Abstract
Dental caries is characterized by the dynamic processes of demineralization and remineralization. Recent advancements in cariology research have introduced sophisticated techniques such as nanoindentation, nano-computed tomography, optical coherence tomography, X-ray photoelectron spectroscopy, atom probe tomography, and transmission electron microscopy, which provide high resolution and sensitive analytics. These methods are particularly valuable for detecting micro- and nano-scale changes in individual lesions, crucial for short-term in vitro studies. They enable assessments of mechanical properties, mineral density, chemical composition, and morphological structure, providing insights into the dynamic nature of caries and aiding in the optimization of caries management strategies and the development of new treatments. Results from these techniques have been validated through in vivo studies by correlating noninvasive imaging with clinical caries progression and monitoring mineral density changes over time. A comprehensive understanding of these techniques is essential to prevent misselection, inappropriate sample preparation, and data misinterpretation. This review focuses on methods applicable to the analysis of early caries lesions based on recent literature and provides detailed insight into a variety of relevant techniques. In addition, it discusses parameters such as main outcome measurements, resolution/magnification, invasiveness, and sample preparation requirements of each approach, thereby providing valuable guidance for researchers planning studies in this field.
Introduction
Dental caries remains a significant global health problem. Caries formation is a dynamic process of demineralization and remineralization, in which the shift in equilibrium to an overall mineral loss leads to the destruction of the structure of enamel and dentin (Abou Neel et al. 2016). Most new developments or strategies for caries management have been initially investigated using in vitro and in situ studies, as the experimental conditions can be well controlled, are less complex and standardized, and the experimental durations are much shorter than in expensive clinical trials (Dohan et al. 2023). Although in vivo caries detection methods such as the International Caries Detection and Assessment System, DIAGNOdent, and quantitative laser fluorescence are useful for clinical assessment, they lack the sensitivity to detect subtle mineral changes occurring in short-term studies (Castilho et al. 2016) and are unsuitable for fundamental research on the dynamics of mineral dissolution/reprecipitation and other mechanisms that underpin tooth decay. Laboratory-based techniques, which can precisely quantify mineral, mechanical, and chemical changes at the lesion level, are essential for understanding caries dynamics and developing new therapies for the repair of cavities, for example, by developing new therapies to promote the regeneration of the mineral (Shao et al. 2019). These findings serve as a foundation for refining and validating clinical detection tools, ultimately bridging the gap between laboratory research and clinical application.
Over the past decade, classic measuring techniques such as transverse microradiography (TMR), polarized light, and microhardness approaches have been used to assess changes in mineral content. TMR is regarded as the gold standard for quantifying subsurface mineral distributions and changes (Huysmans and Longbottom 2004) but requires thin, flat slices (around 100 µm) for accurate measurements (Lo et al. 2010). In contrast to noninvasive methods such as computed tomography (CT) or optical coherence tomography (OCT), TMR involves more invasive sample preparation. There are also a number of new techniques that have been adapted for mineralization research in cariology. Notably, these techniques have been widely applied in other research disciplines and have recently been introduced into dental research due to their distinct advantages, including high resolution, exceptional sensitivity, reliability, and nondestructive analysis. These techniques also cover a broad range of measurements, such as determining mechanical properties, mineral density, chemical characteristics, and morphological and structural characterization at different levels. They can detect changes at the micro- and nano-scale levels in a single lesion, which is particularly beneficial for short-term in vitro studies. However, a lack of comprehensive understanding of the fundamental mechanisms and implications of the techniques could lead to the misselection of assessments, inappropriate preparation of samples, and misinterpretation of data. The latest review in English on mineralization research techniques for caries lesions was published in the 1990s (Arends and ten Bosch 1992). Consequently, this review now aims to provide updates on contemporary laboratory assessments used in mineralization research, and methods have been included when applied to caries or caries-like lesions. We have categorized the techniques according to their primary areas of assessment, namely, mechanical properties, mineral density, and chemical characterization. It should be noted that this classification is not strictly exclusive, as some techniques serve multiple purposes. A summary of the key information on these technologies is also provided in Table 1.
Comparison of Advanced Laboratory Techniques for Assessing Mineralization.
Nanoindentation
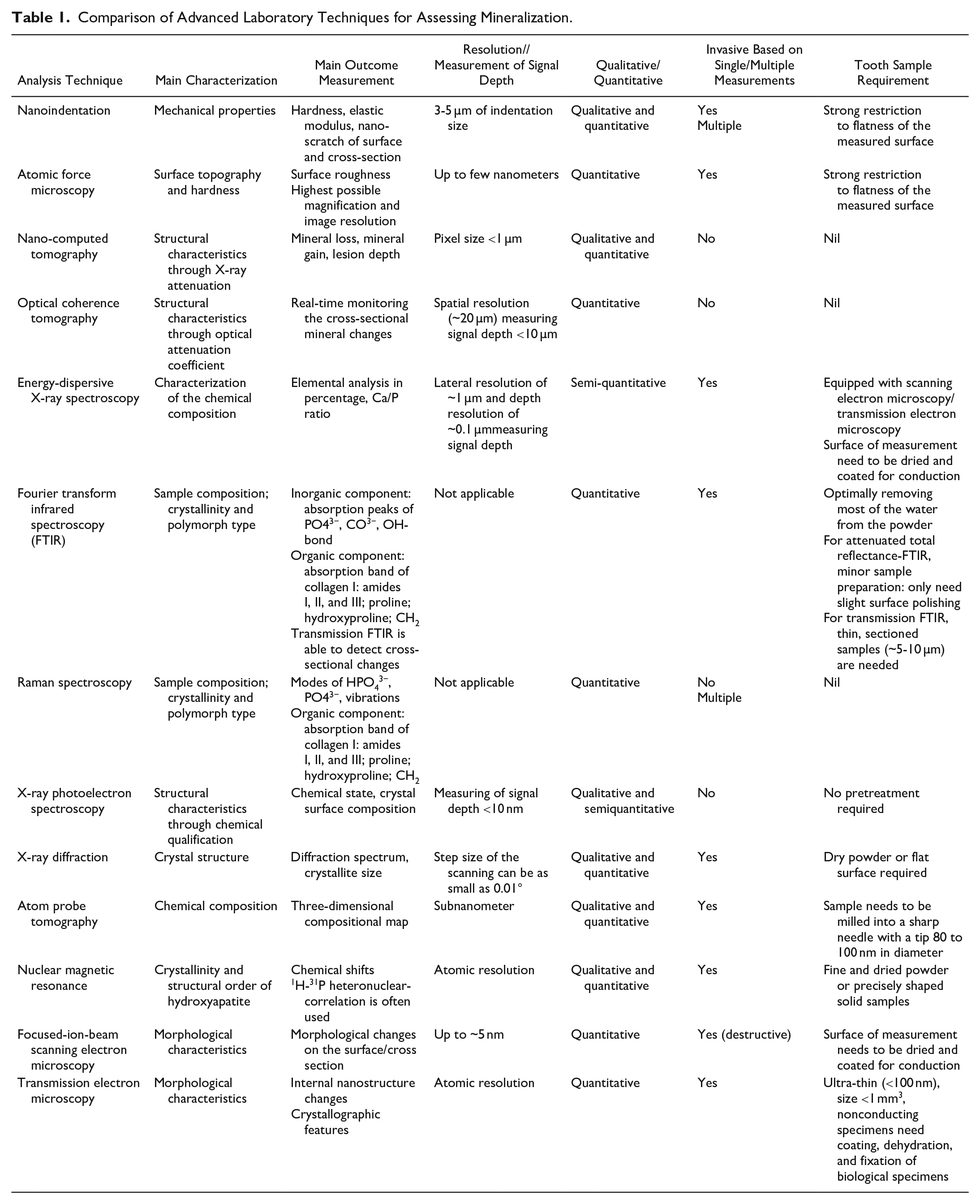
In in vitro studies, hardness tests are the most common method conducted to assess the mechanical properties of enamel and dentin. Although the calculation of mineral content from hardness may not be realistic, it is understood to provide a reasonable indirect method of tracking changes in the mineral content of enamel and dentin. The microhardness test is the most common hardness test so far. Usually, a prescribed load is applied to an indenter (a Knoop or Vickers diamond) that probes into the surface of a specimen by optical imaging. Different from the microhardness, the determination of the nanoindentation hardness usually uses the depth-sensing indentation technique, with a diamond tip being pressed into the specimen to a given maximum depth or load and then removed. During this process, both the load on the indenter and the displacement of the indenter are recorded synchronously (Qian et al. 2005). From such a load-depth curve, the hardness and Young’s modulus of the specimen can be determined using the Oliver-Pharr method (Oliver and Pharr 1992) (Fig. 1A). Obviously, the advantage of this method is that it does not need to image the residual indent for the evaluation of hardness, which facilitates property measurements from microscopic indentations. Another advantage of nanoindentation is that the hardness is measured using a Berkovich-type diamond tip, which is calibrated by a standard indentation procedure prior to testing, which allows for fully estimated error determination. Nanoindentation allows the setting of indentations performed with a small load to analyze specific areas (3-5 µm indentation), which enables the investigation of minor changes in different structures (Joves et al. 2014) (Fig. 1B).
(
Two different types of hardness measurements for caries lesions are commonly used in the literature. Surface hardness is where the indenter load is perpendicular to the polished tissue surface, and cross-section hardness refers to analyses in which the indenter load is parallel to the tissue’s anatomical surface. The main concern of surface hardness measurement in teeth is that the outer surface is too curved to allow for a perpendicular indenter measurement of the surface. During the process of polishing a curved surface to achieve a flat one, areas with greater curvature will inevitably experience more reduction than others, potentially leading to variations in the measured hardness values. This is because the material removal may expose subsurface layers with different properties. In addition, surface hardness measurement assesses only the superficial hardness changes of the caries lesion surface, according to the depth of the indenter. Cross-section hardness requires dissecting the samples perpendicularly to the tooth surface. A flat surface can be obtained by cutting with a fine diamond blade and polishing using silicon papers to P4000 grit, therefore increasing the accuracy of the hardness test (Nath et al. 2024). Furthermore, the cross-section hardness test allows measurements at different depths in the lesion body, and this is more informative than the surface hardness test (Huang et al. 2010).
Atomic Force Microscopy
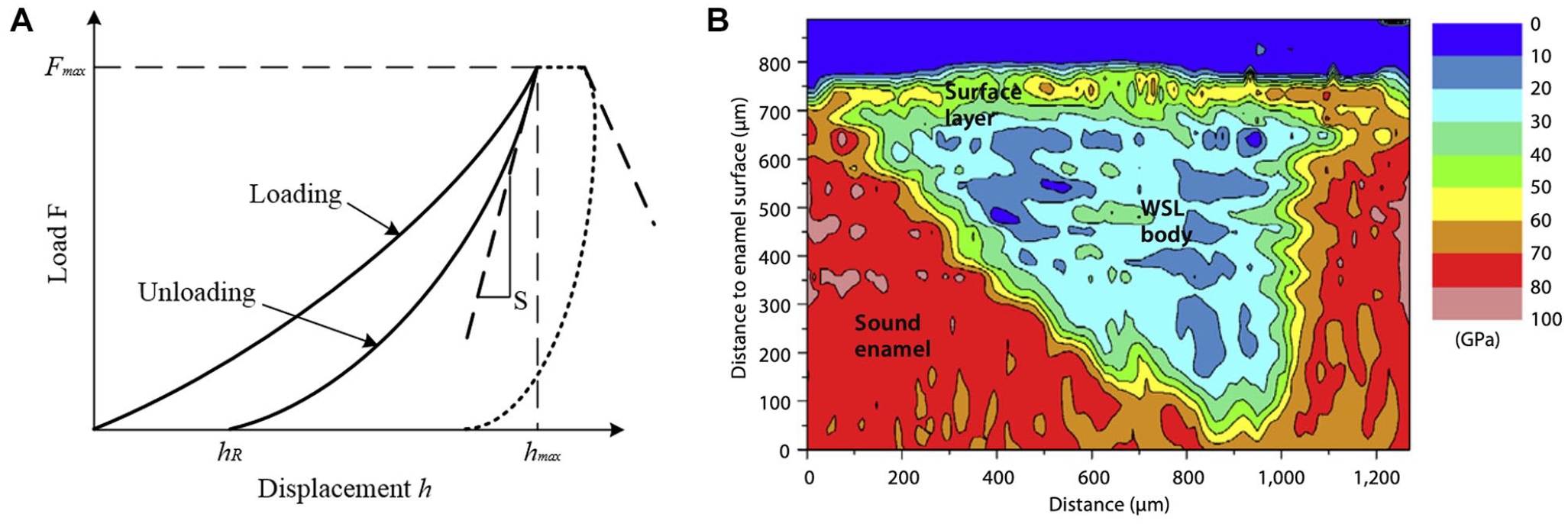
Atomic force microscopy (AFM) is used to measure the topography of a tooth sample surface by assessing the force between a probe and the sample (Picoș et al. 2021). The cantilever of the probe is subjected to either a force or a resonance frequency. By tracking the displacement of the probe in contact mode or the changes in amplitude in noncontact mode, a detailed surface topography of the sample is obtained. However, contact-mode AFM offers higher image quality with fewer artifacts by maintaining continuous contact, ensuring high resolution while minimizing distortion from lateral forces (Silikas et al. 2001).
AFM can be used to assess various mechanical properties such as hardness, elasticity, adhesion, and stiffness. This is achieved through techniques such as force-distance curves, in which the force applied by the probe and the resulting deformation of the sample are measured, allowing for the determination of mechanical characteristics at the nanoscale (Fig. 2). The prior scanning of the surface with AFM, combined with the application of nanoindentation, can be used to reveal the structure and determine the mechanical properties of mineralized dental tissues (Habelitz et al. 2001). Notably, strong restriction to flatness of the measured surface is usually expected for sample preparation for both contact and noncontact modes (Müller-Renno and Ziegler 2024).
Atomic force microscopy micrographs of (
Nano-CT
Nano-CT is an advanced imaging technique that provides high-resolution, 3-dimensional (3D) insights into dental hard tissues, making it highly valuable in the study of caries in vitro. Unlike traditional micro-CT, nano-CT offers a more refined spatial resolution, enabling analysis at the submicron focal spot scale (<1 µm) (Haugen et al. 2020). This enhanced resolution allows for detailed 3D visualization of caries lesions, early enamel demineralization, and subsurface microstructural changes that are crucial for understanding caries progression at a subclinical level. In recent years, nano-CT has been used in dentistry in the characterization of resin composites (Haugen et al. 2020), dentinal tubule occlusion (Menzel et al. 2023), and microcrack formation (Pinto et al. 2023). The mineral density calibration is usually completed using reference phantoms, which consist of hydroxyapatite (HA) discs with known HA concentrations and mineral density, respectively. A linear correlation exists between gray-scale value and mineral density and is determined using 2 or more HA phantoms (Neves Ade et al. 2010).
Moreover, nano-CT allows researchers to analyze the porosity and density changes within the lesion, providing insight into the diffusion pathways that play a role in demineralization. This capability supports the development of more targeted anticaries therapies by enhancing the understanding of lesion composition and progression at an unparalleled level of detail. In addition, it can produce 3D images with features as small as a few hundred nanometers. Nano-CT can also produce quantitative data, such as the volume occupied by occluding particles within tubules. However, precise imaging methods such as nano-CT are limited to analyzing only a small region of the specimen, which may not reliably represent the entire sample’s treatment. Therefore, examining multiple specimens and integrating complementary techniques is essential to obtain statistically relevant data.
OCT
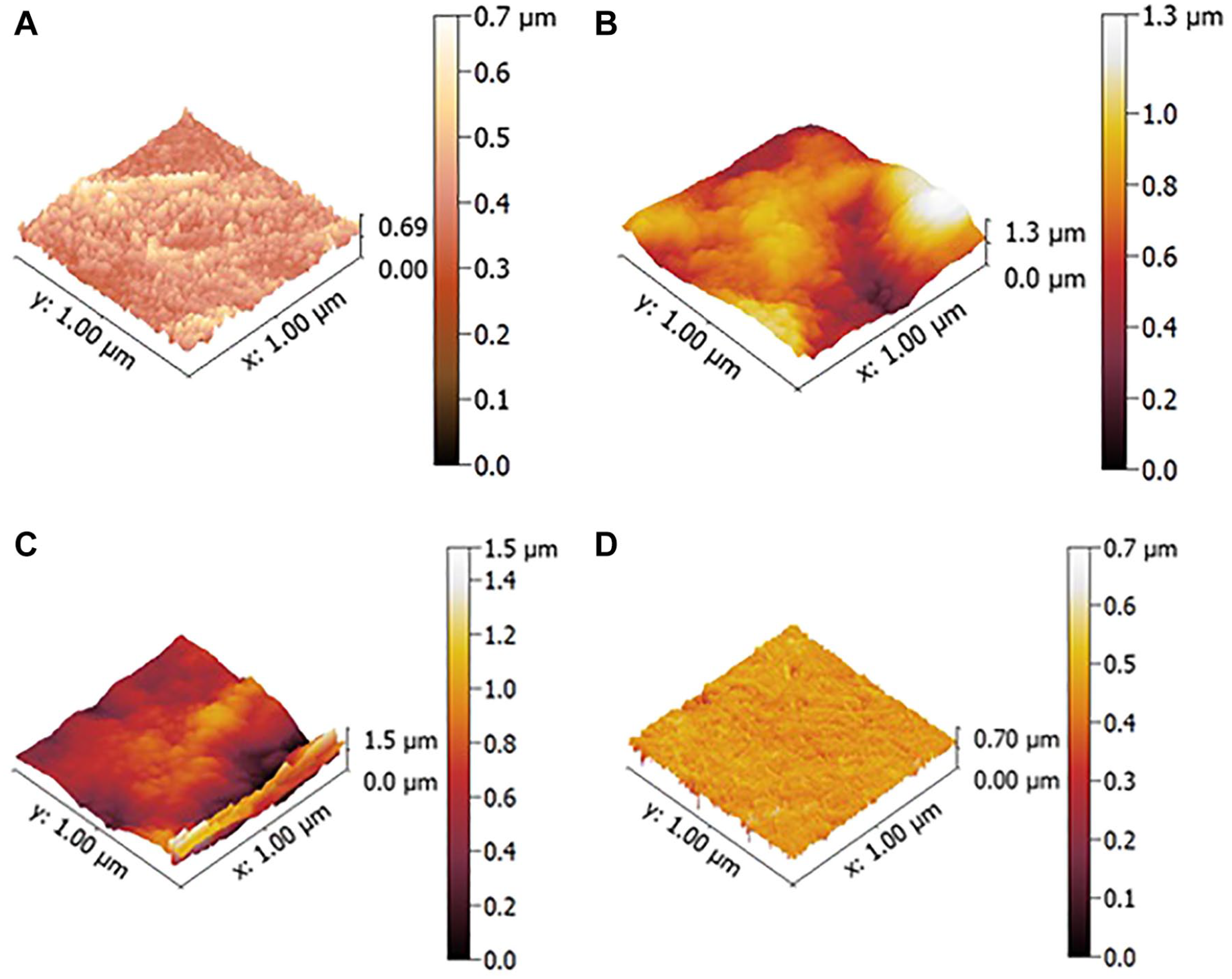
OCT provides tissue section analysis in a noncontact and noninvasive manner (Yavuz and Kargul 2021). The device measures the time delay and intensity of light scattered or reflected from biological tissues, resulting in tomographic imaging of their internal structure. It provides real-time cross-sectional images of internal structures and measures the reflectivity of light from lesion areas over their depth (Yang et al. 2023). In addition, parameters including depth of demineralization and degree of mineral loss can be further quantified using computer processing techniques (Kang et al. 2012). OCT serves as a noninvasive method in dentistry for clinical caries diagnosis, crack and fracture detection, evaluation of restoration, and in vitro assessment of enamel and dentin demineralization. Swept-source OCT (SS-OCT) is a variant of OCT in which the light source is a tunable laser that sweeps the wavelength over a certain range. SS-OCT enhances caries, tooth cracks, and restoration defect detection with improved resolution and enables high-speed 3D imaging for dental diagnostics (Bakhsh et al. 2011). However, intensity-based OCT does not have the capability to measure the birefringence changes of enamel, which seem to influence the diagnostic accuracy for incipient enamel caries (Shimada et al. 2022). Polarization-sensitive OCT (PS-OCT) uses multichannel interferometers to exploit the polarization properties of the tissue, including birefringence and diattenuation (Lee et al. 2014). By detecting changes in birefringence, PS-OCT is useful for assessing initial enamel demineralization and collagen integrity (Kang et al. 2012). The outcomes of SS-OCT and PS-OCT are shown in Fig. 3. Limitation of OCT in assessment of demineralization is its restricted penetration depth in dental hard tissues, caused by signal attenuation due to scattering and absorption. In particular, the structure of carious lesions will induce increased scattering. The strong reflectivity of the tooth surface can obscure signals from deeper lesions, making it challenging to accurately assess lesion severity (Fried et al. 2002).
(
Energy-Dispersive X-Ray Spectroscopy
Energy-dispersive X-ray spectroscopy (EDS) is a semi-quantitative microanalytical technique used to detect the elements present in the studied material. As the detector is a component of scanning and transmission electron microscopes, this technique is often coupled with the morphological and structural characterization of the specimen by electron microscopy. EDS relies on the emission of X-rays from the specimen upon interaction with the incoming electron beam. The energy of the X-rays is characteristic for each element, allowing the compositional characterization in a qualitative and semi-quantitative way, with a typical lateral resolution of ~1 µm and depth resolution of ~0.1 µm (Gonçalves et al. 2021). EDS analysis allows both the collection of spectra from a specific region and the chemical mapping of a surface, showing, for example, the distribution of elements such as calcium, phosphorus (as a proxy for phosphate), and other key elements present in caries lesions (Kale et al. 2022).
EDS is widely used to estimate the composition of the mineral by assessing the calcium-to-phosphorus (Ca/P) ratio of the lesion (Zeitouny et al. 2020), which is correlated with the different forms of calcium phosphate present. These semi-quantitative measurements derive from the weight/atomic percentage of the elements detected in the assessed area, although this is not the absolute amount of the chemical elements present. This approach also requires instrument calibration and well-polished surfaces to avoid loss of signal caused by sample topography. Consequently, caution should be taken when using EDS for quantitative analysis (Khanduri et al. 2020).
Fourier Transform Infrared Spectroscopy
Fourier transform infrared spectroscopy (FTIR) is a widely used technique to identify the chemical composition of specimens based on the interaction of specific chemical functional groups using infrared radiation. FTIR provides a cost-effective, highly sensitive, and straightforward analytical approach to monitor the changes in the chemical structure of the caries lesions. Peak intensity and area are the most commonly used measurements in FTIR quantitative analysis. The area under a band correlates directly with the concentration of the chemical species responsible for that band (Khan et al. 2018). However, using ratios between reference areas or peak heights is sometimes preferred due to uncertainties stemming from sample variation (Ou-Yang et al. 2001). Type I collagen constitutes the main organic component of dentin. In FTIR spectra, the primary infrared absorption band of collagen is Amide I, while the phosphate band signifies the mineral matrix. The absorbance areas ratio between amide I and HPO42− indicated the demineralization of the dentin (Niu et al. 2021). For enamel (Fig. 4A), HA crystals form the largest component, and FTIR is known to be highly sensitive for detecting changes in enamel erosion, in which the phosphate content is changed, and its peak is shifted (Kim et al. 2016).
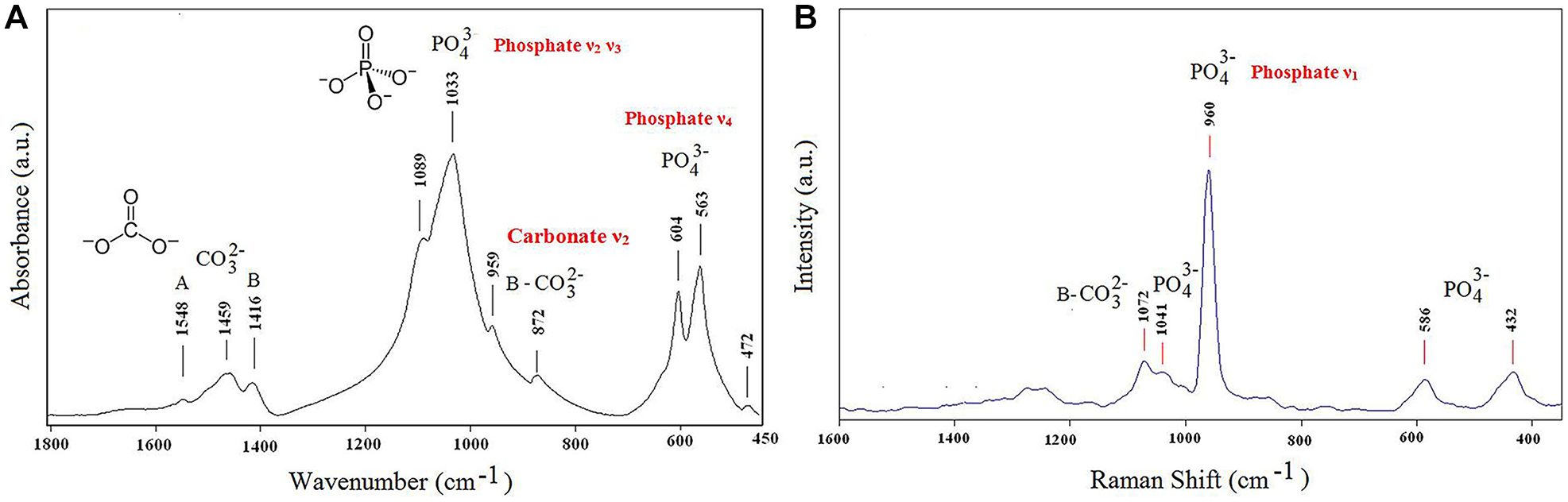
Representative (
The commonly used types of FTIR in mineralization research include attenuated total reflectance-FTIR (ATR-FTIR) and transmission FTIR. ATR-FTIR spectroscopy enables the lesion surface to be positioned directly against the diamond crystal of the ATR unit using a consistent pressure (Akgun et al. 2021). It requires only minimal sample preparation, with slight surface polishing for good contact with the ATR crystal. It can be used on thick samples without resin embedding (Lebon et al. 2014). While transmission FTIR requires thin, sectioned samples (~5-10 µm), usually embedded in resin, it offers higher spectral resolution and can probe deeper into the tissue. It is less commonly used due to more complex sample preparation but proves valuable in detailed cross-sectional chemical profiling of lesions (Mei et al. 2013). In addition, removal of water content from the samples before analysis is essential because water has broad IR absorbance, which can overlap and obscure key bands such as amide bands (Magalhães et al. 2021).
Raman Spectroscopy
Raman spectroscopy is a vibrational spectroscopic technique permitting the investigation of organic and mineral species at micron resolution (Kerr et al. 2016). It involves observing changes in molecular polarization when photons interact with vibrating molecular bonds, generating a spectrum. This nondestructive method of analysis provides direct data on the local atomic structure and chemical and molecular composition of biological samples (Seredin et al. 2020). In addition, Raman spectroscopy can be scaled down to handheld devices while preserving a high level of sensitivity (Marin et al. 2020).
Shifting in Raman spectra reveals vibrations in PO43− and CO32−, arising from carbonated HA (Fig. 4B), the mineral phase in enamel and dentin, which may indicate the substitution of the hydroxyl group (Ramakrishnaiah et al. 2015). Mineral-based spectral parameters have been used to investigate the structural changes in the tissue, namely, the carbonate to phosphate band area ratio (Akgun et al. 2021). The collagen signal in Raman spectroscopy, in addition to peaks corresponding to proline and hydroxyproline, also contains amide III, amide II, and amide I peaks. Changes in amide I and III have been shown to be related to dentin caries lesions (Benson and Cohen 1979). Raman spectroscopy is capable of providing quantitative insights into the crystalline structure of the phosphate groups and the loss of mineral fraction concerning the organic collagen matrix (Marin et al. 2020).
X-Ray Photoelectron Spectroscopy
X-ray photoelectron spectroscopy (XPS) utilizes an electron beam inside the X-ray source and focuses it onto an X-ray anode made of aluminium. The deceleration of the electrons on the anode leads to the production of X-rays. This X-ray beam is monochromatic and focused onto the sample. XPS is suited for the elemental analysis of surfaces, chemical state identification of surface species, and in-depth composition profiles of elemental distribution in relatively thin films (Hanawa et al. 1987). Given that the maximum information depth of the XPS method is limited to 10 nm, XPS analysis has been primarily used to quantify the chemical elemental composition and assess the HA’s chemical response at a superficial level (Yu et al. 2018).
X-Ray Diffraction
X-ray diffraction (XRD) is a method used to determine the atomic and molecular structure of a crystal by diffracting a beam of X-rays (Fatimah et al. 2021). XRD is a versatile technique used for both qualitative and quantitative analysis of crystalline materials. Quantitative analysis involves the determination of lattice parameters, lattice strain, and the presence of defects, while qualitative analysis involves phase identification, phase composition percentage, crystallite size, and orientation (Fatimah et al. 2021). For tooth specimens, it is commonly used to observe the crystal structure and gather the diffraction patterns of the HA crystal reflection on the surface of tooth specimens (Niu et al. 2021). The crystallite size is calculated by using the Scherrer equation from the XRD pattern (Zalite et al. 2022). The primary limitations of XRD are the potential destruction of the tooth structure and difficulty examining different layers of the tooth in the preferred orientation.
Atom Probe Tomography
Atom probe tomography (APT) is an emerging technique that provides chemical composition maps of materials in 3D, with sub-nanometer spatial resolution. Specimens must be prepared in the shape of a sharp needle with the radius at the tip in the range of 100 nm. An electrostatic field is applied to promote the ionization and desorption of the atoms from the material, toward a detector. The detector then measures the time of flight of the ions, which is used to determine the mass-over-charge ratio, and the (x, y) position of the ion impact on the detector, which is used to derive the respective position on the surface of the sample (Gault et al. 2021). APT has been used to analyze the hierarchical structure of human enamel, demonstrating the presence of ion gradients in the individual HA nanocrystals, with implications for their mechanical and chemical properties, such as solubility at low pH (DeRocher et al. 2020). While APT shows great promise in cariology research, it is a destructive technique and requires extensive specimen preparation and, as such, is low throughput.
Nuclear Magnetic Resonance
Solid-state nuclear magnetic resonance (NMR) is an analytical technique that uses the magnetic properties of atomic nuclei to detect the resulting changes (Zia et al. 2019). By rotating the sample, the magic-angle spinning (MAS) of NMR significantly enhances spectral resolution by narrowing the broad NMR lines.
The crystallography of apatite is a key factor of NMR in analyzing de-/remineralization. Two-dimensional (2D) 1H-31P heteronuclear-correlation MAS-NMR has been used to investigate the crystallographic differences in apatite (Hiraishi et al. 2022). Furthermore, MAS-NMR analysis has also been used to examine the influence of fluoride, strontium, and borate ions on the crystallization process during apatite precipitation, offering insights into how these ions may modulate remineralization efficacy (Hiraishi et al. 2024). In addition, 19F exhibited favorable intrinsic NMR properties because of its 100% natural isotopic abundance. 19F MAS-NMR analysis was used to characterize the solid-phase precipitation of fluoridated apatite on the enamel surface (Mohammed et al. 2013). However, NMR requires small, dry, and precisely shaped samples, making sample preparation challenging. Biological samples such as teeth usually need to be pulverized and dried for analysis, which also makes it impractical to localize lesions spatially.
Focused-Ion-Beam Scanning Electron Microscopy
Focused-ion-beam scanning electron microscopy (FIB-SEM) is a technique that is widely used in materials sciences and is being increasingly used in dentistry due to the capability of obtaining high-resolution (~5 to 10 nm) images of cross-sectioned samples (House et al. 2022). The instrument has a beam of gallium ions that is used to mill the specimen, while high-resolution imaging is undertaken using an SEM, as described previously. FIB-SEM offers several advantages over standard SEM, as by using the gallium beam to cut the sample, it enables the imaging and elemental composition determination by EDS of a cross section of the specimen, rather than analysis of only the surface. Importantly, sequential milling and imaging can be performed to yield a 3D reconstruction of the sample. Finally, the ion beam can also be used to cut ~80- to 100-nm-thick lamella for transmission electron microscopy (TEM). FIB-SEM has been used to investigate carious lesions on dentin from human teeth in 3D, in particular cross-sectional changes, such as penetration of minerals into dentinal tubules, the degree of integrity of the dentinal tubules, and the extent of remineralization in caries-arrested dentin (Yoshihara et al. 2020).
TEM (with Selected Area Electron Diffraction)
TEM is a microscopy technique that uses high-energy electrons passing through a sample to create images. It provides very high resolution (0.1-0.2 nm), allowing detailed views of enamel and dentin structures. TEM can show the shape of enamel rods and dentin tissue and how they are affected by caries. High-resolution TEM (HR-TEM) and selected area electron diffraction can also reveal the crystal structure and quality of HA minerals. These methods are useful for studying the dynamics of de-/remineralization of the tooth (Shao et al. 2019). TEM analysis revealed the morphology of nanocrystals and provided an excellent illustration of the nature of HA crystallite anisotropy in de-/remineralized enamel and dentin (Schwendicke et al. 2019).
Since TEM imaging requires electrons to pass through the sample, ultra-thin sections (less than 100 nm) need to be prepared using an ultramicrotome or FIB-SEM. The specimen block should be no larger than 1 mm³, as larger samples are difficult to image. TEM also operates under high vacuum, so complex preparation steps are needed before resin embedding and sectioning (Park et al. 2016). Although cryo-TEM is an option, it involves advanced freezing and cryo-sectioning techniques and needs to be performed at liquid nitrogen temperatures.
One limitation of TEM is that it produces 2D images of 3D structures, making it hard to understand the spatial relationships in complex tissues such as enamel and dentin. To overcome this, transmission electron tomography can be used, in which the sample is tilted and imaged from multiple angles to create a 3D view (Nudelman et al. 2011). This technique could also help in studying caries lesions in human teeth.
Conclusions
This review critically examined common laboratory methods used for evaluating mineralization in early caries lesions. It reviewed more than 10 techniques that encompass multiaspect evaluations, including mechanical properties, mineral density, chemical, and morphological characterizations of lesions at macro, micro, and nano levels. In addition, the authors emphasized the importance of assessing changes in the lesion body, particularly alterations in cross-sectional areas. A combined analytical approach offers a more comprehensive understanding of caries progression compared with simple surface measurements, which may lack accuracy, depth, and clinical significance.
This review will be particularly beneficial for in vitro studies testing the mineralization effects of interventions/factors, mechanistic studies explaining clinical results, and validity tests of different in vitro models. These methods offer high accuracy and sensitivity to detect subclinical changes, thus guiding the subsequent clinical research steps. The main information on the technologies is summarized in Table 1. We also acknowledge potential challenges, such as the unavailability of equipment, lack of technical support, and relatively high costs associated. Nevertheless, the authors propose that this article provides valuable information for researchers in selecting appropriate methodologies for assessing caries lesions that match the objectives of their studies.
Author Contributions
Y. Fu, contributed to data acquisition and analysis, drafted and critically revised the manuscript; F. Nudelman, contributed to conception, drafted and critically revised the manuscript; Y. Zhang, K.C. Li, contributed to data acquisition, critically revised the manuscript; M. Ekambaram, contributed to data interpretation, critically revised the manuscript; P.R. Cooper, contributed to conception, critically revised the manuscript; M.L. Mei, contributed to conception, design, data acquisition, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
