Abstract
The gradual phaseout of dental amalgam has contributed to a significant increase in the use of resin-based materials. While these materials offer several desirable properties, concerns persist regarding their biocompatibility, particularly the release of bisphenol A (BPA). BPA is an endocrine-disrupting chemical linked to adverse effects on human health, including reproductive, developmental, and metabolic disorders. Although food contact materials are the primary source of human BPA exposure and the contribution of dental materials is minor, the associated risks cannot be dismissed due to BPA’s nonmonotonic dose-response relationship. In 2023, the European Food Safety Authority proposed a 20,000-fold reduction in the tolerable daily intake of BPA to 0.2 ng/kg body weight, citing immune system effects at extremely low doses. This proposal has sparked regulatory and scientific debate, as adopting such a stringent limit would effectively ban the use of BPA in food contact materials and many other products. Given this context, it is essential to assess the release of BPA from dental materials both in vitro and in vivo. However, data interpretation is complicated by methodological inconsistencies, including variations in material composition, specimen preparation, choice of extraction media, experimental duration, and analytical methods. In addition, pivotal differences in reporting results make it difficult to synthesize findings and draw reliable conclusions. This review examines the controversy surrounding BPA, critically evaluates evidence on its release from dental materials, and explores mitigation strategies. By highlighting gaps in knowledge and proposing future research directions, this review aims to provide clinicians, researchers, and policymakers with a clearer understanding of BPA-related complexities, ultimately contributing to patient safety and material innovation.
Introduction
Bisphenol A (BPA) is a synthetic organic compound used across various industries, particularly in the food sector, where it is a key component of polycarbonates and epoxy resins used in food contact materials. Other sources of human exposure to BPA include nonfood consumer products (e.g., thermal paper, toys, cosmetics), health care applications, medical devices, and dental materials, as illustrated in Fig. 1 (Geens et al. 2012). Due to its widespread application and endocrine-disrupting properties, BPA contamination in the environment and its potential impact on human health have drawn significant attention from researchers, regulators, and the public (Vogel 2009).
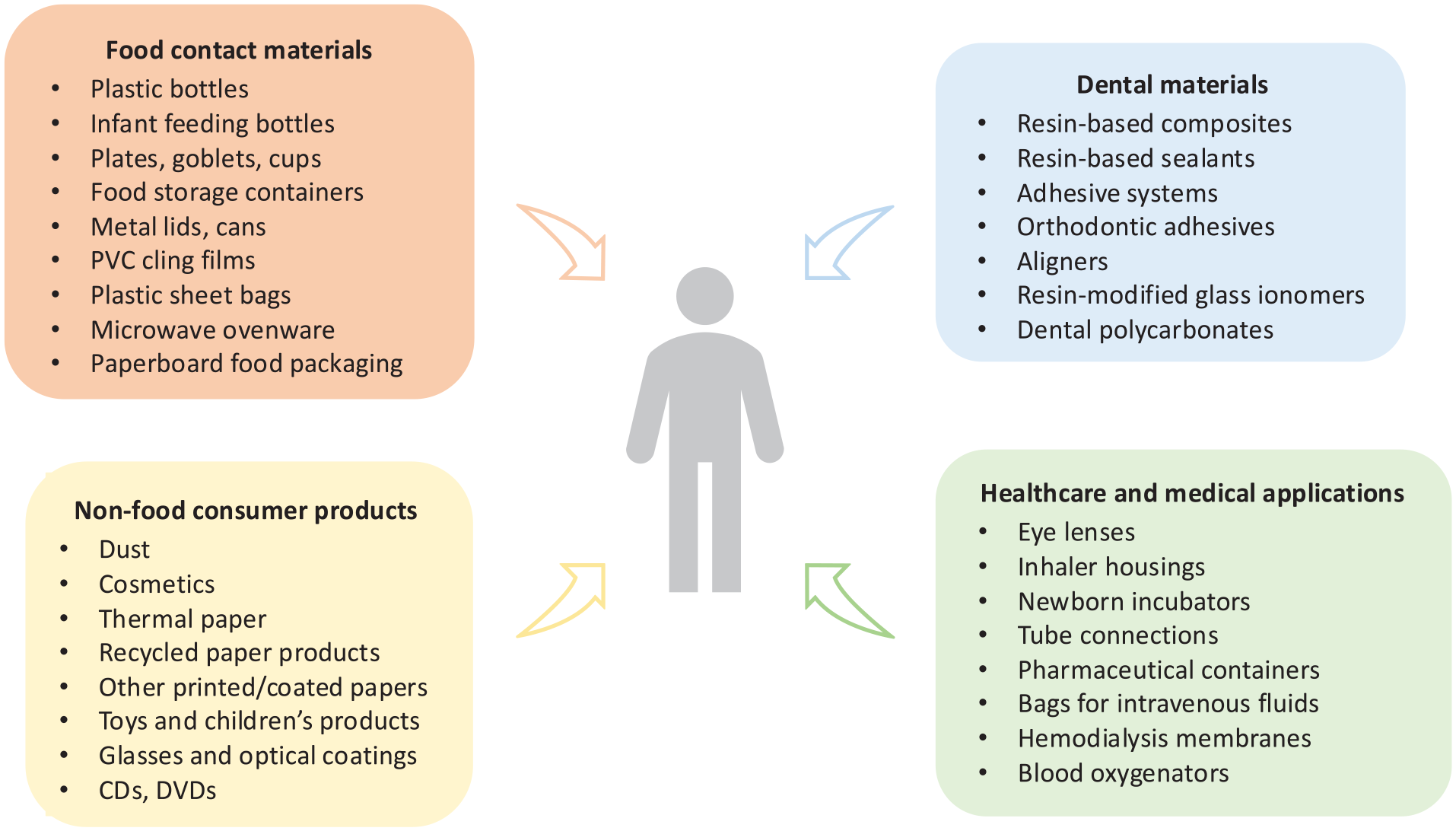
Human exposure to bisphenol A: overview of sources (Geens et al. 2012; EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids 2015).
In dentistry, the relevance of BPA is linked to the increasing use of resin-based materials. Until relatively recently, dental amalgam was the primary restorative material for posterior teeth. However, this has changed due to the improved properties of resin-based composites and global initiatives such as the Minamata Convention on Mercury, which have encouraged the gradual phaseout of amalgam (Mackey et al. 2014). Paradoxically, while amalgam has been restricted due to environmental and potential health concerns, resin-based dental materials could face similar scrutiny due to the presence of BPA in some commonly used monomers (e.g., bisphenol A-glycidyl methacrylate; Bis-GMA). Although BPA is covalently bonded in these monomers, it can still be released over time through degradation and hydrolysis (Fig. 2) (De Nys, Duca, et al. 2021). Consequently, dental materials are frequently cited as a (potential) source of BPA.
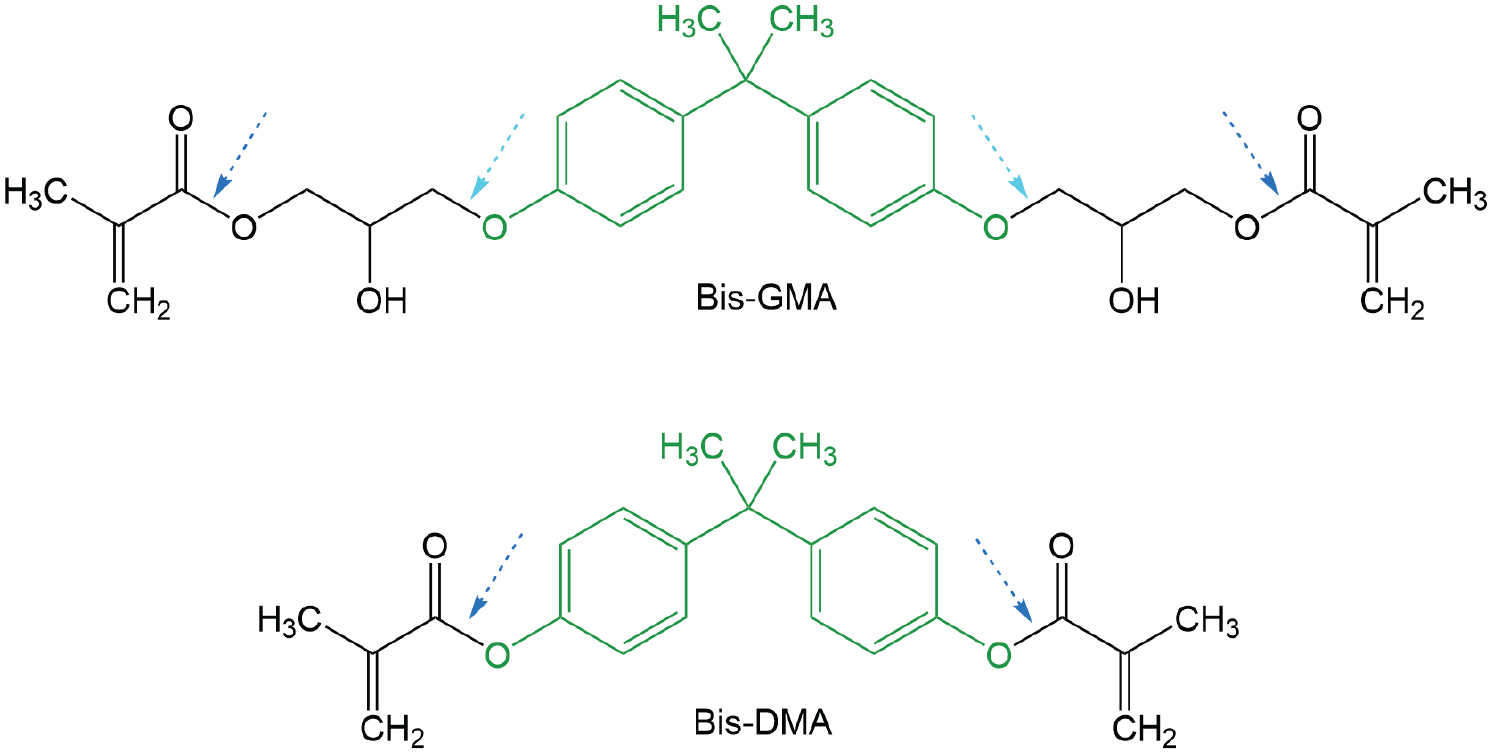
Examples of bisphenol A (BPA)–derived monomers, bisphenol A-glycidyl methacrylate (Bis-GMA) and bisphenol A dimethacrylate (Bis-DMA). BPA (highlighted in green) is incorporated into the structure via an ether bond (light blue arrow) in Bis-GMA, which is more resistant to hydrolysis than the ester bond (dark blue arrow) in Bis-DMA. Although Bis-GMA also contains an ester bond (dark blue arrow) linking the glycidyl group to the methacrylate moiety, its cleavage is less significant, as it does not lead to the release of free BPA.
This review was prompted by 2 recent events: the proposal of the European Food Safety Authority (EFSA) to reduce the tolerable daily intake (TDI) of BPA by 20,000 times, that is, to 0.2 ng/kg body weight per day (EFSA Panel on Food Contact Materials, Enzymes and Processing Aids et al. 2023), and the European Union’s (EU’s) ban on the use and export of amalgam, effective from January 2025 (Council of the EU 2024). The primary objective of this review is to summarize the controversy surrounding BPA, its associated health risks, and the evolving regulatory landscape. Furthermore, the review critically evaluates the existing evidence on the release of BPA from dental materials, emphasizing methodological heterogeneity and variability in reporting. Finally, the review explores mitigation strategies and directions for future research and development within the field of dental materials. This should provide a clearer understanding of BPA-related complexities, ultimately contributing to improved patient safety and material innovation.
Endocrine-Disrupting Properties and Adverse Effects
BPA (2,2-bis(4-hydroxyphenyl)propane, CAS No. 80-05-7) is a crystalline substance consisting of 2 phenolic rings linked by a propane bridge. Its molecular weight is 228.29 g/mol, and given its logKOW of 3.321, BPA is more soluble in fats than in water (Liu et al. 2009). Due to its aromatic structure, featuring a hydroxyl group in the paraposition and methyl groups on the second carbon of the propane bridge, BPA can bind to various hormone receptors (Wetherill et al. 2007), thus being classified as an endocrine disruptor (Kavlock et al. 1996).
The estrogenic properties of BPA were first described in the 1930s (Dodds and Lawson 1936). At the time, studies showed that BPA’s affinity for nuclear estrogen receptors (ERα and ERβ) was approximately 10,000 times lower than that of estradiol (Vogel 2009). However, it was later revealed that BPA can activate other estrogen-responsive pathways with potency comparable to estradiol, including membrane estrogen receptors and the G-protein–coupled estrogen receptor (GPR30) (Rochester 2013). Furthermore, BPA has a high affinity for the estrogen-related receptor gamma (ERRγ), which is crucial for brain and lung development (Okada et al. 2008). Given these findings, BPA should not be classified as a weak xenoestrogen, a claim further supported by animal studies demonstrating organ-specific estrogenic effects (Rochester 2013). In addition to its estrogenic activity, BPA can bind to androgen receptors, where it blocks the effects of natural androgens (Wetherill et al. 2007).
The impact of BPA exposure on the reproductive system has been extensively studied, with research suggesting an association between BPA exposure and reduced fertility, altered sexual development, ectopic pregnancy, and an increased risk of hormone-sensitive cancers (Rochester 2013). Developmental effects have been documented in animal models, including an increased risk of low birth weight and neurodevelopmental alterations, such as increased anxiety and altered social behavior. In humans, epidemiological studies have also indicated that BPA may be linked to developmental disorders, but it is generally difficult to disentangle confounding factors (Ma et al. 2019).
Beyond the reproductive system, BPA has also been associated with metabolic disorders. For instance, by disrupting the activity of intracellular aromatase, BPA can contribute to lipogenesis and the differentiation of fibroblasts into adipocytes, thereby being classified as an obesogenic compound (Alonso-Magdalena et al. 2006). Other actions of BPA include disruption of thyroid hormone homeostasis, increased insulin resistance, and contribution to type 2 diabetes. There is also growing recognition of BPA’s immunomodulatory effects (Ma et al. 2019).
The evaluation of endocrine-disrupting effects is complicated by BPA’s nonmonotonic dose-response relationship, where low doses may lead to biological effects that are not observed at higher doses (Vandenberg et al. 2012). This challenges the traditional toxicologic principle that “the dose makes the poison” and suggests that even minimal BPA exposure might induce clinically significant outcomes (Teeguarden and Hanson-Drury 2013). Furthermore, the effects of BPA may be amplified by other endocrine disruptors (Kortenkamp 2007).
Given this complexity, establishing safe exposure thresholds remains questionable, but caution should be prioritized especially with regard to vulnerable populations (Valentino et al. 2016). Fetuses, infants, and young children are particularly susceptible to BPA exposure and may experience disproportionate effects due to developing organ systems. Although the understanding of cellular and tissue-level effects of BPA is incomplete, even minor hormonal fluctuations during critical windows of development can result in long-term physiological changes (Valentino et al. 2016).
Regulations of BPA
Regulatory agencies worldwide began evaluating BPA exposure levels in the early 2000s, after BPA was classified as an endocrine disruptor (Kavlock et al. 1996) (Fig. 3). Prompted by mounting epidemiological evidence, regulators applied the precautionary principle and progressively tightened exposure limits. Canada was the first country to declare BPA a toxic compound in 2008 and imposed a ban on BPA in baby bottles, followed by the EU in 2011 and the United States Food and Drug Administration (FDA) in 2012 (Rogers et al. 2013). At that time, the TDI for BPA was set at 50 µg/kg body weight per day, but in 2015, EFSA lowered it to 4 µg/kg body weight per day (EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids 2015).
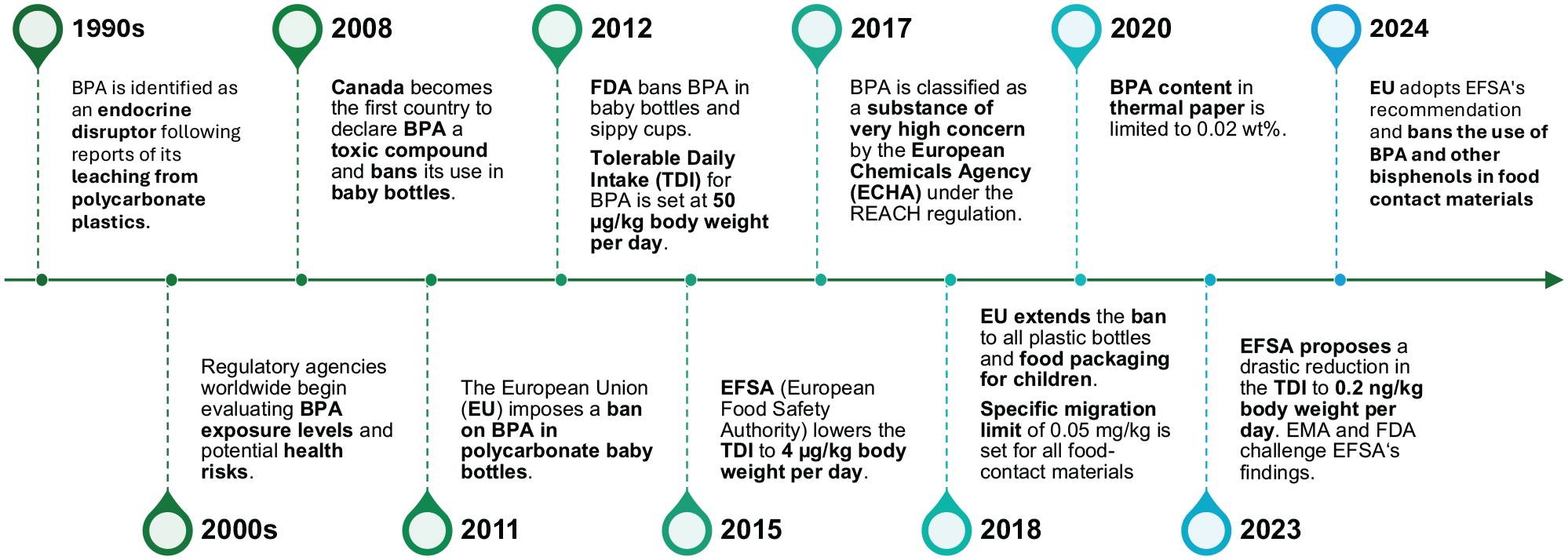
Timeline of BPA regulations.
European regulators continued imposing tighter controls after the European Chemicals Agency classified BPA as a substance of very high concern in 2017. This prompted further evaluations with potential future restrictions under the European Commission Regulation 1907/2006, also known as REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals). The ban on BPA in polycarbonate baby bottles was extended to all plastic bottles and food packaging for children by the European Commission Regulation 2018/213, which also set a specific migration limit of 0.05 mg/kg for all food contact materials. Another regulatory change targeted thermal paper, widely used in receipts and labels, where the XVIIth amendment to the REACH regulation limited BPA content to 0.02 wt% as of January 2020.
The most drastic regulation was introduced in 2023, when EFSA announced the outcomes of its latest reassessment and proposed lowering the TDI to 0.2 ng/kg body weight per day (EFSA Panel on Food Contact Materials, Enzymes and Processing Aids et al. 2023). This proposal indeed triggered dissent from the BPA-dependent industry, but it also ignited a dispute within the scientific and regulatory communities. Notably, the European Medicines Agency and FDA questioned EFSA’s methodology, as it included not only industry-funded studies conducted under good laboratory practice protocols but also published academic findings (Vom Saal et al. 2024). Moreover, EFSA was criticized for its reliance on an intermediate endpoint in an animal study, that is, an increased frequency of Th17 lymphocytes in the spleen of offspring mice (Luo et al. 2016), rather than established apical endpoints with clear human health effects. Critics further pointed out that major toxicologic studies, such as CLARITY-BPA (Consortium Linking Academic and Regulatory Insights on BPA Toxicity), did not show immunotoxicity at these low doses, although Th17 lymphocytes were not specifically evaluated (Prueitt and Goodman 2024). However, EFSA defended its approach and received support from the scientific community (Zoeller et al. 2023; Vom Saal et al. 2024).
Ultimately, the European Commission adopted EFSA’s recommendation and banned the use of BPA in food contact materials in December 2024. For most products, there will be an 18-mo phaseout period, and very limited exceptions will be made where no alternatives exist, to allow industry time to adapt and avoid disruption in the food chain. The ban also includes other bisphenols because of growing concerns that their endocrine-disrupting properties may be even worse than those of BPA (Eladak et al. 2015; Rochester and Bolden 2015). This prevents the so-called regrettable substitution of BPA by alternative bisphenols, i.e., compounds structurally similar to BPA but differing in substituents or the bridging unit between phenolic rings (Rochester and Bolden 2015).
Release of BPA from Dental Materials
Concerns over the exposure to BPA from dental materials were sparked by a study in which 18 volunteers had a resin-based sealant (Delton, Dentsply) applied to all molars (Olea et al. 1996). Samples of their saliva were collected 1 h after application, and unexpectedly high BPA amounts (90–931 µg) were revealed. While these findings were not replicated and some researchers suggested that Olea et al. (1996) may have confused the BPA signal with TEGDMA, as their retention times were similar under the employed experimental conditions (Hamid and Hume 1997), the concerns persist to date.
Nowadays, research concerning the release of BPA from dental materials is centered on resin-based composites, including those for computer-aided design/computer-aided manufacturing. BPA-derived monomers may also be present in adhesive systems, hybrid glass ionomer cements, and resin-based sealants that are primarily used particularly in young children, who are more susceptible to BPA exposure. Fewer studies were dedicated to orthodontic materials (e.g., adhesives, aligners, retainers) and polycarbonates, which may serve as an alternative to poly(methyl methacrylate) for provisional crowns and splints.
Factors Influencing the Release of BPA
In term of composition, polycarbonates have the highest BPA content (Tichy et al. 2021), as they are synthetized through the polymerization of BPA and phosgene (COCl2). However, polycarbonates have not been thoroughly examined, as they are not used as often as resin-based materials containing BPA-derived monomers, such as Bis-GMA, its ethoxylated derivative, propoxylated bisphenol A-dimethacrylate, and bisphenol A-diglycidyl ether. Dental sealants may also contain bisphenol A dimethacrylate, in which BPA is ester bonded. This ester bond is more susceptible to hydrolytic degradation than the ether bonds found in other BPA-derived monomers, making BPA release from sealants potentially higher (Schmalz et al. 1999; Atkinson et al. 2002) (Fig. 2). BPA is primarily released as a result of polymer degradation, but it may also be present in trace amounts as a contaminant from the synthesis of BPA-derived monomers (Fleisch et al. 2010; Löfroth et al. 2019; Kechagias et al. 2020; De Nys, Duca, et al. 2021).
The quality of polymerization is another critical determinant, as it affects both the amount of residual monomers and solvent permeability into the material (Van Landuyt et al. 2011). Polymerization of direct light-curing materials is influenced by various factors, including exposure duration, irradiance, emission spectrum, photoinitiator type, mutual distance, angulation and concentricity of the material and light-curing unit, material thickness and optical properties, and so forth (AlShaafi 2017). However, high-quality polymerization is essential even for polymeric materials manufactured in the laboratory or industrially, as inadequate polymerization increases the risk of BPA release.
Beyond material composition and polymerization, intraoral conditions play a significant role in BPA release, as they facilitate the breakdown of polymer networks (Sakaguchi et al. 2019). Hydrolysis and enzymatic degradation are the primary pathways, whereas acidic and alkaline conditions have been reported to have only a minor effect (De Nys, Duca, et al. 2021). In addition, temperature fluctuations and mechanical stress from mastication can accelerate the chemical and physical degradation of dental polymers. However, the impact of these conditions on the release of BPA remains insufficiently studied, as their accurate simulation in vitro is challenging.
In vitro Studies
While numerous in vitro studies have been performed, the synthesis of their findings is hindered by differences in methodologies and reporting, mainly specimen shape and size, extraction media, duration of exposure, analytical methods, and reported units. Specimen shape and size determine the volume and mass of the material, hence influencing the total amount of leachable BPA. While this seems obvious, if the information is not accurately reported, interpreting the data becomes difficult. Moreover, the specimen surface area affects the rate of BPA release, thus being an important factor in short-term studies (Van Landuyt et al. 2011). In fact, most in vitro studies analyzed eluates collected after 24 h or less, which is of limited relevance, because the rate of BPA release decreases sharply during the first days (Tichy et al. 2022). The measured amounts depend on the extraction medium as well: organic solvents (methanol, ethanol) generally penetrate hydrophobic materials more effectively than aqueous solutions (saliva, artificial saliva, water) do, leading to higher BPA release in organic media (Van Landuyt et al. 2011). Finally, the volume of the extraction medium plays a crucial role; according to the ISO 10993 series of standards, it should be at least 10 mL per 1 g of test material to achieve a maximum concentration gradient in the direction of substance release.
In addition, given the trace amounts of BPA in eluates of dental materials, sensitive analytical methods are required. The eluate is usually separated using chromatography, and individual compounds are subsequently detected by mass spectrometry (MS) or optical methods, for example, ultraviolet-visible spectrometry (UV/VIS). For separation, liquid chromatography (LC) is preferred over gas chromatography (GC), because the temperature of the GC column may initiate the decomposition of monomers such as Bis-GMA (Cavalheiro et al. 2015). The detection of the separated compounds using UV/VIS is based on retention time, as opposed to MS, which identifies compounds based on their mass-to-charge ratio, achieving higher sensitivity and specificity than UV/VIS.
The current gold standard method for BPA analysis is ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS). High operating pressure and ultra-fine particle size enable UHPLC to provide fast, high-resolution separation with greater sensitivity. This is followed by MS/MS, which further increases specificity and sensitivity by using 2 or more mass spectrometers in sequence (Hope et al. 2016). While UHPLC-MS/MS achieves lower limits of quantification (LLOQ) around 0.1 ng/mL (Becher et al. 2018), the detection of BPA can be further improved by derivatization (e.g., using pyridine-3-sulfonyl chloride [LLOQ 0.073 ng/mL]) (De Nys et al. 2018) or dansyl chloride (LLOQ 0.042 ng/mL) (Kolatorova Sosvorova et al. 2017). The value of LLOQ is important because it represents the minimum concentration at which reliable quantification is possible. When BPA levels approach or drop below LLOQ, the reliability of the analysis is decreased. Despite its importance, some studies did not report LLOQ at all, and some methods (with LLOQ as high as 0.5 μg/mL) are not suitable to detect trace amounts of BPA.
Ultimately, inconsistent reporting of results across studies presents another major challenge for interpretation. Some authors express BPA concentrations per volume of extraction media (e.g., µg/L, µg/mL, ng/mL, mmol/L, ppm, ppb), while others report BPA amounts relative to surface area (ng/cm², mmol/mm², pmol/mm²) or weight (µg/g, ng/g, pg/mg) of test specimens. Although conversion may seem straightforward, it requires information on specimen mass, volume, and/or surface area and the volume of extraction medium, which is often missing. To address this issue, standardizing the unit µmol/mm² was proposed in a meta-analysis of components released from dental resin–based materials, as a positive correlation with specimen surface area was found (Van Landuyt et al. 2011). However, most studies included in the meta-analysis used an exposure of 24 h, and since the total amount of leachable components depends on material volume/mass, the correlation with surface area would likely weaken with increasing exposure duration.
A comprehensive overview of in vitro studies is beyond the scope of this review. However, Tables 1 to 3 present the selected results of UHPLC-MS/MS studies (Becher et al. 2018; De Nys et al. 2018; De Nys, Putzeys, et al. 2021; Tichy et al. 2021; De Nys et al. 2022; Tichy et al. 2022), a meta-analysis of BPA release from dental resin–based materials (Van Landuyt et al. 2011), and an HPLC-MS study of various materials (Mazzaoui et al. 2002). To allow for their comparison, the reported values were converted into the amount of released BPA per specimen volume. Conversion into amounts relative to specimen mass would have been preferred, but it was not applicable for all included studies.
Overview of BPA Release from Resin-Based Composites after 24 h.
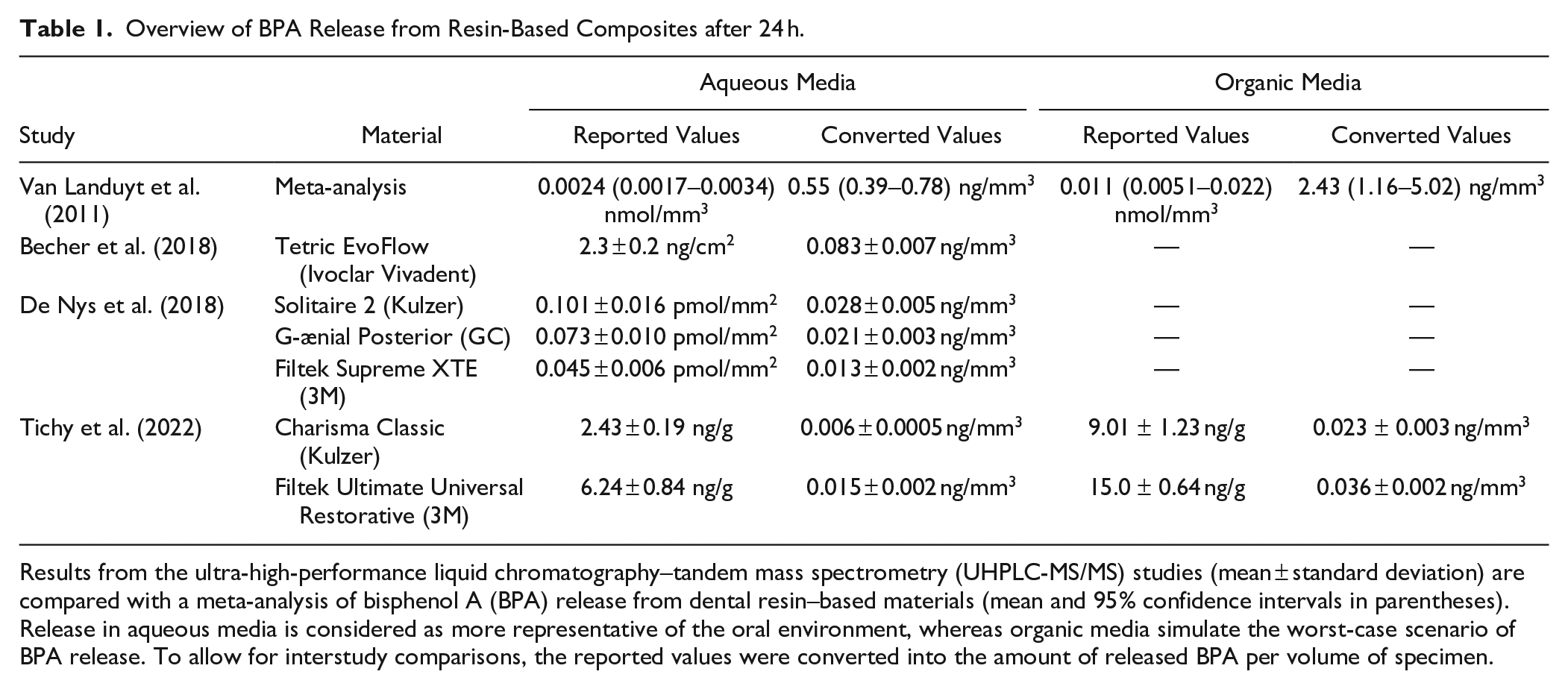
Results from the ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS) studies (mean ± standard deviation) are compared with a meta-analysis of bisphenol A (BPA) release from dental resin–based materials (mean and 95% confidence intervals in parentheses). Release in aqueous media is considered as more representative of the oral environment, whereas organic media simulate the worst-case scenario of BPA release. To allow for interstudy comparisons, the reported values were converted into the amount of released BPA per volume of specimen.
Overview of Long-Term BPA Release from Resin-Based Composites.
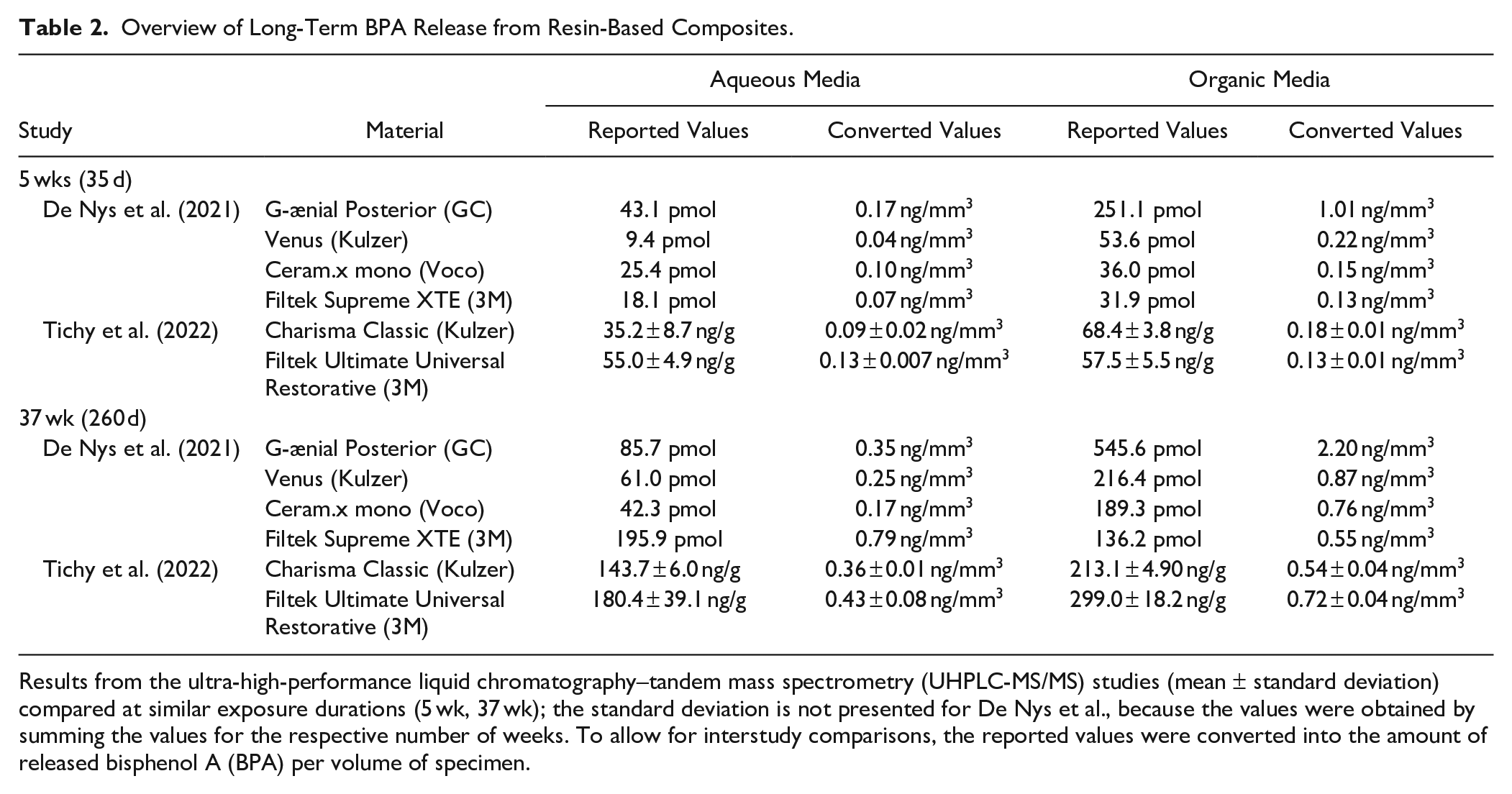
Results from the ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS) studies (mean ± standard deviation) compared at similar exposure durations (5 wk, 37 wk); the standard deviation is not presented for De Nys et al., because the values were obtained by summing the values for the respective number of weeks. To allow for interstudy comparisons, the reported values were converted into the amount of released bisphenol A (BPA) per volume of specimen.
Overview of Short-Term BPA Release from Sealants, Adhesives, Orthodontic Adhesives, Glass Ionomers, and Polycarbonates in Aqueous Media.
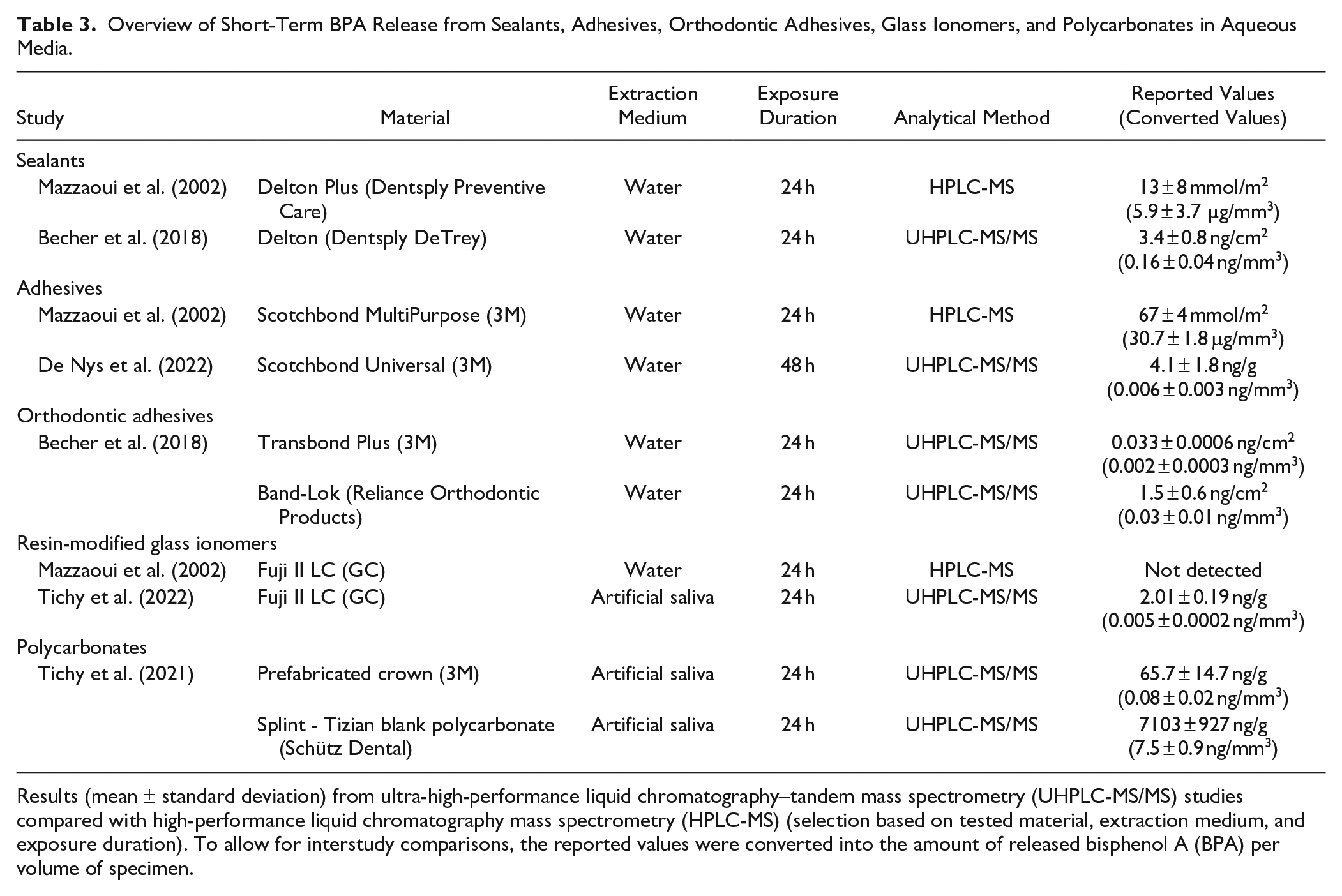
Results (mean ± standard deviation) from ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS) studies compared with high-performance liquid chromatography mass spectrometry (HPLC-MS) (selection based on tested material, extraction medium, and exposure duration). To allow for interstudy comparisons, the reported values were converted into the amount of released bisphenol A (BPA) per volume of specimen.
Tables 1 and 2 are dedicated to the release of BPA from resin-based composites. After 24 h (Table 1), the released amounts ranged from 0.01 to 0.1 ng/mm3, which is approximately 10-fold lower than in the meta-analysis of studies using less sensitive and specific methods (Van Landuyt et al. 2011). While the rate of BPA release decreases after the first day, BPA is continuously released in low doses (Table 2). The presented results also confirm that the release in organic media is higher than that in aqueous media (De Nys, Putzeys, et al. 2021; Tichy et al. 2022).
Table 3 summarizes the short-term BPA release from other materials in aqueous solutions, which have been used more commonly than organic media. The importance of accurate analytical methods is demonstrated by discrepancies between UHPLC-MS/MS results and the HPLC-MS study (Mazzaoui et al. 2002), which either measured much higher values or did not detect BPA at all. Compared with resin-based composites, more BPA was released from the Delton sealant, but the release from adhesives, orthodontic adhesives, and a resin-modified glass ionomer was similar or lower (Becher et al. 2018; De Nys et al. 2022; Tichy et al. 2022). Polycarbonates were found to release much higher amounts of BPA if they were milled or 3D printed, as opposed to prefabricated crowns (Tichy et al. 2021).
In summary, future studies should evaluate BPA release from dental materials not only after 24 h but also over extended periods. They should enable trace-level detection by employing state-of-the-art analytical methods, such as UHPLC-MS/MS, and report the LLOQ value. Ideally, both aqueous and organic extraction media should be used, as aqueous solutions better simulate the oral environment, while organic solvents represent the worst-case scenario of BPA release. The volume of the extraction medium, specimen shape, dimensions, and mass must be clearly reported to allow for the conversion of absolute amounts of released BPA to values relative to surface area, volume, or mass. While Van Landuyt et al. (2011) previously recommended standardizing the unit µmol/mm², we suggest using ng/g. This unit better reflects long-term release, which is more closely related to specimen mass and volume than surface area, and it is more practical for correlating BPA exposure with TDI in clinical and regulatory contexts.
In Vivo Studies
In vivo studies conducted after the report of Olea et al. (1996) have consistently confirmed transient increases in BPA concentrations in saliva and other body fluids following the application of dental resin–based materials. Nevertheless, BPA levels returned to baseline within 24 h (Arenholt-Bindslev et al. 1999; Joskow et al. 2006; Zimmerman-Downs et al. 2010; Lee et al. 2017; Berge et al. 2019), except for orthodontic retainers (Raghavan et al. 2017). Long-term elevation of BPA levels was also reported by a cross-sectional study, which showed that urinary concentrations of BPA were significantly higher in children who had 11 or more surfaces previously restored with sealants and resin composites compared with those with zero restorations (Chung et al. 2012). However, no significant difference was observed in the group with 1 to 10 restored surfaces (Chung et al. 2012), and other studies did not find a significant positive association between dental sealants or restorations and urinary BPA levels (McKinney et al. 2014; Snoj Tratnik et al. 2019).
The ambiguity in these findings likely arises from strong confounding factors, particularly dietary BPA exposure, which is the primary source of human BPA intake. As indicated by in vitro studies, the amounts of BPA released from dental materials are substantially lower than the estimated daily exposure (EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids 2015), making it difficult to distinguish the in vivo effect from baseline even if highly sensitive analytical methods are used. This complicates efforts to link BPA exposure from dental materials to any adverse effects on human health. While current evidence does not justify changes in clinical practice (McKinney et al. 2014), potential risks cannot be entirely dismissed due to the nonmonotonic dose-response relationship.
Mitigation Strategies and Future Perspectives
The growing awareness of BPA’s potential risks has prompted various strategies to minimize exposure from dental materials. One approach involves refining their chemistry to either reduce or eliminate BPA. For resin-based composites, manufacturers have introduced products labeled as “BPA-free,” which were found to release significantly less BPA than composites containing BPA-derived monomers (Tichy et al. 2022). However, the complete replacement of BPA-derived monomers is difficult as they have very favorable physical, chemical, and mechanical properties. Another approach aims at the development of materials with completely different monomer structures, such as ormocers (organically modified ceramics), which were introduced in the 1990s in an attempt to reduce polymerization shrinkage. The precursors of ormocers are alkoxysilanes, with the resulting material consisting of silica domains to which polymerizable (meth)acrylate groups are attached (Moszner and Salz 2001).
Clinical best practices also play an important role. Dentists should follow manufacturers’ instructions for use, especially in terms of light-curing protocols, as polymerization is a crucial factor that determines the amount of residual monomers (Van Landuyt et al. 2011). This involves selecting a light-curing unit with a suitable emission spectrum and light intensity as well as optimizing the curing process, mainly ensuring adequate exposure duration, minimizing distance from the cured material, and checking the angulation and concentricity of the light beam (AlShaafi 2017). The release of monomers, which could degrade into BPA, can also be reduced by adequate management of the oxygen-inhibited layer (e.g., final curing through a glycerin gel) (Marigo et al. 2019). Furthermore, employing barriers such as a rubber dam during restoration finishing and removal is suitable to limit the ingestion of material residues.
More caution might be required for appliances with a large surface area, as they could be a relevant source of BPA. For orthodontic retainers and aligners, the information about BPA release is scarce and conflicting, so their safety remains questionable until high-quality studies are conducted (Peter et al. 2023). On the contrary, polycarbonate splints were found to release considerable amounts of BPA (Suzuki et al. 2000; Watanabe 2004; Tichy et al. 2021); therefore, using other types of splints should be preferred. In general, the use of BPA-free alternatives should be considered, particularly for high-risk groups such as pregnant women or children. If patients are concerned about the release of BPA, clinicians should educate them accordingly.
Looking forward, future regulations of BPA may play a decisive role even for dental materials. This could lead to stricter control (e.g., mandatory testing and reporting of BPA release) but also a complete ban of BPA-derived materials. Researchers and manufacturers should, therefore, continue exploring alternative monomers as well as strategies to reduce their release. In addition, large-scale epidemiological studies are needed to clarify the association of BPA exposure from dental materials with health outcomes. These studies should be conducted in the long term, in realistic clinical scenarios, and they should employ state-of-the-art analytical methods.
Conclusion
The ongoing debate over BPA and its potential health risks has led to increasingly stringent regulations. While numerous studies confirm that BPA can be released from dental polymer materials, measured levels are generally far below the estimated daily exposure from other sources. However, the interpretation of published findings is complicated by methodological heterogeneity, making it difficult to draw definitive conclusions about clinical significance. In addition, the nonmonotonic dose-response relationship raises concerns that even low levels of exposure may have biological effects. To mitigate potential risks, dentists should make informed decisions regarding material selection and adhere to best clinical practices.
Author Contributions
A. Tichy, contributed to conception, design, data acquisition and interpretation, drafted the manuscript; T. Srolerova, contributed to conception, design, data acquisition and interpretation, critically revised the manuscript; F. Schwendicke, contributed to conception and design, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation (project DFG KR 5457/1-1) and Charles University (project Cooperatio 207030 Dental Medicine/LF1).
