Abstract
This 30-mo double-blind randomized clinical superiority trial aimed to assess the effectiveness of semiannual application of 38% silver diamine fluoride (SDF) solution in preventing early childhood caries in primary upper anterior teeth. The active comparator was 5% sodium fluoride varnish (FV). The primary outcome was the mean number of new carious tooth surfaces per child at the 30-mo follow-up. Children aged 3 to 4 y were block randomized to receive semiannual application of SDF or FV on their primary upper anterior teeth over a 30-mo period in kindergarten. Parents completed a validated questionnaire to collect information on child oral health–related behaviors and sociodemographic characteristics. In an intention-to-treat analysis, multilevel logistic regression with a generalized estimating equation (GEE) model was performed. At baseline, 730 children with 17,480 sound tooth surfaces were recruited (SDF group,
Introduction
The primary upper anterior teeth exhibit a higher prevalence of early childhood caries (ECC) among preschool-aged cohorts in Hong Kong, and lesions are readily detectable through clinical examination (Gao et al. 2018; Duangthip et al. 2019). According to the World Dental Federation (FDI) Oral Health Atlas, most children with ECC never receive treatment (FDI World Dental Federation 2024). Conventional dental care in most parts of the world cannot cope with the high ECC prevalence, particularly due to a shortage of resources (Beaglehole et al. 2009). Caries prevention is thus extremely important. The FDI advocates the use of fluoride for the prevention of ECC (FDI World Dental Federation 2018). The current standard of professional care for caries prevention is to use 5% sodium fluoride varnish (FV) 2 to 4 times per year. A systematic review by the Cochrane Collaboration concluded that the application of FV can reduce caries in primary teeth development by 37% (Marinho et al. 2013). An alternative solution is 38% silver diamine fluoride (SDF), which contains a higher content of fluoride (44,800 ppm fluoride) than 5% FV (22,600 ppm fluoride) to promote remineralization (Mei, Chu, et al. 2013). In addition, SDF includes silver, which inhibits growth of cariogenic biofilms (Mei, Li, et al. 2013). However, SDF has a metallic taste, which may cause discomfort for patients.
Clinical studies suggested that SDF prevented ECC development on adjacent sound teeth when SDF was applied to carious lesions in children (Chu et al. 2002; Fung et al. 2018). Milgrom and Chi (2011) advocated for SDF as an important prevention-centered caries management strategy during critical early childhood periods. A literature search in 2020 found no well-designed clinical trial of a clinically meaningful duration assessing the caries-preventive effect of SDF on sound primary teeth (Gao et al. 2020b). Therefore, the objective of this study was to assess the caries preventive effect of the application of SDF on sound primary upper anterior teeth among 3- to 5-y-old children for 30 mo. We hypothesized that SDF would be superior to FV.
Method
This report follows the CONSORT 2010 checklist (Consolidated Standards of Reporting Trials; Schulz et al. 2010).
Trial Design
This stratified-randomized, active-controlled clinical trial was based on a double-blind, parallel-assigned design with 1:1 allocation to 2 arms. The local institutional review board (UW-18-619) approved the study. The trial was prospectively registered at ClinicalTrials.gov (NCT04075474). The full protocol was published in an open access journal (Gao et al. 2020b).
Participants, Setting, and Location
The study was conducted in kindergarten classrooms. The parents were approached by a trained assistant who explained the study and gained consent. Parents were told that only the primary upper anterior teeth would be treated and that they would receive information about the need for treatment of the other teeth. A copy of the consent form is in Appendix 1. The first child was enrolled on October 22, 2020, and the last child was evaluated on October 11, 2023. The inclusion criteria were children aged 3 to 4 y who were generally healthy and attending the first year of kindergarten. Children were excluded if they were uncooperative or were taking long-term medications such as methylphenidate (Leung et al. 2008). The water supply in Hong Kong contains 0.5-ppm fluoride.
Questionnaire
A validated questionnaire (Fung et al. 2018; Gao et al. 2020b) was completed by the parents to collect information about their children’s oral health–related behaviors and sociodemographic characteristics. Items included details about the child’s bottle-feeding habits, snacking frequency, and toothbrushing practices at baseline. Additionally, information on the parents’ education levels and monthly family income was collected.
Clinical Examination
One trained examiner performed the baseline and follow-up examinations. The 6 primary upper anterior teeth were the studied teeth for intervention. They were cleaned and dried with gauze before caries diagnosis. The examiner used a front-surface dental mirror equipped with LED intraoral illumination and a World Health Organization (WHO) Community Periodontal Index periodontal probe for caries diagnosis. Caries was diagnosed at the cavitation level according to criteria recommended by the WHO (2013). The caries status of each studied tooth was recorded at the tooth surface level by assigning a dmfs score (decayed, missing, or filled surface; Chen et al. 2021). The overall caries experience for each child was then evaluated at the tooth level by calculating a dmft index (decayed, missing, and filled teeth) based on the status of all 20 primary teeth. For lesion activity assessment, a caries lesion was diagnosed as arrested if its surface was smooth and hard on probing. A lesion was recorded as active if it was soft on probing (Chu et al. 2002).
The child’s oral hygiene was assessed per the Visible Plaque Index (Ainamo and Bay 1975). Examinations were conducted at 6-mo intervals postbaseline. The examiner (F.M.Z.) completed standardized caries diagnosis training and underwent calibration with an experienced examiner (C.H.C.) to ensure diagnostic validity. The dentist reexamined 10% of the children at each follow-up visit to monitor intraexaminer agreement on the dmfs score, dmft score, and Visible Plaque Index score. Cohen’s kappa values for assessing the caries status and caries experience were at least 0.90 for every examination period.
Intervention
The children received an application of either 38% SDF (treatment group) or 5% FV (active control group). The SDF or FV was applied once every 6 to 30 mo since the first application. At each treatment session, a trained dentist used a microbrush to apply SDF (lot 21109, Advantage Arrest; Elevate Oral Care) or FV (Duraphat Varnish; Colgate-Palmolive) according to the assigned treatment group after the oral examination. Each tooth surface (sound or decayed) of the 6 primary upper anterior teeth was dry-brushed before receiving the application. Typically, each tooth surface received 1 SDF application, but the proximal surfaces of adjacent teeth were treated with a single shared application between them. On average, each child had SDF or FV applied on 18 surfaces of the 6 primary upper anterior teeth. This means that, in this trial, a child could receive 4.8 mg of fluoride in the SDF group or 9.1 mg of fluoride in the FV group every 6 mo (Yan et al. 2022).
The materials were donated by the manufacturers and were stored according to their recommendations. The manufacturers provided a certificate of analysis.
Outcome
The primary outcome was the increment of carious tooth surfaces, measured by the mean number of new carious tooth surfaces per child at 30 mo. The secondary outcome was the incidence of carious tooth surfaces, measured by the odds ratio of carious tooth surface incidence among the primary upper anterior teeth at 30 mo. Other outcomes included the proportion of new carious tooth surfaces at each interim time point.
Sample Size Calculation
The sample size calculation was based on the primary outcome. Drawn from a previous clinical trial (Chu et al. 2002), the mean ± SD number of new carious tooth surfaces found in the 62 children receiving SDF was 0.47 ± 0.87, and that in the 61 children receiving FV was 0.70 ± 0.84 at the 30-mo review. This difference corresponded to a prevented fraction of 33% more in the SDF group when compared with the FV group, which is considered clinically significant. With statistical power set at 0.9 and a type I error at 5% for a 2-sided test, the required sample size to test the primary outcome was 292 for each group. Additionally, for the sample size calculation based on the secondary outcome, the application of FV was estimated to reduce primary tooth caries development by 37% (Marinho et al. 2013). Based on the same prevented fraction of 33% and an intraclass correlation coefficient of 0.3 (Masood et al. 2015) at the surface level, with an average of 23 tooth surfaces per child (yielding a design effect of 7.6) and with a statistical significance level of 5% and a power of 80%, the minimum sample size required was calculated to be 79 children per group. Therefore, the necessary sample size was 292 for each group. To accommodate a 20% loss to follow-up, this 30-mo clinical trial recruited a total of 730 children at baseline examination.
Randomization: Sequence Generation, Allocation Concealment, and Implementation
The children were first categorized as having either an increased caries risk, defined as having a caries experience (dmft >0), or a low caries risk with no caries experience (dmft = 0). The children were then randomized within strata into the SDF or FV group in blocks of 8. The random number sequence was kept by a statistician, and opaque sealed envelopes were used to conceal the allocation sequence until the interventions were applied. The same treatment was performed on all the upper anterior tooth surfaces of the child at each visit.
Concomitant Treatment
The SDF was used to prevent and arrest caries in primary upper anterior teeth but not in posterior teeth. An individual report was given to parents regarding their children’s caries status (Appendix 2) after every examination, and parents were encouraged to consult a private dentist for treatment of children’s carious molars at their own cost during the 30-mo study.
Blinding
Neither the examiner nor the children or their parents were informed of the treatment group allocation for the preventive intervention provided during the study. Another operator applied SDF or FV after the oral examination. An unblinded research assistant would disclose treatment allocation to the parents who requested the treatment history of their child, and the child would be excluded from the study.
Masking
Due to the nature of the treatment, it was not possible to implement masking.
Harms
Harms were not systematically assessed. The parents were provided a 24-h mobile contact number to call if there were any problems or questions.
Statistical Analysis
In this study, a modified intention-to-treat analysis method was used to retain data only from participants who completed the final examination. To maintain high statistical power, missing data for secondary outcomes prior to the final examination were addressed by the last observation carried forward method. Baseline characteristics between the groups were analyzed by Student’s
The primary outcome was the number of new carious tooth surfaces per child at 30-mo evaluation, which was compared between groups by Student’s
All analyses were performed in SPSS version 28.0 (IBM Corporation). Statistical significance was defined as a 2-sided
Results
A total of 730 children (365 per group) from 13 kindergartens were recruited at baseline. The current study analyzed 14,904 tooth surfaces from 621 children who completed the 30-mo study. The loss to follow-up at the child level was 16% (58/365) in the SDF group and 14% (51/365) in the FV group (
Children’s Sociodemographic Characteristics and Oral Health–Related Behaviors at Baseline.
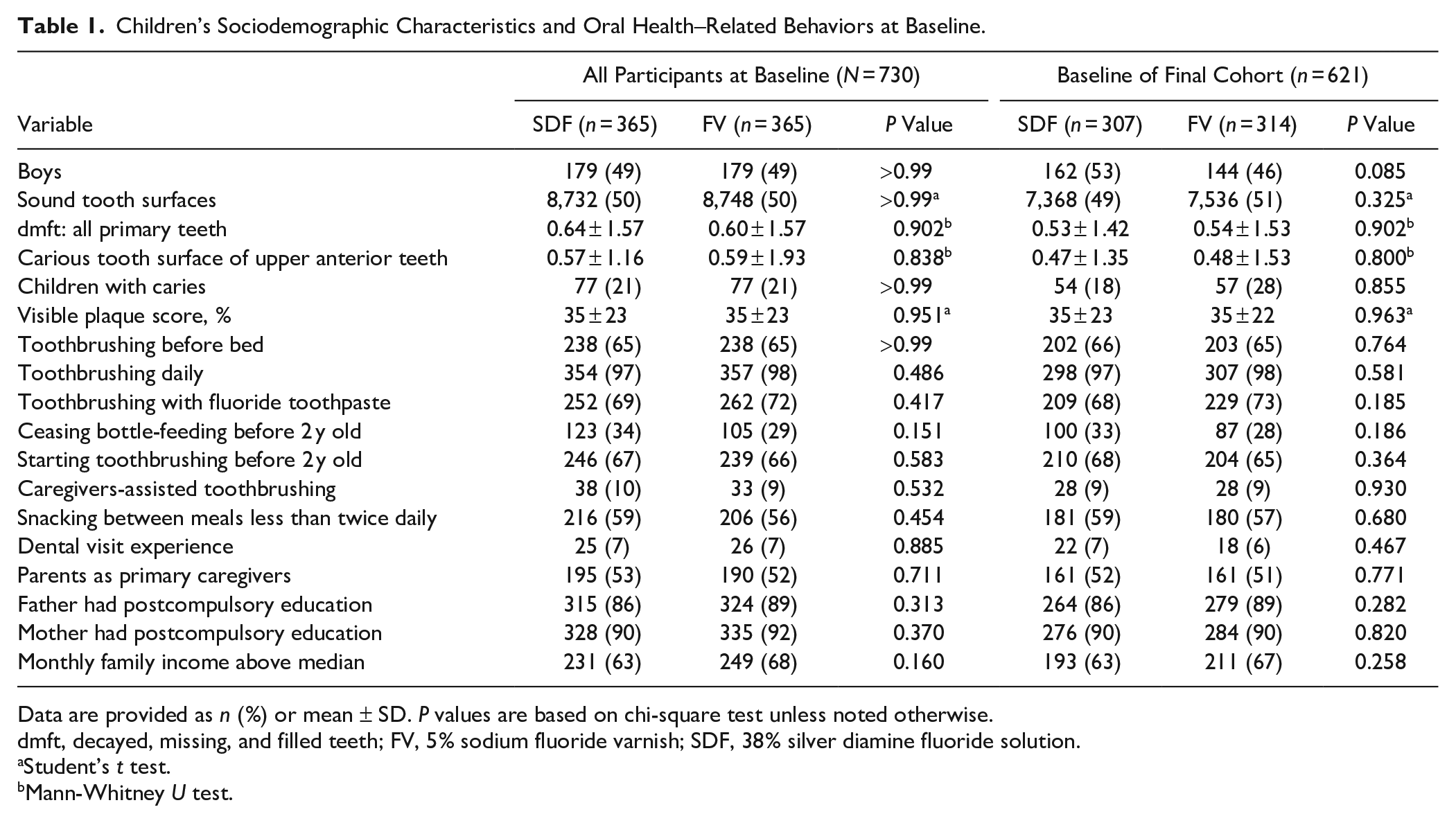
Data are provided as
dmft, decayed, missing, and filled teeth; FV, 5% sodium fluoride varnish; SDF, 38% silver diamine fluoride solution.
Student’s
Mann-Whitney
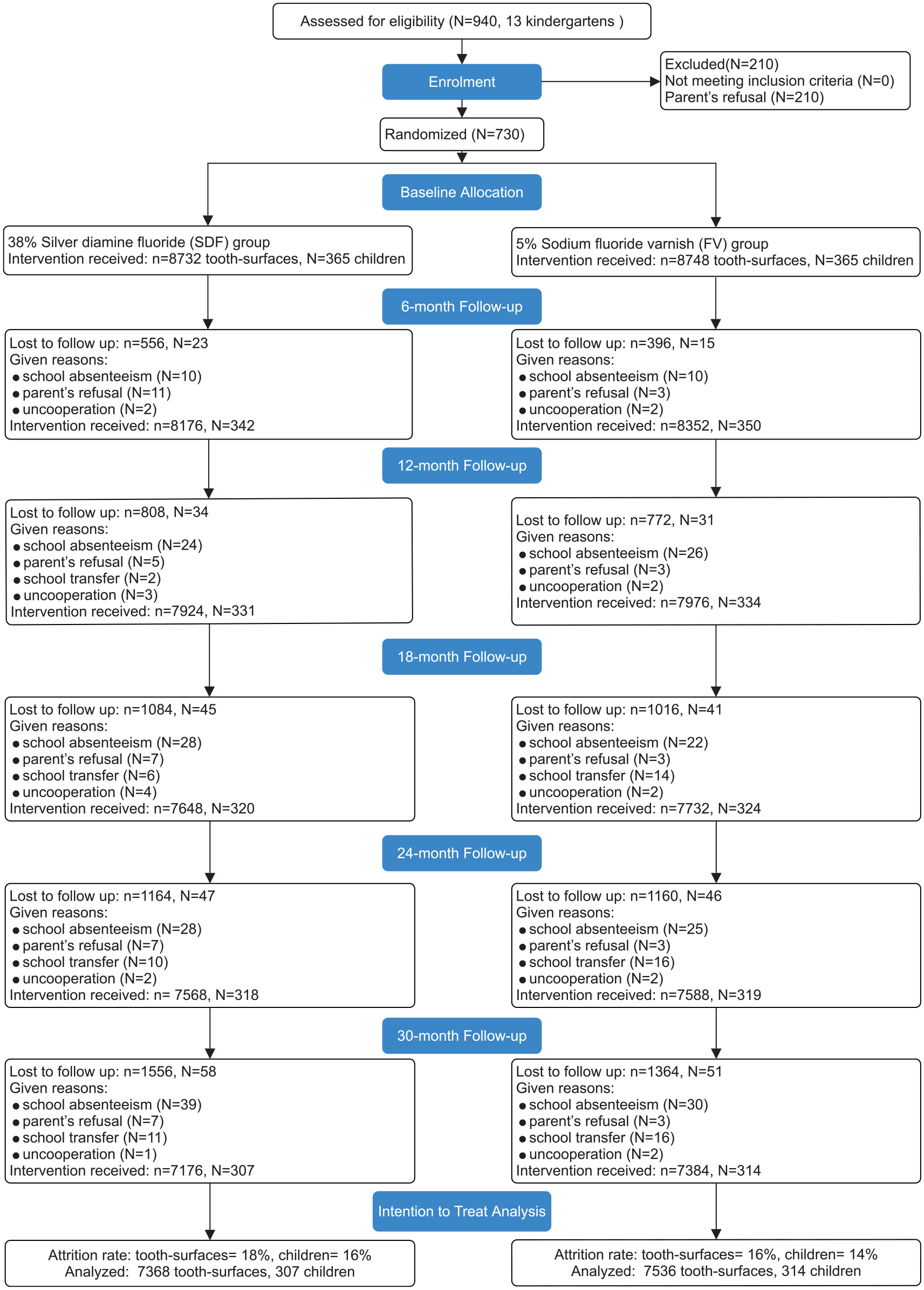
CONSORT flow diagram.
By the 30-mo examination, the mean incident distribution of new carious tooth surfaces was 0.35 ± 1.09 for SDF vs 0.54 ± 1.50 for FV (Mann-Whitney
Carious Tooth Surface Development in Primary Upper Anterior Teeth: SDF vs FV over 30 mo.
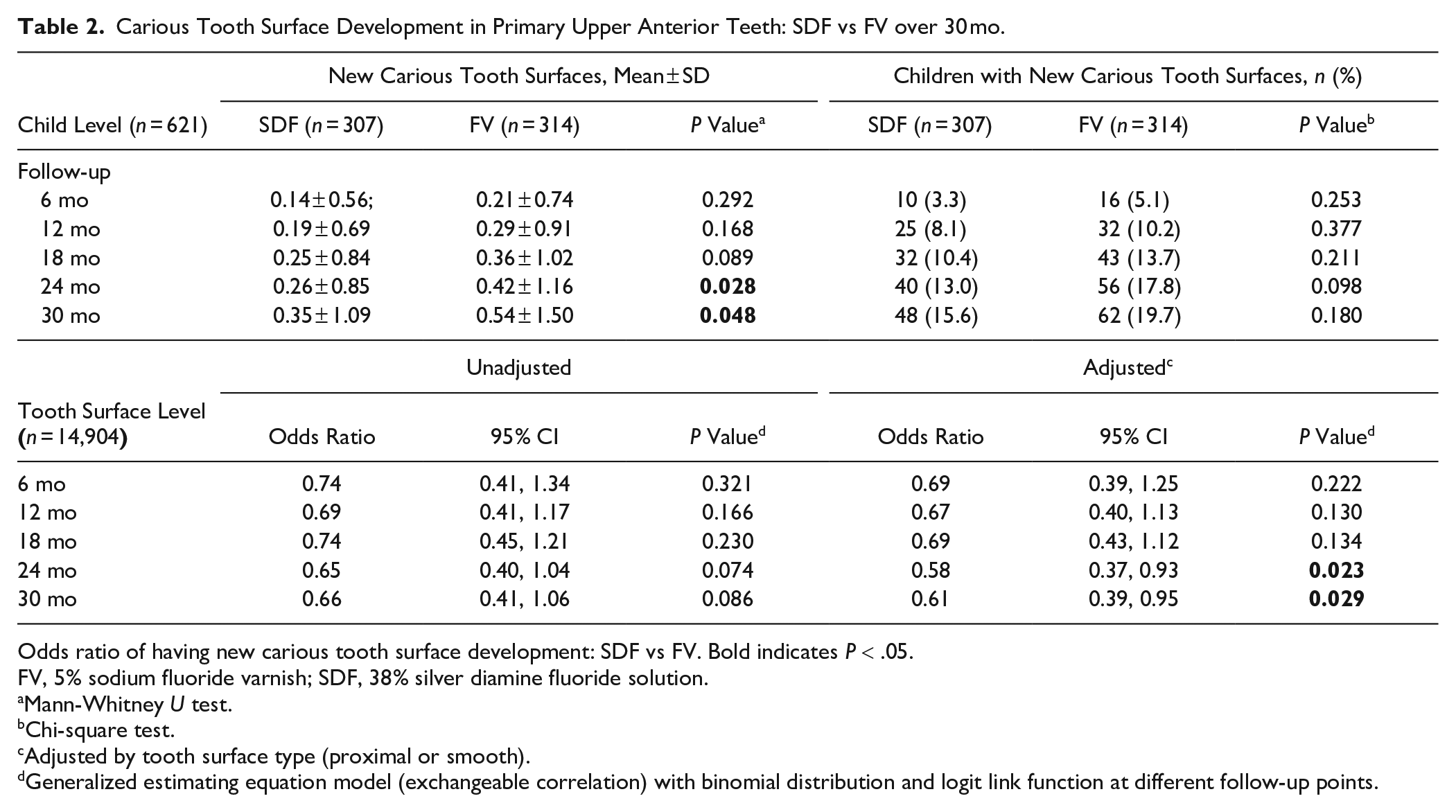
Odds ratio of having new carious tooth surface development: SDF vs FV. Bold indicates
FV, 5% sodium fluoride varnish; SDF, 38% silver diamine fluoride solution.
Mann-Whitney
Chi-square test.
Adjusted by tooth surface type (proximal or smooth).
Generalized estimating equation model (exchangeable correlation) with binomial distribution and logit link function at different follow-up points.
After adjusting for clustering and confounding (tooth surface type), a significant difference between the groups was observed in odds of developing new carious lesions at the tooth surface level at 30 mo (Table 2). The results were similar at 24 mo. At 30-mo follow-up, the percentage of new carious tooth surfaces developed was 1.5% (108/7,224) in the SDF group and 2.2% (166/7,384) in the FV group. Table 3 shows the final multivariable model of GEE analysis confirming that the SDF group had a lower risk than the FV group in developing new carious tooth surfaces (adjusted odds ratio, 0.61; 95% CI, 0.39 to 0.95;
Predicted Probability and Adjusted Odds Ratio of Variables on Carious Tooth Surface Development at 30-mo Follow-up (14,904 Tooth Surfaces from 621 Children).
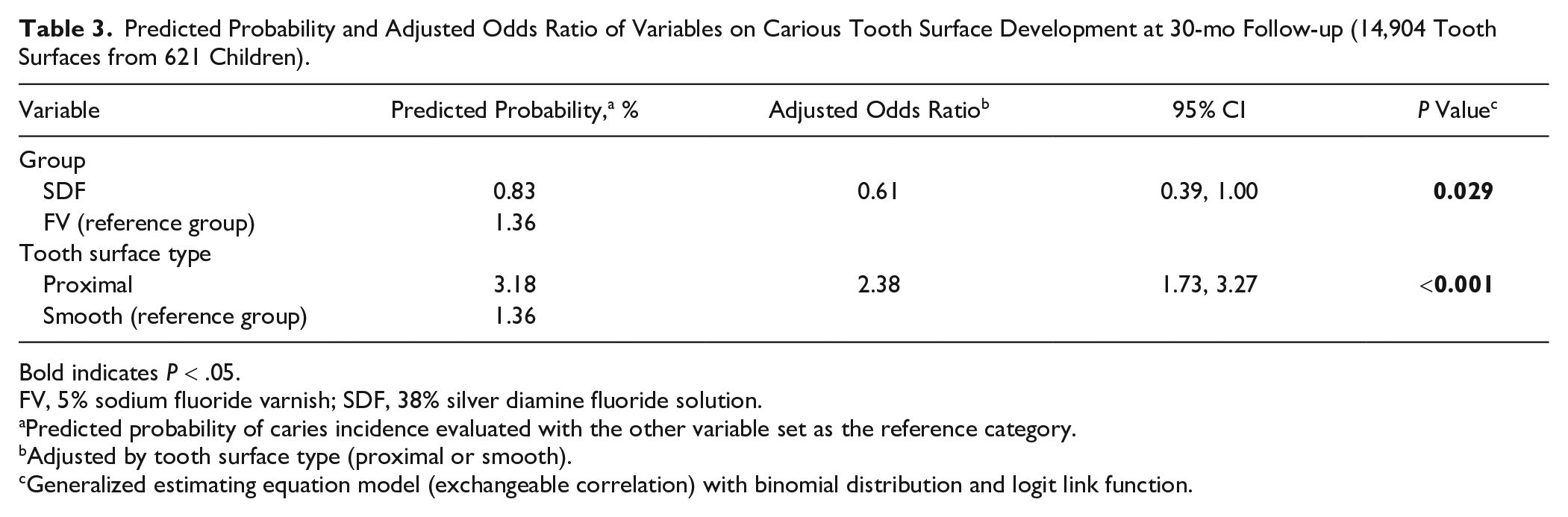
Bold indicates
FV, 5% sodium fluoride varnish; SDF, 38% silver diamine fluoride solution.
Predicted probability of caries incidence evaluated with the other variable set as the reference category.
Adjusted by tooth surface type (proximal or smooth).
Generalized estimating equation model (exchangeable correlation) with binomial distribution and logit link function.
Discussion
The present study is the first long-term clinical trial to examine the effectiveness of SDF in preventing ECC in primary upper anterior teeth. The current study was designed as a superiority trial. SDF has a dual mechanism of action, combining the remineralizing ability of fluoride and the antibacterial properties of silver. The SDF at 38% also has a higher fluoride concentration (44,800 ppm) than 5% FV (22,600 ppm). This clinical trial confirms that the semiannual application of SDF is more effective than FV in preventing caries in primary upper anterior teeth when used for >24 mo for proximal and smooth tooth surfaces. The hypothesis was accepted that SDF outperformed FV at the child and tooth surface levels.
ECC is highly prevalent in anterior teeth at younger ages (Gao et al. 2018; Duangthip et al. 2019). However, this is age dependent and not necessarily at age 3 to 5 y; thus, the results of this study may not apply to younger children. Nonetheless, proximal tooth surfaces are at higher caries risk than smooth tooth surfaces because they are in close contact with each other, making them difficult to clean, more likely to trap food, and prone to plaque buildup. If the proportion of proximal to smooth tooth surfaces differed between the treatment groups at baseline, not adjusting for this difference could bias the results. To ensure accurate results, the analysis adjusted for the type of tooth surface in our analysis via the GEE model. This adjustment isolated the caries-preventing effect of SDF from the natural risk differences between tooth surfaces. The adjusted odds ratio was 0.61 (
This study had an expected and acceptable loss to follow-up. In Hong Kong, about 10% of families move between districts, causing their children to change kindergartens (Gao et al. 2020a). The analysis used data from children who attended the final examination. The analysis utilized a modified intention-to-treat analysis instead of a per-protocol analysis because the proportion of children deviating from the protocol was similar in both groups. This method allowed direct observation of the final outcomes, without needing to estimate missing data, thereby improving the accuracy of the evaluation.
A systematic review and meta-analysis on the caries-preventive effects of SDF was published in 2019 (Oliveira et al. 2019). The review found that annual applications of 38% SDF led to a 54% reduction (95% CI, 27% to 73%) in the incidence of new caries as compared with quarterly applications of 5% FV. However, the studies included in the review showed significant heterogeneity. They differed in follow-up duration (range, 12 to 36 mo), SDF concentrations (12%, 30%, or 38%), dentition type (primary or permanent teeth), and participant age range (3 to 15 y). Another concern is that while SDF was applied to carious teeth, the outcome measured was the development of caries on sound teeth. Two 12-mo clinical trials were published in 2023 investigating the use of SDF to prevent ECC (Jain et al. 2023; Zheng et al. 2023). Conducting clinical trials for caries prevention is challenging because cavitated carious lesions take time to develop (Chesters et al. 2004). A review concluded that 12-mo clinical trials are too short to adequately assess interventions for caries prevention (Chesters et al. 2002). Therefore, this study addressed the need for longer follow-up periods with appropriately large sample sizes to gather enough data for meaningful analysis of preventive effects.
The safety of topical SDF use in preschool children has been demonstrated in numerous studies (Milgrom et al. 2018; Ellenikiotis et al. 2022; Zheng et al. 2023), with analyses indicating minimal risk of fluoride or silver toxicity; a 20-kg child, for instance, would not reach toxic levels even with nearly 400 applications (Yan et al. 2022). This is particularly relevant in Hong Kong, where young children are at higher risk of ECC in their anterior teeth (Duangthip et al. 2019). To further ensure safety in our study and considering potential challenges with a child’s cooperation and parental acceptance, SDF was intentionally limited to the primary anterior teeth. Consequently, the intermittent application schedule (18 applications over 30 mo) provided a substantial safety margin against dental fluorosis, and notably, no parents reported any adverse effects.
A recent Cochrane review highlighted the risk of detection bias in studies using SDF to arrest caries (Worthington et al. 2024). SDF causes carious lesions to turn black but does not discolor sound teeth. This trial focused on detecting cavitated lesions rather than arrested lesions, and the examiner was carefully trained. Previous studies have shown very high reliability in assessing cavitated lesions, with kappa values consistently >0.90. In this trial, a single examiner performed all clinical assessments. To ensure diagnostic validity, the examiner underwent rigorous calibration against a gold standard reference examiner’s assessments prior to study initiation, establishing consistent interpretation of diagnostic parameters. Intraexaminer reliability was ensured through repeated examinations at each visit and biannual recalibration. While caries diagnosis at the cavitation level is generally reliable, there remains a potential examiner effect on the outcome.
The incidence of carious tooth surfaces was relatively low in both groups, which decreases the statistical power and increases the risk of a type II error. This low incidence leads to greater variability and uncertainty in the estimates of carious tooth surface incidence. To address this, GEE analysis was used to reduce variability or random error by adjusting for tooth surface type, making it easier to detect true differences. Another limitation of this study is that some children visited a dentist and received dental treatment; however, the number of such children was small. The results after exclusion of these children were the same.
This study employed a 2-arm design without a control group (no treatment) for ethical reasons. While most researchers consider FV effective in preventing ECC, the absence of a no-treatment comparison makes it impossible to determine whether either intervention was more effective than no treatment. This study found that SDF is more effective than FV in preventing ECC on the upper anterior teeth. However, these results may not be applicable to posterior teeth. Another 30-mo clinical trial to study the preventive effect of SDF on occlusal (fissure) caries in the primary molars of young children is underway (Duangthip et al. 2022).
There is increasing global acceptance and use of SDF for caries management. The WHO has included SDF in its Model List of Essential Medicines for children and adults (WHO 2021). This inclusion aims to enhance the accessibility of SDF and reduce the burden of dental caries. This study provides valuable information on the use of SDF for the prevention of ECC in young children, particularly in community care settings, which should help boost acceptance.
Conclusion
When applied every 6 mo, SDF provided superior caries prevention after 24 mo for proximal and smooth tooth surfaces on primary upper anterior teeth when compared with FV in young children.
Author Contributions
F.M. Zheng, data acquisition and analysis, drafted and critically revised the manuscript; I.G. Yan, E.C.M. Lo, data analysis and interpretation, critically revised the manuscript; D. Duangthip, data acquisition, critically revised the manuscript; S.S. Gao, C.H. Chu, contributed to conception and design, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-pdf-1-jdr-10.1177_00220345251363837 – Supplemental material for Randomized Clinical Trial on Caries Prevention of Silver Diamine Fluoride
Supplemental material, sj-pdf-1-jdr-10.1177_00220345251363837 for Randomized Clinical Trial on Caries Prevention of Silver Diamine Fluoride by F.M. Zheng, I.G. Yan, D. Duangthip, E.C.M. Lo, S.S. Gao and C.H. Chu in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345251363837 – Supplemental material for Randomized Clinical Trial on Caries Prevention of Silver Diamine Fluoride
Supplemental material, sj-pdf-2-jdr-10.1177_00220345251363837 for Randomized Clinical Trial on Caries Prevention of Silver Diamine Fluoride by F.M. Zheng, I.G. Yan, D. Duangthip, E.C.M. Lo, S.S. Gao and C.H. Chu in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank Dr. Hollis Haotian Chai for assistance with data recording, Ms. Samantha K.Y. Li, and Mr. Sicheng Wu for their statistical advice. They are also grateful to the kindergarten teachers, children, and parents for their valuable support in this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received support from the Research Grant Council General Research Fund (17100019).
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
