Abstract
Mandibular retrognathia is a common craniofacial anomaly characterized by a posteriorly positioned mandible, which can affect a person’s oral function, esthetics, and quality of life. While facial characteristics are widely acknowledged to have a hereditary component, the etiology of this skeletal anomaly is poorly understood. This study aims to identify genetic loci associated with mandibular retrognathia and help elucidate its causes. Using the FinnGen cohort (2,647 cases, 497,020 controls), a genome-wide association study identified 2 regions of genome-wide significance with lead single-nucleotide polymorphisms rs227727 at NOG locus and rs7225448 at SOX9 locus. Interestingly, both associations were driven by results from females. The rs227727 association was replicated in an independent cohort from Iceland. Functional annotation revealed that rs227727 disrupts an enhancer regulating NOG, a gene important for normal bone and cartilage development and previously implicated in craniofacial anomalies. Morphological analyses of Nog-/- mice revealed mandibular retrognathia-like features, including reduced mandibular length and condylar width. Variants in linkage disequilibrium with rs7225448 were located within distal enhancers near SOX9, a transcription factor essential for cartilage development. SOX9 mutations are known to cause Robin sequence with its constituent severe mandibular retrognathia. In addition, regional analysis revealed variation in the prevalence of mandibular retrognathia across Finland’s 18 administrative regions, which was correlated especially with the rs227727 risk allele regional prevalence. These findings highlight genetic contributors to mandibular retrognathia and emphasize the importance of long-range regulatory elements in craniofacial development. By integrating genetic, functional, and epidemiological data, this study enhances our understanding of the genetic architecture underlying craniofacial anomalies.
Introduction
Craniofacial development is coordinated by an intricate series of events that require fine-tuned synergistic actions in the growth of the craniofacial complex to accomplish proper function as well as a unique facial appearance. Facial characteristics are strongly determined by genetic factors, and several genetic loci have been associated with certain facial traits such as nose and eye shape in recent genome-wide association studies (GWASs) (Cha et al 2018; Xiong et al 2019). Mandibular retrognathia, characterized by a posteriorly positioned mandible relative to the maxilla, is a common craniofacial anomaly with significant implications for esthetics, function, and quality of life.
The prevalence of malocclusion traits varies widely between countries due to genetic predisposition and specific environmental factors (Moreno Uribe and Miller 2015). Mandibular retrognathia can result from small size and/or abnormal growth of the mandible, often leading to Class II occlusion, in which the mandibular first permanent molar tooth is positioned distally compared with the maxillary first permanent molar (Angle 1899). The prevalence of Class II occlusion varies among different populations, and in Finland, the reported prevalence ranges from 12% to 19% (Myllärniemi 1970; Kerosuo et al 1991; Lombardo et al 2020). Women are more often observed to have Class II occlusion and increased overjet compared with men in Sweden and Finland (Ingervall et al 1978; Mohlin 1982; Krooks et al 2016). Conversely, men are more frequently observed to have Class III malocclusion and anterior crossbite than women are (Salonen et al 1992).
GWASs have identified hundreds of variants associated with craniofacial traits, many of which likely influence regulatory element activity and gene expression levels (Cha et al 2018; Xiong et al 2019; White et al 2021). Various genes, genetic factors, and signaling pathways that take part in mandibular development and disorders have been studied extensively in laboratory animal models (Manocha et al 2019). However, to our knowledge, no large-scale GWASs have been performed for the search for genetic risk factors that specifically predispose to mandibular retrognathia.
Our GWAS aimed at identifying genetic risk factors that contribute to mandibular retrognathia in a large sample in the Finnish population using the FinnGen study. We report novel associations between mandibular retrognathia and variants located in enhancers near the NOG (Noggin) and SOX9 (SRY-Box Transcription Factor 9) genes. The lead variant near NOG is a common, noncoding variant that alters the function of an enhancer (Leslie et al 2015). Using Nog-/- mice, we find evidence that deregulation of NOG may lead to changes in the morphology of the mandible and condyles. The associated region within the SOX9 locus resides 735 kb upstream of SOX9 but downstream of the enhancers identified in Robin sequence, a developmental condition with the central triad of cleft palate, glossoptosis, and small retrognathic mandible (Gordon et al 2009).
Subjects and Methods
Phenotype Definition in the FinnGen Cohort
FinnGen (https://www.finngen.fi) is a large biobank initiative in Finland, which combines the genome data of 500,000 Finns with health registry data (Kurki et al 2023). Demographic and clinical data are derived from nationwide electronic health registers. The study included 500,348 participants from FinnGen Data Freeze 12, from which 282,064 were females and 218,284 were males. All participants had a Finnish genetic ancestry determined by a principal component analysis of their genotypes. Ethnic Finns were defined as individuals whose relative probability of being part of the FIN cluster was >95% (Kurki et al 2023).
Cases with mandibular retrognathia were defined from the electronic health records in primary and hospital care, using diagnosis code K07.13 of the Finnish version of ICD-10, available since 1996. Mandibular retrognathia is typically diagnosed in Finland after thorough clinical and radiographic examination by a specialist orthodontist. Cephalometric analysis is performed using lateral cephalograms. The SNB (sella-nasion-B-point) angle, which reflects the anteroposterior position of the mandible relative to the cranial base; the SNA (sella-nasion-A-point) angle, indicating the position of the maxilla relative to the cranial base; the ANB angle (SNA-SNB); and the lengths of the maxilla and mandible are used to support the diagnosis of mandibular retrognathia. After exclusion of cases with orofacial clefts (ICD-10 codes Q35, Q36, and Q79), the cohort consisted of 2,647 cases (1,854 females, 793 males) and 497,020 controls (279,952 females, 217,335 males). The median age at first event was 12.4 y for females and 11.6 y for males. Cases that had undergone bilateral sagittal split ramus osteotomy for the correction of retrognathic mandible were identified using the operational code EDC10 (959 cases, 1,688 controls).
Phenotype definition in the Icelandic cohort is described in the Appendix Methods. Genotyping, quality check, and imputation as well as genetic and bioinformatic analysis, analysis of Nog-/- mandibles, and ethics are described in the Appendix Methods.
Results
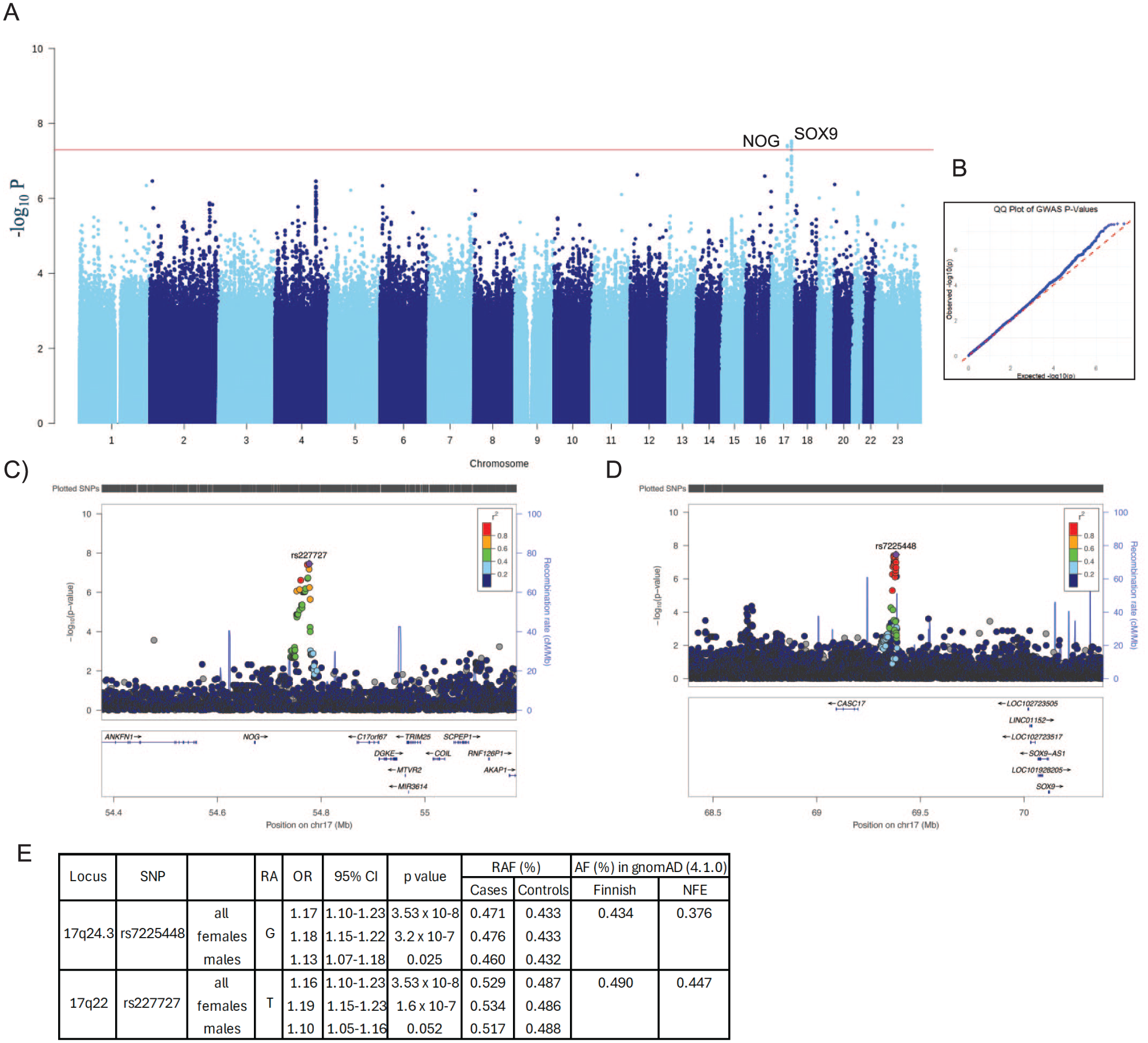
Two Loci Near NOG and SOX9 Are Associated with Mandibular Retrognathia
We conducted a GWAS of mandibular retrognathia in the Finnish population using the FinnGen study with 2,647 cases with mandibular retrognathia and 497,020 ethnicity-matched population controls (FinnGen Data Freeze 12). We identified 2 regions with genome-wide significant (P < 5 × 10−8) associations with mandibular retrognathia. Both loci are located in the long arm of chromosome 17 (Fig. 1). The region 734 kb upstream of SOX9 with lead single-nucleotide polymorphism (SNP) rs7225448 (17-71385926-A-G, P = 3.5 × 10−8, odds ratio [OR] = 1.16) consists of 4 significant and a credible set of 16 variants in high linkage disequilibrium (LD) spanning 16 kb (Appendix Table 1). The other region with lead SNP rs227727 (17-56699594-A-T, P = 3.5 × 10−8, OR = 1.16) is located 80 kb downstream of the NOG gene and consists of 2 significant and 15 credible variants spanning 25 kb (Fig. 1). To assess the robustness of these findings, we reanalyzed the data using the SAIGE method (Zhou et al 2018), which is well-suited for handling case–control imbalance and sample relatedness. The results obtained from the SAIGE analysis were consistent with those from Regenie, including the lead SNPs (rs227727, rs7225448), effect sizes (0.155, 0.155), standard errors (0.028, 0.029), and P values (4.15e-8, 5.79e-8), respectively. These findings support the overall validity of the association signals, although minor differences in significance levels were observed as expected when using a different method.
(
Population attributable risks for these associations were estimated as 13.8% for rs7225448 and 15.1% for rs227727 and altogether as 26.8%, suggesting a remarkable contribution to mandibular retrognathia on the population level.
When the cohorts were divided into subgroups by gender, females showed a more significant association and higher effect size than males did at both SOX9 (rs7225448: females P = 3.2 × 10−7, OR = 1.18; males, P = 0.025, OR = 1.13) and NOG loci (rs227727: females P = 1.6e-7, OR = 1.19; males P = 0.052, OR = 1.10, RAF cases = 0.517, RAF controls = 0.488) (Fig. 1E and Appendix Fig. S1). The difference between risk allele frequencies (RAFs) in cases and controls was larger for females (0.043 with rs227727 and 0.047 with rs7225448) than for males (0.025 with rs227727 and 0.022 with rs7225448), even though, as expected, the RAFs for all females and all males were equal.
To test whether the loci identified represented a correlation to phenotype severity, we further divided the retrognathia cohort into subtypes of cases that had undergone mandibular sagittal split osteotomy (959 cases) and those that had not (1,688 controls) and performed a GWAS with these cohorts (Appendix Fig. S2). The variants rs227727 and rs7225448 did not show association in this GWAS (rs227727: P = 0.675, OR = 1.026; rs7225448: P = 0.138, OR = 1.097), even though the RAFs were increased among the surgical cases as compared with the mandibular retrognathia cases only. However, the lack of association in the surgical subgroup analysis may reflect limited statistical power due to the smaller cohort sizes rather than a true absence of effect.
Mandibular retrognathia showed statistically significant positive genetic correlations with several malocclusion diagnoses such as deep bite, distal bite, and overjet as well as with temporomandibular joint disorders (Fig. 2). Mandibular retrognathia showed negative correlation with crossbite and mandibular prognathia, as expected (Fig. 2). Mandibular retrognathia can be caused by condylar resorption, which can be associated with anterior open bite. Despite this, no significant genetic correlation was observed between mandibular retrognathia and open bite (P = 0.141).
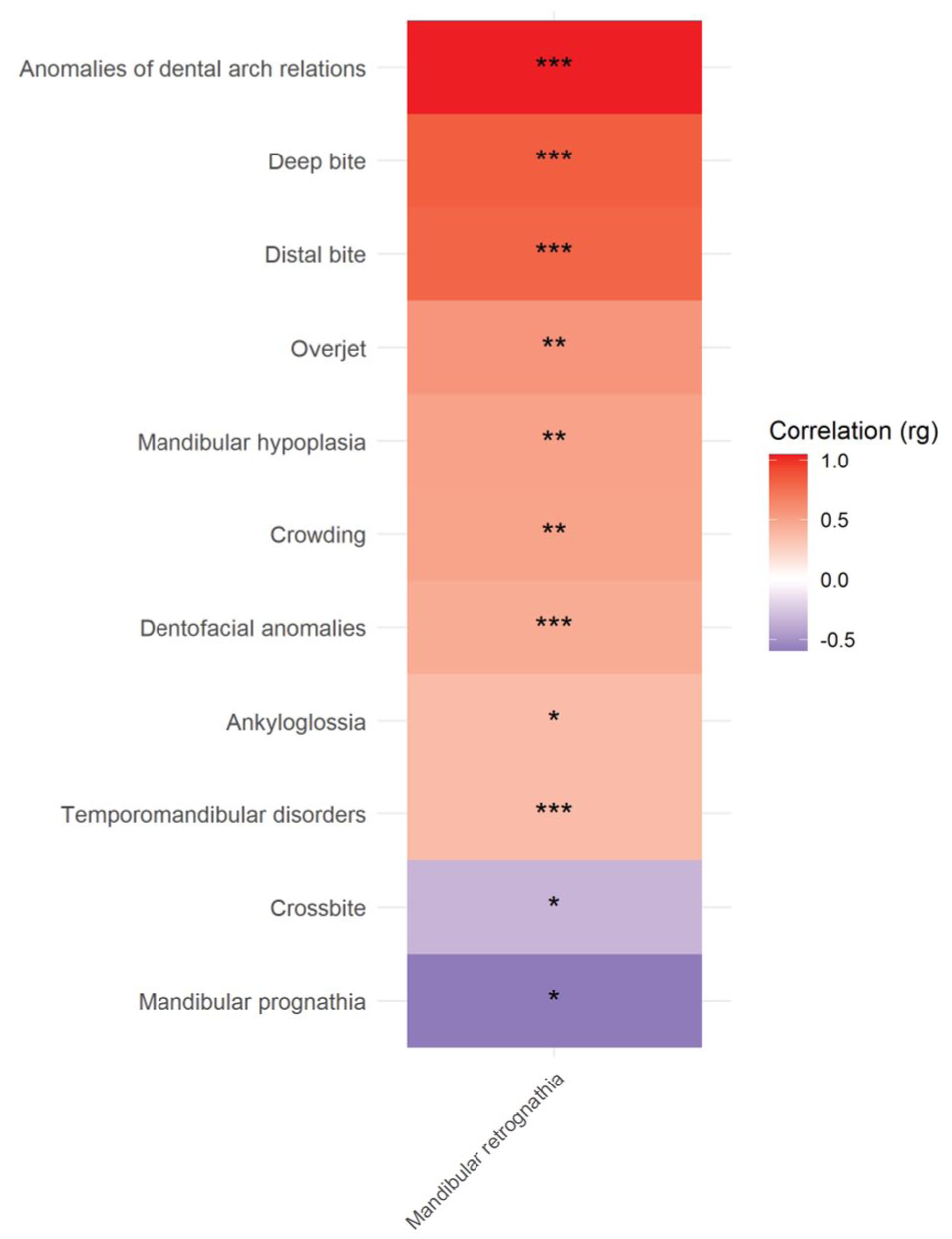
Genetic correlations (rg) between craniofacial and dental-related phenotypes and mandibular retrognathia analyzed using LD score regression (LDSC). Positive correlations (red) indicate shared genetic architecture, while negative correlations (blue) suggest inverse relationships.
Phenome-wide association study with rs227727 showed statistically significant association with height, and several types of arthroses in addition to mandibular retrognathia as discovered in the current study (Appendix Figs. S3 and S4). These findings highlight the shared genetic architecture between mandibular retrognathia, dental phenotypes, and developmental phenotypes such as height.
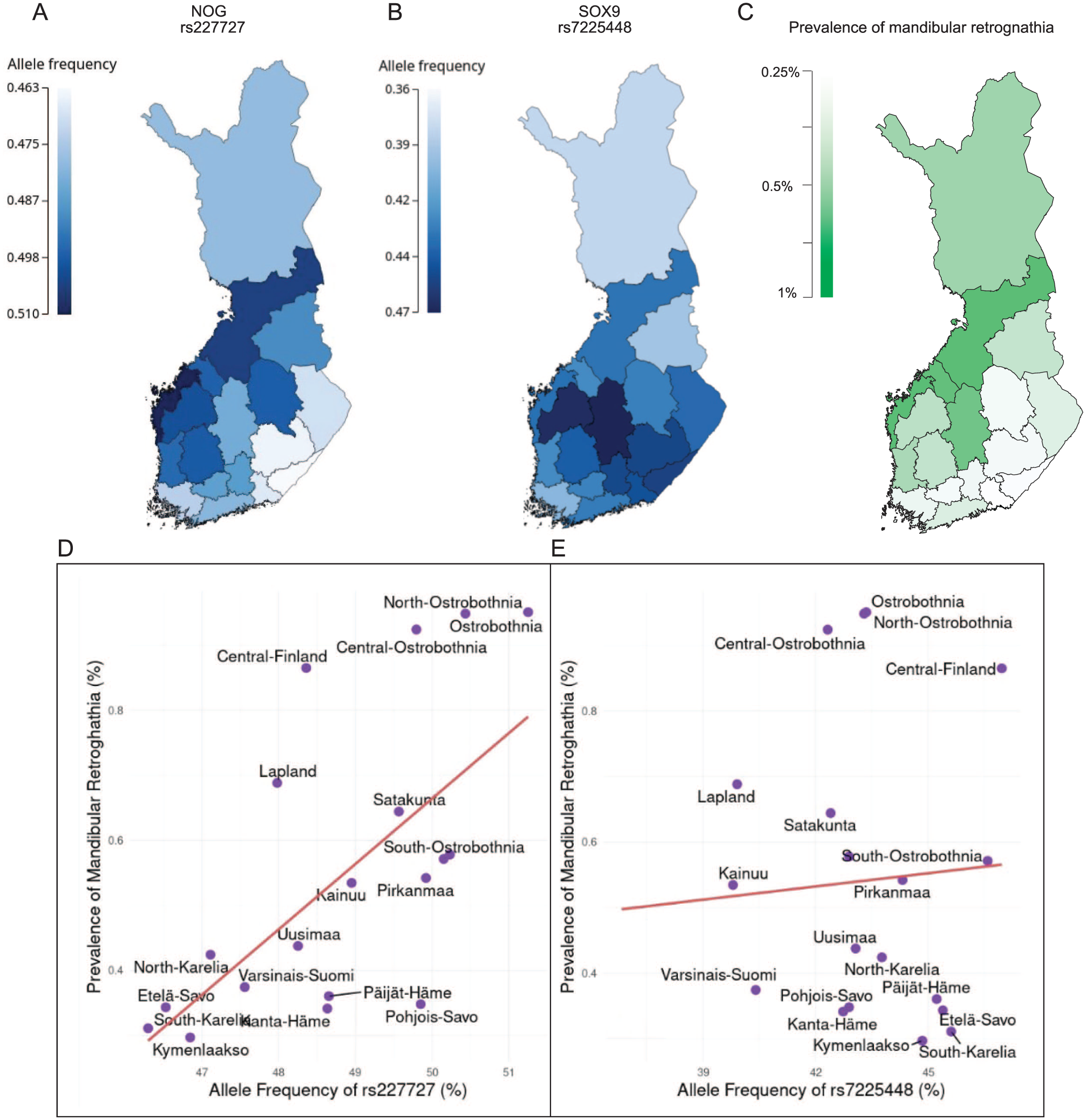
Regional Correlation of Allele Frequencies and Mandibular Retrognathia Prevalence
To test whether the distribution of the risk alleles co-localized with the prevalence of mandibular retrognathia across Finland, we estimated the distribution of allele frequency using region-level birthplace registry data of FinnGen study subjects. The RAF of rs227727 ranges from 0.463 to 0.510 in Finland, and it was higher in the western regions of the country (Fig. 3A), correlating (Pearson’s r = 0.619, P = 0.00361) with a high prevalence of mandibular retrognathia (Fig. 3C). With few exceptions, the RAFs of the lead SNPs among cases were higher than in controls in all regions (Appendix Table 2). The RAF of rs7225448 ranges from 0.36 to 0.47 in Finland and does not correlate significantly (Pearson’s r = 0.0733, P = 0.759) with mandibular retrognathia prevalence (Fig. 3B and Appendix Table 2).
(
Regulatory Activity of Credible Set Variants
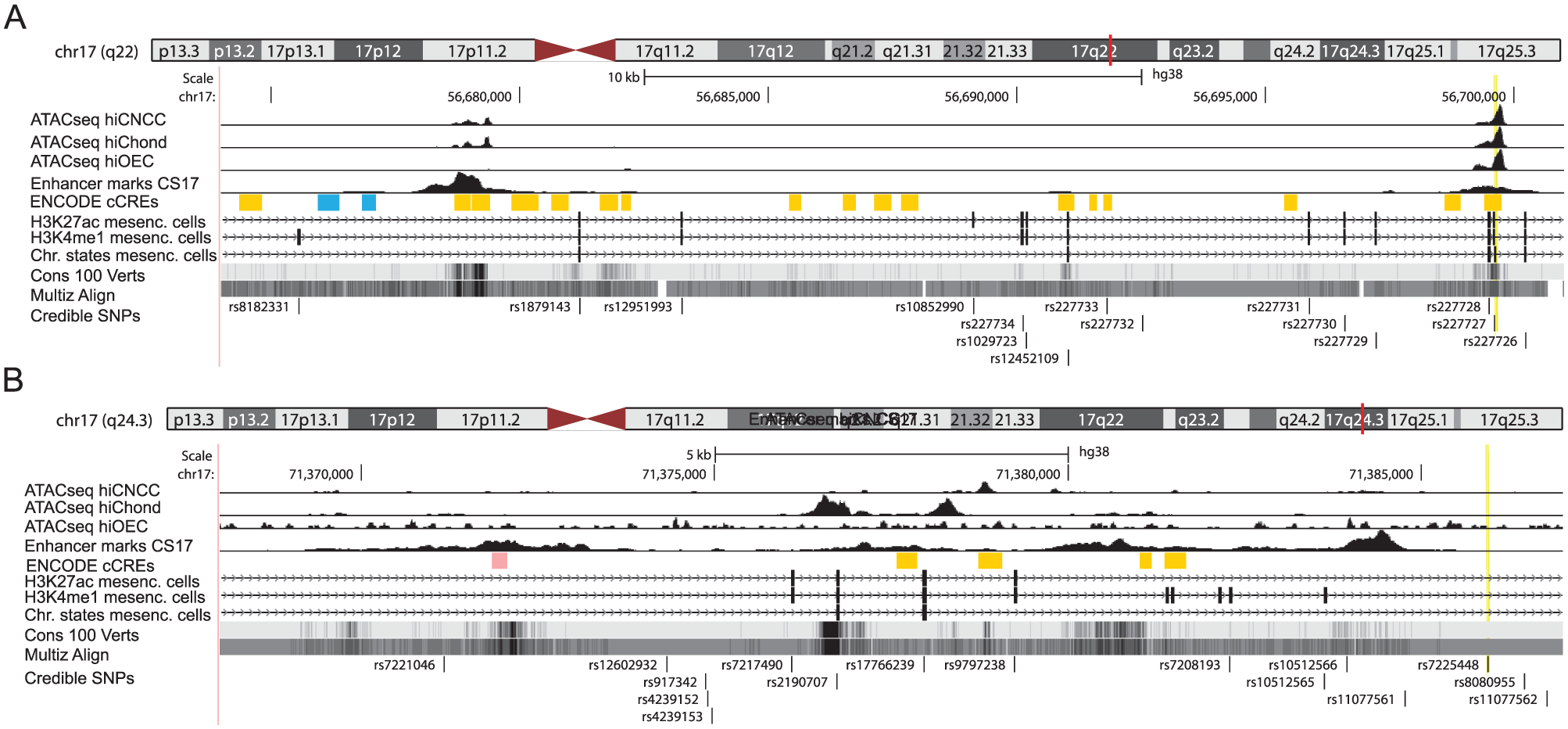
To evaluate the variants in the credible set for possible regulatory variants, we analyzed their positions relative to chromatin marks that correlate with open chromatin and active enhancer function (Fig. 4). Two regions with regulatory features were located within the NOG region, one of which overlapped with the lead SNP rs227727. Rs227727 is located in a multispecies conserved regulatory sequence (Fig. 4A), associated with nonsyndromic cleft lip and palate, and has previously been reported to alter the function of a NOG enhancer (Leslie et al 2015). While the lead SNP rs7225448 did not associate with obvious regulatory function, several credible variants in LD with the lead SNP in the middle of the SOX9 association region co-localized with reported indications of distal enhancer marks and open chromatin especially in chondrocytes and osteoblasts and affected transcription binding motifs (Fig. 4B). Of special interest is rs2190707, which localizes in a multispecies conserved sequence with enhancer marks and open chromatin. Concerning both associated regions, near NOG and SOX9, these features were specifically identified in cells involved in the development of the lower jaw, namely, oral epithelial cells, neural crest cells, and chondrocytes.
(
Replication in an Independent Icelandic Cohort
To replicate the association signals identified in the mandibular retrognathia cohort, a GWAS was conducted in an independent Icelandic cohort comprising 283 retrognathia cases and 246,436 population controls. The variant rs227727, located near NOG, showed a nominally significant association with mandibular retrognathia (P = 0.036282, OR = 1.2), with the direction of the effect being consistent across the datasets. Furthermore, rs227727 showed a negative effect in relation to mandibular prognathia (P = 1.4E-5, OR = 0.7). In contrast, the variant rs7225448, located near SOX9, did not show a significant association (P > 0.05) with mandibular retrognathia in the Icelandic cohort.
Nog Affects Condylar and Mandible Size
Mice deficient in Noggin, an antagonist of bone morphogenetic protein (BMP) signaling and coded by Nog, show craniofacial abnormalities due to excessive cartilage formation and failure to initiate proper joint development (Tylzanowski et al 2006). During mandibular development, Meckel’s cartilage undergoes significant enlargement in Nog-/-, which adversely affects the growth of the mandible as well as the formation of the temporomandibular joint (Wang et al 2013). We analyzed Alcian blue–Alizarin red staining of E18.5 mandibles of Nog-/- and wild-type littermate controls and found that inactivation of Nog results in a significant reduction in mandibular length and condylar process width (Fig. 5). Furthermore, the morphology and orientation of the mutant mandibular processes were notably altered. Specifically, the coronoid and condylar processes were narrower and oriented caudally, while the angular process appeared more rounded and enlarged relative to those in wild-type mice. During the development, Meckel’s cartilage demonstrated increased prominence in the Nog-/- mandible when compared with the wild-type littermate across developmental stages E14.5 to E16.5. Despite these changes, a progressive reduction in the length of the Nog-/- mandible was observed, in contrast to the wild-type littermate (Fig. 5C). These structural changes highlight the role of Nog in regulating mandibular length and morphogenesis.
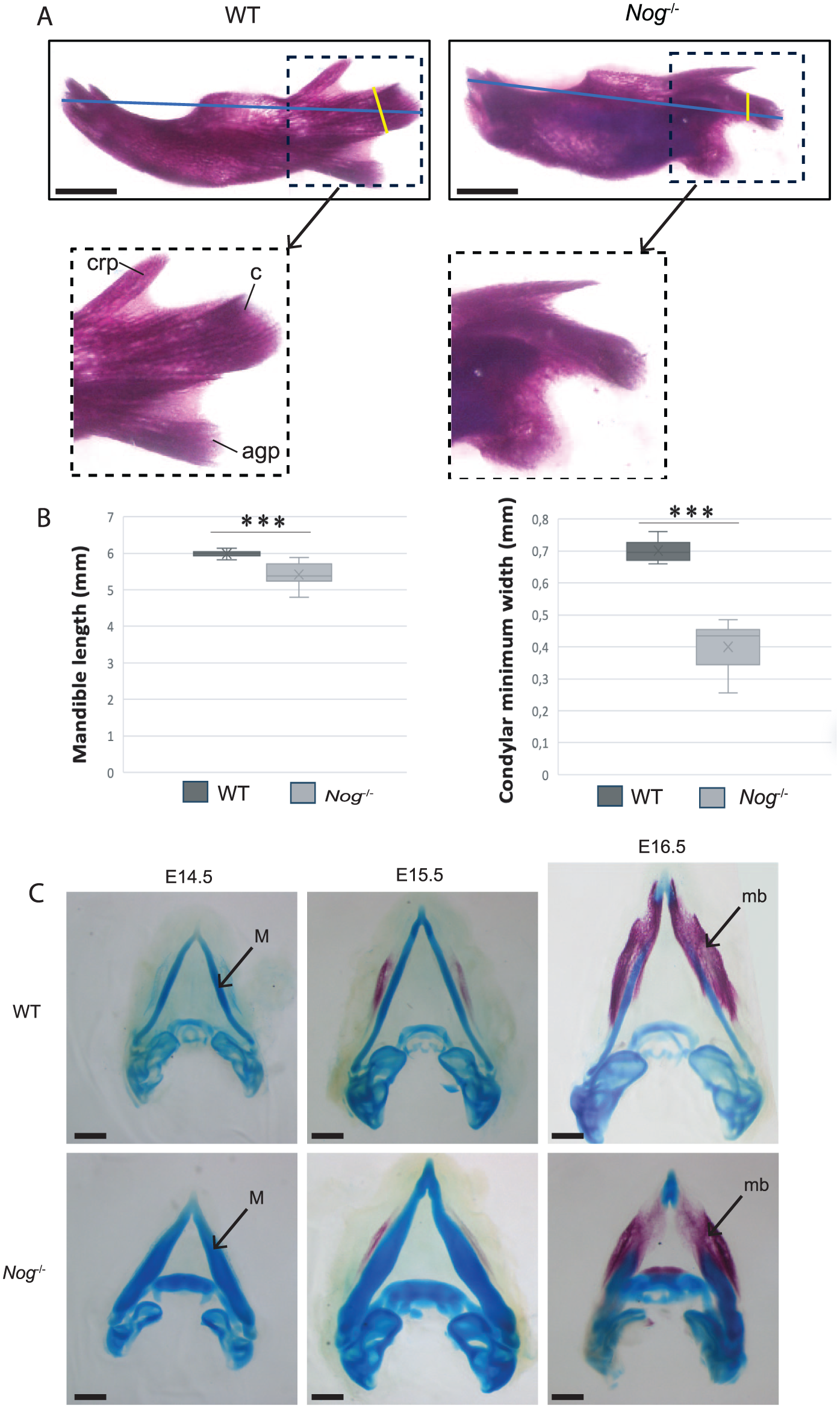
(
Discussion
We conducted a large-scale GWAS into dental malocclusion and identified 2 loci associated with mandibular retrognathia. Consistent with the relatively common prevalence of the phenotype, both lead SNPs have high RAFs. Both loci are in intergenic regions known to regulate genes (NOG and SOX9) critical for Meckel’s cartilage development and bone growth.
Evidence shows that the common variant rs227727, 110 kb downstream of NOG, reduces NOG enhancer activity (Leslie et al 2015). This risk variant was associated with nonsyndromic cleft lip/palate and its functionality shown by reduced Nog expression in the oral epithelial and mesenchymal cells. Nog antagonizes BMP signaling during bone development (Wan et al 2007), and NOG mutations cause multiple synostosis syndrome 1, which includes facial abnormalities (Takahashi et al 2001). Nog null allele mice exhibit excessive chondrocyte proliferation and Meckel’s cartilage expansion (Wang et al 2013). Meckel’s cartilage is a first branchial arch embryonic structure that contributes to the primary cartilaginous skeleton. However, it is a transient structure thought to contribute little to the postnatal mandibular bone, which is primarily formed by intramembranous ossification together with endochondral bone formation in secondary cartilaginous processes, including the condylar process (Svandova et al 2020). Even though Nog-/- mice exhibit an expansion in Meckel’s cartilage, we show that these mice exhibit a reduction in mandibular length, which is consistent with the mandibular retrognathia phenotype (Wang et al 2013).
In line with previous literature on NOG’s role in endochondral ossification, cartilage homeostasis, and joint development, our PheWAS analysis revealed significant associations of rs227722 with height and several types of arthroses. Experimental models of osteoarthritis have demonstrated that administration of Noggin protein attenuates cartilage degeneration and bone destruction (Chien et al 2020). Consequently, altered Nog levels, resulting from variants affecting enhancer regions, may impair cartilage function and predispose individuals to osteoarthritis. In addition, our data on rs227727 revealed a statistically significant association with height, aligning with previous findings for rs227724—also located near NOG—which is among the hundreds of variants reported to be associated with height (Lango Allen et al 2010). These findings highlight the notion that NOG is not only playing a role in bone growth but also in joint formation and joint function and may be involved in broader developmental traits such as height determination (Brunet et al 1998; Wen et al 2021).
The lead SNP, rs7225448, is in an intergenic region 735 kb upstream of SOX9. While rs7225448 did not locate with signs of active enhancer function in the relevant tissues, several variants in LD did. SOX9 has a well-documented role in cartilage development, and haploinsufficient SOX9 mutations cause campomelic dysplasia (Foster et al 1994) with a spectrum of phenotypes including cleft palate and micrognathia. In addition, Robin sequence, characterized by a small retrognathic mandible, which results in a downward and backward displacement of the tongue and consequent cleft palate, can be caused by inhibition of enhancer elements located up to 1.45 Mb upstream of SOX9 (Benko et al 2009; Gordon et al 2009). SOX9 variants have also been associated with facial shape variation in several GWASs investigating normal-range facial morphology in individuals of European descent (Xiong et al 2019; Hoskens et al 2021; White et al 2021).
Analysis of the far upstream (1.45–1.25 Mb) enhancers important in Robin sequence has indicated that their inactivation in cranial neural crest cells (CNCCs) leads to a moderate reduction of the SOX9 expression (Long et al 2020). Analogous manipulations in mice affected the development of mandibular ramus and condylar process, indicating an increased sensitivity of mandibular development to a reduced SOX9 dosage. This ultra-long-range regulation has been explained by multiloop DNA folding with enhancers clustered centrally to contact the SOX9 promoter (Chen et al 2023). The association presented in our study suggests another distal enhancer element 735 to 750 kb upstream of SOX9, which may become most relevant with advancing differentiation in chondrocytes. Our in silico analysis supports the associations and possible functional effect of rs7225448 or variants in LD on the development of mandibular retrognathia. However, this needs to be validated in future experimental studies on animal models.
Inactivation of Sox9 in the CNCCs results in an absence of condylar cartilage and temporomandibular joint malformation (Wang et al 2011). This finding aligns with our observation that retrognathic mandible is correlated with temporomandibular joint disorders. From a clinical perspective, it was not unexpected that we discovered a genetic correlation between retrognathic mandible and additional sagittal malocclusions, namely, increased overjet and distal bite, as well as a negative genetic correlation with mandibular prognathia. The observed genetic correlation with deep bite may result from a skeletal reduction in the lower anterior vertical face height or closing growth rotation of the mandible. Alternatively, it could be a secondary consequence of the retrognathic mandible, whereby the lower incisors fail to occlude with the upper incisors and consequently overerupt, causing deepening of the curve of Spee.
The Finnish population has genetic differences both along the east–west and south–north axes, and the differences are visible in genes as well as in disease heritage. This is due to the strong genetic drift and regions of genetic isolations that have occurred in Finland over hundreds of years (Peltonen et al 1999). Here we observe a remarkable north–west to south–east declining gradient in the prevalence of mandibular retrognathia that correlates with a similar gradient of the RAF of rs227727. In Finland, public health care provides free services for children younger than 18 y. The diagnosis of mandibular retrognathia is based on cephalometric analysis by orthodontists educated by a common curriculum minimizing regional diagnostic differences. The observed correlation supports the role of rs227727 in the etiology of mandibular retrognathia. Although the allele frequency differences between cases and controls observed for both lead SNP risk alleles were modest, the differences were present in almost all regions of Finland and may represent a biologically relevant signal when considered in the context of shared polygenic risk. Geographic variation in phenotype prevalence is therefore likely driven by the cumulative effect of many common variants.
Differences in mandibular size and position between females and males are influenced by factors involving hormonal, muscular, and skeletal growth differences. Both SOX9 and NOG were more strongly associated with mandibular retrognathia in females than in males. Although there may exist a lower level of ascertainment among males, it should not be expected to affect the RAF among them. We suggest that the identified variants have a weaker effect on male mandibular development due to other etiological factors including hormonal growth regulation during puberty. In addition, although condylar resorption was not associated with mandibular retrognathia in our study, it may contribute to the observed gender differences. Condylar resorption occurs more frequently in women than in men and has been associated with variations in sex hormone levels, particularly estrogen, which is thought to play a role in the pathogenesis of the condition (Nicolielo et al 2017). Altogether, the results of this study emphasize the effect of genetic differences between females and males that further contribute to why females have a more retrognathic mandible.
Replication in an independent Icelandic cohort provided additional support for the association of rs227727 with mandibular retrognathia. Specifically, the association was nominally significant, and the direction of effect was consistent across the discovery and replication datasets, suggesting that this association may generalize beyond the Finnish population. In contrast, the variant rs7225448 did not replicate in the Icelandic cohort, which may reflect differences in statistical power or population-specific genetic architecture.
The FinnGen project systematically identifies known risk associations for multiple traits and has the potential to discover common genetic variants that are also present in pan-European populations (Kurki et al 2023). It is well established that current analysis tools, together with relatedness matrix correction and principal component adjustment, effectively account for within-population stratification. Although allele frequencies can differ between ancestry groups, the underlying biological mechanisms are largely shared across populations. Both lead SNPs are present worldwide and reflect a polygenic risk for mandibular retrognathia. The findings of this study contribute to our understanding of the development of mandibular retrognathia and highlight the role of genetic predisposition to this common craniofacial anomaly. Further research into the molecular mechanisms underlying mandibular development may provide insights into novel therapeutic approaches for treating this condition.
Author Contributions
E. Juuri, S. Strausz, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; Md. R. Hasan, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; M. Hongqiang, contributed to data acquisition, critically revised the manuscript; T. Jónsson, Á. Thordarsson, G. Auðólfsson,D. Gudbjartsson, contributed to conception and design, data acquisition, critically revised the manuscript; H. Stefansson, contributed to conception and design, data analysis, and interpretation, critically revised the manuscript; P. Nieminen, H.M. Ollila, D.P Rice, contributed to conception and design, data analysis, and interpretation, drafted and critically revised the manuscript. All authors gave their final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251397375 – Supplemental material for Variants Near NOG and SOX9 Are Associated with Mandibular Retrognathia
Supplemental material, sj-docx-1-jdr-10.1177_00220345251397375 for Variants Near NOG and SOX9 Are Associated with Mandibular Retrognathia by E. Juuri, S. Strausz, Md. R. Hasan, M. Hongqiang, T. Jónsson, Á. Thordarsson, G. Auðólfsson, D. Gudbjartsson, H. Stefansson, P. Nieminen, H.M. Ollila and D.P. Rice in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345251397375 – Supplemental material for Variants Near NOG and SOX9 Are Associated with Mandibular Retrognathia
Supplemental material, sj-pdf-2-jdr-10.1177_00220345251397375 for Variants Near NOG and SOX9 Are Associated with Mandibular Retrognathia by E. Juuri, S. Strausz, Md. R. Hasan, M. Hongqiang, T. Jónsson, Á. Thordarsson, G. Auðólfsson, D. Gudbjartsson, H. Stefansson, P. Nieminen, H.M. Ollila and D.P. Rice in Journal of Dental Research
Footnotes
Acknowledgements
We want to acknowledge the participants and investigators of the FinnGen study. The FinnGen project is funded by 2 grants from Business Finland (HUS 4685/31/2016 and UH 4386/31/2016) and the following industry partners: AbbVie Inc., AstraZeneca UK Ltd, Biogen MA Inc., Bristol Myers Squibb (and Celgene Corporation & Celgene International II Sàrl), Genentech Inc., Merck Sharp & Dohme LCC, Pfizer Inc., GlaxoSmithKline Intellectual Property Development Ltd., Sanofi US Services Inc., Maze Therapeutics Inc., Janssen Biotech Inc, Novartis AG, and Boehringer Ingelheim International GmbH. The following biobanks are acknowledged for delivering biobank samples to FinnGen: Auria Biobank (www.auria.fi/biopankki), THL Biobank (www.thl.fi/biobank), Helsinki Biobank (www.helsinginbiopankki.fi), Biobank Borealis of Northern Finland (https://www.ppshp.fi/Tutkimus-ja-opetus/Biopankki/Pages/Biobank-Borealis-briefly-in-English.aspx), Finnish Clinical Biobank Tampere (www.tays.fi/en-US/Research_and_development/Finnish_Clinical_Biobank_Tampere), Biobank of Eastern Finland (www.ita-suomenbiopankki.fi/en), Central Finland Biobank (www.ksshp.fi/fi-FI/Potilaalle/Biopankki), Finnish Red Cross Blood Service Biobank (www.veripalvelu.fi/verenluovutus/biopankkitoiminta), Terveystalo Biobank (www.terveystalo.com/fi/Yritystietoa/Terveystalo-Biopankki/Biopankki/), and Arctic Biobank (https://www.oulu.fi/en/university/faculties-and-units/faculty-medicine/northern-finland-birth-cohorts-and-arctic-biobank). All Finnish Biobanks are members of BBMRI.fi infrastructure (https://www.bbmri-eric.eu/national-nodes/finland/). Finnish Biobank Cooperative-FINBB (https://finbb.fi/) is the coordinator of BBMRI-ERIC operations in Finland. The Finnish biobank data can be accessed through the Fingenious® services (![]() ) managed by FINBB. We also thank Airi Sinkko and Johanna Pispa for excellent technical help.
) managed by FINBB. We also thank Airi Sinkko and Johanna Pispa for excellent technical help.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by grants from HUS Helsinki University Hospital Research Foundation: Y2519SU008 (D.P.R.) the Finnish Dental Society Apollonia (E.J. and S.S.), Instrumentarium Science Foundation and Academy of Finland #340539 (H.M.O.).
Data Availability Statement
GWAS summary stats will be made available on an open access GWAS catalog postacceptance. All other data that support the findings of this study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
