takatoh et al 2021), contains many masseter and tongue-protruding premotor neurons. While tongue movements were generated during CMA stimulation, we did not observe any tongue protrusion, which probably explains the smaller increase in c-Fos staining observed here.
In contrast, JuxtV and IntV were rarely responsive to electrical stimulation of the Vtr, which instead elicited neuronal calcium responses mostly in PCRt and SupV. In agreement with previous reports that a proportion of SupV neurons have intrinsic rhythmogenic properties (Bourque and Kolta 2001; Hsiao et al 2007; Nonaka et al 2012), we found that about 30% of its neurons responded to electrical stimulation of the Vtr with rhythmic activity, while such a pattern was seen in 75% of SupV neurons, mostly in its medial part, when a rhythm-inducing stimulus (BAPTA injection) was applied to NVsnpr.
Trigeminal sensory nuclear complex
Mercer Lindsay et al (2019) previously identified the oralis and interpolaris subdivisions of the trigeminal spinal nucleus as key components of circuits that drive coordinated orofacial behaviors. However, their investigation did not include much of NVsnpr. Here, we detected more significant increases of activity in NVsnpr than in NVspo after cortical stimulation but surprisingly few neuronal responses after stimulation of the Vtr and local BAPTA applications. This may be due to the low calcium signal characteristic of the GCaMP6f (Gamage et al 2023) or to the possibility that the activity of rhythmic neurons in NVsnpr does not require calcium (Morquette et al 2015).
Astrocyte activation
Astrocytes throughout the brainstem slices showed calcium responses following Vtr stimulation or BAPTA application, confirming their broad responsiveness within the masticatory CPG. The 3 types of Ca2+ responses (sustained, transient, and repetitive) observed may reflect the same types seen in neurons. This finding aligns with our prior work showing that NVsnpr neuronal rhythmicity and bursting are dependent on astrocytic Ca2+ and coupling (Morquette et al 2015; Condamine et al 2018). Despite this, c-fos activation was rare after cortically driven RJMs (data not shown), suggesting a difference from neuronal c-fos activation, which is rarely induced by simple membrane depolarization and more often linked to proliferation, damage, or plasticity in astrocytes (Cruz-Mendoza et al 2022).
Sensory and cortical integration
There seems to be a convergence of sensory and cortical inputs in the PCRt and at the junction of JuxtV and SupV (Fig 5). Those areas contain an important overlap of interneurons directly connected to sensory and cortical inputs. It is difficult to conclude how these different inputs are respectively processed. In the rat, very few rhythmic bursts were recorded in neurons from PeriV, which led us to believe that these premotor areas do not generate masticatory rhythm but may instead relay the rhythmic pattern generated elsewhere or integrate it to other inputs before relaying it to the MNs (Bourque and Kolta 2001). There was a sparse, but still significant, NVsnpr activation, in which intrinsically rhythmic neurons are found. PCRt showed a strong activation in response to sensory and cortical stimulation in our experiments, which agrees with the evidence, in the rat, that this area receives corticobulbar projections as well as projections from jaw muscle spindle afferents in the trigeminal mesencephalic nucleus and from SupV (Shammah-Lagnado et al 1992). Given the high degree of interconnectivity between neurons in PeriV, PCRt, and the TNSC, we presume that cortical inputs as well as rhythm-determining inputs can be rapidly distributed to all neurons in the network.
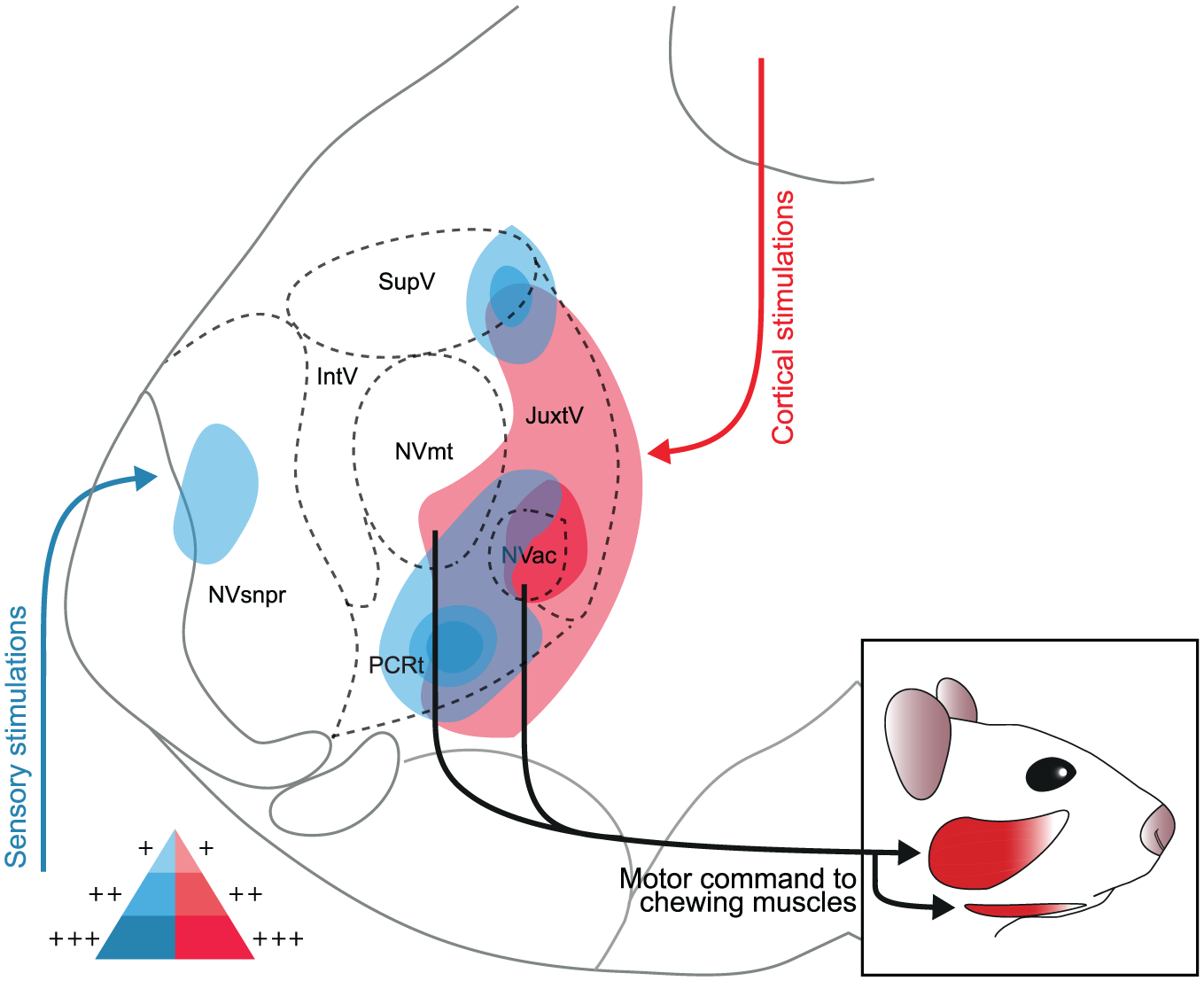
Schematic representation of the areas activated during the stimulation of cortical or sensory inputs. Stimulations of the cortical masticatory area (in red) mainly engaged the JuxtV, PCRt, NVac, and NVmt on both sides of the brainstem, while stimulation of the Vtr (in blue) engaged mainly the NVsnpr, IntV, SupV, and PCRt. The areas with high saturation represent those with the highest cell density. For both types of stimulations, an overlap of activity is identified in the PCRt and at the junction of SupV and JuxtV, which could indicate a convergence of cortical and sensory inputs in these areas.
Conclusion
Mastication is more than just a step in food preparation for digestion and has been linked to important functions such as regulation of memory, vigilance, stress, and anxiety (Sketchley-Kaye et al 2011; Allen and Smith 2012; Fukushima-Nakayama et al 2017), emphasizing the need to better understand the circuitry underlying it.
The results presented here suggest that cortical and sensory stimulation activates distinct neuronal populations with some overlap particularly in the PCRt, which may function as an integration hub, further highlighting the role of this area in orofacial coordination.
Author Contributions
D. Falardeau, O. Y. Sanvi, S. Dubois, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; D. Verdier, contributed to conception and design, data analysis and interpretation, drafted and critically revised the manuscript; A. Kolta, contributed to conception and design, data interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-3-jdr-10.1177_00220345251407574 – Supplemental material for Cortical and Sensory Integration in Brainstem Circuits for Mastication
Supplemental material, sj-docx-3-jdr-10.1177_00220345251407574 for Cortical and Sensory Integration in Brainstem Circuits for Mastication by D. Falardeau, O. Y. Sanvi, S. Dubois, D. Verdier and A. Kolta in Journal of Dental Research
Supplemental Material
sj-docx-4-jdr-10.1177_00220345251407574 – Supplemental material for Cortical and Sensory Integration in Brainstem Circuits for Mastication
Supplemental material, sj-docx-4-jdr-10.1177_00220345251407574 for Cortical and Sensory Integration in Brainstem Circuits for Mastication by D. Falardeau, O. Y. Sanvi, S. Dubois, D. Verdier and A. Kolta in Journal of Dental Research
Supplemental Material
sj-pdf-1-jdr-10.1177_00220345251407574 – Supplemental material for Cortical and Sensory Integration in Brainstem Circuits for Mastication
Supplemental material, sj-pdf-1-jdr-10.1177_00220345251407574 for Cortical and Sensory Integration in Brainstem Circuits for Mastication by D. Falardeau, O. Y. Sanvi, S. Dubois, D. Verdier and A. Kolta in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345251407574 – Supplemental material for Cortical and Sensory Integration in Brainstem Circuits for Mastication
Supplemental material, sj-pdf-2-jdr-10.1177_00220345251407574 for Cortical and Sensory Integration in Brainstem Circuits for Mastication by D. Falardeau, O. Y. Sanvi, S. Dubois, D. Verdier and A. Kolta in Journal of Dental Research
Footnotes
Acknowledgements
We thank Dr. Fanny Gaudel for her contribution to the design of the figure and Yang Tian Lu for conducting 2 immunohistochemistry experiments presented in the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (180522).
Data Availability Statement
All data are available in the main text or the supplementary materials. All data supporting the findings of this study are included in the accompanying dataset file.
A supplemental appendix to this article is available online.
