Abstract
Several empirical observations strongly suggest that salivary function is regulated by the circadian clock. Salivary volume, electrolytes levels, and saliva protein composition all show 24-h cycle fluctuations. The exact effects of circadian disruption on salivary gland (SG) physiology and its potential role in salivary pathologies have not been elucidated. Here, we examined the effects of circadian disruption on SG structure, functional gene and protein expression, and immune status using several circadian knockout (KO) mice models where we targeted the following canonical clock genes: brain and muscle ARNT-like 1 protein (Bmal1) KO, Period2 (Per2) KO, Cryptochrome1 (Cry1) KO, Cryptochrome2 (Cry2) KO, and Cryptochrome 1 and 2 double KO (DKOCry). All mice were females of young and old age, and data were compared with wild type (control) mice. Our results showed that circadian dysregulation had limited effects on stimulated salivary secretion and SG structure. Yet, circadian disruption significantly affected the expression of several key salivary markers, including mucins, amylase, and aquaporins, in young and aged SGs with increased amylase and acidic mucin production observed in several KO models. In addition, targeting of clock genes has resulted in subtle alterations of the salivary immune microenvironment with increased lymphocyte infiltration and upregulated levels of proinflammatory cytokines. These immune shifts were more pronounced in aged glands with the most proinflammatory phenotypes observed in DKOCry and Bmal1KO mice. Collectively, our results implicate the circadian clock in the intricate temporal regulation governing SG function. Our data also suggest that circadian dysregulation may predispose to increased tissue stress and inflammation. Exploration of salivary system chronobiology represents a new avenue for salivary disease prevention and treatment.
Background
The circadian clock, a natural 24-h internal system found in most organisms, governs almost all physiologic processes, enabling adaptation to environmental changes (Adeola et al. 2019). In mammals, this system is centered in the suprachiasmatic nucleus, which oversees subsidiary clocks in various brain regions and organs (Reppert and Weaver 2002). At the molecular level, this system is maintained and controlled by a diverse group of transcription factors expressed by clock genes (Papagerakis et al. 2022). The core clock genes include brain and muscle ARNT-like 1 protein (BMAL1), circadian locomotor output cycles kaput protein (CLOCK), periods (PER1/PER2), and cryptochromes (CRY1/CRY2), among others (Said et al. 2020). Transcription-translation feedback loops involving these genes drive the circadian rhythm (Zheng et al. 2014). It is estimated that 43% of all protein-coding genes exhibit circadian oscillations in transcription (Zhang et al. 2014). Disruptions in the circadian clock have been linked to various physiologic and pathologic conditions affecting varied health outcomes, including metabolic disorder, cancer susceptibility, and development of autoimmunity (Adeola et al. 2019).
In the context of saliva, several observations strongly suggest that salivary function is regulated by the circadian clock. Multiple reports showed distinct circadian variations in saliva secretion with clear diurnal fluctuations in salivary volume, electrolytes, and protein composition (Dawes 1972; Dawes and Ong 1973; Zheng et al. 2012). There is a clear diurnal variation in salivary immunoglobulin A levels (peaking through the night), which is thought to be mediated by suprachiasmatic nucleus stimulation of the salivary sympathetic nerves (Wada et al. 2017). We and others have highlighted the role of the molecular circadian clock in influencing aquaporin 5 (Aqp5) expression, a key player in regulating water permeability and salivary fluid secretion, where we showed that Aqp5’s expression is influenced by BMAL1-CLOCK heterodimer and that its expression levels exhibit rhythmic patterns, especially in the submandibular gland (Zheng et al. 2012; Satou et al. 2019). However, the exact effects of circadian dysregulation on salivary function and health are still greatly understudied with several unknowns, with no study in the literature having hitherto thoroughly investigated the impact of molecular circadian disruption on salivary function.
This study aims to address this gap by performing a comprehensive analysis of salivary functional gene and protein expression, immune status, glandular structure, and saliva production in 5 mouse knockout (KO) models targeting the following core clock genes: brain and muscle ARNT-like 1 protein (Bmal1) KO, Period2 (Per2) KO, Cryptochrome1 (Cry1) KO, Cryptochrome2 (Cry2) KO, and Cryptochrome 1 and 2 double KO (DKOCry).
Materials and Methods
Mice
This study was approved by the University of Saskatchewan’s Animal Research Ethics Board and adhered to the guidelines set forth by the Canadian Council on Animal Care for humane animal use (protocol 20200010; the approved protocol included the research question, key design features, and analysis plan). We have complied with the ARRIVE 2.0 guidelines (Animal Research: Reporting In Vivo Experiments) for our animal protocol and experimentation (a full checklist is provided in the Appendix). Exclusively female mice aged between 12 and 29 wk were utilized due to the higher prevalence of immune-based salivary gland (SG) pathologies such as Sjögren’s syndrome (SS) in females (Qin et al. 2015). Mice were analyzed from 5 clock gene KO colonies and 1 wild type (WT) colony (serving as control) and categorized as young (12 wk) and middle-aged or aging (29 wk). A total of 48 mice were used (8 mice per genotype group; 4 young and 4 aging). Genotyping protocol and primer sequences are detailed in the Appendix.
Sample Collection
Collections of saliva and blood were scheduled at 2 distinct times: 9:00 to 10:00 a.m. (rest period) and 9:00 to 10:00 p.m. (active period). SGs were harvested, including the submandibular, parotid, and sublingual glands (SLGs). Collection procedures are detailed in the Appendix.
Histologic Staining and Analysis
We utilized hematoxylin and eosin staining, as well as periodic acid–Schiff and alcian blue staining (PAS/AB), to characterize SG structure and morphology (Appendix).
Immunostaining
Anti-CD3 antibody (14-0031-82; Thermo Fisher), anti-amylase (15845-1-AP; Thermo Fisher), and anti-AQP5 (A5-99403; Invitrogen) were used (Appendix).
Quantitative Real-time Polymerase Chain Reaction
We analyzed the gene expression levels of interleukin 17 alpha (IL17A), SS A antigen (SSA/Ro60), tumor necrosis factor alpha (TNFa), Aqp5, amylase (Amy), and mucin 19 (Muc19). Beta-2 microglobulin (B2M) was selected as a reference gene, as it is unaffected by circadian regulation (Appendix).
Quantitative Proteomics Analysis of Mouse Blood Samples
Blood samples from aging mice across all colonies were utilized for the assessment of protein quantities (Appendix).
Statistical Analysis
All statistical analyses were performed by R Studio and SPSS version 29 (Appendix).
Results
Circadian Disruption Effects on Stimulated Saliva Secretion and SG Size
Examination of stimulated salivary secretion uncovered limited circadian-dependent differences after normalizing the saliva levels to body weight. Young Per2KO mice Mean ± SD (5.1538 ± 0.8696 µL/g, P = .041) produced significantly more saliva in the morning as compared with WT, while old Cry1KO mice Mean ± SD (1.5504 ± 0.2547 µL/g, P = .009) had reduced nighttime saliva output (Fig. 1A, B). Gross inspection of the submandibular SGs revealed no significant differences in gland weight among the colonies at 29 wk. In the 12-wk groups, the Bmal1KO glands were noticeably lighter when compared with WT, Per2KO, and CryDKO (P = 0.0922, 0.1101, and 0.0692, respectively; Fig. 1C) with some significant differences in gland area between young clock KO mice and WT controls (Fig. 1D).
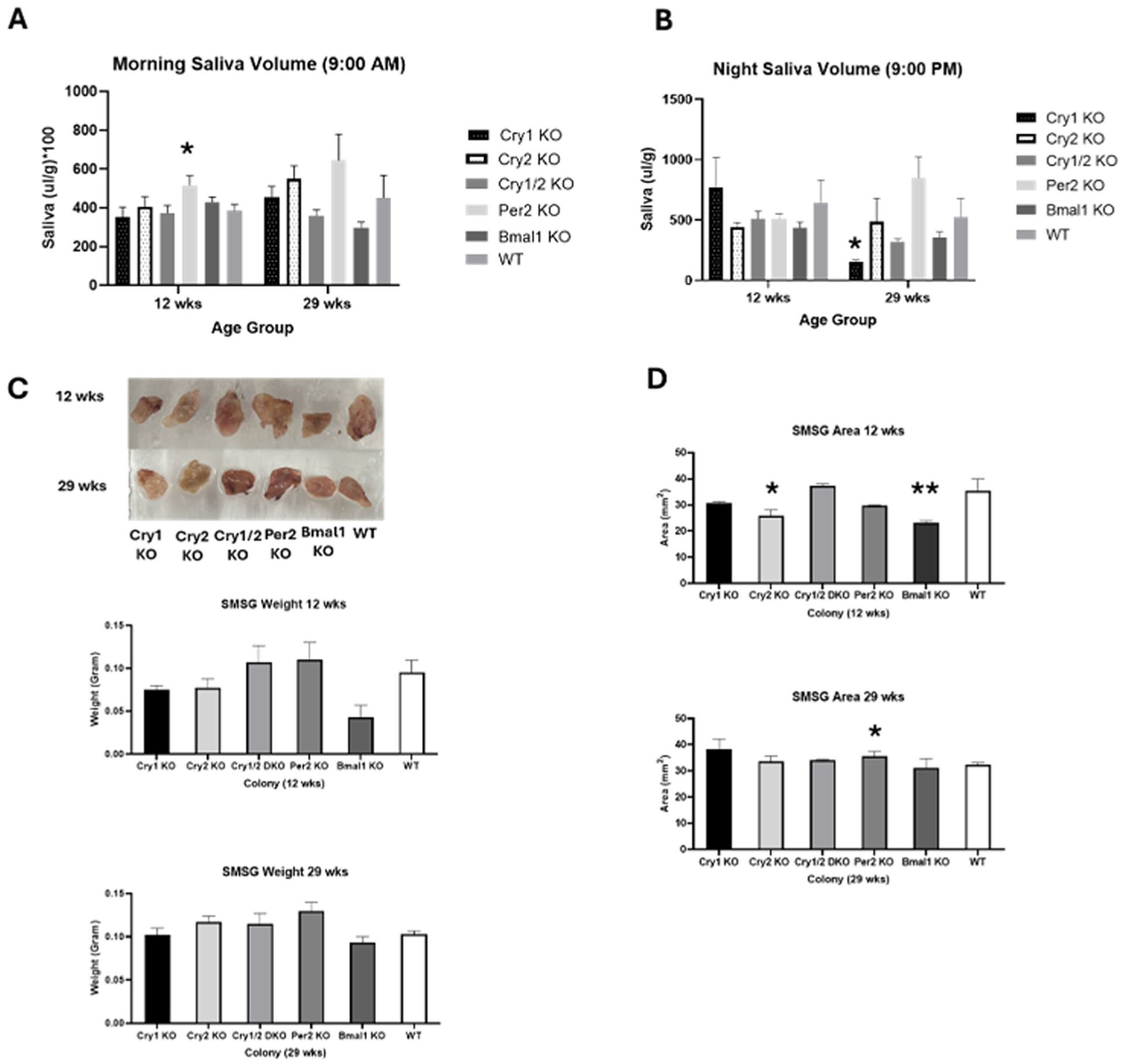
The impact of clock gene knockouts (KOs) on stimulated saliva production and gland size. (
Targeting the Core Clock Genes Resulted in Limited Effects on SG Structure
Analysis of hematoxylin and eosin–stained images did not reveal any major microstructural changes in acinar or ductal cells except for a higher rate of acinar epithelial reduction observed in DKOCry mice (Fig. 2A, B). Focal lymphocytic sialadenitis was assessed by measuring the focus score (mononuclear aggregates per 4 mm2 of glandular area, n = 4 per genotypes; Fig. 2C); generally, our focus scores show that targeting the clock genes, particularly Crys and Bmal1, resulted in mild lymphocytic infiltration that usually does not reach the threshold for pathologic focal lymphocytic sialadenitis. With regard to salivary tissue architecture, our analyses revealed that aging glands show smaller acini accompanied by histologic aspects of atrophy with reduced parenchymal/stromal ratio when compared with their younger counterparts in all of the colonies (Fig. 2D, E). These effects were most pronounced in the Per2KO mice, as older mice showed significantly smaller acini and increased stromal fibrosis when compared with their younger counterparts. No significant differences in acini size and parenchymal/stromal ratio were noted between the clock KO genotypes.
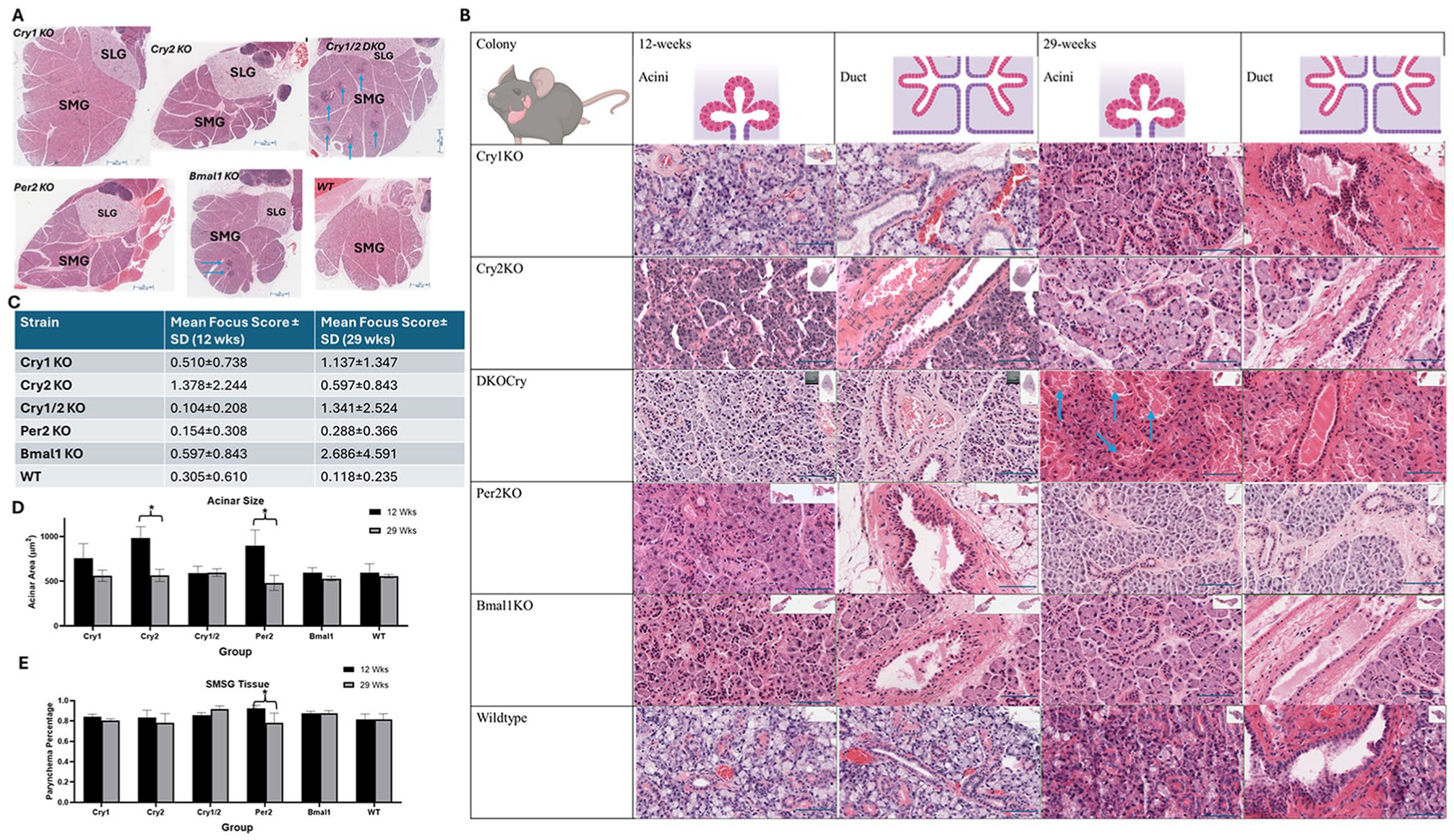
The impact of circadian disruption and aging on salivary gland structure. (
Targeting the Core Clock Genes Differentially Affected Salivary Functional Markers
Our PAS/AB staining and Muc19 gene expression show a substantial increase in acidic mucin production in the SLGs of Per2KO, Bmal1KO, and CryDKO mice when compared with WTs (Fig. 3A–G). Similarly, the 3 colonies showed augmented levels of Amy gene and protein expression in their parotid glands when compared with WTs (Fig. 4A, B). As for Aqp5, only targeting Per2 resulted in a significant reduction of Aqp5 gene and protein expression levels in aging glands (Fig. 4C–E).
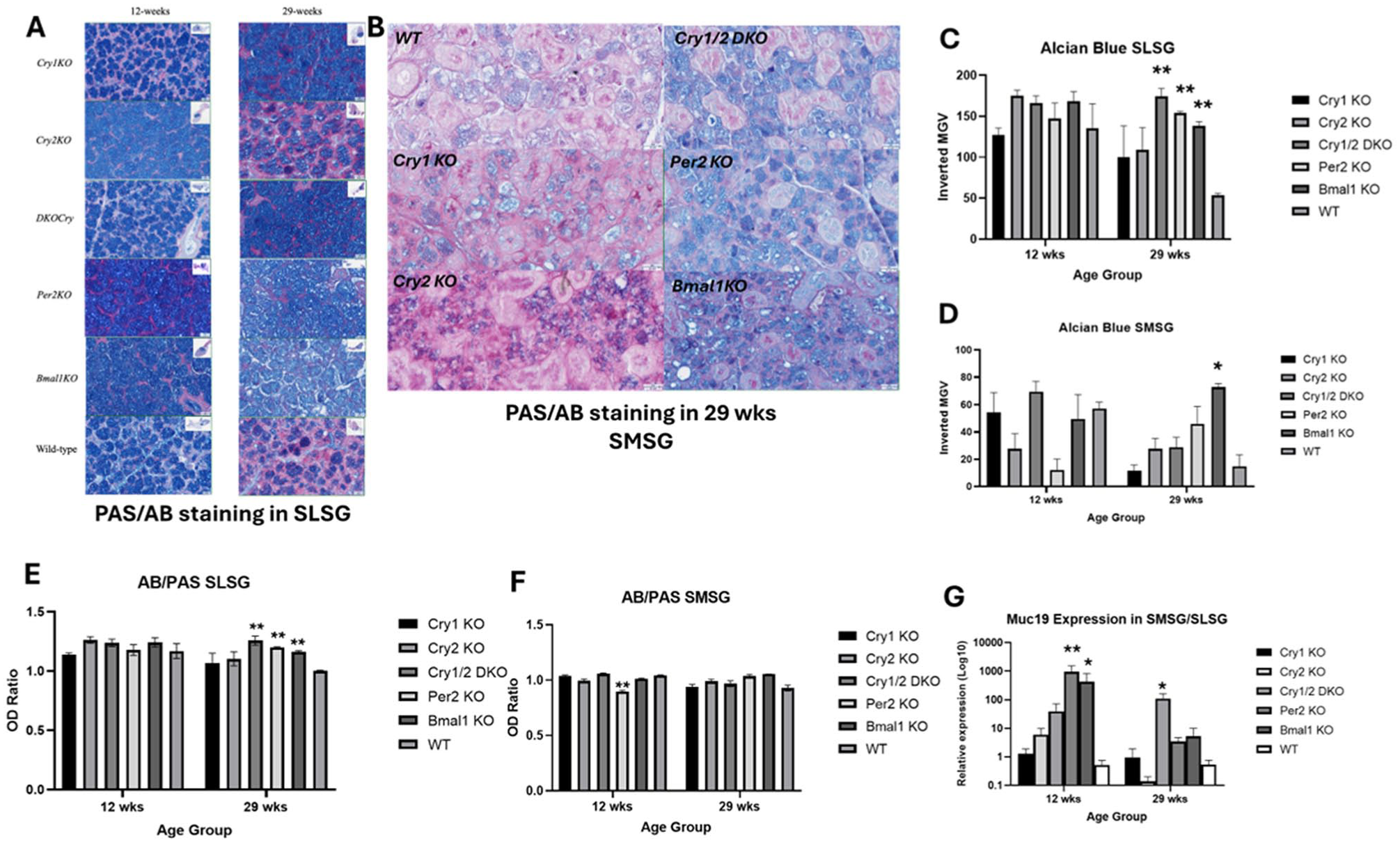
The impact of circadian disruption on salivary mucin production. (
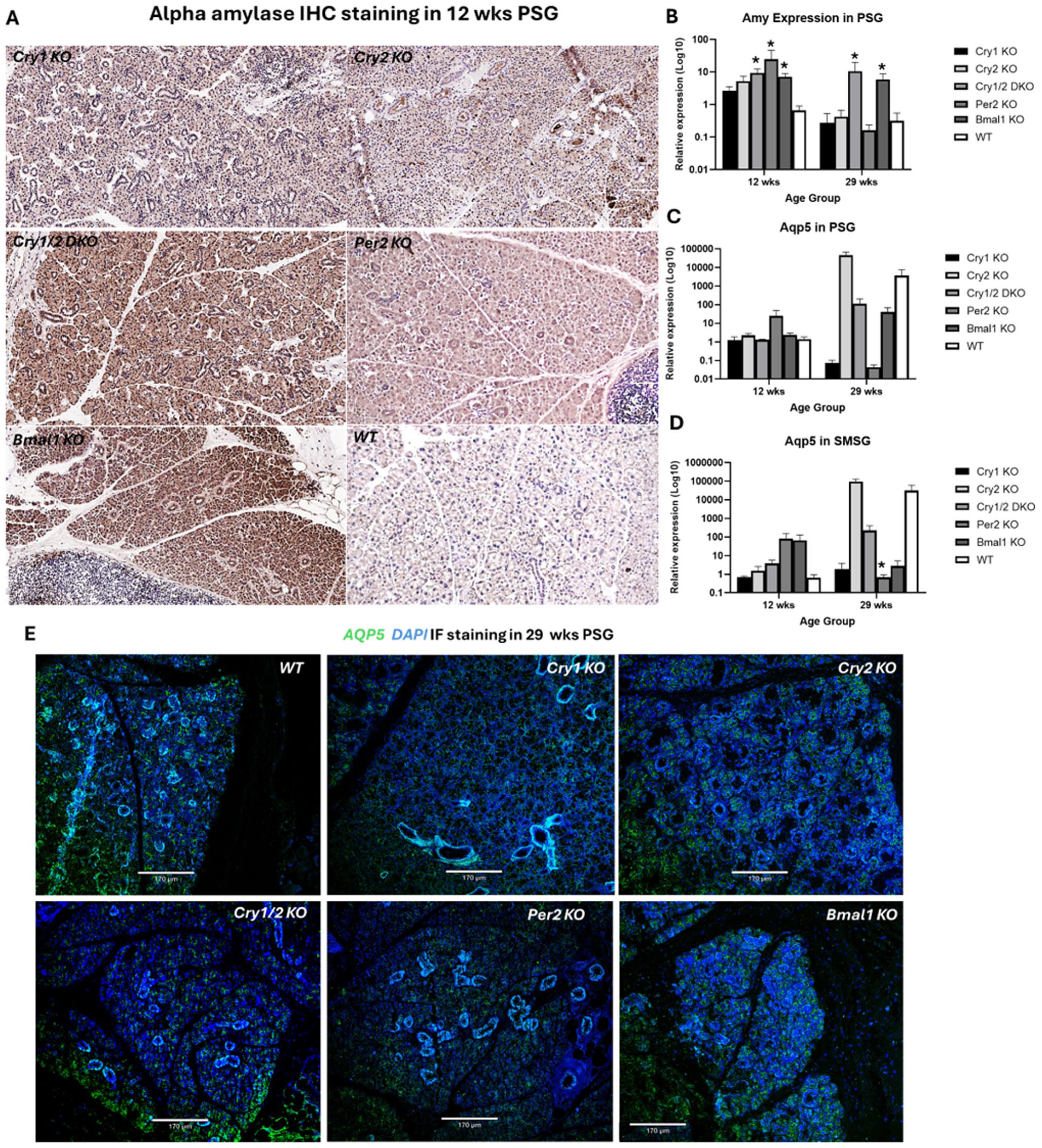
The impact of circadian disruption on salivary functional markers. (
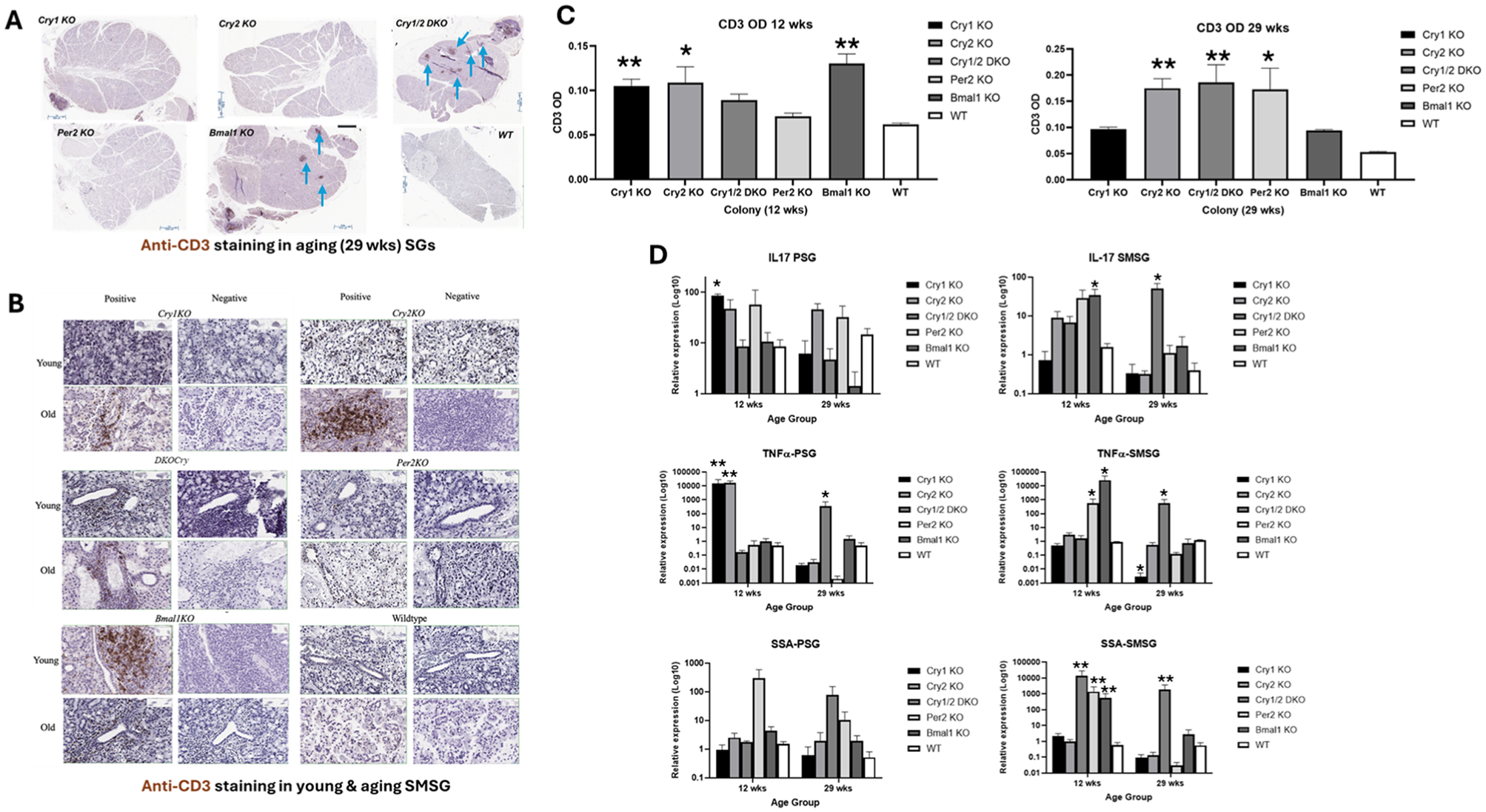
Targeting the different components of the molecular clock effects on immune markers in salivary glands (SGs). (
Targeting Cryptochrome Genes Resulted in Proinflammatory Salivary Features
Our T-cell abundance analysis showed enhanced infiltration of T lymphocytes in the young and aging circadian KO colonies when compared with their WT counterparts with a more pronounced infiltration observed in aging glands (Fig. 5A, B). The colonies with the highest CD3 levels were the young Bmal1KO and aging CryDKO (Fig. 5C). With regard to the cytokine profile, targeting both Cry genes has resulted in significantly higher levels of TNFα, IL-17, and SSA antigen in the submandibular glands (Fig. 5D). Serum proteomics analysis of the aging mice profiling unveiled several altered pathways reflective of immune responses in the different KO models (Appendix Table 2, Appendix).
Discussion
In this study, we unveiled the multifaceted effects of circadian dysregulation on saliva production, glandular structure, functional gene expression, and salivary immune environment.
It is well established that salivary flow follows a distinct circadian rhythm with notable daily fluctuations in salivary flow rate and composition. These rhythms are important for effective nutrition and self-defense functions of saliva, with the flow rate typically increasing during the active phase and decreasing at the rest phase (Nater et al. 2007). Previous studies showed that unstimulated saliva secretion follows circadian rhythms in flow rate and protein, sodium, potassium, calcium, and chloride concentrations in humans (Dawes 1972; Dawes and Ong 1973). However, the impact of dysregulation of the circadian rhythm on salivary secretion has never been investigated. Our data show that targeting different clock genes did not result in clear significant changes in the volume of stimulated saliva collected after parasympathetic stimulation. The lack of significant effects on stimulated salivary flow indicates that targeting the circadian clock does not significantly influence autonomic parasympathetic regulation of salivary function.
Aging SGs in the circadian KO colonies showed increased stromal proliferation (fibrosis), reduced parenchymal tissues, and smaller average acinar size (atrophy) when compared with their younger counterparts. Young Per2KO mice had a smaller mandibular gland with low connective tissue (i.e., high parenchyma/stroma ratio) and larger acini when compared with their WT littermates, while older Per2KO mice demonstrated the exact reverse phenotype. The effects of the circadian rhythms on gland size and structure have been reported in other endocrine and exocrine glandular tissues (Ulrich-Lai et al. 2006; Kalsbeek et al. 2012; Bedrosian et al. 2016; Lee et al. 2022). For example, it was shown that chronic disruption of circadian rhythms (e.g., shift work and exposure to constant light) can lead to long-term changes in adrenal gland size accompanied by alterations in hormone release and feedback mechanisms (Ulrich-Lai et al. 2006). Our hematoxylin and eosin staining did not reveal any major microstructural changes in acinar or ductal cells except for a higher rate of cellular degeneration observed in DKOCry, suggesting that disrupting the circadian clock does not significantly affect SG development.
Our salivary functional analyses implicate the circadian clock in the temporal regulation of the 3 key SG functions: enzymatic digestion, mucus production, and water transport. Indeed, our quantitative real-time polymerase chain reaction and immunohistochemistry analyses revealed that Bmal1KO and DKOCry mice expressed significantly higher levels of Amy when compared with WT in young and aging mice. For the Per2KO mice, the effects appear to be age specific, with significantly higher levels of Amy in younger glands and lower levels in aging glands when compared with WTs. The circadian nature of amylase secretion is very well documented and often aligns with the anticipated time of food intake that is driven by clock gene activity (Nater et al. 2007; Out et al. 2013). The increased amylase production noted in our mice suggests an increased sympathetic drive in these colonies, a phenomenon that is observed in individuals with circadian disruption. Indeed, previous studies have shown that shift work or jet lag can cause increased sympathetic nervous system activation with increased serum norepinephrine levels (Aziz et al. 2021; Wang et al. 2021). The impact of the circadian disruption on amylase was also reported in older adults and attributed to the increased sympathetic drive observed with increasing age and reduced physical activity (Strahler et al. 2010).
Acidic mucin production was significantly upregulated in the aging Bmal1KO, Per2KO, and DKOCry mice. Indeed, our gene expression level analysis of Muc19 (the major gel-forming mucin in the saliva of mice, particularly in SLG) and PAS/AB staining revealed a higher mucin level in the SLG of the 3 KOs when compared with WTs. Unlike amylase, there is limited specific information about the circadian rhythm of salivary mucin expression, and this is the first study showing that salivary mucin production is influenced by the clock genes. Increased production of acidic mucins in the SGs is commonly observed during periods of tissue stress, inflammation, or infection. For example, in CCL28-deficient mice, which have impaired mucosal immunity, acidic mucin production is reduced, and this is associated with increased bacterial invasion (Kaibori et al. 2024). Furthermore, proinflammatory cytokines (e.g., TNF-α and IL-17, which are increased in these colonies) are known to stimulate mucin synthesis and can shift mucin production toward more acidic forms as part of the tissue’s reaction to immune activation and injury (Kaibori et al. 2024). Based on the aforementioned, the observed increase in SLG acidic mucins in the circadian KOs likely reflects a direct effect of clock gene loss on mucin gene regulation and an indirect effect via increased inflammatory signaling and tissue stress.
Our data suggest that the Aqp5 gene is a downstream target of PER2 as Per2KO showed marked decrease in Aqp5 in aging parotid and submandibular SGs. AQP5 water channels provide a major route for osmotically driven water movement in acinar and intercalated ductal cells (Flodby et al. 2017). Our group first demonstrated that the expression levels of Aqp5 showed regular oscillatory patterns under light/dark and complete-dark conditions and Bmal1 overexpression resulted in increased Aqp5 expression levels (Zheng et al. 2012). In a subsequent study, Satou et al. (2019) also observed a circadian rhythmic pattern in Aqp5 mRNA expression in rat whole SGs. Interestingly, the Aqp5 rhythmic expression patterns were synchronous with Per2 expression, which further suggests that Per2 acts as a direct regulator of AQP5 (Satou et al. 2019). Conversely, Uchida et al. (2018) suggested that the daily rhythm of Aqp5 expression is predominantly controlled by the central clock in the suprachiasmatic nucleus rather than by the autonomous salivary molecular clock, suggesting that more studies are needed to conclude how the clock exactly regulates Aqp5 expression. No significant changes in amylase, mucin, or aquaporin expression were noted in Cry1KO and Cry2KO mice, suggesting that elongating or shortening the circadian period (noted in people with morning and night preference chronotypes, respectively) does not appear to significantly alter saliva enzymatic, mucinous, and/or watery composition.
With regard to the immune markers and features in SGs after circadian disruption, we found that all our circadian KO colonies showed a higher degree of T-cell infiltration when compared with WTs. The increase was more pronounced in aging glands, and the colonies with the highest lymphocytic infiltration were the DKOCry (in the aging group) and the Bmal1KO (in the young group). The chronic phlogistic infiltrates were mostly concentrated surrounding interlobular collecting and intralobular striated ducts. The circadian clock plays a substantial role in regulating lymphocyte trafficking and infiltration as lymphocyte migration occurs in a circadian manner (Druzd et al. 2017). Similar to our observed immune phenotype in the aging DKOCry SGs, Cao et al. (2017) reported increased infiltration of leukocytes in adult CryDKO mice lungs and kidneys. Expression of IL-17 and TNF-α was significantly upregulated in the aging DKOCry SGs. The circadian clock plays a crucial role in regulating the expression of IL-17 and TNF-α. Indeed, core clock genes such as retinoic acid receptor–related orphan receptor alpha (Rorα) and nuclear receptor subfamily 1 group D member 1 (NR1D1) were found to induce the expression of IL-17A and IL-17F in Th17 cells (Amir et al. 2018). The interaction between TNF-α and the circadian clock appears to be bidirectional with each influencing the other’s function and expression (Cermakian et al. 2014). It was previously reported that the absence of Cry1 and Cry2 leads to increased levels of serum TNF-α (Cavadini et al. 2007; Cermakian et al. 2014). Our results further consolidate the Cry-mediated regulation of TNF-α as targeting either Cry1 or Cry2, individually or together, resulted in significant upregulation of TNF-α mRNA levels. We also assessed the expression levels of the SSA antigen. Anti-SSA/Ro antibodies are found in approximately 50% to 70% of patients with SS, and their presence is among the classification criteria of the American College of Rheumatology and European Alliance of Associations for Rheumatology for primary SS (Baldini 2022). Similar to IL-17 and TNF- α, the highest upregulation in SSA antigen levels was noted in the aging DKOCry SGs. However, further assessment is still needed to comprehensively evaluate any SS-like pathologic changes. This includes measuring anti-SSA/Ro antibodies in serum samples (which could not be assessed reliably here due to serum sample volume constraints) as tissue expression alone does not fully capture autoimmune status. Additionally, performing flow cytometry or immunostaining on SG tissues is essential to clearly identify and quantify the infiltrating immune cell populations, such as the different T-cell subsets, B cells, germinal centers (BCL2 and 6), and other immune cells (e.g., CD21-positive follicular dendritic cells). In a recent study, Liu et al. (2022) demonstrated the potential links between SS and the clock as they showed that Cry1, Cry2, and Bmal1 gene expression was significantly reduced in the SGs of patients and NOD mice. In addition, they showed that melatonin treatment in NOD mouse models nearly restored clock gene activity in SGs, reduced immune cell infiltration, and decreased proinflammatory responses (Liu et al. 2022).
Each circadian KO model used in this study carries distinct systemic phenotypes, which must be considered when interpreting the observed changes in the SGs. Cry1KO and Cry2KO mice display altered circadian periodicity but generally retain rhythmicity: Cry1KO mice have a shortened circadian period, while Cry2KO mice have a lengthened period. Both are viable and fertile and exhibit only mild metabolic or behavioral changes under standard conditions (van der Horst et al. 1999). In contrast, DKOCry and Bmal1KO mice are completely arrhythmic in constant darkness, with profound systemic effects. DKOCry mice are viable but display metabolic syndrome features, immune dysregulation, and increased tumor susceptibility (van der Horst et al. 1999). Bmal1KO mice exhibit the most severe systemic phenotype, including complete loss of circadian rhythmicity, premature aging, infertility, metabolic syndrome, and reduced life span (Bunger et al. 2000). Per2KO mice typically show a shortened free-running period, but many—especially females—become arrhythmic after prolonged constant darkness, with increased cancer risk and metabolic disturbances (Bae et al. 2001). The phenotypes that we observed in the SGs of these models most likely reflect an interplay of local (gland-intrinsic) circadian disruption and systemic consequences of global clock gene loss. Our findings of altered expression of the salivary markers (i.e., the reduction of Aqp5 in Per2KO glands and increased acidic mucins and amylase) support a direct local role for clock genes in SG homeostasis. However, the immune alterations observed (increased T-cell infiltration and elevated IL-17 and TNF-α) are likely amplified by systemic immune dysregulation characteristic of global circadian disruption. Indeed, DKOCry and Bmal1KO mice are known to develop systemic immune abnormalities, including heightened autoimmunity and altered cytokine profiles, which can predispose to or exacerbate local glandular inflammation. The observed increase in lymphocytic infiltration and proinflammatory gene expression in these models’ SGs is consistent with this systemic immune activation.
In conclusion, our comprehensive investigation has unveiled a complex interplay between circadian rhythms and salivary function, with potential implications for SG immune regulation. While the specific mechanisms may vary among species, the fundamental role of the circadian clock in regulating physiologic processes is conserved across mammals, including humans. Therefore, it is plausible that similar disruptions in circadian rhythms could affect human SG health and immune function. Our findings open new avenues for understanding SG physiology and for the identification of clock genes as key regulators of SGs, rendering them an excellent potential therapeutic target for chronotherapy in salivary disorders.
Author Contributions
H. Mortazavi, R. Said, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; G.S. Katselis, P. Chumala, contributed to data acquisition, critically revised the manuscript; G. Pannone, contributed to data interpretation, critically revised the manuscript; S. Papagerakis, P. Papagerakis, contributed to conception and design, data interpretation, drafted and critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251372506 – Supplemental material for Effects of Circadian Misalignment on Young and Aging Salivary Glands
Supplemental material, sj-docx-1-jdr-10.1177_00220345251372506 for Effects of Circadian Misalignment on Young and Aging Salivary Glands by H. Mortazavi, R. Said, G.S. Katselis, P. Chumala, G. Pannone, S. Papagerakis and P. Papagerakis in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the University of Saskatchewan’s VPFR–Centennial Enhancement Chair in One Health. H.M. received an Anatomy, Physiology, and Pharmacology devolved scholarship from University of Saskatchewan during this research.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
