Abstract
Aging is a systemic immunomodulatory condition that makes organisms more susceptible to infection-driven periodontitis and associated periodontal tissue loss. Therefore, modulation of the bacterial-induced inflammatory host response could potentially target the pathophysiological systemic and local crosstalk and resulting tissue homeostasis in aged organisms. However, underlying pathways connecting nutritional modulation with periodontal disease pathology and whether an aged organism benefits from specific immunomodulatory nutritional components are not known. Based on its potent immunomodulatory function, we determined whether nutritional interventions with dietary monounsaturated fatty acid (FA) oleic acid (OA [C18:1]), a main component of Mediterranean-style diets, as compared with a Western-style diet component saturated FA palmitic acid (PA [C16:0]) could modify the response to periodontal infection in a murine periodontal Porphyromonas gingivalis–inoculation model comparing old (final age of up to 2 y) and young (21 wk) mice. Local fibroblastic response and circulating regulatory immune-competent lipid mediators (LMs) were determined to provide insights into underlying pathways and how diet and age affect inflammation and resolution of the periodontal infection. The dietary intake of PA enhances periodontal bone destruction in response to P. gingivalis infection in old mice, whereas an OA-enriched diet (ED) has a protective effect. Specifically, PA-ED enhances the inflammatory profile of the periodontal microenvironment and primes gingival fibroblasts, prominent regulators within local tissue homeostasis, toward an inflammatory phenotype. Moreover, systems-wide serological lipidomic analyses of LMs and their respective stimulation of osteoclast differentiation revealed 10-hydroxydocosahexaenoic acid, associated with OA intake, as a protective candidate for age- and nutrition-dependent regulation of osteoclast-driven bone loss. Given the rising prevalence of periodontitis in the aging population, incorporating OA-rich foods could offer promising strategies for addressing the sequelae of aging and promoting periodontal health.
Keywords
Introduction
In periodontal disease (PD), a dysbiotic oral microbiome in association with hyperinflammatory host response and nonresolving inflammation fosters a microenvironment with host-mediated destruction of tooth-supporting tissues, including gingival tissues, periodontal ligament (PDL), and alveolar bone. Subsequently, inflammation and dysbiosis reinforce each other in a vicious cycle (Lamont et al. 2018). PD is especially prevalent in aged individuals, and due to demographic developments with growth in the aging population, it will increase further in coming years (Trindade et al. 2023). Various systemic factors modulate periodontitis risk (Darby 2022) by either influencing dysbiosis or the inflammatory host response. Aging is an immunomodulatory systemic condition that makes organisms more susceptible to infection-driven loss of tissue homeostasis, thereby supporting periodontitis prevalence in aged organisms (Trindade et al. 2023). Therefore, novel supportive treatments could focus on immunomodulation, and recent human and animal studies highlighted nutrition as a potent immunometabolic intervention to reduce inflammatory tissue damage (Bartha et al. 2022; Kruse et al. 2022; Doding, Hufner, et al. 2023).
Prominent representatives of anti-inflammatory nutrition include Mediterranean-style diets (MD) that offer many health benefits (Bartha et al. 2022; Kruse et al. 2022; Doding, Hufner, et al. 2023). They are characterized by a high intake of plant food, with oleic acid (OA)–rich olive oil as main fat source, moderate dairy, and limited fish and poultry consumption. Monounsaturated fatty acid (FA) OA (C18:1) serum levels correlate negatively with periodontal tissue loss in patients following an anti-inflammatory diet (Kruse et al. 2022). In contrast, Western-style diets (WD) high in saturated fats, processed meat, red meat, butter, high-fat dairy products, eggs, and refined grains (Clemente-Suarez et al. 2023) are considered “sterile” proinflammatory stimuli (Christ et al. 2018). WD contain high levels of saturated FAs, specifically palmitic acid (PA [C16:0]) correlating positively with PD (Ramirez-Tortosa et al. 2010).
In particular, diets high in saturated FAs such as PA have been linked to disrupted lipid and bone metabolism, contrary to MD-style OA-enriched diets (OA-ED) (Drosatos-Tampakaki et al. 2014). In a murine oral infection model with periodontitis key pathogen Porphyromonas gingivalis, young obese and normal-weight OA-fed mice displayed comparatively lower inflammation, less alveolar bone loss, and enhanced osteoclast abundancy in femoral bone as compared with mice on isocaloric WD-style PA-enriched diets (PA-ED) enriched diets (Muluke et al. 2016; Doding, Zimmermann, et al. 2023).
PA is metabolized to ceramides, lysophosphatidylcholine, and cytotoxic diacylglycerides, inducing inflammatory reactions through different mechanisms, potentially resulting in apoptosis (Drosatos-Tampakaki et al. 2014). OA not only reduces inflammation (Vassiliou et al. 2009) but mitigates lipotoxicity by promoting triglyceride sequestration and storage, preventing tissue accumulation of harmful lipid-intermediates, and, consequently, reducing osteoclast differentiation (Drosatos-Tampakaki et al. 2014). In osteoblasts, conversion from PA to OA-treatment restores differentiation capacity and reduces IL-6–dependent osteoclastic differentiation (Drosatos-Tampakaki et al. 2014; Doding, Zimmermann, et al. 2023). Furthermore, OA-ED is linked to increased circulating proresolving molecules, including docosahexaenoic acid (DHA)–derived specialized proresolving mediator (SPM) resolvin D4 (RvD4) and DHA-metabolite 4-hydroxydocosahexaenoic acid (4-HDHA), which improved oral trabecular bone in young normal-weight animals inoculated with P. gingivalis (Doding, Hufner, et al. 2023). 4-HDHA and RvD4 directly improved osteoblast mineralization in inflammatory conditions (Doding, Zimmermann, et al. 2023), whereas PA incubation increased bone-resorbing osteoclast activity (Drosatos-Tampakaki et al. 2014), ultimately affecting bone homeostasis within the local microenvironment. Human studies highlighted SPM’s proresolving activity, reporting elevated SPM markers after successful therapy in the gingiva of periodontitis patients (Ferguson et al. 2020).
Resident cells such as gingival fibroblasts (GFbs) are indirect modulators of oral bone integrity and inflammatory response. Thus, GFbs define complex signaling networks such as cytokines interleukin (IL)–6, IL-1β, tumor necrosis factor–α (TNF-α), and prostaglandin E2 (PGE2) production (Wielento et al. 2023) and secretion of bone modulators (e.g., osteoclastic stimulator receptor activator of NF-κB ligand [RANKL]/osteoclastogenesis inhibitor osteoprotegerin [OPG]) within the local microenvironment in periodontal infection. Proinflammatory TNF-α promotes cell migration, tissue destruction, and osteoclast activation (Garlet 2010) and is elevated in lipopolysaccharide (LPS)–stimulated human GFbs and gingival tissues of periodontitis patients (Kim et al. 2022).
PA induces TNF-α–mediated inflammation and osteoclastogenesis, highlighting its role in periodontitis pathogenesis (Muluke et al. 2016). Furthermore, GFbs express CXCR2-chemokines (CXCL1, CXCL2) that recruit immune cells, such as neutrophils, to the site of inflammation (Williams et al. 2021), underscoring their crucial role in periodontitis progression and tissue homeostasis.
Increased periodontitis prevalence with age results in global health issues (Trindade et al. 2023). Generally, immunosenescence and inflammaging reflect immune system changes that affect tissue homeostasis and cellular function, increasing periodontitis susceptibility in age (Ebersole et al. 2016; Lee et al. 2022). Moreover, immunosenescence drives a rise in senescent cells exhibiting a proinflammatory secretory phenotype, with IL-6 contributing to immune dysfunction and driving chronic inflammation (Almanan et al. 2020; Lee et al. 2022).
Personalized treatments aim to address the sequelae of aging using modifiable factors that affect local and systemic disease crosstalk. Nutritional strategies could support individualized periodontal patient care since WD with high levels of saturated fats are common. Notably, diets enriched with saturated fats are associated with PD progression in the elderly (Iwasaki et al. 2011). Still, regulatory pathways serving as basis for preventive algorithms and cellular response patterns in periodontal tissue require investigation. Whether aged organisms could benefit from specific nutritional components or substitution is not known. Based on potency in immunomodulation, we determined whether interventions with dietary OA compared with a WD-component PA modify periodontal infection response and alveolar bone loss. Fibroblastic response and circulating immune-regulatory lipid mediators (LMs) were determined to provide insights into defining pathways and how diet and age drive the resolution of periodontal infection in age.
Materials and Methods
Results
Dietary PA Increases Bone Loss in Older Mice, Whereas OA Protects
To investigate the age-dependent influences of FAs on oral bone in infection, young (5 wk) and old (78 wk) C57BL6 mice received identical diets either supplemented with PA (PA-ED), OA (OA-ED), or a normal standard diet (ND) for 16 wk and were infected with P. gingivalis/placebo 3 times per week for 5 weeks. All groups demonstrated similar weight gain and were normal weight (Appendix Fig. S2A). While infection had no effect, young OA-ED animals were significantly heavier compared with ND animals (Appendix Fig. S2B). In young animals, serological cholesterol (Appendix Fig. S2C) and triglyceride levels appeared independent from nutritional intake (Appendix Fig. S2D). Although cholesterol (Appendix Fig. S2C) and triglyceride levels (Appendix Fig. S2D) were significantly enhanced in Aged infected PA-ED (PA-EDo/P.g.) animals compared with NDo/P.g., no differences in the infected FA groups (PA-EDo vs. OA-EDo) were detectable. However, young animals on OA-ED (OA-EDy) showed significantly improved bone microarchitecture compared with NDy and PA-EDy in response to oral P. gingivalis infection (Fig. 1A, B). In old P. gingivalis-infected animals, bone volume/total volume remained stable in OA-ED conditions, whereas PA-ED significantly reduced the bone microarchitecture in periodontally infected animals as compared with ND (Fig. 1B).
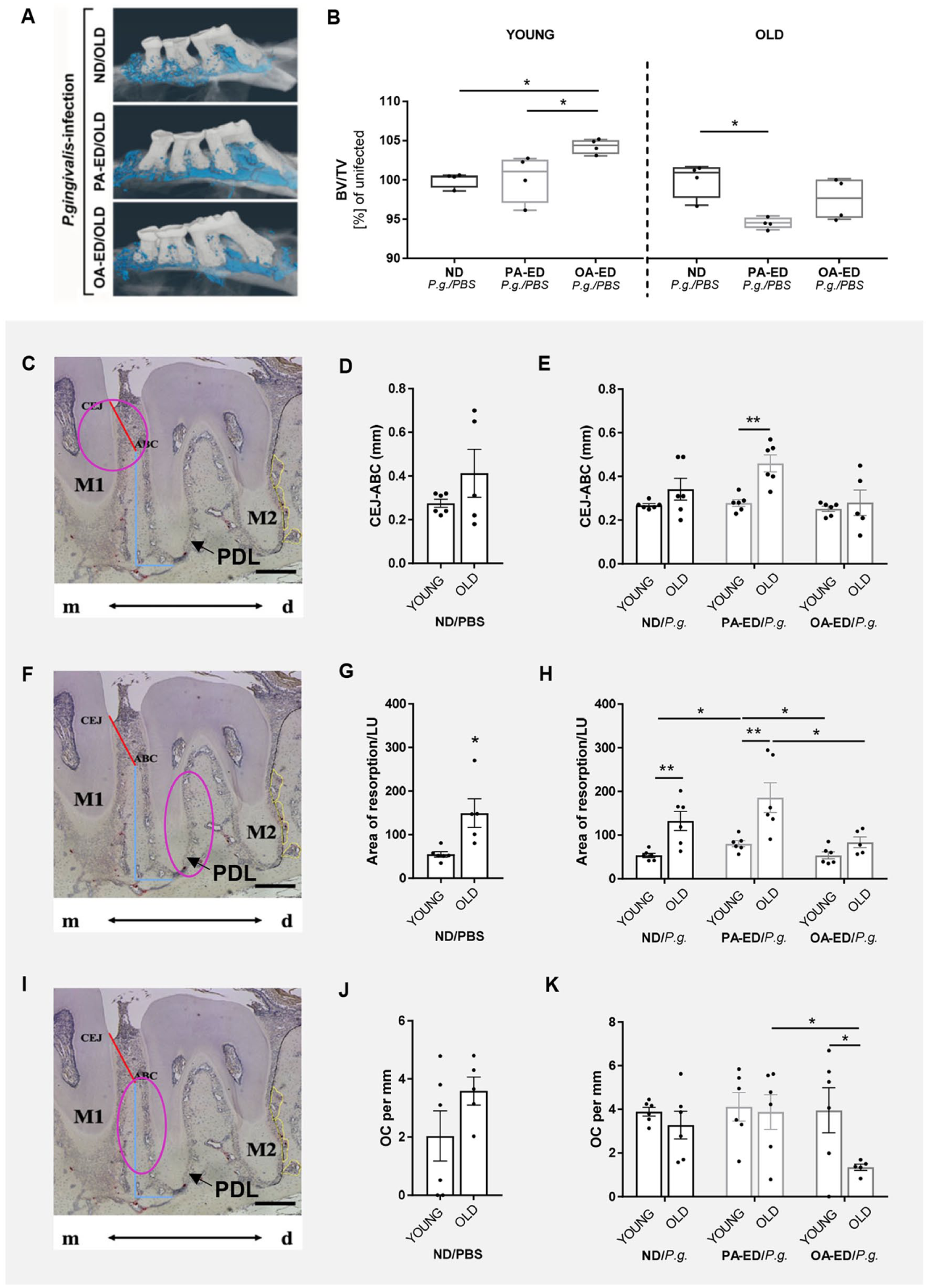
Palmitic acid exacerbates age-driven bone loss, while oleic acid supports bone integrity in periodontal infection. Young (21 wk) and old (>20 mo) C57BL6JRj were fed with normal diet (ND), a palmitic acid–enriched diet (PA-ED), or an oleic acid–enriched diet (OA-ED) for 16 wk and infected with control/placebo (PBS) or Porphyromonas gingivalis (P.g.) for 5 wk. (
Histomorphometry revealed that the distance from the cementoenamel junction to the alveolar bone crest (CEJ-ABC) at the bone peak between the first and second molars (Fig. 1C) remained unchanged during aging in noninfected (Fig. 1D) and infected ND groups (Fig. 1E). However, PA-ED intake significantly intensified P. gingivalis–induced bone resorption in older animals compared with young animals, whereas OA-ED intake did not (Fig. 1E).
Resorption lacunae between M2 and M3 (Fig. 1F) increased age dependently in the bone surrounding the PDL space (Fig. 1G). Furthermore, in young groups, P. gingivalis infection of PA-ED mice (PA-EDy/P.g.) significantly enlarged the cumulative lacunae area compared with NDy/P.g. and OA-EDy/P.g. mice (Fig. 1H). Of note, old OA-ED animals inoculated with P. gingivalis (OA-EDo/P.g.) presented with lower cumulative lacunae areas compared with PA-EDo/P.g. (Fig. 1H). Overall, the resorption area was significantly larger in aged animals under NDo/P.g. and PA-ED/P.g. as opposed to young equivalents (Fig. 1H).
Although tartrate-resistant acid phosphatase (TRAP)–stained bone-resorbing osteoclasts (OCs) in the mesial M2 root region (Fig. 1I) demonstrated no impact of age in control conditions (Fig. 1J), OA-ED intake significantly reduced osteoclast numbers in OA-EDo/P.g. compared with OA-EDy/P.g. animals (Fig. 1K). In this context, OA-EDo/P.g. depicted significantly reduced osteoclasts (OCs) numbers compared with PA-EDo/P.g. animals, underlining OA-ED’s protective properties in age (Fig. 1K).
Dietary FAs Stimulate the Expression of Bone-Regulatory Markers across Age Groups
To determine whether inflammatory and bone homeostatically active cytokines, prominent regulators in periodontal cascades, could be targets for nutritional immune modulatory lipids at different ages, the expression was determined in gingivae overlaying the alveolar bone and periodontal defects. Young and old mice were fed ND or FA-ED for 16 wk and orally infected with P. gingivalis or placebo. Four days after the final infection, gingival expression patterns were examined in young (Fig. 2A–E), in age comparison (Fig. 2A′–E′), and in aged animals (Fig. 2A″–E″). Gingival Ifng expression appears unaffected by nutrition, age, or infection (Fig. 2A/A′/A″), whereas PA-EDy animals exhibited an infection-dependent increase in gingival Cxcl2 expression (Fig. 2B) and a trend toward increased Il1b (P = 0.06) (Fig. 2C), leading to the highest Cxcl2 (Fig. 2B) and Il1b (Fig. 2C) expression levels in PA-EDy/P.g. animals compared with NDy/P.g. and OA-EDy/P.g. In contrast to infected PA-EDy groups, the dietary intake of OA-ED appeared to decrease Il1b expression in young P. gingivalis–infected OA groups (P = 0.08) (Fig. 2C). Of note, the gene expression levels were comparable in young and old ND/control/placebo (PBS) animals (Fig. 2B′/C′), and FA- and infection-induced changes were completely abolished in aged animals (Fig. 2B″/C″). Dietary intake of PA significantly increased Il6 in aged PA-EDo/PBS groups compared with ND and OA-ED groups (Fig. 2D″). Conversely, Tnf expression was downregulated in OA-EDy/P.g. groups compared with PA-EDy/P.g. and respective controls (Fig. 2E).
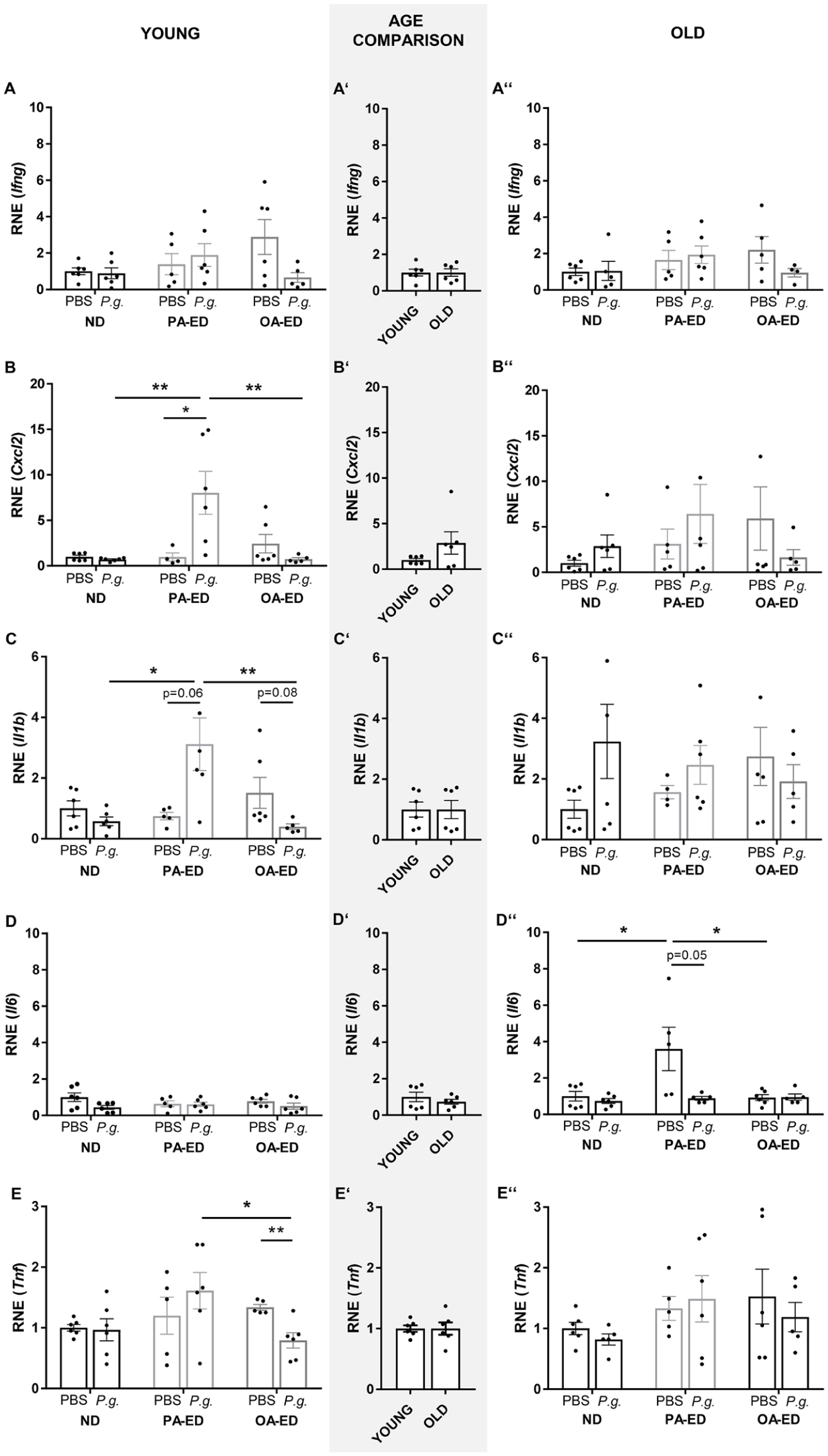
Age and diet influence the expression of inflammatory and bone-regulatory markers. Quantitative polymerase chain reaction (qPCR) analyses of gingival tissue samples of the respective groups are shown in age comparison. Young (21 wk) and old (>20 mo) C57BL6JRj were fed with different diets (normal diet [ND], palmitic acid–enriched diet [PA-ED], or oleic acid–enriched diet [OA-ED]) for 16 wk and infected with control/placebo (PBS) or Porphyromonas gingivalis (P.g.) for 5 wk. (
Dietary Lipid-Metabolic FAs Foster a Specific Inflammatory Response in Gingival Fibroblasts Derived from Young and Old Mice
GFbs, prominent in gingiva overlaying alveolar bone, are part of the intricate microenvironment involved in the responsiveness to bacterial insult and the subsequent shift in local tissue homeostasis (Christ et al. 2018). To evaluate FA’s impact on the inflammatory profile and address age-specific modulation, GFbs were isolated from young (4 wk) and old (>18 mo) mice and supplemented with the respective FAs (Fig. 3A) in the presence of periodontal pathogen P. gingivalis–LPS. PA alone increased Il6-gene expression compared with OA pretreatments in fibroblasts derived from young and old mice (Fig. 3B). LPS treatment enhanced the Il6 expression in young PA-treated cells and induced an increased trend in OA-challenged conditions (OA/ctrl vs. OA/P.g., P = 0.06) compared with their respective controls (Fig. 3B). Although old cells showed increased Il6 and Cxcl2 expression in PA/ctrl compared with OA/ctrl cells, additional LPS stimulation increased proinflammatory Il6 and Cxcl2 levels following FA pretreatment compared with respective placebos, with a general trend toward lower levels in fibroblasts derived from older mice across all experimental groups (Fig. 3B, C).
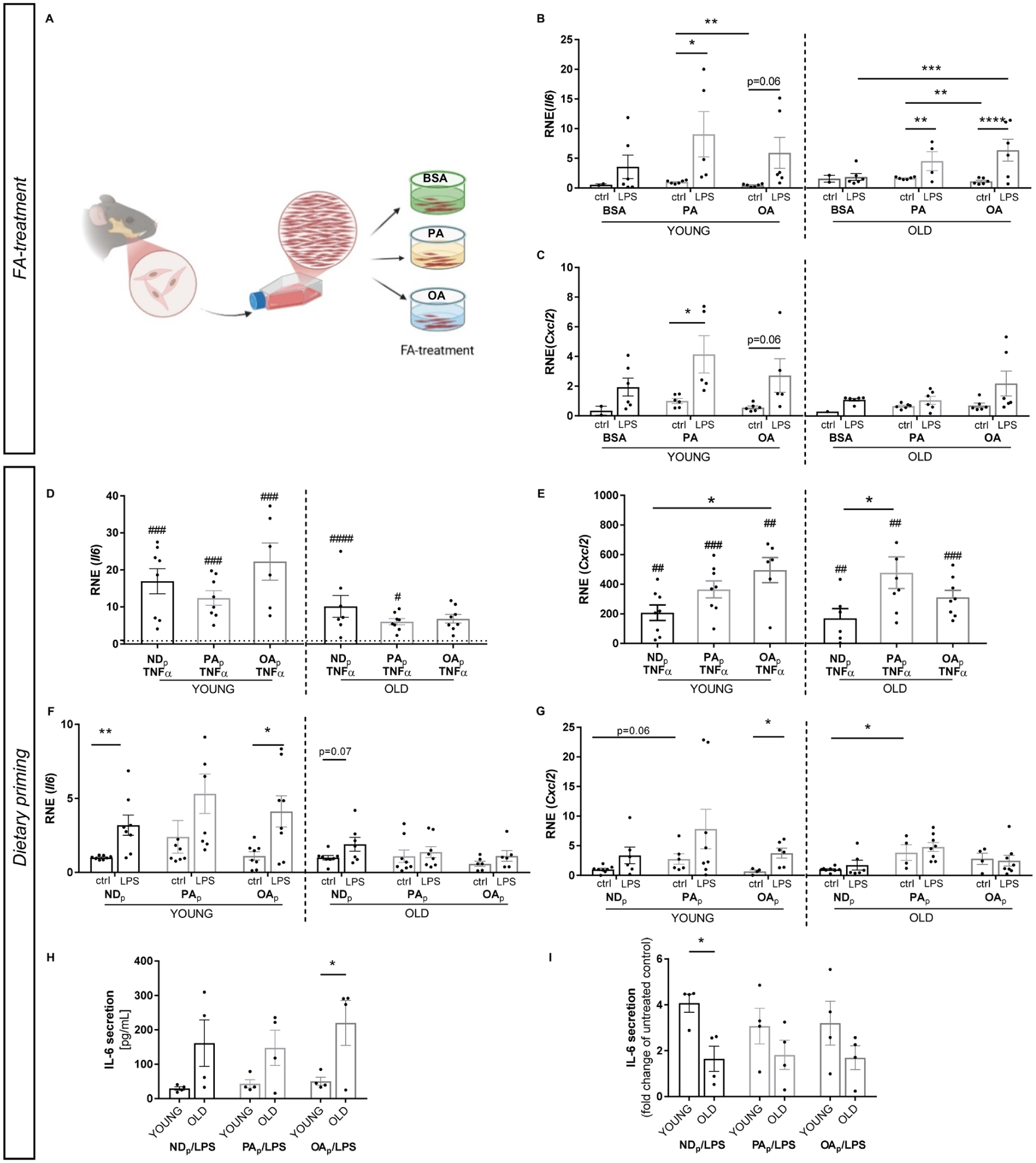
Intake of a palmitic acid (PA)–enriched diet amplifies the inflammatory response in gingival fibroblasts, with age-specific shifts in cytokine dynamics. (
Dietary Intake Highlights Age-Specific Shifts in Cytokine Dynamics
To investigate whether consumption of immune modulatory FAs induces lasting cellular changes or predominantly alters cellular environment in age, in vitro results were complemented by using nutritionally primed GFbs from young and old donors. For this purpose, mice received ND, PA-ED, or OA-ED for 8 wk before GFbs were isolated (Appendix Fig. S1).
To assess whether diets modulate the response in basal inflammatory conditions, incubation with TNF-α was used to mimic a proinflammatory microenvironment. TNF-α stimulation increased Il6 expression in GFbs from young and old mice without detectable changes based on dietary priming in both age groups (Fig. 3D). In fibroblasts derived from young OA-ED–fed animals, Cxcl2 expression was upregulated compared with NDp, while PA-ED (PAp) elevated Cxcl2 expression in older mice compared with NDp (Fig. 3E). Furthermore, TNF-α treatment increased Cxcl2 expression in both age groups (Fig. 3E). Overall, under inflammatory stimulation, Il6 expression was strongly age dependent, whereas Cxcl2 levels changed more in response to priming.
Next, cells were stimulated with P. gingivalis–LPS, and their inflammatory profile was determined. Although no significant differences in the basal Il6 expression of PA-ED–derived (PAp) GFb were detectable (Fig. 3F), Cxcl2 levels were enhanced in dietary primed cells from young and old individuals (young: NDp vs. PAp, P = 0.06) (Fig. 3G). LPS infection increased Il6 expression in young NDp and OAp cells (Fig. 3F). Even though no significant changes in Il6 levels in response to LPS stimulation occurred based on dietary priming, PA-ED (PAp)–derived fibroblasts showed a trend toward higher Il6 expression, specifically in younger cells compared with other dietary groups (Fig. 3F). Thereby, NDp and OAp cells exhibited increased Il6 expression, whereas basal and stimulated expression levels of Il6 were not different in PAp cells (Fig. 3F). In line, OAp-GFbs responded to LPS stimulation with an increased Cxcl2 expression, whereas transcription levels were already higher in PAp cells in basal conditions without significant increase upon LPS stimulation (Fig. 3G). Contrarily, aged cells did not exhibit inflammatory Il6 (Fig. 3F) and Cxcl2 (Fig. 3G) responses to LPS stimulation independent of dietary priming. However, aged PA-ED–primed GFbs demonstrated an increased inflammatory Cxcl2 expression in basal conditions compared with NDp (Fig. 3G).
In addition, cytokine secretion of stimulated GFbs was determined to assess the secretory impact on the microenvironment. Of note, fibroblasts derived from young PA-ED and ND groups appeared to exhibit higher basal IL-6 secretion levels compared with the respective aged equivalents, turning significant in OAp cells (Fig. 3H). LPS treatment induced a higher inflammatory response in cells derived from young donors compared with cells of an aged origin, without changes being associated with dietary priming (Fig. 3I).
Dietary OA Enhances Inflammation Resolution in P. gingivalis Infection
Older mice on OA-ED showed stabilized bone microarchitecture and fewer osteoclasts around PDL when exposed to P. gingivalis infection. Since our previous study highlighted the importance of circulating serum LM such as SPMs in regulating the local cellular microenvironment (Doding, Hufner, et al. 2023), we performed systems-wide lipidomic profiling in the serum of all groups (Fig. 4A).
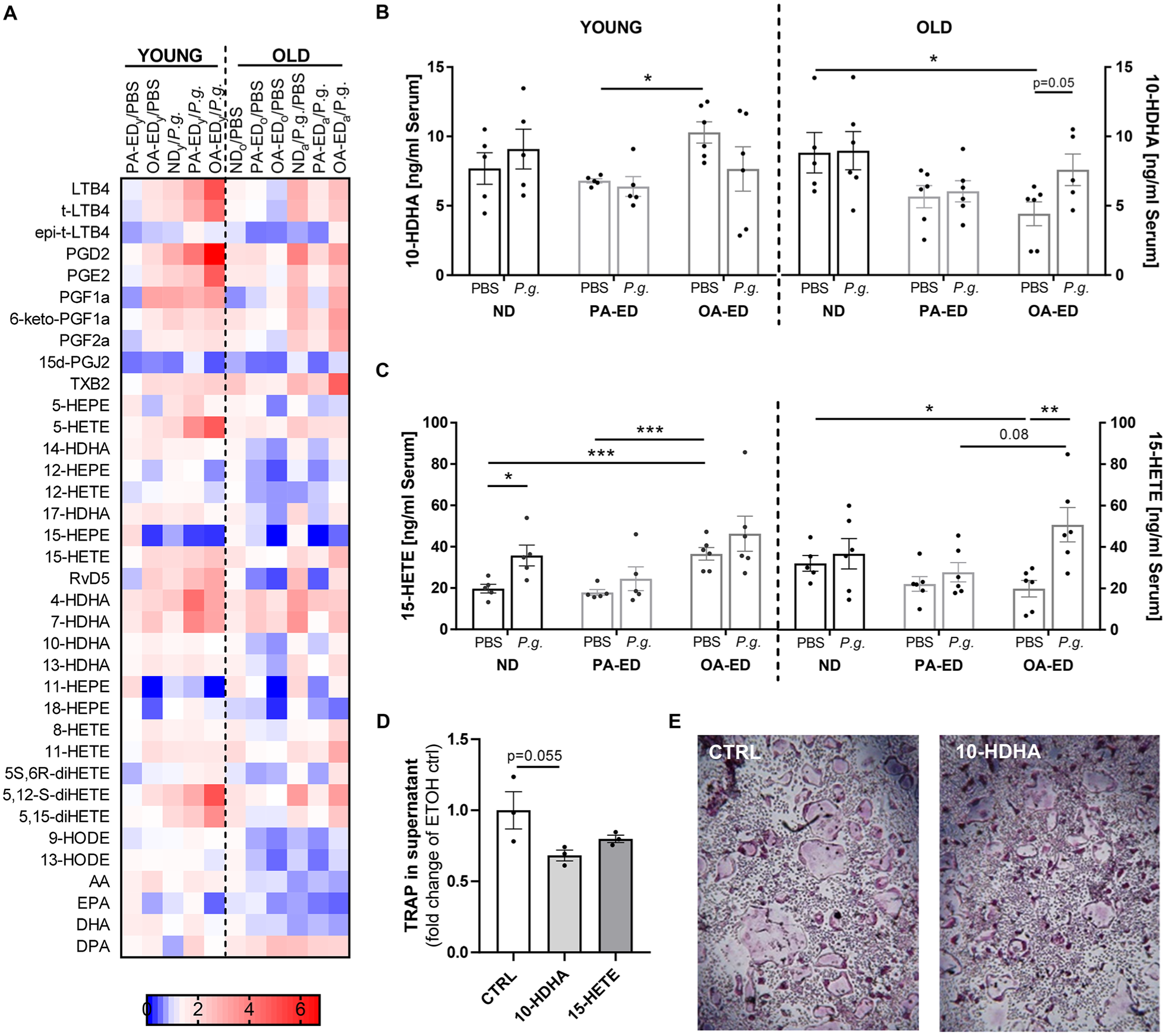
Dietary oleic acid enhances inflammation resolution during P. gingivalis infection, while the docosahexaenoic acid (DHA) metabolite 10-hydroxydocosahexaenoic acid (10-HDHA) reduces osteoclast differentiation. (
The depicted heat map compares different diet and age groups and provides a visual representation of LM concentrations (Fig. 4A). The LM profiles of aged mice show reduced LM secretion after P. gingivalis infection compared with young animals. This is particularly detectable in inflammatory LMs such as PGE2 and PGD2 (Fig. 4A). Moreover, arachidonic acid, the main substrate for proinflammatory mediators, appeared in lower levels in the serum of aged animals (Fig. 4A).
Differences in various LMs based on diet, age, and infection prompted a focus on specific LM classes in subsequent analyses. Although serum levels of anti-inflammatory 10-hydroxydocosahexaenoic acid (10-HDHA) increased under OA-ED in young animals compared with PA-ED (OA-EDy/PBS vs. PA-EDy/PBS), the dietary intake of OA-ED reduced serum levels of 10-HDHA in old animals compared with ND (OA-EDo/PBS vs. NDo/PBS) (Fig. 4B). However, after infection with P. gingivalis, 10-HDHA levels tended to increase in old animals (old: OA-EDo/PBS vs. OA-EDo/P.g.: P = 0.05) but appeared stable or even reduced in young mice (Fig. 4B). OA-ED reduced SPM precursor 15-hydroxyeicosatetraenoic acid (15-HETE) in old animals compared with ND (Fig. 4C). However, in response to bacterial inoculation, old animals exhibited a significant increase in 15-HETE under OA-ED compared with respective controls (Fig. 4C). Lipidomic bone analyses depicted age- and nutrition-dependent changes in femoral LM composition (Appendix Fig. S3A). A comparison between serum and femur showed similar trends associated with nutrition and infection between groups, although the impact was not as pronounced in bone (Appendix Fig. S3B/C).
10-HDHA Reduces Osteoclast Differentiation
To investigate whether respective circulating lipid mediators directly affect cellular resorptive function and gain more insight into the connection between bone metabolism and periodontal homeostasis, primary preosteoclasts were differentiated in the presence of DHA metabolite 10-HDHA and the SPM precursor 15-HETE. The respective TRAP secretion was reduced under 10-HDHA treatment compared with the control conditions (Fig. 4D), which was also reflected in morphological appearance (Fig. 4E) and demonstrated potency of 10-HDHA in regulating cellular differentiation. 15-HETE, an LM increased in inflammatory periodontal conditions (Elabdeen et al. 2013), had no significant effect on TRAP secretion (Fig. 4D).
Discussion
Aging, as systemic condition, increases susceptibility to infection-associated loss of tissue homeostasis, thereby contributing to a high prevalence of periodontitis in age. Our study demonstrated, that the intake of PA-ED by old animals—contrary to young animals—exacerbates the effect of P. gingivalis infection on oral bone. Furthermore, PA-ED elevates both the basal and reactive inflammatory profile of the periodontal microenvironment and primes gingival fibroblasts, key regulators within local tissue homeostasis, toward an inflammatory phenotype. Moreover, serological analyses of LM and their respective stimulation of osteoclast differentiation identified 10-HDHA as protective candidate for age- and nutrition-dependent regulation of osteoclast-driven bone loss.
Periodontitis-associated tissue damage is primarily driven by the host inflammatory response and its resolution (Doding, Zimmermann, et al. 2023). WD, which is high in saturated fats such as PA, sugar, and processed foods, promotes inflammation (Peikert et al. 2024), and epidemiological studies have linked elevated circulating C16:0 levels to increased periodontal attachment loss (Ramirez-Tortosa et al. 2010). In contrast, MD, rich in plant-based foods and olive oil, is considered a supportive therapy for inflammatory diseases (Tsigalou et al. 2020). MD reduces inflammation markers, lowers disease risk, is associated with reduced mortality (Sureda et al. 2018), and may mitigate periodontitis-associated inflammation. Our findings highlight the role of diet in inflammation regulation by emphasizing its potential in modulating age-associated inflammatory conditions, underlining aging as a systemic inflammatory comorbidity. PA-ED exacerbated P. gingivalis–associated bone loss in older, but not younger, animals. Aged animals on PA-ED exhibited significantly greater alveolar bone loss than OA-ED and ND groups did as well as their younger PA-ED counterparts, suggesting age is a critical comorbidity factor. This aligns with human studies linking WD to increased periodontitis risk, particularly in individuals with metabolic conditions (Alhassani et al. 2021).
Aging, or immunosenescence, weakens immune responses due to lifelong antigenic stimulation and stress, leading to chronic inflammation, characterized by basally elevated cytokines (e.g., IL-6, IL-1, or TNF-α), contributing to morbidity, cardiovascular events, neurodegenerative disorders, cancers (Liu et al. 2023), and chronic conditions such as periodontitis (Hajishengallis 2014). Consistently, aged individuals showed reduced inflammatory reactivity to P. gingivalis infection in gingival tissue, indicating immunosenescence. In line with this, GFbs from aged donors exhibited lower responses to FA treatment and LPS stimulation than GFbs from younger animals did. While young GFbs showed elevated basal Il6 and Cxcl2 expression upon PA exposure—suggesting diet-induced chronic low-grade inflammation—aged cells remained unresponsive under both basal and stimulated conditions. This supports the concept of inflammaging, as aging increases baseline IL-6 secretion while diminishing the infection response. Similarly, aged mice display higher systemic IL-6 levels and immune dysfunction, indicating potentially greater susceptibility to periodontitis progression (Almanan et al. 2020). Consistent with previous findings on the trained immunity of immune cells from young WD-fed mice (Christ et al. 2018), we observed prolonged dietary effects on GFb’s inflammatory capacity; however, this was limited to young cells, as aged GFbs showed minimal response to LPS infection. PA directly exacerbates inflammation in primary GFbs under lipotoxic stress (Doding, Hufner, et al. 2023). Given the observed oral bone loss in aged mice, we propose that basal inflammation in aged GFbs is sufficient to amplify inflammatory responses upon infection. This is supported by increased Cxcl2 expression following TNF-α stimulation in aged cells, potentially linking inflammaging to disease progression, as aging is associated with elevated IL-1β and TNF-α, reduced collagen synthesis, and oxidative stress in gingival tissues (Furukawa et al. 2022).
Epidemiological studies suggest olive oil–rich MD as disease preventive and in support of “successful aging” (Foscolou et al. 2019). Generally, advanced age is associated with a significant reduction in alveolar bone, even in clinically healthy periodontium, and may therefore represent an independent systemic risk factor for alveolar bone loss (Damanaki et al. 2018). However, only PA-ED increased alveolar bone loss in our aged P. gingivalis model. Human studies linked OA-rich MD to reduced risk for osteoporosis-related fractures in 55- to 80-y-olds (Garcia-Gavilan et al. 2018), supporting the here described underlying bone-protective pathways and dietary substitution in age.
Considering age as a systemic inflammatory condition, OA-ED appears to protect against inflammation. Previous work demonstrated OA as an attenuator of LPS-driven inflammatory response in murine immune cells (Muller et al. 2021). In an obesity model, OA-ED, in contrast to PA-ED, dampened generalized inflammatory activation and reduced inflammatory gene expression in gingival tissue in periodontal infection (Muluke et al. 2016). In line with this, the current results demonstrate reduced expression of inflammatory markers in the gingival tissue in infectious conditions, underlining OA’s anti-inflammatory properties. In this context, OA, as compared with PA, reduces Il6 and Cxcl2 expression in young GFb. IL-6 and CXCL-2 play a promotional role in osteoclast differentiation (Brylka and Schinke 2019; Doding, Zimmermann, et al. 2023), and a change in the fibroblastic expression pattern could indirectly affect bone resorption. Furthermore, the protective effects of OA-EDs seem to derive from its proresolving properties (Doding, Hufner, et al. 2023). Building on research identifying OA-ED–induced LMs as key factors in inflammation resolution and bone preservation in a murine P. gingivalis model (Doding, Hufner, et al. 2023), we identified 10-HDHA in P. gingivalis–infected older animals, suggesting molecular feedback mechanisms and reinforcing OA’s anti-inflammatory and bone-protective properties. Notably, 10-HDHA is DHA derived, and DHA supplementation reduces gingival bleeding and improves attachment in advanced periodontitis (Stando et al. 2020). In contrast, 15-HETE, which is elevated in P. gingivalis–infected older animals, showed no significant effect on bone metabolism pathways, suggesting its role may be untargeted in the investigated setting of inflammation and remodeling.
Our inoculation model used a single pathogen, which may not capture microbiome complexity. Although P. gingivalis is a key pathogen in periodontitis (Hajishengallis et al. 2012), there is diversity among P. gingivalis strains in virulence (Seers et al. 2020), gingipain distribution (Seers et al. 2020), and interaction patterns with platelets (Jockel-Schneider et al. 2018); all could directly influence the immune response in periodontitis progression. We selected P. gingivalis–W50 based on previous findings pertaining to oral bone loss (Drosatos-Tampakaki et al. 2014; Muluke et al. 2016; Doding, Hufner, et al. 2023) and its significant influence on the gut microbiome and stress response, especially under PA-ED (Doding et al. 2025). Overall, the current findings provide a foundation for additional studies, potentially with a bacterial mixture or ligature model.
Based on previous studies, only male animals were used (Doding, Hufner, et al. 2023; Doding, Zimmermann, et al. 2023). Future studies will address dietary substitution in females to investigate hormonal variations.
Our findings support regulatory pathways and underline age as a systemic disease and risk factor in the complex interplay between diet, inflammation, and periodontal infection. Whereas proinflammatory WD-style PA-ED enhances basal inflammation and promotes infection-driven bone loss in age, anti-inflammatory MD-style OA-ED preserves bone from deleterious actions of age and infection by driving resolution. Given the rising prevalence of periodontitis in aging populations, incorporating OA-rich foods could offer promising strategies for promoting periodontal health.
Author Contributions
A. Döding, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; A. Petzold, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; O. Ciaston, J. Symmank, contributed to data acquisition, critically revised the manuscript; M. Wichmann-Costaganna, P. Schädel, K. Becker, contributed to data acquisition and analysis, critically revised the manuscript; H. Noels, contributed to data analysis, critically revised the manuscript; C. Jacobs, O. Werz, contributed to data interpretation, critically revised the manuscript; U. Schulze-Späte, contributed to conception, design, data interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251369518 – Supplemental material for Fighting Age-Associated Bone Loss in Periodontitis with Dietary Interventions
Supplemental material, sj-docx-1-jdr-10.1177_00220345251369518 for Fighting Age-Associated Bone Loss in Periodontitis with Dietary Interventions by A. Döding, A. Petzold, O. Ciaston, M. Wichmann-Costaganna, P. Schädel, J. Symmank, H. Noels, C. Jacobs, K. Becker, O. Werz and U. Schulze-Späte in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank Ann-Kathrin Bär, Anton Domuncu, Michael Reimann, Jewgeni Len, Katrin von Brandenstein, and Sina Hourtz for technical and experimental assistance in performing the in vitro and in vivo studies.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the Interdisciplinary Center of Clinical Research of the Medical Faculty Jena (IZKF UKJ FF02), the German Federal Ministry of Education and Research (BMBF: 01EC1901B, Project 2) Foundation, “Else Kröner Fresenius Stiftung” within the graduate program “Else Kröner Promotionskolleg JSAM,” Interdisciplinary Center of Clinical Research of the Medical Faculty Jena (doctoral program). The funders played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Ethics Statement
Animal experiments approval No. UKJ-17-036.
Data Availability Statement
Data are available upon request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
