Abstract
Hybrid layers degrade due to endogenous collagenolytic enzymes and adhesive hydrolysis. Adhesive hydrolysis releases by-products that, along with uncured monomers, may have an adverse effect on collagen fibrils and contribute to the dissolution of the hybrid layer in resin–dentin bonds. The aim of this study was to investigate the effects of methacrylate monomers and corresponding by-products on collagen type I. Tendon fibers (TFs) from mouse tail were incubated with BisGMA 0.1%, BisEMA 0.1%, UDMA 0.1%, HEMA 0.1%, TEG-DMA 0.1%, methacrylic acid 0.025% (MAA), pyruvic acid 0.025% (PA), trypsin as positive control (PC), and water/ethanol as negative control for 1 h, 6 h, 24 h, 72 h, and 7 d. At each period, the specimens were tested mechanically (tensile strength and elastic modulus) and the storage medium tested for hydroxyproline (HPY) release and expressed as percentage of collagen solubilization (%CS). The TFs were morphologically analyzed by a Nikon-Eclipse 80i microscope. Incubation media and time affected TFs in different ways. The incubation of TFs with PA or MAA caused significant damage to the structure, reducing properties and increasing %CS to levels similar or higher than that of trypsin PC. Incubation in the monomers BisGMA 0.1%, BisEMA 0.1%, UDMA 0.1%, HEMA 0.1%, and TEG-DMA 0.1% did not cause hydroxyproline release, and their effects on the TF mechanical properties varied and were possibly related to dehydration. Methacrylate monomers and their by-products can adversely affect TF structure and properties. The findings indicate that collagen degradation in resin–dentin bonds can also be caused by by-products of the adhesive.
Introduction
Although the hybrid layer is the foundation of contemporary adhesive dentistry, its long-term stability is considered a weak point of restorations (Tjäderhane 2015). Degradation of this biocomposite formed by dentin collagen and adhesive resin can create interfacial gaps prone to bacterial colonization, leading to restoration failure and recurrent caries (Tjäderhane 2015). Dentin collagen breakdown has been linked to host-derived enzymes such as matrix metalloproteinases (MMPs) and cathepsins (CT), which are present in mineralized dentin and become active upon demineralization during caries or acid etching (Vidal et al 2014; Tjäderhane et al 2015; Scaffa et al 2017). Simultaneously, the adhesive resin undergoes hydrolytic degradation, exposing unprotected collagen fibrils (Ferracane 2006; Tjäderhane 2015; Fugolin and Pfeifer 2017) and releasing monomers and by-products at the interface.
Although enzymatic collagen degradation and polymer hydrolysis occur simultaneously at the resin–dentin interface, they were initially considered independent events (Tjäderhane 2015). However, a later study reported increased MMP activity triggered by polymer hydrolysis via esterase catalysis, particularly in total-etch systems (Serkies et al 2016). Resin–dentin interfaces are also affected by exogenous factors such as cholesterol esterase from Streptococcus mutans and MMPs released by neutrophils and macrophages in saliva, all contributing to composite and adhesive hydrolysis and oxidation (Santerre et al 1999; Ferracane 2006; Huang et al 2018; Gitalis et al 2019; Huang et al 2021).
During dentin bonding, a sequence of molecular-level chemical events occurs at the interface. Subpolymerized adhesive monomers are eluted within the first hour after polymerization (Miletic et al 2009). These free monomers are chemically unstable and rapidly break down into various by-products (Sevkusic et al 2014), including methacrylic acid (MAA) and pyruvic acid (PA), common by-products of monomers in resin-based materials (Reichl et al 2002; Van Landuyt et al 2007; Emmler et al 2008; Van Landuyt et al 2011; Sevkusic et al 2014). Although uncured adhesives and their by-products likely come into direct contact with exposed collagen fibrils during and after bonding (Sevkusic et al 2014; Jiang et al 2017; Schertl et al 2019), their specific effects on collagen remain unaddressed in the context of bonding durability (Van Landuyt et al 2011; Gupta et al. 2012).
Therefore, the present study evaluated the effects of uncured monomers commonly used in the formulation of adhesive systems and their known by-products on the mechanical properties and solubilization of type I collagen. Accordingly, we tested the null hypothesis that monomers and by-products would not have significant effects on the ultimate tensile strength (UTS), elastic modulus (EM), solubilization, and morphological structure of collagen fibers.
Methodology
Tails from 10 female 3-mo-old C57BL/6 mice (from unrelated studies) were used in compliance with ethical guidelines (UBC Animal Care Protocols, Canadian Council on Animal Care, and ARRIVE 2.0). Isolation, preparation, and storage tendon fibers (TFs), primarily composed of type I collagen, are detailed in Appendix.
The detailed methodologies for TFs isolation, solutions preparation, pH monitoring of solutions, TFs incubation, mechanical testing, collagen solubilization through hydroxyproline-based quantification, optical microscopy analysis, data handling, and data correlation are provided in the Appendix.
Results
Ultimate Tensile Strength
The results indicated that both incubation media (P < 0.001; power = 1; F = 38.76) and incubation time (P < 0.001; power = 0.89; F = 4.803) had a significant effect in UTS, but no interaction was observed (P = 0.081; power = 0.38; F = 1.419) (Fig 1A). Incubation of TFs in the positive control (PC) and MAA 0.025% resulted in the lowest values for UTS (P < 0.001). Incubation in MAA 0.025% significantly decreased the UTS over time, especially after 24 h (P < 0.001). Three of the 10 samples had extensive degradation at 7 d of incubation in MAA that could not be tested and were excluded from the statistical analysis. Similarly, specimens incubated in PA 0.025% could not be tested due to complete rupture of the TF structure within the first hour of incubation. Incubation in UDMA 0.1% caused a decrease in UTS over time (P < 0.001; power = 0.891), but the means were not significantly different from negative control (NC) in all incubation periods.
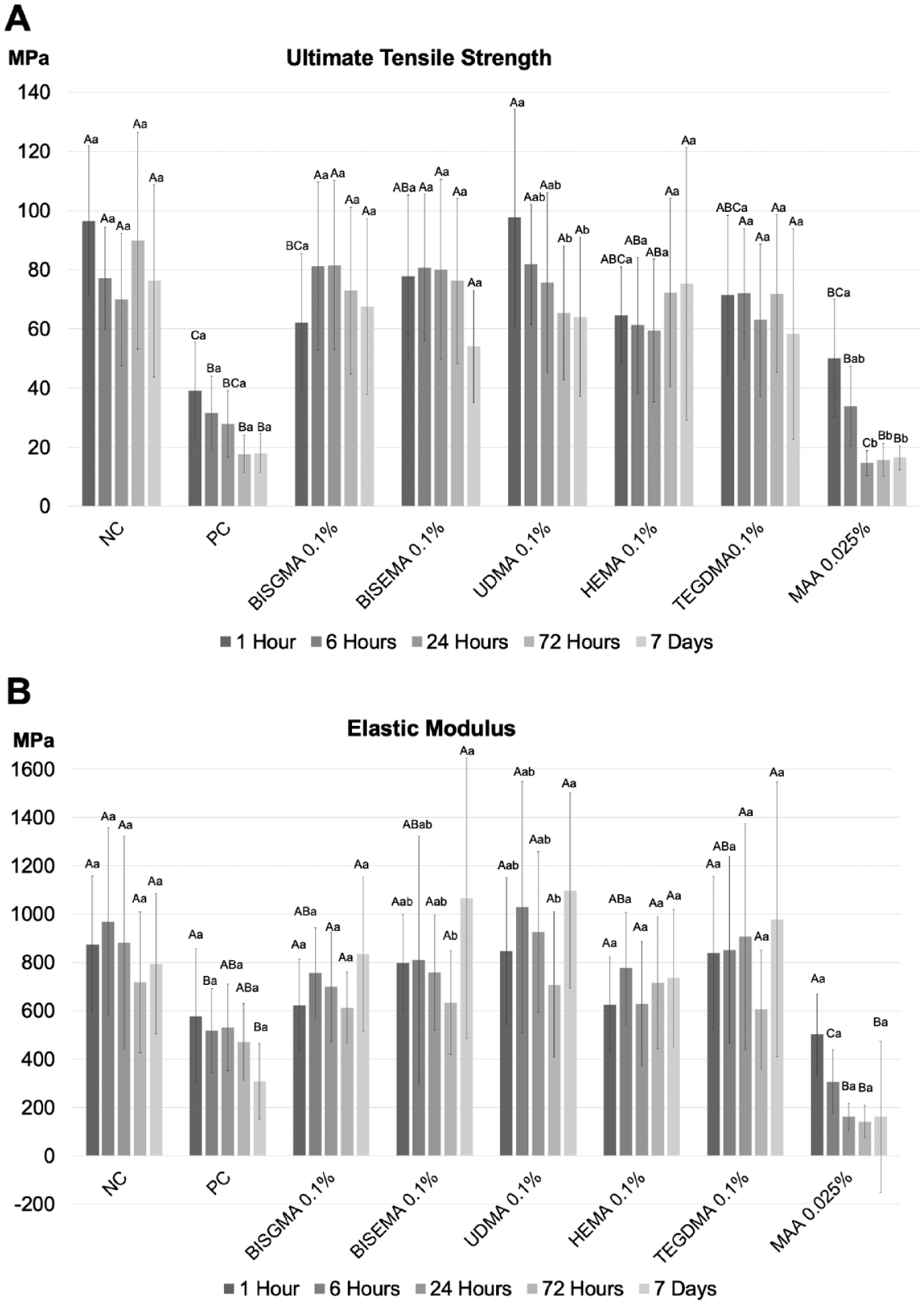
Mean (
Elastic Modulus
The results indicated that both incubation media (P < 0.0001; power = 0.84; F = 25.47) and incubation time (P = 0.003; power = 1; F = 4.16) had a significant effect on EM, with no interaction between the factors (P = 0.19; power = 0.20; F = 1.24). Within the BisEMA 0.1% and UDMA 0.1% incubation groups, the analysis of the TF specimens at different storage times showed a significant decrease in EM after 72 h (P < 0.05). However, at each incubation period, the EM values of these groups were not significantly different from those of the negative control (P > 0.05) (Fig 1B). Furthermore, there was a slight (P > 0.05) increase in EM values for all monomer groups after 7 d of incubation. A significant reduction in the EM values was observed after 6 h of incubation in the PC and MAA groups (P < 0.001). Incubation in MAA resulted in significantly lower values of EM than in PC (P < 0.05, Appendix Table 5) when the groups were compared independent of the incubation time.
Collagen Solubilization
Two-way analysis of variance showed that both incubation media (P < 0.001; F = 65.19) and time (P < 0.001; F = 30.63) significantly affected %CS, with no interaction between factors (P < 0.001; F = 23.48).
PA caused the greatest solubilization at all observed periods: 1 h, 15.2%; 24 h, 17.6%; 7 d, 75.5%; followed by MAA 0.025%: 1 h, 0.2%; 24 h, 1.73%; 7 d, 7.71%; and PC trypsin: 1 h, 2.8%; 24 h, 2.96%; 7 d, 5.56%. The highest solubilization occurred at 7 d. Other monomers caused no significant TF solubilization (Fig 2).
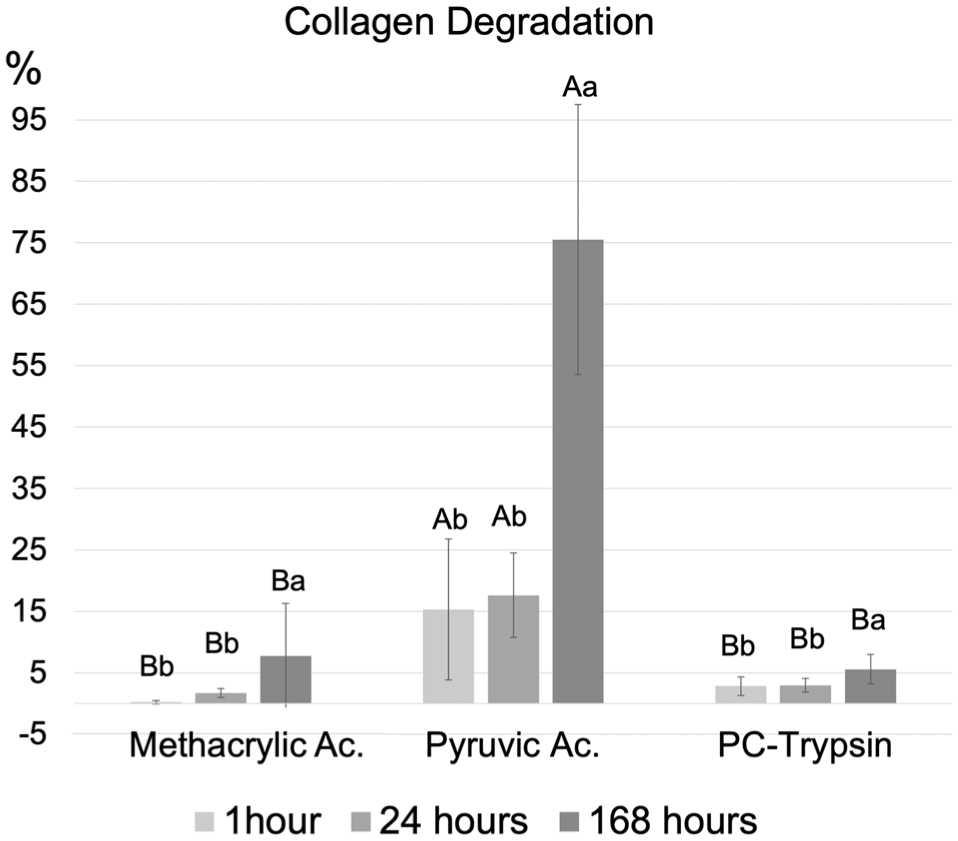
Percentage of collagen degradation after incubation in the solutions. Different lowercase letters indicate a statistical difference between the different incubation times for each by-product solution. Different uppercase letters indicate the statistical difference between by-product solutions and PC-trypsin within the same incubation time. Error bars show the standard deviation. N = 10 for all groups.
Optical Microscopy Analysis
Morphology
Microscopy showed early structural unravelling at 2 min for PA 0.1%, followed by hivelike voids at 5 min (PA) and 10 min (MAA). At 60 min, both by-products induced marked changes: TFs in MAA 0.1% appeared flatter with small voids, while PA 0.1% showed larger, more numerous internal voids (Fig 3).
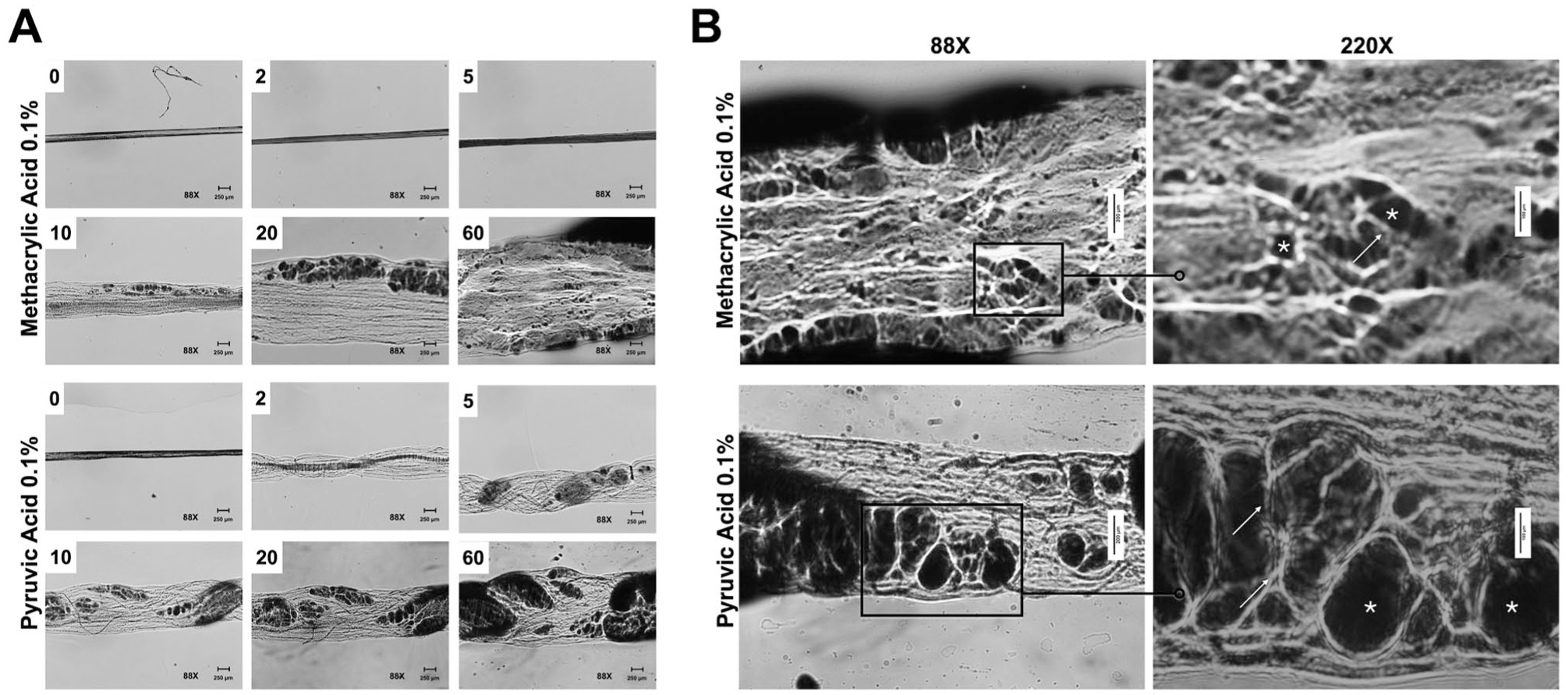
(
Diameter quantification
The diameter of the TFs remained stable in HEMA, BisGMA, and BisEMA (Appendix Table 11, P > 0.05). UDMA, TEGDMA, and NC caused significant increases by 5 min, which then stabilized (P < 0.05). MAA and PA induced greater swelling at 5 and 60 min (P < 0.05); although PA acted earlier, MAA produced larger diameters by 60 min (P < 0.0001; Appendix Table 11)
Discussion
The present study showed that by-products that commonly result from the degradation of methacrylate resins significantly damaged the structure of type I collagen. Both by-products PA and MAA negatively affected the UTS of TFs. The incubation in PA 0.025% severely damaged the TFs to the point that the mechanical test could not be performed within the first hour of incubation. The incubation in MAA 0.025% also resulted in 3 prefailure samples after 7 d of incubation, with most of the reduction in the UTS occurring after 24 h (Fig 1A). Acids are known to induce collagen denaturation by disrupting ionic interactions within the secondary and/or tertiary structure, leading to fiber swelling and unravelling and potentially progressing to hydrolysis (Bogue 1923; Etherington 1977; Nelson and Cox 2012). This effect may also be related to the acid-induced removal of glycosaminoglycans (Screen et al 2005; Toledano et al 2020). Proteoglycans, glycosaminoglycans, and noncollagenous proteins are highly polar matrix components and can be dissociated from collagen-rich tissues under low-pH conditions or with chaotropic agents (Screen et al 2005; Toledano et al 2020). Their removal leads to disruption of the extracellular matrix organization and promotes swelling of collagen fibrils and fibers due to increased water uptake and loss of interfibrillar cohesion (Screen et al 2005). Collagen fiber unravelling has been associated with reduced mechanical properties, which explain our results for the MAA group (Bertassoni and Swain 2017) (Fig 1A).
While low pH has long been recognized as a factor in collagen denaturation and hydrolysis (Bogue 1923; Etherington 1977; Nelson and Cox 2012), our results indicate that proton concentration alone does not fully account for the extent of degradation observed with MAA and PA.
Methacrylate anions and analogs, being α,β-unsaturated carboxylic acids, can participate in Michael-type conjugate additions with nucleophilic residues such as lysine side chains (Sánchez-Roselló et al 2014). Similarly, pyruvate anions, due to their α-keto structure, can react with lysine residues via Schiff base formation—analogous to early-stage Maillard chemistry (Wnorowski and Yaylayan 2003). These specific chemical interactions, which occur independently of enzymes, likely explain why MAA and PA cause collagen damage even at low concentrations. Both have the potential to disrupt intrafibrillar cross-links and structural cohesion, modifying collagen matrices (Wnorowski and Yaylayan 2003; Sánchez-Roselló et al 2014), which suggests that MAA and PA exert additional, anion-specific effects.
Adding to that, Etherington (1977) reported optimal collagen fiber solubilization at pH 3.5. In our study, the 0.025% PA solution had a pH of 3.62 ± 0.17—close to this optimum—and specimens ruptured completely within the first hour, precluding mechanical testing. This group also exhibited the highest %CS (75.5%), particularly after 7 d. In contrast, 0.025% MAA had a higher pH (4.75) and a much lower %CS (7.71%) (Bogue 1923).
Incubation in trypsin 0.01% (PC) and MAA 0.025% resulted in the lowest UTS values in all incubation periods, becoming more significant over time compared with the other solutions (Fig 1A). PC-trypsin acts by removing noncollagenous proteins and glucosamines, causing the unraveling of microfibril bundles in collagen type I, consequently decreasing the mechanical properties (Bedran-Russo et al 2008; Bertassoni and Swain 2017). Insoluble, cross-linked collagen, such as that found in tendons, exhibits significant resistance to enzymatic degradation by trypsin. The highly organized structure impedes trypsin’s access to its cleavage sites. As a result, degradation is typically less than 10% (Gomes et al 2020; Toledano et al 2020; Vallecillo et al 2021). In addition, although mice tendons have a high concentration of collagen, the percentage fluctuates from about 70% to 80% of tendon dry weight (Wang 2006), which constitutes a limitation of our study, as the exact concentration of each fiber could not be determined.
The comonomers used in this study did not result in significant alterations in the UTS as values were like control NC (water/ethanol). No differences in the EM were observed among incubation time for each solution (Fig 1B). UTS and EM in TFs respond differently because they represent distinct mechanical properties. EM reflects the stiffness in the elastic region, largely influenced by early deformation, collagen fiber crimp, and fibril recruitment, while UTS depends on the ultimate failure of fibrils (Screen et al 2005; Ker 2007). Microdamage and collagen cross-linking affect UTS more significantly than EM does, which remains stable as long as early structural mechanisms function properly. Thus, despite being measured from the same specimen, EM is less affected than UTS due to its different biomechanical determinants (Screen et al 2005; Wang 2006; Ker 2007). It is important to note that the EM reflects only the elastic portion of the stress–strain response, whereas the UTS represents the maximum force at fracture, which may extend beyond the elastic region depending on the material’s characteristics. This distinction likely explains the observed differences between EM and UTS measurements (Screen et al 2005; Wang 2006; Ker 2007). Our results showed a decrease in EM after 72 h of incubation in the BisEMA and UDMA solutions. However, since no significant differences were detected compared with the negative control at any incubation time, the extent of this effect was likely minimal and reversible. These findings suggest that short-term exposure to BisEMA and UDMA by-products may induce a subtle, temporary softening of the collagen fiber rather than permanent structural degradation (Wang 2006; Vallecillo et al 2021). The slight increase in EM for the monomer groups observed after 7 d of incubation (Fig 1B) can be attributed to the dehydration of the fibers caused by the media saturated with monomers and solvent (Garcia et al 2005). The use of simulated body fluid, phosphate-based, and saline solutions was unsuitable due to the potential ion deposition on fibers, which could confound mechanical test results. (Panwar et al 2015; Di Foggia et al 2019).
Incubation of collagen fiber with the most common monomers used in dental resins for the periods tested resulted in no significant or detectable damage to the collagen structure or properties. Although no damage was observed, it is relevant to point out that these monomers will eventually degrade, generating by-products such as those investigated in this study, which had a significant effect in damaging collagen fibers. It is speculated that if the specimens were incubated with the monomers long enough for degradation and release of by-products to occur, then evidence of collagen degradation could have been found. However, it is important to note that the study used neat monomers and neat solutions of PA and MAA as the incubation media, rather than a polymerized resin blend undergoing degradation. While neat monomers may be compared with uncured monomers present in adhesive systems, they do not directly replicate the complex interactions and gradual degradation occurring within the adhesive interface. Despite this limitation, the results suggest that exposure to residual monomers and their subsequent degradation products could contribute to collagen breakdown over time, supporting concerns regarding the long-term stability of the adhesive–dentin interface (Van Landuyt et al 2011; Tjäderhane 2015). In addition, although UDMA, TEGDMA, BisEMA, and BisGMA are not considered highly hydrophilic, they contain ethylene oxides, urethane, ether linkages, ester, and hydroxyl groups that attract water molecules, allowing for slow but continuous water sorption over time (Ferracane 2006; Yiu et al 2006; Van Landuyt et al 2007) A particular highlight must be given to HEMA due to its highly hydrophilic nature, as it absorbs water in both cured and uncured conditions, leading to hydrolytic degradation of polymerized adhesive into by-products (Ferracane 2006; Yiu et al 2006; Van Landuyt et al 2007; Van Landuyt et al 2011; Tjäderhane 2015).
With regard to fiber diameter alterations, incubation in MAA 0.1% and PA 0.1% resulted in a significant increase in diameter compared with other solutions at 5-min and 60-min incubation (Appendix Table 11, Figs 1, 2). Considering specifically the characteristics of fiber morphology, the swelling was clearly a result of rapid liquid influx within the fibers as large voids and lacunas were opened by the rupture and unraveling of the fiber’s structure (Etherington 1977; Nelson and Cox 2012) (Fig 3 and Appendix Table 11). However, the 2 acids presented different patterns of morphological change. While PA 0.1% acted fast initiating morphological changes within 1 to 2 min of incubation, MAA started after 10 min of incubation (Fig 3). Over time (60 min), however, fiber diameter was significantly larger for the MAA 0.1% group than for the PA 0.1% group (Appendix Table 11 and Fig 1). The morphological features (Fig 3) indicated that MAA 0.1% caused the flattening of the structure with smaller voids, while PA 0.1% resulted in larger voids and bigger lacunae (Fig 3; Appendix Fig 2). Although trypsin and MAA 0.1% presented similar %CS (Fig 2), significant morphological changes were not observed for PC trypsin (Appendix Fig 1). The trypsin peak effect is usually observed after 48 h of incubation (Bedran-Russo et al 2008; Bertassoni et al 2015; Bertassoni and Swain 2017). Our morphological change analysis followed only up to 60 min. Also, the trypsin enzymatic transformation effect happens on the level of the microfibril bundle (~20 nm structures) of collagen type I (Bertassoni and Swain 2017) not observable under the magnification used in this study. Although our optical microscopy (88×–220×) identified gross swelling and void formation, the lack of high-resolution imaging (eg, scanning electron microscopy [SEM] or transmission electron microscopy [TEM]) remains a limitation of this study. Such techniques would allow direct visualization of nanoscale fibrillar alterations, providing stronger morphological confirmation of the chemical and mechanical findings reported here. Future investigations incorporating SEM or TEM will be important to validate and extend our results by capturing ultrastructural changes in dentin collagen induced by resin by-products.
As expected, the %CS had a direct correlation with the decrease in the fiber mechanical properties (Appendix Tables 6, 7, 8, and 9) with high correlation with UTS (−0.75; P < 0.0001) and EM (−0.70; P < 0.0001). Incubation time was a significant factor affecting UTS, EM, and %CS only for by-product PA 0.025%, MAA 0.025%, and PC-trypsin (Appendix Tables 8, 9, and 10). The other neat monomers tested did not seem to affect the TFs in a short-term incubation time.
Considering the restorative procedure, unprotected collagen in an incompletely infiltrated hybrid layer is already considered a weak link due to exposure to collagenolytic enzymes (Tjäderhane 2015; Scaffa et al 2017). Although the hybrid layer represents a relatively closed environment, residual water and unreacted components within the adhesive interface can promote hydrolytic and oxidative degradation of ester-based monomers and polymer networks over time (Ferracane 2006; Delaviz et al 2014).
These degradation reactions release acidic by-products soon after bonding, which may diffuse through the hybrid layer and interact with exposed collagen fibrils, contributing to structural destabilization (Reichl et al 2002; Van Landuyt et al 2007; Emmler et al 2008; Van Landuyt et al 2011; Sevkusic et al 2014). Our findings suggest that such by-products can severely affect collagen fibrils at the hybrid layer, thereby exacerbating known pathways of collagen and hybrid layer degradation. The concentrations tested in this study are comparable with those eluted from typical adhesive resins and likely present at the resin–dentin interfaces (Appendix Table 2), supporting this mechanism.
The simultaneous occurrence of resin degradation and enzymatic or hydrolytic breakdown of the hybrid layer likely amplifies collagen damage at the interface (Tjäderhane 2015; Huang et al 2021). This process may facilitate endogenous enzymes to access susceptible peptide bonds that would otherwise remain protected until later stages of collagen breakdown (Etherington 1977). As shown by Serkies et al (2016), the total-etch technique creates adhesive–dentin interfaces more susceptible to collagen solubilization, an effect intensified by polymeric degradation (Tjäderhane et al 2015; Serkies et al 2016).
An additional drawback of acidic by-product production is the maintenance of a vicious cycle at low pH, in which hydrolysis of the monomer’s ester bonds is accelerated (Delaviz et al 2014; Fugolin and Pfeifer 2017). Therefore, the persistent decrease in pH within the hybrid layer microenvironment may contribute to continuous damage, inducing gap formation and eventual restoration failure.
In view of the results found in the present study, the acidic by-products might play a role in the hybrid-layer degradation, by swelling and/or predamaging the collagen and, consequently, making it more suitable to proteolytic enzymes (metalloproteinases and CT) attack (Etherington 1977). The low-pH environment also boosts the action of cathepsin K (Garnero et al 1998) and MMPs (Tjäderhane 2015) and contributes to the continuation of polymeric resin degradation (Delaviz et al 2014; Fugolin and Pfeifer 2017). In conclusion, based on the findings of PA and MAA, by-products caused TF solubilization, as observed by fiber hydrolysis, reducing their mechanical properties and affecting collagen morphology. The null hypothesis was partially rejected, as PA and MAA significantly affected the UTS and EM, promoted TF solubilization, and altered collagen morphology. However, no consistent or significant effects were observed in the monomers groups.
Author Contributions
R.V. Rodrigues, contributed to conception and design, data acquisition, analysis, or interpretation, drafted and critically revised the manuscript; A.P. Manso, contributed to design, data analysis, and interpretation, critically revised the manuscript; R.M. Carvalho, contributed to conception and design, data analysis, and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251393865 – Supplemental material for Can Resin Monomers and By-Products Damage Collagen?
Supplemental material, sj-docx-1-jdr-10.1177_00220345251393865 for Can Resin Monomers and By-Products Damage Collagen? by R.V. Rodrigues, A.P. Manso and R.M. Carvalho in Journal of Dental Research
Footnotes
Acknowledgements
We express our thanks to Shelly McErlane (Centre for Comparative Medicine Sciences) for the donation of mouse tails used in this study. This study was supported by UBC Start-Up research funding (RMC, AMP).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
