Abstract
Chronic apical periodontitis (CAP) is a persistent inflammatory condition caused by microbial infections in the root canal system, leading to bone loss and tissue damage. In this study, we tested the hypothesis that maresin 1 (MaR1), a specialized proresolving mediator, facilitates inflammatory resolution and promotes bone healing in CAP. We developed a CAP model in mice through pulp exposure. Animals received intracanal administration of either MaR1 or a vehicle. Micro–computed tomography (micro-CT) was used to analyze lesion size and bone volume changes. Inflammatory cell infiltration was assessed in hematoxylin and eosin–stained sections, and microbial diversity was analyzed using next-generation sequencing. The role of regulatory T cells (Tregs) was further explored through diphtheria toxin–induced depletion of Tregs in Foxp3eGFP/IL17 transgenic mice. All statistical analyses were performed using parametric methods, as confirmed by the Shapiro–Wilk test for data normality. Analysis of variance with Tukey’s post hoc and Bonferroni-corrected t tests was applied. P < 0.05 was considered significant. In 2-dimensional analyses, a significant difference was observed between the control and lesion groups, supporting the validity of the experimental model. MaR1 treatment significantly reduced lesion size (P < 0.0001). The bone volume/total volume ratio was significantly higher in the MaR1 group than in the vehicle group (P < 0.05). Bone mass was reduced in the lesion group, whereas MaR1 treatment significantly alleviated this loss (P < 0.05). The number of inflammatory cells was significantly lower in the MaR1 group compared to the vehicle group (P < 0.05). MaR1 also reduced Enterococcus faecalis, a key pathogen in persistent infections. This study highlights MaR1 as a promising treatment for chronic apical periodontitis, showing benefits in resolving inflammation, preserving bone, and reducing E. faecalis. Unlike conventional therapies, MaR1 supports immune modulation and tissue repair.
Keywords
Introduction
Endodontic infections are characterized by microbial invasion of the dental pulp and surrounding tissues. Despite advancements in root canal therapy, periapical lesions and chronic inflammation continue to pose significant clinical concerns, even after successful microbial control (Artaza et al. 2024). Persistent inflammation may arise when infection is not fully eradicated, or it may persist even after microbial control, if host-mediated inflammation remains unresolved (Henriques et al. 2011).
Specialized proresolving mediators (resolvins, lipoxins, protectins, and maresins) modulate immune responses, facilitate the resolution of inflammation, promote tissue homeostasis, and support healing (Coleman 2010; Hasturk et al. 2015; Kantarci et al. 2018, 2022; Serhan and Levy 2025). Lipoxin A4 and resolvin E1 have been shown to reduce periodontal inflammation and accelerate healing (Hasturk et al. 2007; Kantarci et al. 2015; Van Dyke and Serhan 2003). Maresin 1 (MaR1) is derived from docosahexaenoic acid and biosynthesized by macrophages, exerting potent anti-inflammatory and proresolving actions (Serhan et al. 2009, 2012). MaR1 is biologically and functionally distinct from resolvins due to its macrophage-derived origin, superior ability to enhance efferocytosis, and direct role in supporting tissue repair during the resolution phase (Wang et al. 2020). MaR1 promotes M2 macrophage polarization, limits neutrophil infiltration, induces regulatory T-cell (Treg) expansion, increases anti-inflammatory cytokine production, and suppresses effector T-cell activation (Serhan et al. 2012; Chiurchiù et al. 2016). MaR1 restores stemness and regenerative function in periodontal ligament stem cells under inflammatory stress, thereby enhancing their viability, proliferation, migration, and clonogenicity (Albuquerque-Souza et al. 2020). Thus, MaR1 actively governs resolution biology and accelerates tissue repair (Albuquerque-Souza et al. 2020; Serhan and Levy 2025).
Given the chronic inflammatory nature of chronic apical periodontitis (CAP), we hypothesized that MaR1 promotes inflammation resolution and enhances bone healing primarily through immune modulation. We aimed to evaluate the therapeutic potential of MaR1 in periapical tissues by targeting immune regulation, promoting resolution-driven bone repair, and investigating whether microenvironmental normalization is associated with decreased pathogenic persistence.
Materials and Methods
Animal Model of Periapical Lesion and Diphtheria Toxin–Mediated Treg Depletion
All animal experiments complied with the ARRIVE 2.0 (Animal Research: Reporting of In Vivo Experiments) guidelines and were approved by the Forsyth Institute Animal Care and Use Committee (#24-002). The mouse model of periapical lesions was developed using 8- 12-wk-old male and female mice with equal sex distribution. The researchers conducting treatments, evaluating outcomes, and analyzing data were blinded to the group allocations. Only teeth free from caries, trauma, fractures, or odontogenic anomalies were selected. Systemically healthy animals, assessed by monitoring behavior, activity level, grooming, and weight stability, with no signs of infection in the surrounding gingival tissues, were included. Gingival health was evaluated by intraoral examination under magnification, with no erythema, swelling, or lesions. Two experimental models were employed: the first model involved inducing periapical lesions by exposing the pulp in C57BL/6J mice to elucidate the effects of MaR1 on endodontic infections. To test the role of Treg cells, we used Foxp3eGFP/IL17 transgenic mice. In this strain, Foxp3+ cells (Tregs) express the human diphtheria toxin receptor (DTR), which makes them susceptible to apoptosis upon diphtheria toxin (DT) administration. The transgenic model and DT-mediated depletion protocol have been previously validated for effective and selective Treg ablation in similar immunoinflammatory settings (Alvarez Rivas et al. 2024; Appendix data). Sample size estimation was based on data from Siddiqui et al. (2019), using periapical lesion size as the primary outcome variable. For 2-group comparisons, an effect size of 1.817 was assumed, with a significance level (α) of 0.05 and a statistical power (1 − β) of 80%. Based on these parameters, the minimum required sample size was calculated to be 6 subjects per group. The bilateral maxillary first molars were selected as the experimental sites, and the sample size was defined as the number of teeth. Sample sizes per group were as follows: control (n = 12), lesion (n = 8), MaR1 (n = 20), vehicle (n = 24), DT lesion (n = 8), DT MaR1 (n = 5), and DT vehicle (n = 8). A total of 85 teeth were analyzed across all groups.
Induction of Periapical Lesions
Following intraperitoneal injection of a ketamine/xylazine mixture (87 mg/kg and 10 mg/kg, respectively) and immobilization, pulp exposure was performed on maxillary first molars using a 1/4 round carbide bur (Henry Schein) mounted on a variable-speed electric handpiece (Aseptico). Continuous water irrigation was maintained (flow rate: 50 mL/min) using sterile saline to prevent overheating of the teeth. A surgical microscope (Seiler Revelation) was used to ensure precise pulp exposure (Goldman et al. 2021). Postoperative analgesia was provided via subcutaneous injection of buprenorphine (3.25 mg/kg; Ethiqa XR). To minimize potential confounding, the same single-dose regimen was uniformly administered across all experimental groups; its pharmacologic effect was limited to the immediate postoperative phase. The exposed pulps were left open to the oral environment for 21 d to induce periapical lesions.
Application of Maresin 1 or Vehicle via Root Canals
The root canals were mechanically cleaned to a depth of 1 mm using #6-8 C-type files (Dentsply Maillefer). Irrigation was performed with 0.05 mL of distilled water at a target flow rate of 1 mL/min, and the flow rate was manually standardized using a volume–time calibration method through 34-gauge needles (1.5 mm long) connected to a 1-mL syringe. The operator maintained a consistent flow by timing the passive delivery of 0.05 mL over approximately 3 s, which was precalibrated prior to experimentation. The same operator performed the manual standardization procedure throughout the study to minimize interoperator variability. The root canals were dried with sterile paper points (Henry Schein) for 20 s.
The treatment phase included 4 main experimental groups: MaR1, vehicle, control, and lesion. The control group consisted of untreated healthy teeth, with no pulp exposure or intracanal application. The lesion group underwent pulp exposure but received no treatment. Maresin 1 (10 µM) or vehicle was applied as a single intracanal dose on day 1, immediately after pulp exposure. The lesion was allowed to progress for 21 d, followed by a 4-wk healing period. At the end of the experimental period (day 49), mice were euthanized by exposure to 100% carbon dioxide for 5 min. Death was confirmed by the absence of respiration and heartbeat, followed by decapitation. The MaR1 group received 10 µM MaR1 (Cayman Chemical) via root canal delivery, while the vehicle group received a 10% v/v ethanol in phosphate-buffered saline (PBS) solution using the same delivery method. Root canals were accessed using a 34-gauge needle, and cavities were sealed with resin-modified glass ionomer cement (Ionoseal; VOCO GmbH). Three additional groups were included under the Treg-depletion model using Foxp3eGFP/IL17 transgenic mice—DT lesion, DT MaR1, and DT vehicle—following the same procedures.
Micro–Computed Tomography Analysis
To assess the impact of CAP on the periapical area and measure lesion formation and bone remodeling, the maxillae were scanned using micro–computed tomography (micro-CT) (Scanco Medical uCT40). In the 2-dimensional (2D) analysis, lesion areas within the apical third of the roots were measured in square millimeters using ImageJ software (National Institutes of Health; Appendix Fig. 1). For the 3-dimensional (3D) analysis, Amira 3D software (Thermo Fisher Scientific) was used to calculate the bone volume/total volume (BV/TV) ratio, bone mineral density (BMD), and bone mass within a standardized bounding box (Appendix Fig. 2). A standardized bounding box was applied in the 3D analysis to minimize anatomical variability among specimens.
Histological Analysis and Inflammatory Cell Quantification
Following micro-CT analysis, maxillae were demineralized in 20% EDTA (pH 7.4) for 3 wk at room temperature. Samples were cryoprotected by sequential incubation in 15% and 30% sucrose solutions at +4°C and subsequently embedded in optimal cutting temperature compound (Fisher Healthcare). Cryosections were obtained using a cryostat (Leica CM1850) (10 µm thickness) and stained with hematoxylin and eosin (H&E). Three roots (mesiobuccal, distobuccal, and distopalatal) were examined under 10× and 20× magnification. Inflammatory cell quantification was performed under 20× magnification using a standardized rectangular region. Inflammatory cells were counted from 3 to 4 distinct regions adjacent to the apex. Preliminary calculations revealed no significant variation across regions; therefore, standardized counting from a representative apical region was used. The consistency of this approach was verified by comparing the original values with their means, which showed no change in the overall pattern across groups (Appendix Fig. 3, Appendix Table 1).
Periapical Microbial Profiling
16S next-generation sequencing (16S NGS) was performed to evaluate the microbial composition of periapical endodontic lesions. One sample was collected per group from an independent animal, with no longitudinal sampling; each sample represents an independent biological replicate. Following euthanasia, first molar teeth were extracted, and periapical lesions were sampled using sterile paper points. Samples were stored at –80°C until DNA extraction. Paper point tips were eluted into 150 μL PBS, and DNA was purified using the MasterPure Complete DNA Purification Kit (Epicentre) according to the manufacturer’s protocol. DNA concentration and purity were determined using a NanoDrop 2000 spectrophotometer (Thermo Scientific), and the samples were stored at –80°C until sequencing.
Statistical Analysis
All statistical analyses were performed using parametric methods, as data normality was confirmed using the Shapiro–Wilk test. For comparisons involving more than 2 groups, 1-way analysis of variance was performed to evaluate overall differences among the groups. Tukey’s multiple-comparison test was used as a post hoc analysis to identify pairwise group differences while controlling for type I error across comparisons. For prespecified comparisons between 2 independent groups of interest within the same data set, unpaired 2-sample t tests were used, with the Bonferroni correction applied to adjust for multiple testing. All statistical tests were 2-tailed, and a corrected P value of less than 0.05 was considered statistically significant.
Results
Effect of Maresin 1 in Chronic Apical Periodontitis
The experimental design for the CAP model is shown in (Fig. 1A). The 2D analysis revealed a statistically significant difference between the control group (n = 12) and the lesion group (n = 8), confirming the reliability of the experimental model. Representative 2D and 3D images for the treatment groups are shown in Figure 1B. Lesion sizes in the MaR1 group (n = 20) were significantly smaller compared to the vehicle group (n = 24) (P < 0.0001) (Fig. 1C). The 3D analysis results were consistent with the 2D findings. The BV/TV ratio in the MaR1 group was significantly higher compared to the vehicle group (P < 0.05), indicating reduced bone loss (Fig. 1D).
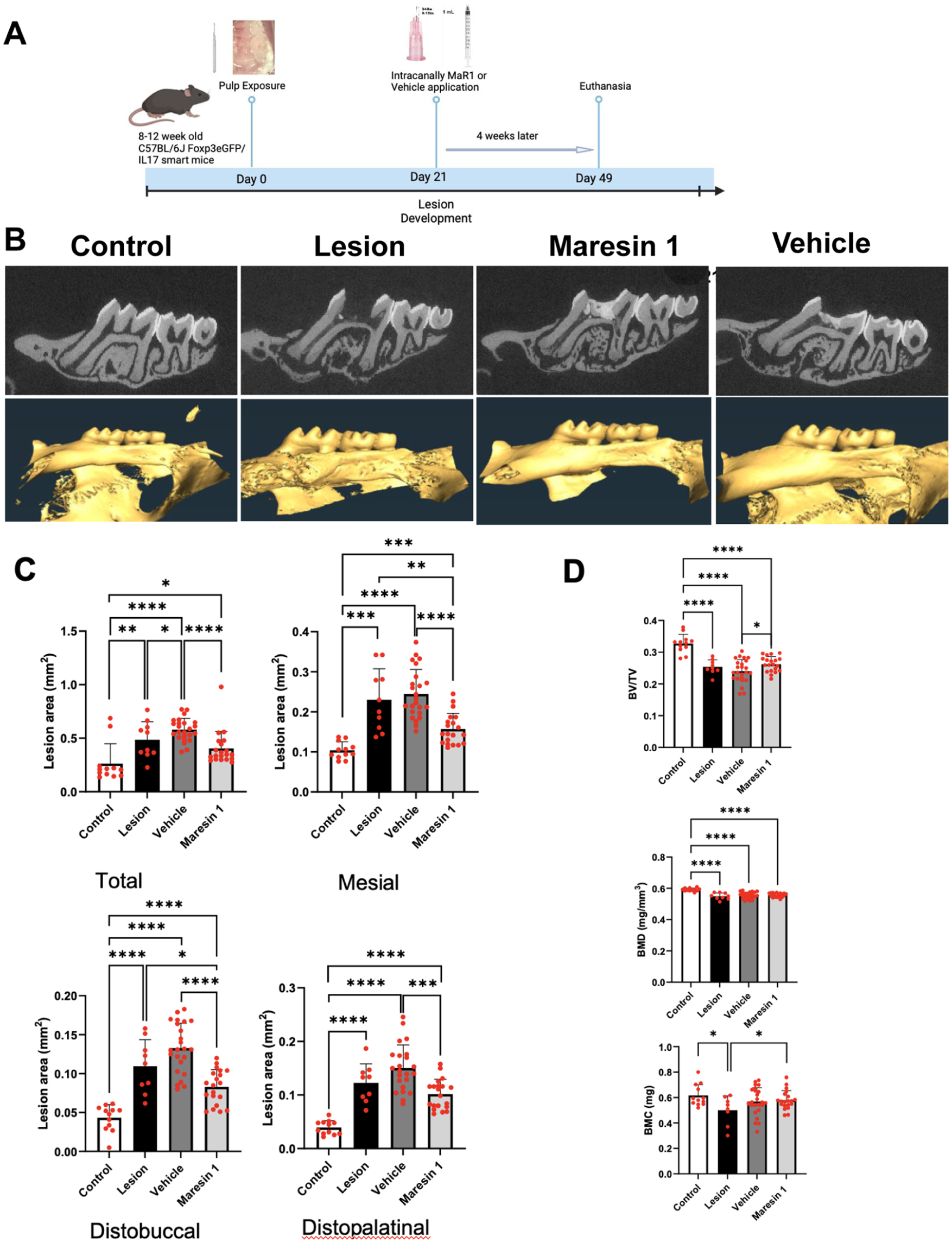
Micro-CT assessment of lesion dimensions and bone structural parameters following Maresin-1 treatment in the chronic apical periodontitis model. (
In BMD measurements, a statistically significant difference was observed between the control group and all other groups (P < 0.0001). In the bone mass analysis, the lesion group showed a significant reduction compared to the control group (P < 0.05). MaR1 treatment resulted in a significant increase in bone mass compared with the lesion group (P < 0.05), indicating its protective effect against bone loss and suggesting that MaR1 reduces lesion size and preserves bone integrity in CAP. In parallel, inflammatory cell counts were significantly lower in the MaR1 group than in the vehicle group (P < 0.05), indicating reduced cellular infiltration in the apical region (Fig. 2).
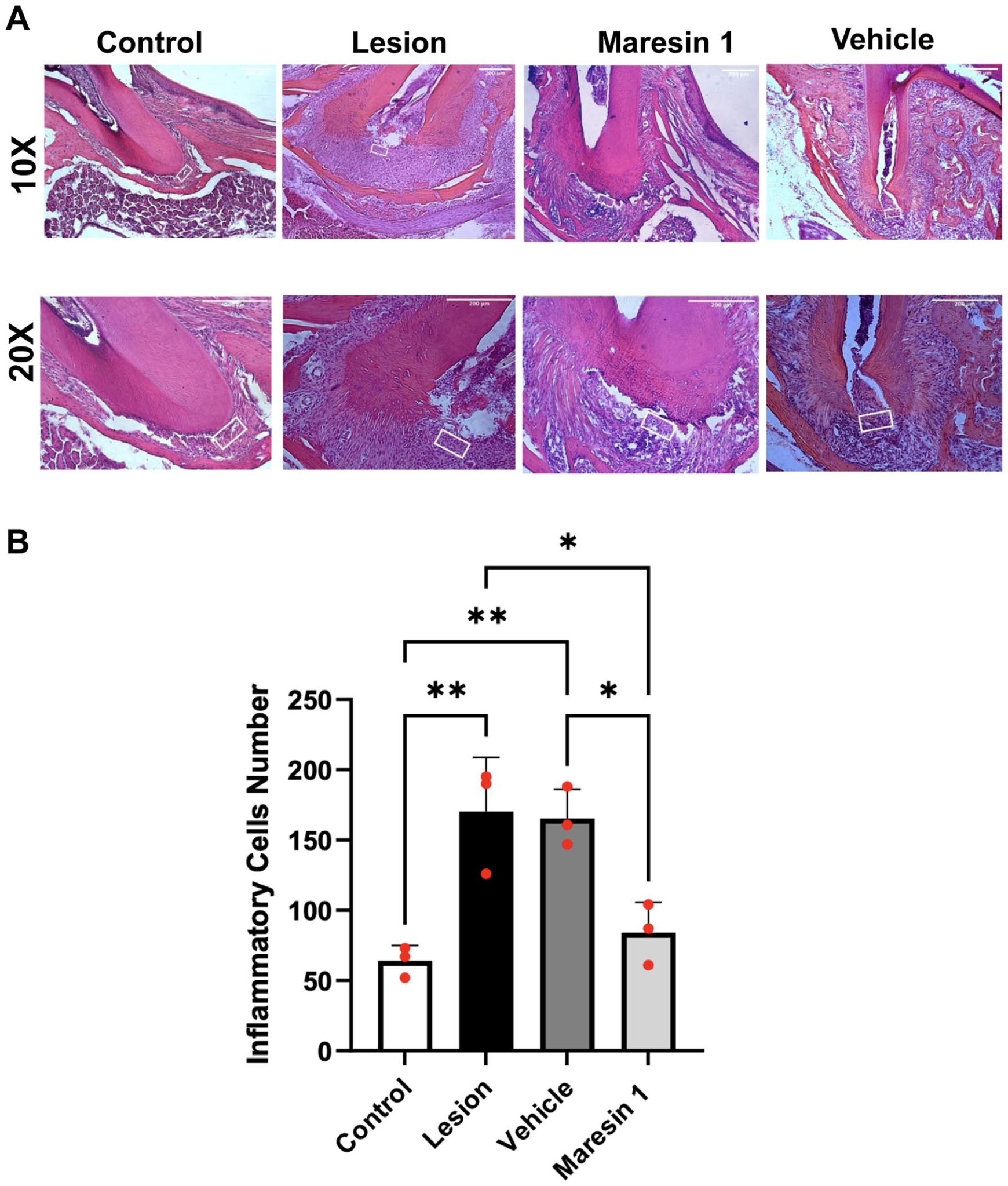
Histological assessment and quantitative measurement of inflammatory cell infiltration in the chronic apical periodontitis model. (
Role of Tregs in Chronic Apical Periodontitis and Inflammatory Resolution
The roles of Tregs in CAP and its inflammatory resolution are shown in Figure 3A. Representative 2D and 3D images for the treatment groups are shown in Figure 3B. The 2D analysis showed that the DT group (n = 8) had a smaller lesion area than the non-DT group, and the difference was statistically significant (P < 0.05). On the other hand, the 3D analysis demonstrated significantly higher BV/TV ratios in the DT lesion group (P < 0.01), indicating reduced bone loss compared to the non-DT lesion group (Fig. 3C, D). While no statistically significant differences were observed in 2D analyses between DT-treated and non-DT groups, MaR1-treated groups displayed smaller lesion sizes than their vehicle-treated counterparts (non-DT, P < 0.0001; DT, P < 0.01) (Fig. 3C). BV/TV ratios in non-DT groups (MaR1 and vehicle) were significantly higher compared to their DT-treated counterparts (P < 0.05) (DT MaR1, n = 5; DT vehicle, n = 8). BMD analysis indicated that vehicle-treated animals without DT had significantly higher BMD values than the DT vehicle groups (P < 0.05). The MaR1 group displayed higher BMD values than the vehicle group (Fig. 3D). MaR1 treatment also increased bone mass compared with the DT-treated groups, but the magnitude of the impact was lower than in the DT animals, suggesting a limited effect of MaR1 in the absence of Tregs (Fig. 3D).
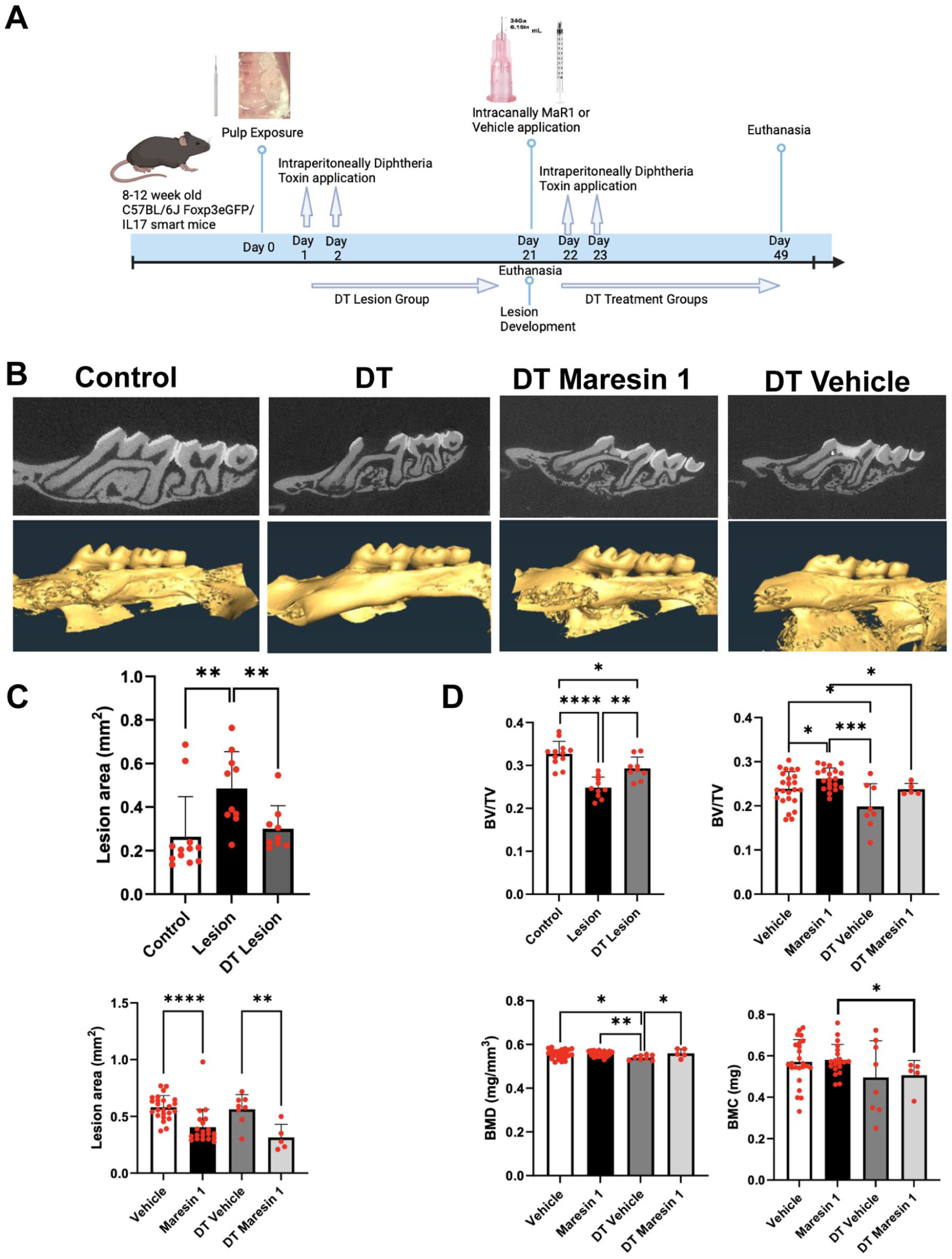
Effect of Treg depletion and Maresin-1 treatment on lesion size and bone parameters in the chronic apical periodontitis model. (
Histological analysis revealed that the number of inflammatory cells in DT-treated lesion groups was lower than in non-DT lesion groups, although this difference was not statistically significant (Fig. 4). Inflammatory cell counts were reduced in the MaR1-treated group compared to the vehicle-treated group (P < 0.01), further confirming its anti-inflammatory effects.
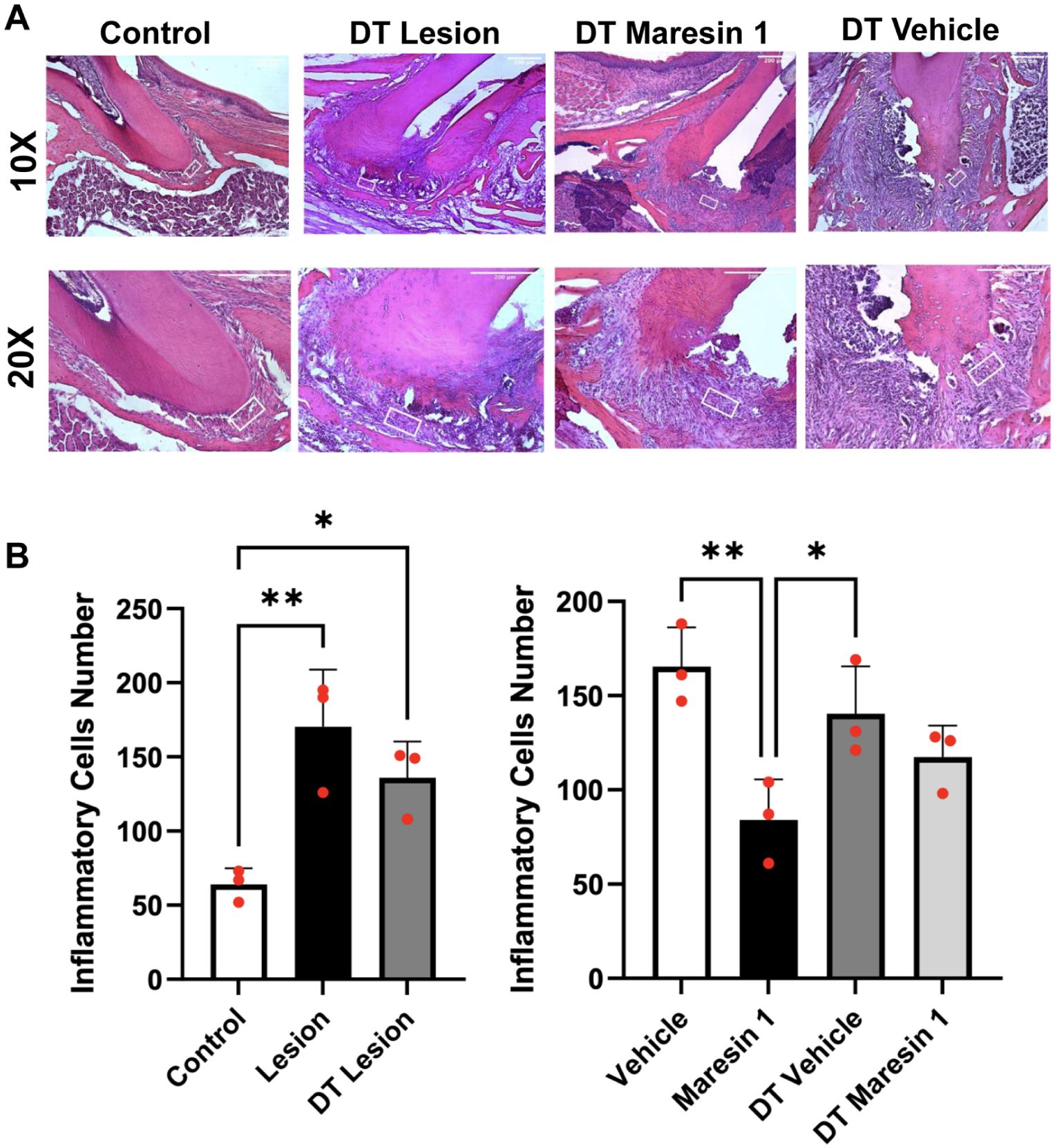
Histological assessment and quantitative analysis of inflammatory cell infiltration in Treg-depleted and non–Treg-depleted chronic apical periodontitis groups. (
Microbial Composition of Periapical Lesions
The 16S NGS analysis revealed that MaR1 treatment reduced the relative abundance of E. faecalis by 54% and 49% in the DT-treated and non-DT-treated groups, respectively, compared with the vehicle. The reduction in Enterococcus faecalis was more pronounced in DT-treated animals, suggesting that Tregs may contribute to its persistence in CAP (Fig. 5A).
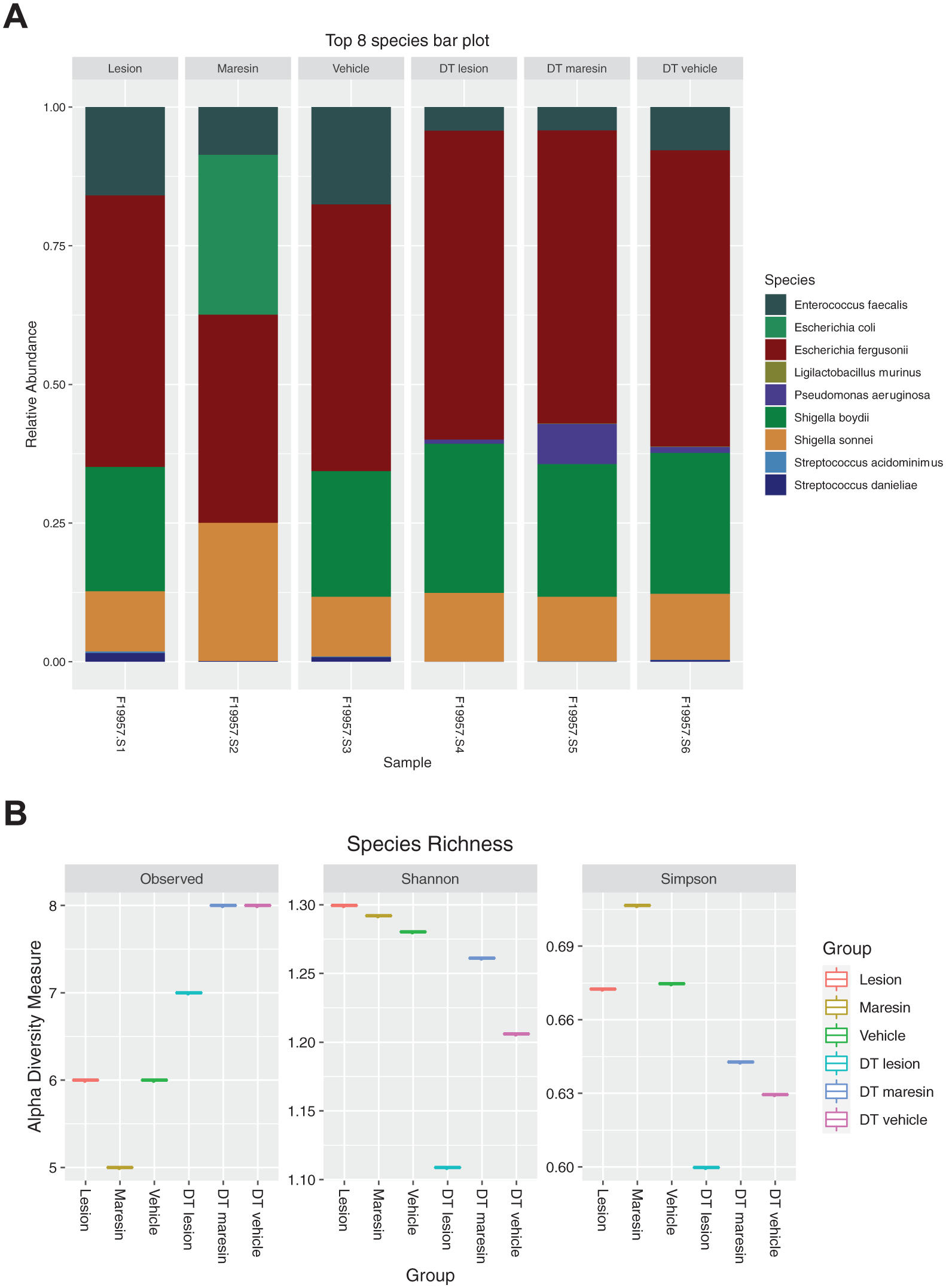
Microbial composition and alpha diversity analysis demonstrating the impact of Treg depletion and Maresin-1 treatment on periapical microbiome structure. (
Discussion
Chronic apical periodontitis is a persistent inflammatory condition that results from the progression of pulp infections into the periapical tissues. Rodent models have been widely employed to investigate their pathophysiology and to evaluate potential therapeutic interventions (Goldman et al. 2021; Mohaved et al. 2020; Nair 2004; Stashenko et al. 1998). The present study underscored the critical importance of resolving inflammation in CAP management, emphasizing the potential of MaR1 as a novel therapeutic agent for promoting tissue healing and microbial balance.
The observed reduction in lesion size and bone loss in MaR1-treated groups indicated that MaR1 actively facilitated inflammatory resolution and bone regeneration. Unlike conventional anti-inflammatory agents, which primarily suppress immune responses, MaR1 promotes tissue repair by modulating immune responses to resolve chronic inflammation while supporting healing. MaR1 exerts its potent actions through the G protein–coupled receptor LGR6, which is expressed on human neutrophils (PMNs), monocytes, and macrophages (Chiang et al. 2019). These findings were consistent with prior research on specialized pro-resolving mediators (SPMs), which have demonstrated dual anti-inflammatory and proresolving properties in periodontal models in response to lipoxins and resolvins (Hasturk et al. 2007; Kantarci et al. 2015; Van Dyke and Serhan 2003).
The immune system plays a central role in managing endodontic infections by facilitating the clearance of pathogens while preserving the structural integrity of host tissues. A dysregulated immune response can lead to irreversible damage in pulp and periapical tissues, contributing to the chronic nature of the condition (Rotstein and Ingle 2019). Regulatory Tregs play a vital role in maintaining immune homeostasis by suppressing excessive inflammatory responses within the immune system. The transcription factor FoxP3 is crucial for the differentiation and function of Tregs, serving as a key regulator of immune responses across various inflammatory diseases (Campos et al. 2015; O’Garra and Vieira 2004).
Our study demonstrated the role of Tregs in modulating periapical inflammation. The depletion of Tregs through DT administration resulted in smaller lesion sizes and altered inflammatory responses, suggesting that Tregs play a crucial role in immune regulation and tissue healing in CAP.
Although direct assessment of Th17 activity was not conducted in this study, previous findings suggested that Treg depletion enhanced Th17 responses, contributing to more effective microbial clearance and reduced chronic lesion formation (Noack and Miossec 2014). These differential outcomes between the Treg-depleted and nondepleted groups highlighted the critical role of immune regulation in tissue healing and underscored the therapeutic efficacy of MaR1 across different immunological contexts.
MaR1 significantly reduced inflammatory cell infiltration in periapical lesions, suggesting its direct role in controlling excessive immune responses. This finding is particularly relevant for chronic endodontic infections, where uncontrolled inflammation can lead to irreversible tissue damage. By promoting the expansion of Tregs and reducing effector T-cell activation, MaR1 supported immune homeostasis, thereby mitigating the risk of chronic inflammation (Campos et al. 2015; Chiurchiù et al. 2016). We previously reported that Tregs underwent phenotypic changes and exhibited increased IL-17 expression during periodontitis; resolvin E1 (RvE1) restored Treg frequency to healthy levels and reestablished the Treg/effector T-cell balance (Alvarez et al. 2021). In our model, Treg suppression reduced lesion size, and MaR1 restored this balance.
The primary aim of this study was to evaluate the overall inflammatory burden and tissue response. We used H&E-stained sections, which remain a widely accepted first-line method in experimental periapical models to assess the extent and distribution of inflammation (Goldman et al. 2019; Aubeux et al. 2021). This approach provided a reliable basis for comparing treatment outcomes across groups, particularly in studies focusing on global inflammatory resolution.
MaR1 treatment reduced the relative abundance of E. faecalis, a key pathogen associated with persistent endodontic infections, by 54% and 49% in the DT-treated and non-DT-treated groups, respectively, compared to the vehicle. However, this reduction should not be interpreted as evidence of direct bactericidal activity, as no in vitro or mechanistic antimicrobial assays were performed in the present study. Instead, the observed microbial shift is more likely secondary to immune-mediated resolution and restoration of a less inflammatory microenvironment, which may reduce ecological support for E. faecalis persistence (Wu 2023). Consistent with this interpretation, the reduction in E. faecalis was accompanied by a proportional increase in less pathogenic commensal taxa (Fig. 5B), suggesting microenvironmental normalization rather than direct antimicrobial suppression. Future studies involving direct testing of MaR1 against E. faecalis in vitro or in vivo will be needed to determine whether MaR1 exerts any primary antimicrobial effect beyond its established proresolving functions.
In addition to resolving inflammation, MaR1 has been shown to restore the regenerative properties of periodontal ligament stem cells under inflammatory conditions, suggesting potential applicability in regenerative endodontic procedures (Albuquerque-Souza et al. 2020).
Although this study demonstrated a reduction in inflammation and improvement in periapical healing following MaR1 administration, direct assessment of bone remodeling activities, such as osteoclast or osteoblast function, was not performed. Future studies utilizing techniques such as tartrate-resistant acid phosphatase staining or osteogenic marker analysis are needed to elucidate the regenerative effects of MaR1.
The 16S NGS analysis indicated that DT treatment increased E. faecalis levels. DT is known to suppress Tregs, leading to immunosuppression and potentially increasing the risk of infections. However, studies indicate that Tregs may have varying effects depending on the pathogen. For E. faecalis, Tregs may both promote and suppress its colonization. Our findings showed that E. faecalis failed to colonize after MaR1 treatment, suggesting that MaR1 limits E. faecalis survival. Components of E. faecalis may also promote Treg development. For example, PEF7121, obtained from E. faecalis cell wall extract, induced IL-10 and inhibited macrophage and T-cell functions, promoting Treg development (Sparo et al. 2014). These data suggested that E. faecalis and Treg cells may have a mutual relationship, in which E. faecalis can contribute to Treg development, facilitating its colonization in the absence of active inflammation. Additionally, recent studies have shown that RvE1 promotes pulp regeneration and modulates inflammation in dental models, supporting the broader therapeutic potential of SPMs in endodontic applications (Wu et al. 2023).
Conclusion
These findings suggested that MaR1 is a promising therapeutic agent for managing CAP by promoting inflammation resolution, supporting bone preservation, and contributing to pathogen control, including reductions in E. faecalis levels. In contrast to traditional therapies that focus solely on eliminating infection, MaR1 actively modulates the immune response and promotes tissue repair.
Author Contributions
E. Demir, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; I. İlgenli, contributed to conception and design, data interpretation, drafted and critically revised the manuscript; Z. Guney, contributed to acquisition, analysis, and interpretation, critically revised the manuscript; S. Mohammadi, K. Lauwens, N. Yakar, contributed to acquisition, and interpretation, critically revised the manuscript; C. Alvarez Rivas, contributed to conception and design, data acquisition, and interpretation, critically revised the manuscript; O. Unlu, contributed to conception and design, critically revised the manuscript; A. Kantarci, contributed to conception and design, data interpretation, critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251399558 – Supplemental material for Maresin 1 Resolves Inflammation and Aids Bone Healing in Periapical Lesions
Supplemental material, sj-docx-1-jdr-10.1177_00220345251399558 for Maresin 1 Resolves Inflammation and Aids Bone Healing in Periapical Lesions by E. Demir, I. İlgenli, Z. Guney, S. Mohammadi, K. Lauwens, N. Yakar, C. Alvarez Rivas, O. Unlu and A. Kantarci in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institute for Health Research (RF1AG062496) and the Scientific and Technological Research Council of Turkiye (TUBITAK BIDEB 2214-1059B142300611). This research was conducted as part of E. Demir’s endodontic specialization thesis.
Data Availability
The data generated in this study, including full-resolution micro-CT and histological image files, are not deposited in a public database. Deidentified summary data supporting the findings of this study are included within the article and the Appendix. No additional publicly shareable data are available.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
