Abstract
The human papillomavirus (HPV) is the most common sexually transmitted infection of the 21st century, infecting an estimated 630 million people worldwide and contributing to approximately 4.5% to 5% of all cancers. The incidence of HPV-associated oropharyngeal cancer is increasing. Yet, population screening for oral HPV infection is not recommended by the Food and Drug Administration due to the low prevalence and lack of a universally accepted screening strategy or tool. The study aims to evaluate the link between cervical and oral HPV infection in women and identify high-risk groups. The protocol was registered on PROSPERO (CRD42023477695). The systematic search was performed in 3 databases (PubMed, EMBASE, and CENTRAL) on January 3, 2025. Forty-one analytical observational studies reporting on oral HPV status in women with and without cervical HPV positivity were included. Effect sizes, including proportions and odds ratios with 95% confidence intervals (CIs), were analyzed using a random-effects model. Location, population characteristics, and sampling methods were used to perform the subgroup analyses. The meta-analysis revealed an odds ratio of 2.22 (CI: 1.52–3.25) for oral HPV in women with cervical HPV positivity compared with those without it. The prevalence of oral HPV was significantly higher in cervical HPV-positive women (13%, CI: 8%–20%) than in HPV-negative women (4%, CI: 2%–7%). In addition, the prevalence of oral HPV was even higher in cervical HPV-positive women with positive cytology (17%, CI: 6%–39%). Risk-of-bias assessment indicated low risk among all studies using Quality in Prognosis Studies and moderate to low risk among studies assessed with the Joanna Briggs Institute Prevalence Critical Appraisal Tool. Our findings support the association between cervical HPV positivity and elevated risk for oral HPV infection. Moreover, the highest prevalence of oral HPV carriage was observed in women with positive cytology. As 1 in 8 women with cervical HPV carries oral HPV, screening should be considered for this at-risk population.
Keywords
Introduction
The human papillomavirus (HPV) is the most common sexually transmitted infection of the 21st century, infecting an estimated 630 million people worldwide and contributing to approximately 4.5% to 5% of all cancers (Roman and Aragones 2021; Shapiro 2022). There are approximately 200 different genotypes of HPV, of which more than 20 are known or probable carcinogenic (Shapiro 2022). Almost all sexually active individuals will contract HPV at some point in their lives (Chesson et al. 2014; Perkins et al. 2023).
HPV infects the micro-traumatized epithelium during sexual contact, where the basement membrane is exposed. Due to cell-mediated immune mechanisms, asymptomatic high-risk HPV infection can be cleared within a period of time. If the immune system cannot eliminate HPV, it can lead to malignant transformation and cancer (Berman and Schiller 2017; Perkins et al. 2023). Apart from its well-known association with cervical cancer, HPV is also implicated in malignancies of the oropharynx as well as the anus, vagina, vulva, and penis (Pinkiewicz et al. 2022).
Oropharyngeal squamous cell carcinoma (OPSCC) has one of the most rapidly increasing incidence rates among all cancers in high-income countries (Lechner et al. 2022). Globally, approximately 85,000 new cases of OPSCC are diagnosed each year, with an estimated 22,000 being HPV positive. The male-to-female ratio is approximately 4:1 (Berman and Schiller 2017). The global burden of HPV-associated OPSCC varies significantly across regions, with higher incidences reported in North America, Europe, and Oceania. In the United States, the age-standardized incidence rate (ASIR) for males is 3.41 per 100,000, while for females, it is 0.71 per 100,000. Similarly, in Europe, the ASIR is 1.72 per 100,000 for males and 0.41 for females (Ndon et al. 2023). The incidence of HPV-positive OPSCC is rising rapidly in high-income countries. For instance, in the United States, the proportion of OPSCC cases attributed to HPV increased from 16% in the 1980s to 73% by the mid-2000s (Berman and Schiller 2017). Similar trends are observed in Europe, with HPV-positive OPSCC cases comprising approximately 41.9% of all OPSCCs (Ndon et al. 2023). Notably, while HPV-positive OPSCC has historically been more prevalent in men, recent data suggest a significant increase in its occurrence among women as well. Between 1995 and 2012, the proportion of p16-positive OPSCCs in women increased from 29% to 77% in certain regions of the United States (Ndon et al. 2023). While cervical cancer rates have declined in developed countries due to effective prevention programs, the incidence of HPV-positive oropharyngeal cancer (OPC) is increasing and exceeding cervical cancer (Roman and Aragones 2021).
Despite this trend, there are significant gaps in standardized screening methods for oral HPV, and the Food and Drug Administration (FDA) does not support population-based oral HPV screening, citing low prevalence and lack of a universally accepted screening strategy or tool (Sullivan-Chang et al. 2017). Furthermore, no OPC precursor lesion has been clearly identified, and the multistep process of oropharyngeal carcinogenesis has not yet been clarified (Berman and Schiller 2017). Given these challenges, the identification of high-risk populations is crucial for targeted follow-up and implementation of advanced prevention strategies.
Several new articles have been published since the previous meta-analysis, in which the pooled prevalence of oral HPV reached 15.5% (confidence interval [CI]: 6.9%–24.2%) in cervically positive women (Termine et al. 2011). Moreover, the direct comparison between cervically HPV-positive and -negative women has not been previously studied. We aimed to investigate whether cervical HPV positivity increases the risk of oral HPV infection and to assess the prevalence of oral HPV among cervical HPV-positive and -negative women.
Methods
The present systematic review and meta-analysis were conducted based on the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guideline and the Cochrane Handbook. The study protocol was registered on PROSPERO (registration number CRD42023477695).
For our clinical questions, we used the following frameworks: the CoCoPop framework to assess the prevalence of oral HPV infection in women with cervical HPV (condition: oral HPV, context: cervical HPV, population: women) and the PECO framework to examine whether cervical HPV infection increases the risk of oral HPV infection in women (population: women, exposure: cervical HPV positive, comparison: cervical HPV negative, outcome: oral HPV infection) (Moola et al. 2015).
Eligibility Criteria
Observational cross-sectional and cohort studies were included, reporting on cervical and oral HPV infection in women and providing data on the concordance of HPV infection between the 2 body parts. Finally, we followed stricter inclusion criteria, as the genotypes in the Gardasil 9 HPV vaccine had to be tested.
One of the primary outcomes was the odds ratio (OR) of oral HPV positivity in women with cervical HPV infection compared with a cervical HPV-negative control group; the other primary outcome was the prevalence of oral HPV positivity in women with and without cervical HPV infection. Secondary outcomes were the prevalence and OR of oral HPV positivity in women with positive cytology and the sex worker population, divided by oral sampling method, continent, and low-risk, high-risk genotype of HPV with a particular focus on HPV 6, 11, 16, 18, 31, 33, 45, 52, 58 distribution.
The following studies were excluded: case reports, conference abstracts, reviews, and studies that did not report cervical and oral HPV concordance or did not respond to our request, as well as studies that did not test all the genotypes of the Gardasil 9 vaccine.
Sources of Information and Search Strategy
The systematic search was conducted in Medline (via PubMed), Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) on November 3, 2023. No filters or language restrictions were applied to the search. The search was updated on January 3, 2025.
The following search terms were used in the systematic search: (HPV OR human papillomavirus* OR human papilloma virus*) and (cervix OR cervixes OR cervices OR cervical OR genital* OR endocervix OR endocervical OR ectocervix OR ectocervical) and (oral OR mouth OR oropharynx OR oropharyngeal OR mesopharyngeal). See Appendix Table 1 for the full search terms.
Selection Process
Selection was performed by 2 independent reviewers (A.E.M. and F.H.O.). After removal of duplicates, articles were screened by title and abstract, and then those that met our inclusion criteria were selected based on full texts.
Data Collection Process and Data Elements
Two authors (A.E.M. and F.H.O.) independently collected data from eligible articles. The following data were extracted: first author, year of publication, study location, DOI, study type, study period, study population characteristics, oral sampling method, cervical cytology result, HPV DNA detection method, number of cervically or orally positive and negative women, and number of cervical and oral low-risk, high-risk, and unknown HPVs, along with, separately, the number of the 9 genotypes (HPV 6, 11, 16, 18, 31, 39, 45, 52, 58) contained in the Gardasil 9 vaccine at the cervix and in the oral region.
Quality of Assessment and Quality of Evidence
Two authors (A.E.M. and F.H.O.) independently assessed the risk of bias based on the Cochrane Collaboration, using the Joanna Briggs Institute Prevalence Critical Appraisal Tool (JBI) and the Quality in Prognosis Studies (QUIPS) tools. The JBI has 9 criteria for target population and study setting. Each criterion is scored as “yes,” “no,” “unclear,” or “not applicable” based on the information provided in each study, with a total score potential of 9 points. A higher score indicates a lower risk of bias. The QUIPS tool contains 6 domains of prognostic factors, each scoring as “low,” “moderate,” “high,” or “no information” based on the information provided.
We evaluated the quality of evidence for the outcomes based on the principles of the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) Working Group (Schünemann et al. 2013). Using the GRADEPro Guideline Development Tool (Schünemann et al. 2013), we created the Summary of Findings tables (Appendix Table 6 and 7).
Synthesis Methods
As considerable between-study heterogeneity was assumed in all cases, a random-effects model was used to pool effect sizes.
The ORs and proportions were used as effect size measures with 95% CIs. To calculate study ORs and pooled ORs, the total number of patients and those with the event of interest in each group separately was extracted or calculated from the studies where available. The results show the odds of an event of interest in the experimental group compared with the odds of an event of interest in the control group. To calculate study proportions and pooled proportions, the total number of patients and those with the event of interest were extracted from each study.
Results were considered statistically significant if the pooled CI did not contain the null effect value. We summarized the findings of the meta-analysis in forest plots. Where applicable—the study number is sufficiently large and not too heterogeneous—we also reported the prediction intervals of results (i.e., the expected range of effects of future studies).
In addition, between-study heterogeneity was described by the Higgins and Thompson I2 statistics (Higgins and Thompson 2002). Small study publication bias was assessed by visual inspection of Funnel plots and calculating the Peters (modified Egger’s) test p-value (Peters et al. 2006). Potential outlier publications were explored using different influence measures and plots following the recommendation of Harrer et al. (2021).
All statistical analyses were performed with R (R Core Team 2021) using the meta (version 6.5-0; Balduzzi et al. 2019) package for basic meta-analysis calculations and plots and the dmetar (Harrer et al. 2019) package for additional influential analysis calculations and plots. Additional details on calculations, data synthesis, publication bias assessment, and influential analyses can be found in the Supplementary Material (Additional Synthesis Methods).
Results
Search and Selection
Our systematic literature search identified a total of 10,545 studies, of which 39 were found suitable for qualitative and quantitative synthesis. In addition, 2 more studies were found eligible by citation search. The PRISMA flowchart of the search is shown in Figure 1.
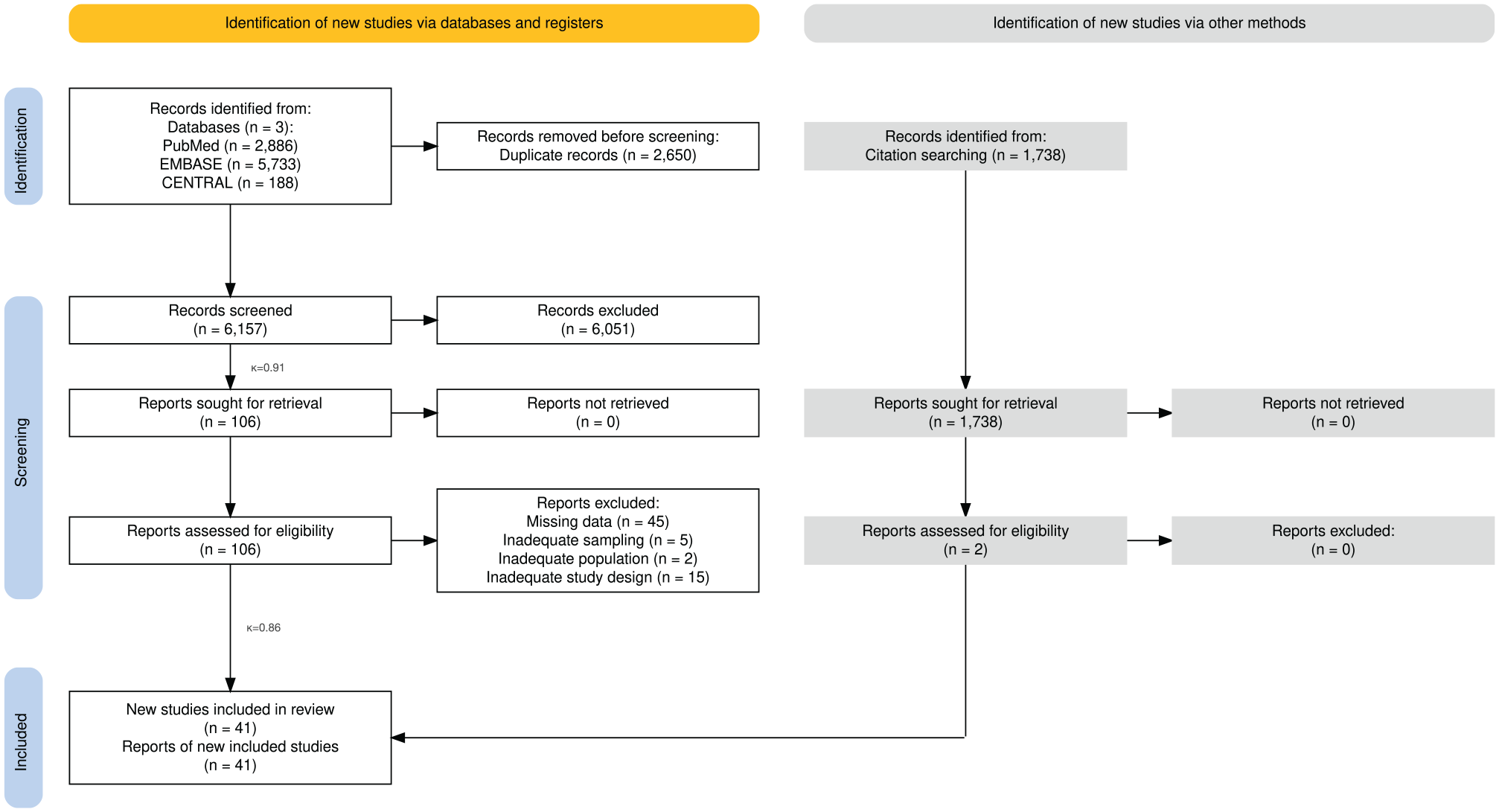
PRISMA 2020 flowchart of the study screening and selection process.
Main Characteristics of the Included Studies
Most studies were cross-sectional, conducted in different geographical regions, including Europe, North and South America, Africa, and Asia between 2004 and 2020. The baseline characteristics of enrolled studies are detailed in Appendix Table 3.
OR of Oral HPV in women with and without Cervical HPV
Thirty-four studies, including 5,276 women, reported oral HPV carriage in the cervical HPV-positive and cervical HPV-negative groups. The overall OR for oral HPV positivity was 2.22 (CI: 1.52–3.25; I2 = 39% [CI: 7%–59%]), as shown in Figure 2.
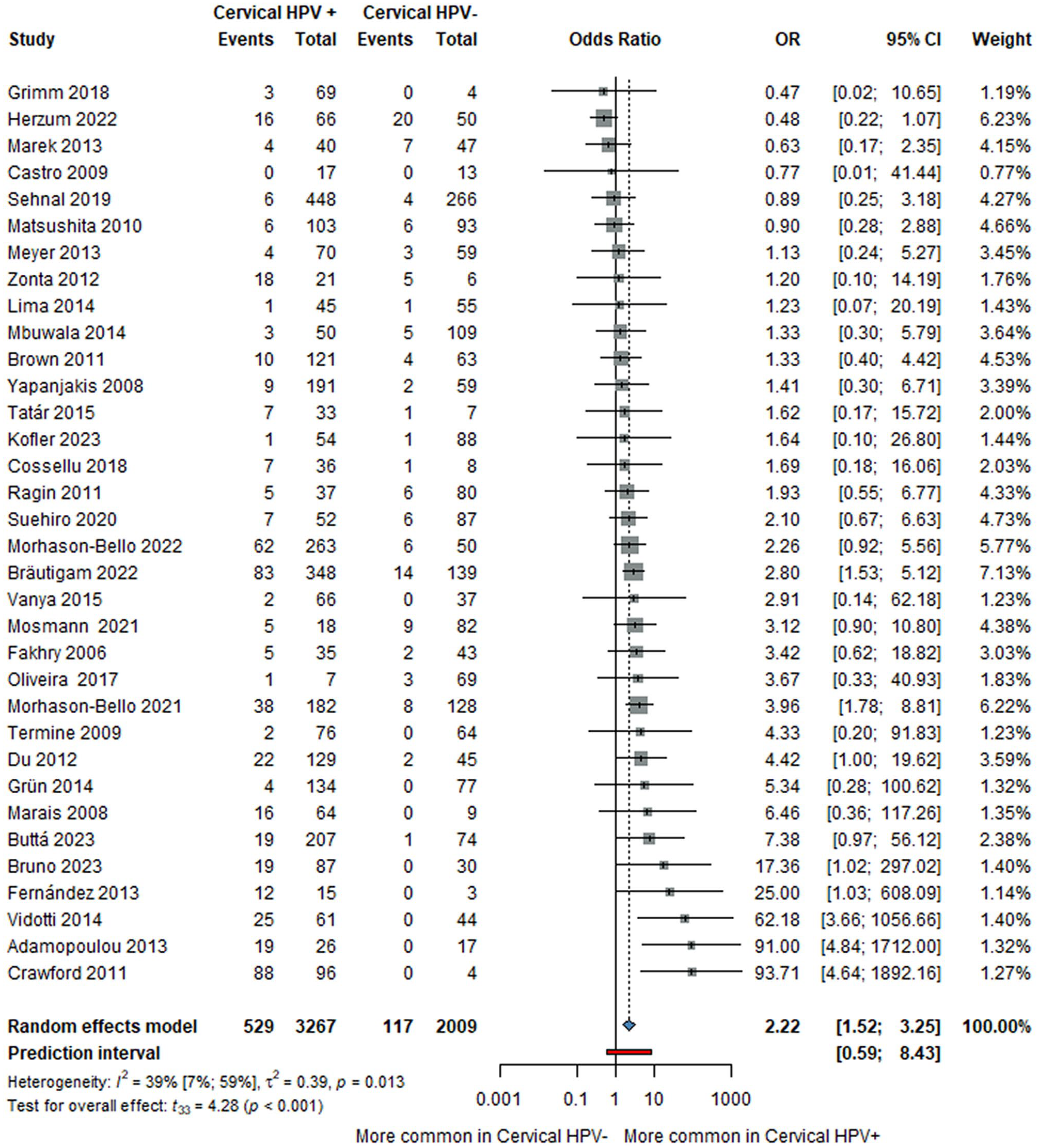
Forest plot showing the odds ratio for oral HPV in women with and without cervical HPV. CI, confidence interval; HPV, human papillomavirus; OR, odds ratio.
Prevalence of Oral HPV in Women with and without Cervical HPV
The overall prevalence of oral HPV in the cervical HPV-negative group was 4% (CI: 2%–7%; I2 = 66% [CI: 51%–76%]), including 34 studies with 2,009 women, while the overall prevalence of oral HPV in the cervical HPV-positive group was 13% (CI: 8%–20%; I2 = 91% [CI: 88%–93%]), including 41 studies with 4,146 women. Details are shown in Figures 3 and 4.
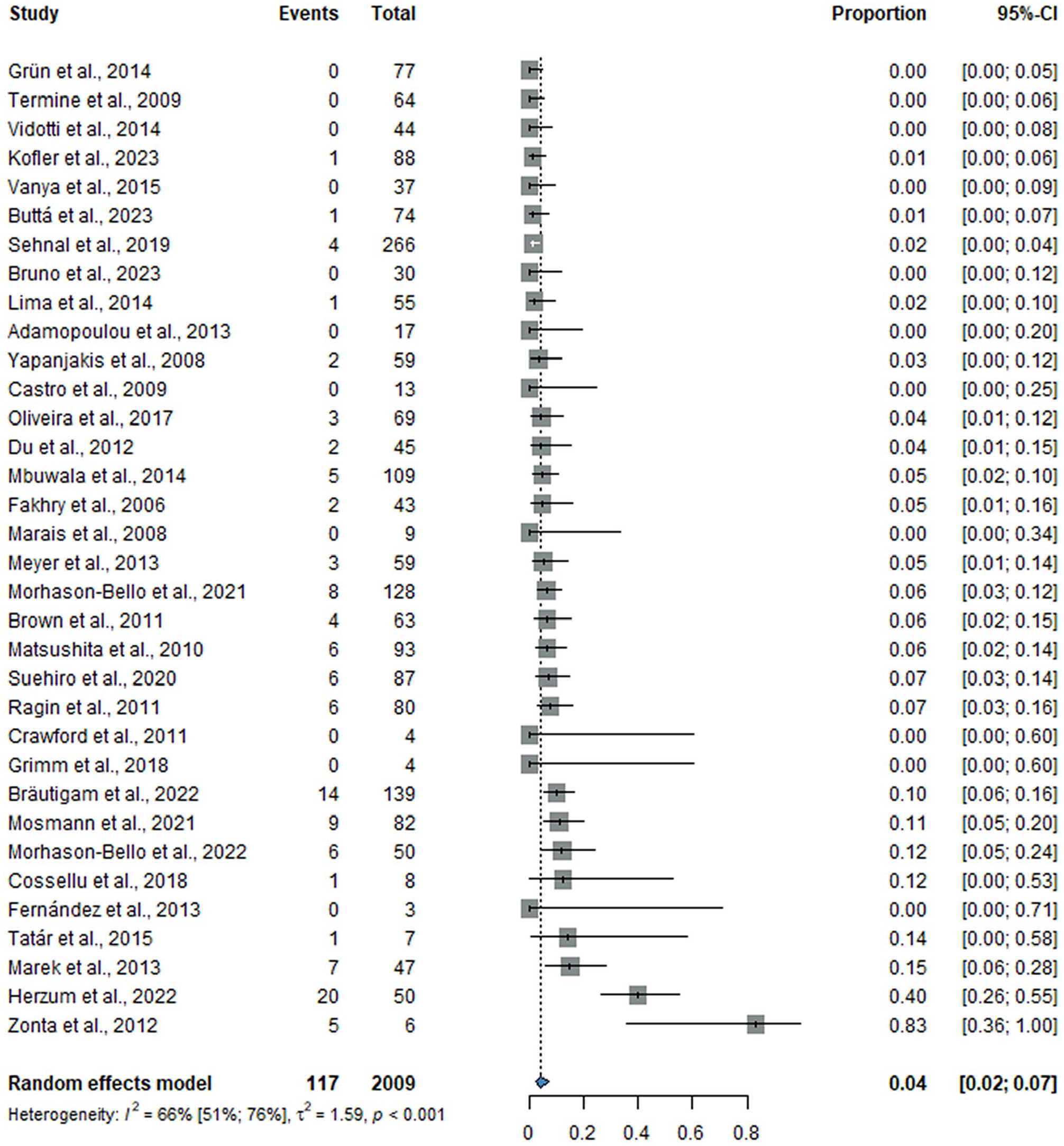
Forest plot showing the prevalence of oral HPV in women without cervical HPV. CI, confidence interval; HPV, human papillomavirus.
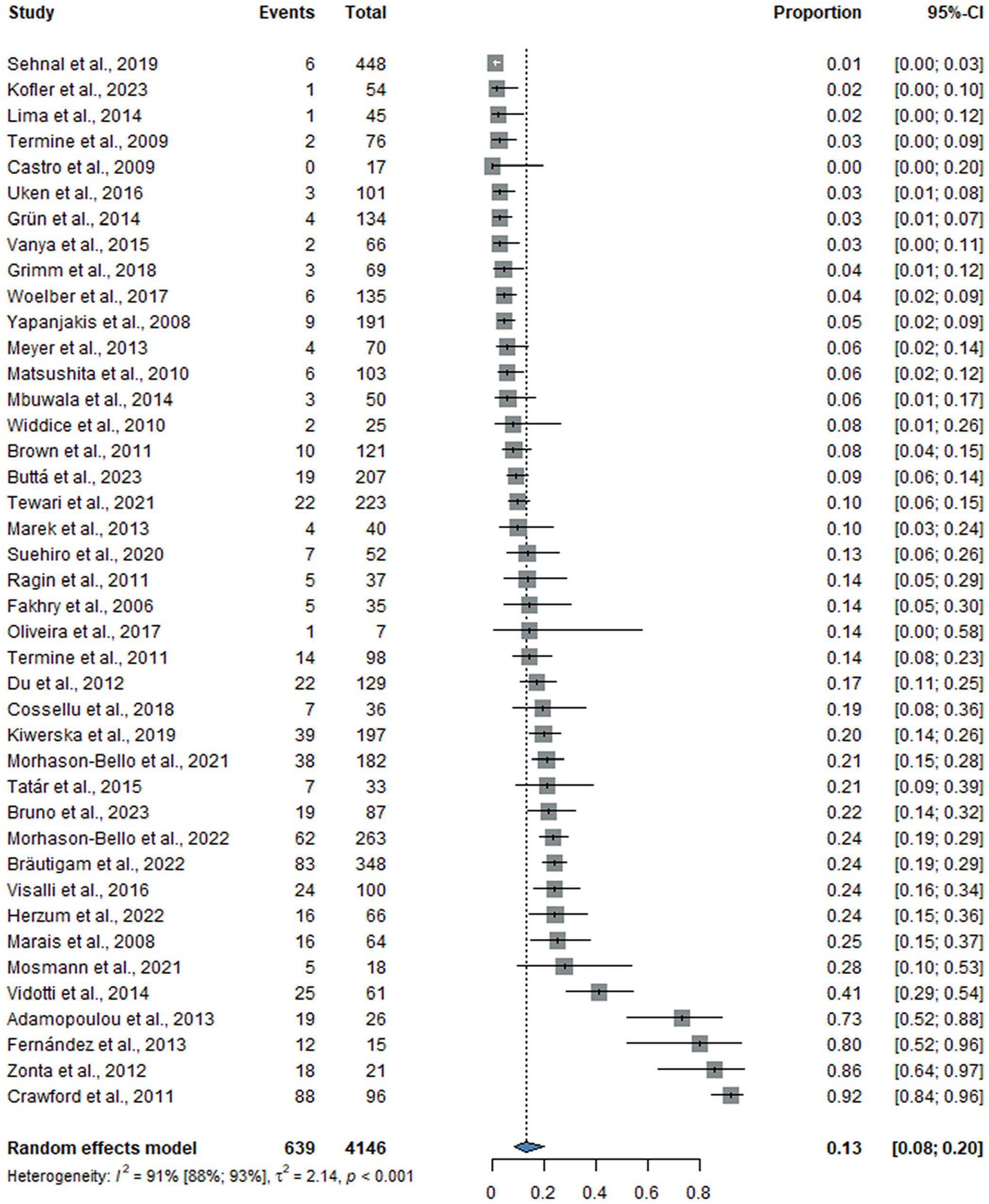
Forest plot showing the prevalence of oral HPV in women with cervical HPV. CI, confidence interval; HPV, human papillomavirus.
Prevalence of Oral HPV in Women with Cervical HPV and Positive Cytology
The overall prevalence of oral HPV in the cervical HPV-positive group with positive cytology was 17% (CI: 6%–39%; I2 = 95% [CI: 92%–96%]), including 14 studies with 1,186 women, as shown in Figure 5.
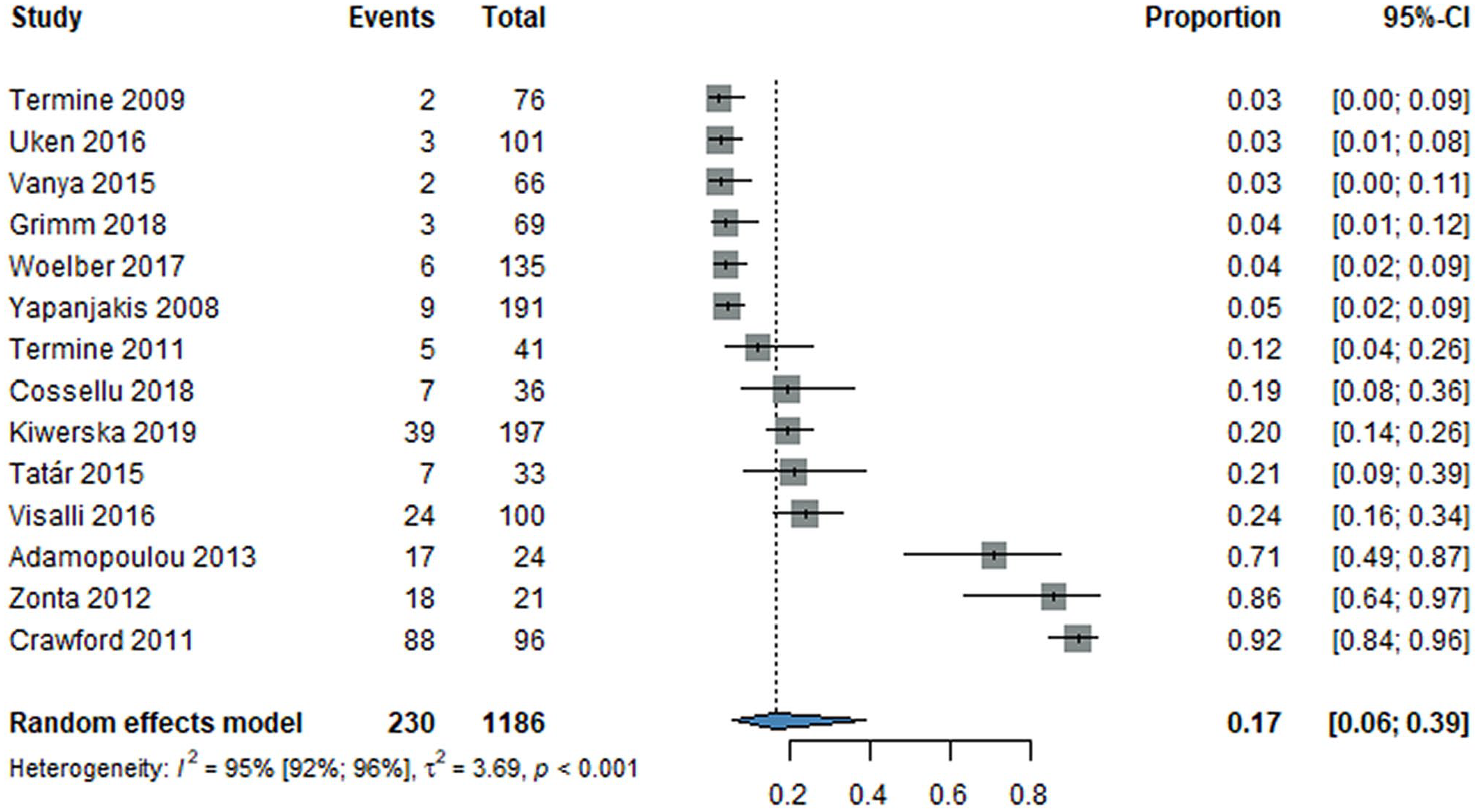
Forest plot showing the prevalence of oral HPV in women with cervical HPV and positive cytology. CI, confidence interval; HPV, human papillomavirus.
Subgroup Analyses
Multiple analyses were performed to assess ORs and proportions by continent, oral sampling technique, sex worker population, and different genotype distribution (Appendix Figs. 2–9, 36, 37). The rinsing technique (OR = 3.07, CI: 1.26–7.50; I2 = 43% [CI: 0%–75%]) yielded higher odds than the brushing method did (OR = 2.20, CI: 1.21–4.01; I2 = 47% [CI: 13%–68%]), but the difference between the groups did not reach statistical significance (p = 0.482). Comparing brushing sites, we obtained higher odds in the oral cavity (OR = 2.92, CI: 1.82–4.68; I2 = 0% [CI: 0%–60%]) versus the oropharynx (OR = 2.05, CI: 0.63–6.66; I2 = 38% [CI: 0%–71%]) but without reaching statistical significance between the groups (p = 0.525). In terms of distribution between continents, oral HPV prevalence was highest in North America and lowest in Europe. In the sex worker population, we obtained lower numbers than in our general population in terms of odds (OR = 1.39, CI: 0.60–3.20; I2 = 0% [CI: 0%–85%]) and proportion in cervically HPV-positive women (12%, CI: 4%–30%; I2 = 86% [CI: 67%–94%]).
Risk-of-Bias Assessment
All studies assessed using the QUIPS tool had a low risk of bias, while studies assessed using the JBI had a score of 6 or higher, indicating a moderate or low risk of bias. The results of the risk-of-bias assessment are shown in Appendix Tables 4 and 5.
Publication Bias and Heterogeneity
Most results showed moderate to high heterogeneity. The reasons for heterogeneity could possibly be explained by inconsistent oral sampling methods and different virus detection techniques, as well as heterogeneous populations in terms of age, global and individual differences in sexual practices, and partner HPV status.
The Peters test was performed for ORs and proportional analyses. The main test results suggest no significance, indicating no evidence of funnel plot asymmetry and publication bias (p = 0.1592, p = 0.0651, p = 0.1591, and p = 0.1931). A leave-one-out sensitivity analysis was also performed for the ORs and proportions, and no studies seem to have affected the outcome. The results of the Peters test, funnel plots, and leave-one-out analyses are shown in Appendix Figures 10 to 13 and 23 to 26.
Quality of Evidence
Appendix Tables 6 and 7 present the summary of finding tables. We assessed our outcomes (oral HPV positivity in women with cervical HPV, without cervical HPV, and with cervical HPV and positive cytology). All of our findings were classified as low or very low in the certainty of evidence ranking due to the nonrandomized nature of the trials, high heterogeneity, and wide confidence intervals.
Discussion
Our study showed a strong association between cervical and oral HPV infection. The results show that women with cervical HPV have 2.22 (CI: 1.52–3.25) times higher odds of developing oral HPV infection. One in 8 women with cervical HPV has oral HPV infection (13%, CI: 8%–20%), while 1 in 25 women without cervical HPV carries the virus orally (4%, CI: 2%–7%). In addition, the prevalence of oral HPV in women with cervical HPV and positive cytology is even higher, 17% (CI: 6%–39%).
In our meta-analysis, we observed some outliers that may be due to small population size, different countries, different patient characteristics, oral sampling techniques and kits, more sensitive nested polymerase chain reaction technique, population with positive cytology, and population with other risk factors (smokers, contraceptive users, prisoners, and sex workers) (Crawford et al. 2011; Zonta et al. 2012; Adamopoulou et al. 2013; Fernández et al. 2013; Vidotti et al. 2014). Several risk factors have been identified for cervical HPV, including smoking and oral contraceptives (Mazarico et al. 2014; Xu et al. 2018; Asthana et al. 2020). Female prisoners may be at risk for more frequent oral sexual activity (Yap et al. 2010). We found no significantly higher results when we looked at the population of sex workers, who are much more exposed to HPV due to the large number of sexual partners. This could presumably be explained by the fact that sex workers use condoms during oral sex more often than the general population does, but there are conflicting findings in the literature (Brown et al. 2011; Magalhaes et al. 2019; Lee et al. 2021).
The highest oral HPV carriage was in women with cervical HPV positivity and positive cytology. Virtually all positive cervical cytology cases are caused by HPV infection; in cases in which cytological lesions are already observed, HPV has been present on the cervix for a relatively longer period of time (Berman and Schiller 2017; Perkins et al. 2023), presumably allowing more time for infection of the mucosa of the oral cavity and oropharynx.
Inadequate oral hygiene, which leads to disrupted oral microbiome, periodontitis, and periodontal pockets, may contribute to conditions that support HPV infection and persistence, potentially serving as reservoirs for the virus (Chow et al. 2010). A comprehensive cross-sectional analysis identified poor oral health, measured by various indicators, as a risk factor for oral HPV (Bui et al. 2013). Furthermore, several studies have pointed to the higher prevalence of HPV in potentially malignant and malignant oral disorders (Bouda et al. 2000; Szarka et al. 2009; Mazul et al. 2017).
Smoking contributes to the persistence of oral HPV and is more frequently observed in patients with HPV-related OPC. In the study, multivariate analysis revealed that the most significant prognostic factor for survival in HPV-driven OPC was smoking status at the time of diagnosis. Specifically, smokers had a hazard ratio of 0.25 compared with nonsmokers, indicating a substantially better survival outcome for nonsmokers (Kero et al. 2014; Mirghani et al. 2018).
Although this study focused only on women, the exposure of men to HPV should not be ignored. The predominance of men is not fully understood. This is probably explained by the fact that HPV copy numbers are much higher in the cervix than in the penis. Therefore, heterosexual men are exposed to a much higher number of viral copies during oral sex than heterosexual women are (Berman and Schiller 2017). This is supported by a study in which the standardized incidence of tonsil or tongue cancer was 2.7:1 in husbands of women with cervical cancer, compared with 2.4:1 in husbands of women with cervical intraepithelial neoplasia (Hemminki et al. 2000).
Currently, there is no known precursor lesion for HPV-induced OPC, and the multistep process of carcinogenesis remains unclear. In addition, no reliable oral HPV detection tests are available. The most commonly used methods include brush or swab sampling and the rinse technique. In the brush and swab technique, cells are collected directly from the mucosa of the oral cavity and/or oropharyngeal region. In contrast, the rinse technique involves collecting cells from the detached surface using a fluid medium, such as water, mouthwash, or physiological saline (Dona et al. 2019). A study on oral rinses collected from individuals undergoing tonsillectomy revealed HPV positivity in 10.3% of the samples, while none of the resected tonsil tissues tested positive for HPV (Grun et al. 2017). Another study revealed lower HPV detection rates in oropharyngeal and oral brushings compared with oral rinses, with poor concordance between methods, especially for HPV-positive cases. Oral rinses identified more HPV types, but their origin remained unclear (Dona et al. 2019).
Therefore, the main preventive option against HPV-positive OPCs is HPV vaccination, the first of which was approved in 2006 (Berman and Schiller 2017; Shapiro 2022). In 2020, the FDA approved the Gardasil 9 HPV vaccine for the prevention of HPV-positive oropharyngeal and other head and neck cancers (Markowitz and Schiller 2021). Australia was one of the pioneers in implementing a gender-neutral vaccination program, with high uptake rates of 75.9% for boys and 80.2% for girls completing the 3-dose regimen. By contrast, only half of adolescents in the United States had completed the recommended vaccination regimen by 2018, and nearly one-third remained unvaccinated (Lechner et al. 2022). A study from Costa Rica observed a 93.3% reduction in oral HPV 16 and 18 infections in young women who were vaccinated against HPV, reinforcing the potential of vaccination to reduce OPC risk (Herrero et al. 2013). A study analyzing 1.67 million Swedish girls and women from 2006 to 2017 reported a significant reduction in the population-level risk of invasive cervical cancer (Lei et al. 2020). Expected herd immunity is still far away, with coverage in the World Health Organization regions between 29% and 60% instead of the desired 80% in 2020, and vaccine uptake has declined in many high-income countries (Shapiro 2022). Despite documented evidence of immunity to reinfection, immunity after natural HPV infection is incomplete and poorly understood. In contrast, immunity from the HPV vaccine gives about 90% protection against HPV infection for at least 15 years (Perkins et al. 2023).
A concept in which different medical professions need to work together and act in an interdisciplinary way to control HPV effectively is possible. A woman with a positive cervical HPV result and/or cytological dysplasia could be referred to an otorhinolaryngologist and/or dentist with the appropriate knowledge and closely followed for possible progression of HPV infection. As it is sexually transmitted, partner involvement, screening, and follow-up would also be essential. There are several known risk factors for oral HPV carriage, including smoking, sexual behavior (greater number of sexual partners and oral sexual activity), stress, and HPV infection at another anatomical site or partner site (Shigeishi and Sugiyama 2016; Lugovic-Mihic et al. 2021; Dzundova et al. 2023). All these factors should be considered during follow-up.
Strengths and Limitations
Regarding the strengths of our analysis, we followed our preregistered protocol, and a rigorous methodology was applied. We presented worldwide data, considered HPV detection by polymerase chain reaction technique, set a minimum genotype requirement for inclusion, and performed subgroup analysis.
Among the limitations of this work, it should be noted that there is no standardized oral sampling method for HPV screening and that different HPV test kits were used in the studies. Although our study shows a strong association between oral and cervical HPV infection, a meta-analysis cannot replace primary studies. Supervised observational studies would be needed to more accurately investigate the nature of HPV infection.
Implications for Practice and Research
Implementing scientific findings in practice is crucial for the benefit of patients (Hegyi et al. 2020; Hegyi et al. 2021). There is a need to raise awareness of oral HPV infection, its risk for cancer, and more prevention programs. Women and men should be encouraged to take the HPV vaccine, which also protects against HPV-positive OPC (Shapiro 2022).
Women with cervical HPV positivity and/or cervical dysplasia should be encouraged to undergo oral HPV screening with their partners. In addition, policymakers should reconsider recommending oral HPV screening in high-risk populations. The effectiveness of oral HPV testing in the early detection and management of OPC is essential, and there is a need to develop a validated, standardized oral HPV sampling technique and tool.
Conclusion
Our study provides clear evidence that cervical HPV positivity increases the risk of oral HPV infection. Women with cervical HPV positivity and/or cervical dysplasia are a high-risk group that should undergo oral HPV screening and should be followed up more closely with their partners. However, given the challenges related to the lack of a standardized screening tool for oral HPV infection, further research is needed to develop such tools for effective screening.
Author Contributions
A.E. Mózes, contributed to conception and design, data acquisition and interpretation, drafted the manuscript; F.H. Olasz, contributed to data acquisition, critically revised the manuscript; P. Martineková, P. Hegyi, contributed to conception and design, data interpretation, critically revised the manuscript; S. Kiss-Dala, contributed to data analysis and interpretation, critically revised the manuscript; A. Bródy, D. Végh, Á. Zsembery, contributed to data interpretation, critically revised the manuscript; N. Ács, N.K. Rózsa, contributed to conception and design, data interpretation, drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251337071 – Supplemental material for Cervical HPV Positivity Elevates the Risk for Oral HPV Infection: A Systematic Review with Meta-Analysis
Supplemental material, sj-docx-1-jdr-10.1177_00220345251337071 for Cervical HPV Positivity Elevates the Risk for Oral HPV Infection: A Systematic Review with Meta-Analysis by A.E. Mózes, F.H. Olasz, P. Martineková, S. Kiss-Dala, A. Bródy, D. Végh, Á. Zsembery, P. Hegyi, N. Ács and N.K. Rózsa in Journal of Dental Research
Footnotes
Acknowledgements
We sincerely thank Dr. Zsuzsanna Ragó for her invaluable support and guidance throughout this research. This work would not have been possible without her contribution. We also greatly appreciate Dr. Eszter Szalai for her significant support and contributions to this work. Her assistance was highly appreciated throughout the research process.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval was required for this systematic review with meta-analysis, as all data were already published in peer-reviewed journals. No patients were involved in the design, conduct, or interpretation of our study. The datasets used in this study can be found in the full-text articles included in the systematic review and meta-analysis.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
