Abstract
The development of craniofacial bones and teeth relies heavily on the Wnt signaling pathway, yet the specific mechanisms and Wnt variants involved remain under continual investigation. Using publicly available single-cell sequencing data from the mouse incisor, we reveal Wnt1 expression across dental structures and investigate its role using a Col1a1-dependent Wnt1 transgenic mouse model. Inducing Wnt1 early on affects craniofacial bone without disturbing tooth development, but prolonged embryonic induction leads to postnatal mortality with osteopetrosis-like bone overgrowth and malformed teeth. While tooth formation was initially unaffected by postnatal Wnt1 induction, prolonged activation impaired tooth root formation and odontoblast differentiation, resulting in shortened roots and thinner dentin. Three-dimensional micro–computed tomography quantification reveal that both embryonic and postnatal activation of Wnt1 significantly increase neural crest–derived craniofacial bone volume, whereas mesenchymal-derived craniofacial bones are unaffected. Importantly, osteoclastogenesis is suppressed by Wnt1 in a dose-dependent manner, revealed through bulk RNA sequencing and in vitro experiments. These findings emphasize the differential effects of Wnt1 on bone development based on origin and highlight its role in modulating osteoclast activity, indicating broader implications for craniofacial development and potential therapeutic avenues.
Introduction
Craniofacial development is a complex process involving the intricate interplay of various tissues, signaling pathways, and molecular mechanisms, one of which is the Wnt signaling pathway. The Wnt pathway is a fundamental and evolutionary conserved pathway that plays a crucial role in various developmental processes. It consists of 2 main branches, the canonical or Wnt/β-catenin–dependent pathway and the noncanonical or β-catenin–independent pathway. The latter can be divided into the planar cell polarity pathway and the Wnt/Ca2+ pathway (Komiya and Habas 2008).
The Wnt pathway is activated by a family of secreted glycoproteins encoded by wingless/Wnt genes. To date, 19 Wnt ligands have been identified in mice and humans (Buechling and Boutros 2011).
Traditionally, Wnts are known to primarily engage with Frizzled (Fzd) receptors and the co-receptors LRP5/6 (Ren et al. 2021). However, Wnts can also interact with ROR family proteins (ROR1 and ROR2) and RYK in noncanonical Wnt signaling (Green et al. 2014), PTK7 in planar cell polarity signaling (Berger et al. 2017), and MuSK in neuromuscular junction formation (Banerjee et al. 2011). Since the canonical Wnt pathway fulfills a key role in the development of neural crest cells (Wu et al. 2003), which later give rise to most of the facial mesenchyme and subsequently the craniofacial skeleton and dental mesenchyme (Achilleos and Trainor 2015), misregulation of the pathway has been linked to genetic syndromes with bone and tooth malformations and different syndromes. With regard to craniofacial bone development, the complexity is underscored by the different embryonic origins and ossification mechanisms (Wei et al. 2017). For example, inactivation of the Wnt signaling pathway leads to craniofacial defects (Ko and Sumner 2021). Furthermore, postnatal tooth development and homeostasis are also heavily dependent on Wnt signaling, with misregulation leading to periodontal ligament disturbance, ankylosis (Wu et al. 2019), and growth failure (Zhang et al. 2013). Furthermore, homozygous mutations of Wnt3 lead to tetramelia syndrome (Niemann et al. 2004), mutations in Wnt5a to Robinow syndrome (Person et al. 2010), mutations in SOST to Gorlin syndrome (Gorlin et al. 2001), and mutations in Wnt7a to Fuhrmann syndrome (Woods et al. 2006). Meanwhile, Wnt10a mutations have been shown to affect bone and teeth formation such as odontoonychodermal dysplasia (Adaimy et al. 2007), Schopf–Schulz–Passarge syndrome (Bohring et al. 2009), and tooth agenesis (Kantaputra and Sripathomsawat 2011). Furthermore, mutations in Wnt1 led to recessive osteogenesis imperfecta (Pyott et al. 2013) and early onset of osteoporosis (Laine et al. 2013), and they play an essential role in bone homeostasis (Joeng et al. 2017).
We recently identified Wnt1 as a bone anabolic Wnt ligand in long bones (Luther et al. 2018) and in alveolar bone and cementum formation during tooth maintenance (Nottmeier et al. 2021).
However, despite these findings, the precise role of Wnt1 in craniofacial bone development and tooth formation in the context of embryonic and early postnatal development remains to be elucidated. Especially because Wnt1 is a key regulator of neural crest cells, which constitute most of the facial mesenchymal cells (Echelard et al. 1994), we expect Wnt1 to fulfill an important role in craniofacial development. Using a published single-cell RNA sequencing dataset from the growing mouse incisor (Krivanek et al. 2020), we identified a subset of Col1a1-positive cells that co-expressed Wnt1. Therefore, to investigate the embryonic and postnatal roles of Wnt1, in this subset of cells that accounted for osteoblasts, osteocytes, odontoblasts, and cementoblasts, we used inducible Wnt1-transgenic mice under the influence of the 2.3kb Col1a1 fragment promoter. Embryos and pups were analyzed at E15, 5, P0, P14, and P28 to reveal the functions of Wnt1 in craniofacial bone and tooth development. Inducing Wnt1 expression in Col1a1-expressing cells enables us to evaluate the potency of Wnt1 in mesenchymal cells, offering insights into its therapeutic potential in craniofacial bone and tooth development.
Methods
Methods are described in the appendix. Animal experiments were conducted in accordance with the ARRIVE2.0 guidelines.
Results
Short-Term Wnt1 Induction during Embryonic Development Affects Craniofacial Bone but Not Tooth Development
Analysis of a publicly available single-cell RNA sequencing dataset generated from the cervical loop from an adult murine incisor highlighted that Wnt1 expression is present throughout the dental pulp, dental epithelium, alveolar osteocytes, and osteoblasts (Fig. 1A–C). This expression was confirmed by in situ hybridization for Wnt1 in the adult mouse incisor (Fig. 1D). The mouse incisor combines features of the cap stage (stem cell maintenance) and bell stage (cellular differentiation) while continuously cycling through aspects of these stages due to its uninterrupted growth. To investigate the role of Wnt1 in this osteocyte and osteoblast subset, we used an inducible Wnt1 transgenic mouse model (hereafter called Wnt1Tg), which included a doxycycline-dependent Tet-off system (Fig. 1E–I).
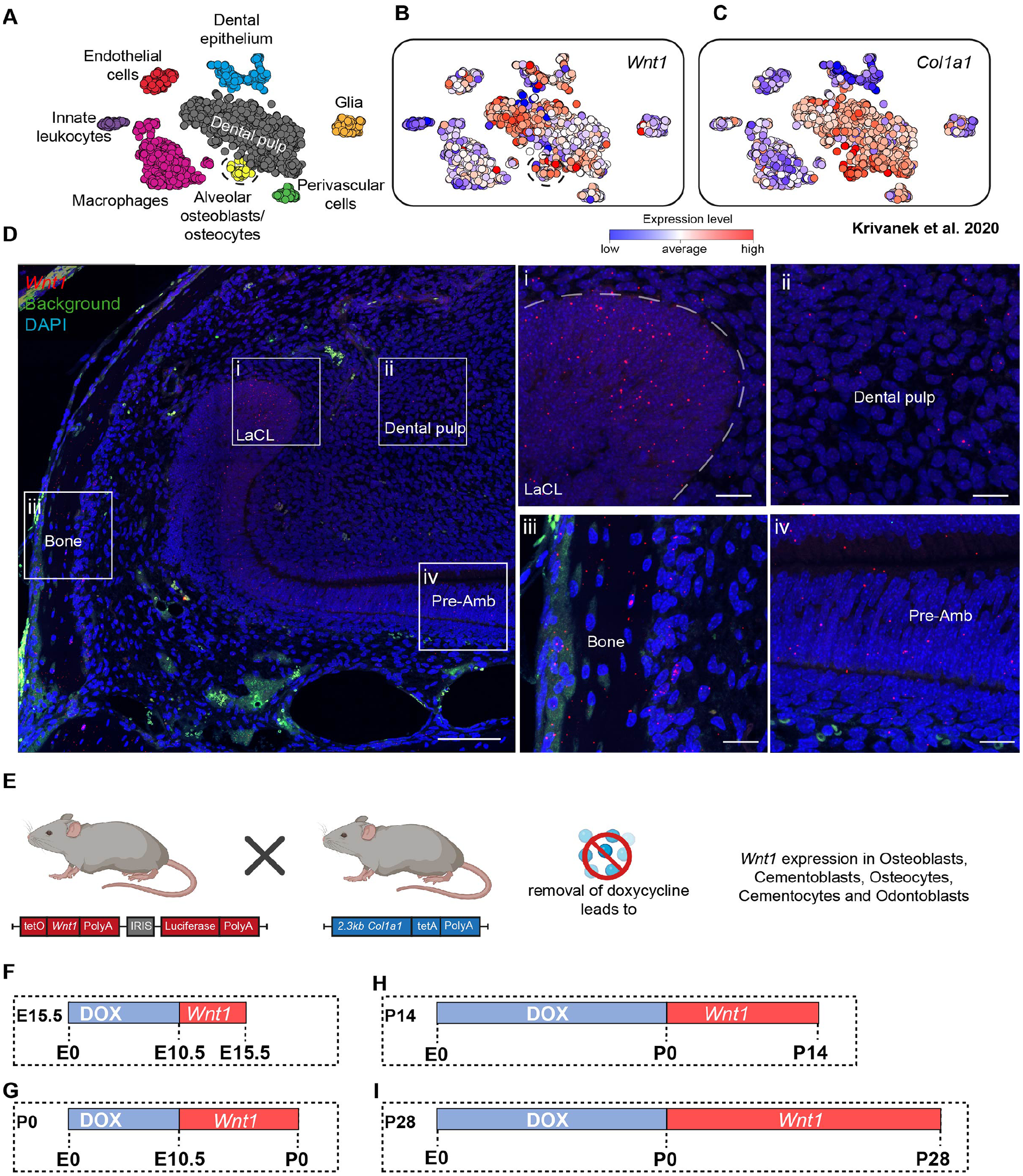
Wnt1 expression in the murine incisor and experimental overview. (
To study the effects of Wnt1 activation during embryonic bone and teeth formation, we first examined the individual stages of wild-type development using micro–computed tomography (micro-CT). Phosphotungstic acid was used to stain soft tissues, enabling 3-dimensional (3D) segmentation of the mandibular bone and dental primordia (Appendix Fig. 1). Next, we used Wnt1Tg mice to investigate the effects of Wnt1 in Col1a1-expressing cells during embryonic development. When Wnt1 was initiated on the day of mating, no transgenic offspring were born (Appendix Fig. 2). Since Wnt1 plays a fundamental role during neural crest migration (Ikeya et al. 1997), these results were anticipated. We then induced Wnt1 at day E10.5, when neural crest cell migration is complete (Serbedzija et al. 1992), and analyzed transgenic mice at E15.5 (Appendix Fig. 3A–F), the stage when tooth development can be first well distinguished.
Micro-CT with phosphotungstic acid contrasting was used to analyze mandibular bone and incisors in E15.5 Wnt1Tg and control embryos. While the shape of the mandibular bone remained unchanged (Appendix Fig. 3A–B), its volume and thickness increased significantly (Appendix Fig. 3D, and Fig. 4C and E). Dental primordia of the lower incisor and molar appeared normal, and incisor volume showed no significant difference (Appendix Fig. 4F). These findings highlight Wnt1 as a bone-anabolic ligand during early embryonic development without affecting initial tooth development.
Wnt1 Induction at E10.5 Alters Craniofacial Bone and Tooth Morphogenesis by P0 and Leads to Postnatal Lethality
Next, we evaluated later stages of tooth development to determine if they were affected by Wnt1 activation (Fig. 2A and Appendix Fig. 4). To our surprise, Wnt1 activation starting at E10.5 resulted in P0 pups dying shortly after birth. In-depth analysis using micro-CT coupled with histology revealed an altered skull shape in P0 Wnt1Tg mice with a recessed lower lip and a steep eruption angle of the lower incisors (Appendix Fig. 4A). Further analysis of the mandible length and height revealed a shorter mandible in P0 Wnt1Tg mice compared with controls (Appendix Fig. 4B). Nevertheless, bone volume was increased in most of the neural crest–derived bones, including the mandible, maxilla, and frontal bone (Fig. 2A and B and Appendix Fig. 5A). Interestingly, mesoderm-derived bones such as the parietal and occipital bone exhibited no increase in bone volume (Fig. 1A and B). However, in situ hybridization of Wnt1 and Col1a1 revealed that all major bones (neural crest– and mesoderm-derived bones) express Wnt1 and Col1a1 (Appendix Fig. 6). In terms of teeth, the lower incisor was significantly shortened compared with controls (Fig. 2C and F). Histology analysis revealed that odontoblasts were altered in Wnt1Tg mice (Appendix Fig. 7C and D). Despite no increased expression of Wnt1 in ameloblasts in Wnt1tg mice (Appendix Fig. 7E), enamel of the incisor was missing (Appendix Fig. 7F and G). When investigating the molars, a similar but reduced phenotype was detected. This was manifested in later erupting M2 molars and M1 cusps, where the volume of Wnt1Tg molars was significantly decreased (Fig. 2C and G). In addition, tartrate-resistant acid phosphatase (TRAP) staining revealed a severe inhibition of osteoclastogenesis in P0 Wnt1Tg compared with controls (Fig. 2D and H and Appendix 7A and B).
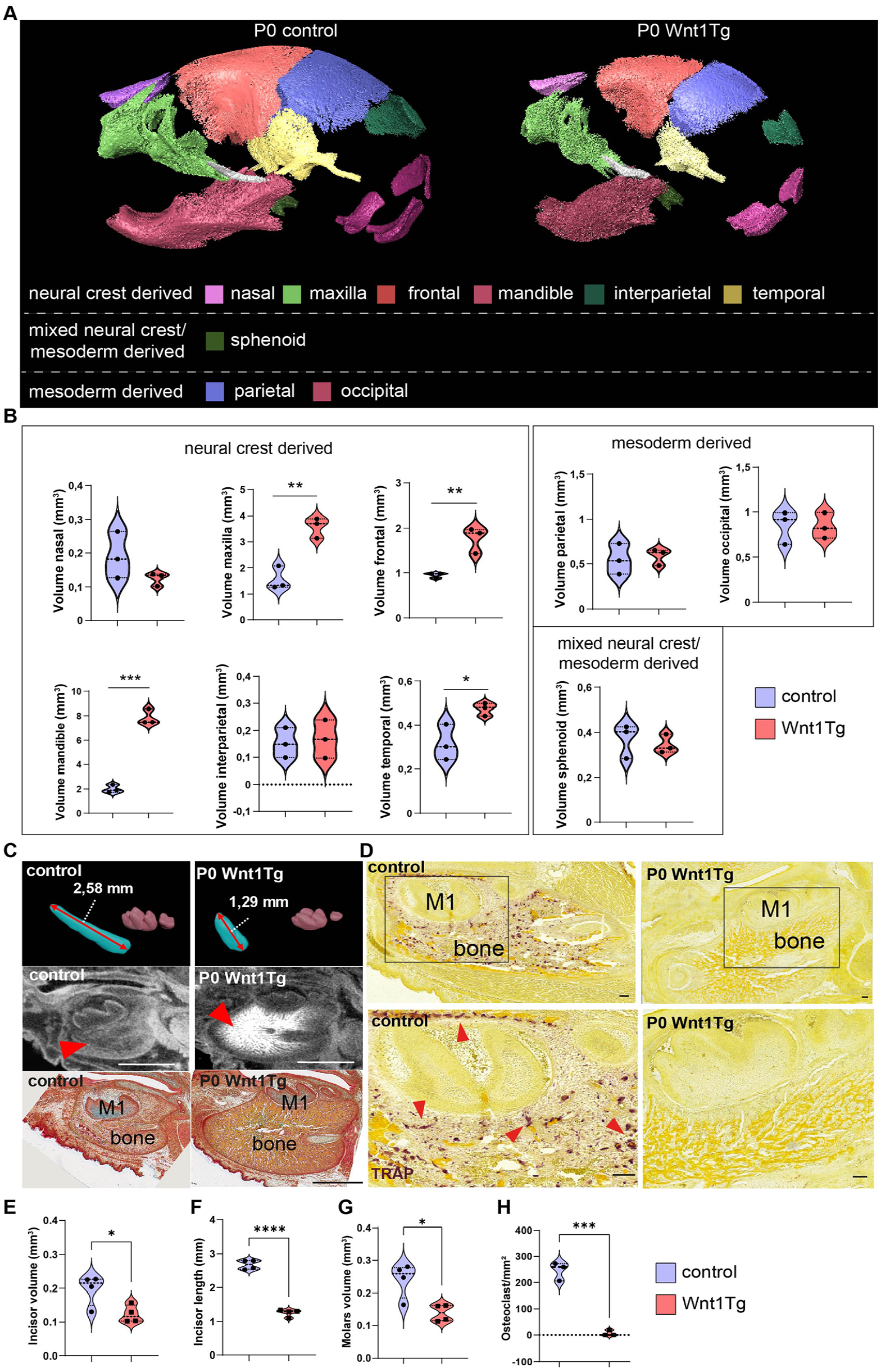
Bone and tooth malformations in P0 Wnt1Tg mice following 8 days of Wnt1 activation during embryonic development. (
Postnatal Overexpression of Wnt1 Affects Tooth Roots and Surrounding Bone Structures but Not Crown Formation
To investigate the specific effect of Wnt1 activation during the early phase of craniofacial development, postnatal activation (starting at P0) of Wnt1 was initiated and mice were sacrificed at P14 or P28 (Appendix Fig. 8 and Fig. 3). Analysis of the skull wall thickness of P14 and P28 animals revealed thickened neural crest–derived bones in Wnt1Tg mice compared with controls (Appendix Fig. 5B and C), which was confirmed using volume measurements of all major neural crest– and mesoderm-derived bones (Appendix Fig. 8A and B and Fig. 3A and B). At P14, 3D segmentation of the molars and incisors coupled with pentachrome staining confirmed the excessive bone formation with no alteration in odontoblasts or ameloblasts (Appendix Fi. 8C and Appendix Fig. 9A and B). Trabecular bone of the mandible increased significantly, while cortical bone remained unaffected (Appendix Fig. 9C and E). Remarkably, the increase in bone mass after 14 d was minor compared with 8 d induced P0 pups (Appendix Fig. 5A and B). Contrary to our observation in P0 pups, quantification of TRAP staining showed no inhibition of osteoclastogenesis in P14 Wnt1Tg versus control (Appendix Fig. 8D and G).
At P28, molars showed no significant difference in volume, but molar roots were significantly shorter (Fig. 3C–G). Pentachrome staining of the first molar revealed differences in tooth morphology and ectopic pulp calcification and significantly thinner dentin (Appendix Fig. 10A and B). Trabecular bone of the mandible increased significantly, while cortical bone remained unaffected (Appendix Fig. 10C–E). TRAP staining revealed that Wnt1 activation did not exert an overall suppressive effect on osteoclast formation (Fig. 3D). Quantification across the entire mandible did not show statistically significant differences in osteoclast numbers (Fig. 3H).
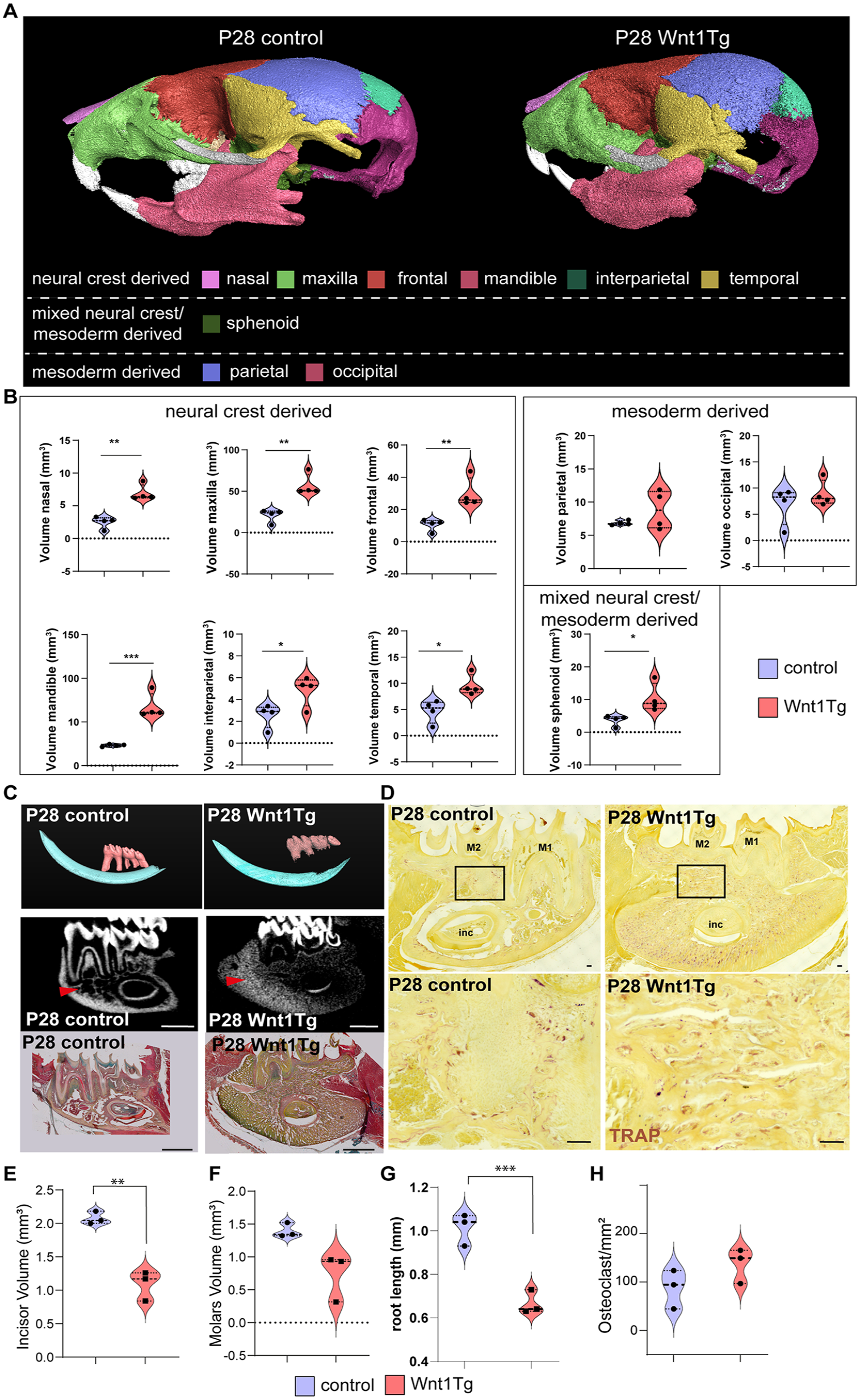
Long activation of Wnt1 after birth leads to craniofacial bone and tooth root alterations. (
Molecular Insights into Wnt1 Activation and Its Effects on Bone and Odontogenesis at Key Postnatal Stages
To understand the molecular differences, we performed RNAseq analysis of mandibles from P0, P14, and P28 Wnt1Tg and control mice (Fig. 4). At P0, osteoclast markers, such as Ctsk and Acp5, were significantly impaired alongside markers of bone ossification and remodeling such as Spp1 and Mmp13 (Fig. 4A). To compare the biological processes affected by Wnt1 activation in P0 pups, we performed Gene Enrichment Ontology (GO) analysis (Appendix Fig. 11B and 12D). The results support our observation that Wnt1 affects odontogenesis and bone mineralization.
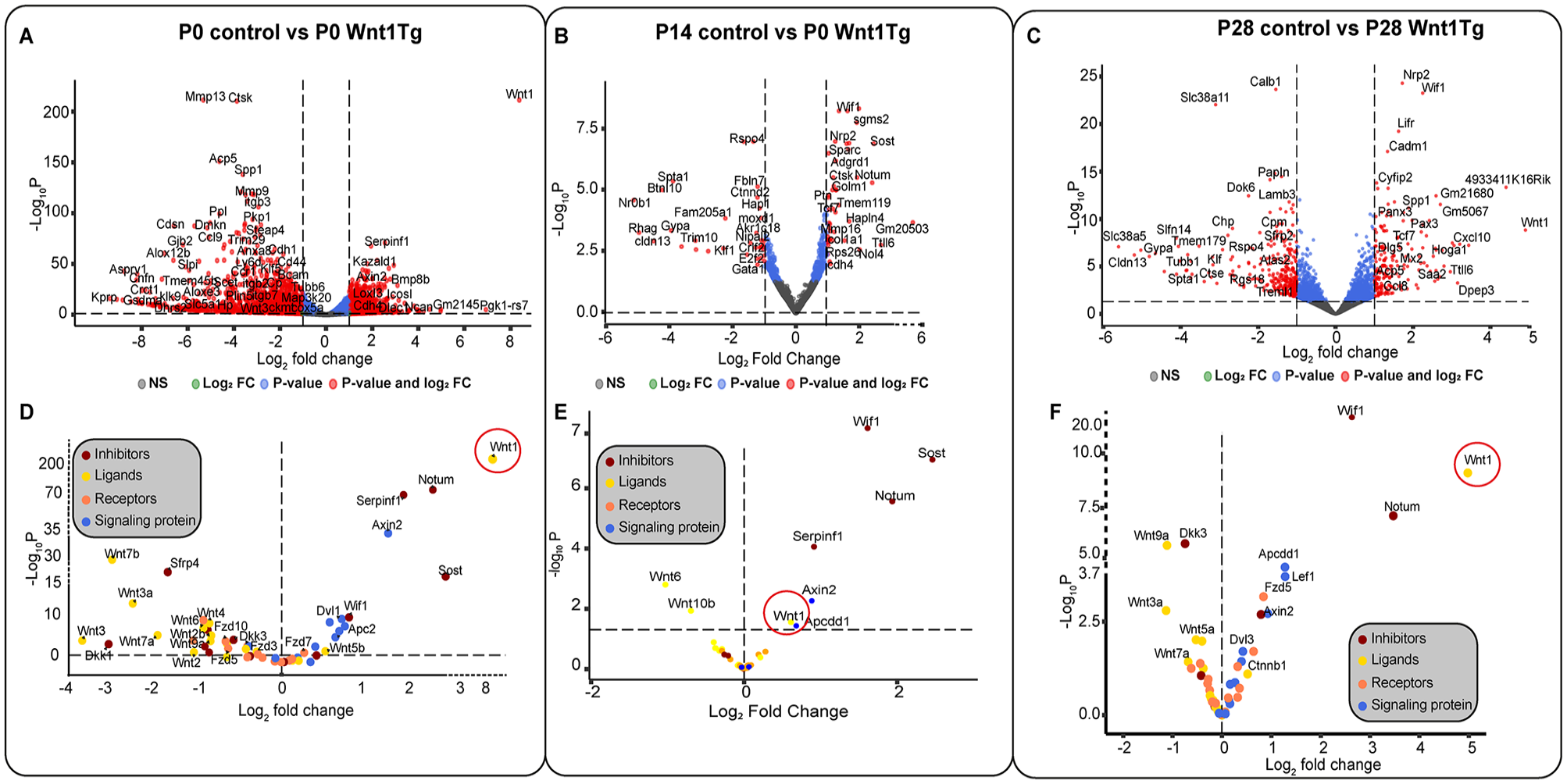
Molecular insights into Wnt1 activation at developmental stages P0, P14, and P28. (
Using Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis, we identified altered pathways including the Wnt pathway, and osteoclast differentiation (Appendix Fig. 12C). A deeper analysis of the canonical Wnt pathway revealed an 8-fold increase in Wnt1 expression, which overactivated downstream components such as Axin2, Dvl1, and Dvl2 along with the suppression of most Wnt ligands and Wnt receptors (Fzd8, Ror1, Fzd10, Fzd3, Musk, Ror2, Fzd5) and an upregulation of Wnt signaling inhibitors and antagonists such as Sost, Serpinf1, and Notum (Appendix Fig. 11C–G and Appendix Fig. 12A). The osteoclast differentiation pathway was inhibited with a decrease in the expression of secreted M-CSF along with Nfatc1, Rankl, and osteoclast-specific proteins such as Ctsk and TRAP (Appendix Fig. 12B). These results support our histological observations indicating a strong suppression of osteoclasts.
RNAseq analysis from P14 Wnt1Tg and controls showed increased activity in Wnt pathway inhibitors (Wif1, Tcf7, Sost, Notum) and bone-related genes (Ctsk, Sparc, Tmem119, Mmp16) (Fig. 4B and Appendix Fig. 13).
GO analysis revealed that clusters related to bone mineralization, ossification, odontogenesis, osteoclast differentiation, bone remodeling, and resorption were affected (Appendix Fig. 13B and Appendix Fig. 14D). Furthermore, KEGG analysis highlighted fewer affected pathways than P0 Wnt1Tg, but the Wnt pathway was still significantly activated (Appendix Fig. 13C and D and Appendix Fig. 14A and C). Moreover, analysis of the osteoclast differentiation pathway indicated upregulation of Nfatc1, in addition to Rankl and Opg expression, accompanied by high expression of the osteoclast-specific proteins Ctsk and Trap; meanwhile, M-CSF levels remained unchanged (Appendix Fig. 14B).
RNAseq analysis from P28 Wnt1Tg and controls revealed upregulation of Wif1, Lifr, and Panx3. Meanwhile, Sfrp2 was significantly downregulated (Fig. 4C). GO analysis indicated that the affected genes exhibited clusters resembling those found in P0 Wnt1Tg and P14 Wnt1Tg, focusing on bone mineralization, bone ossification, and odontogenesis (Appendix Fig. 15B, Appendix Fig. 16D). Moreover, KEGG analysis showed the same affected pathways as in P14 Wnt1Tg and P0 Wnt1Tg (Appendix Fig. 16C). Detailed analysis of the Wnt pathway in P28 Wnt1Tg revealed higher activation compared with P14 Wnt1Tg and lower activation compared with P0 Wnt1Tg (Fig. 4A–C). The heat map and volcano plot of the Wnt pathway components indicated upregulation of only Serpinf1 and Notum in P28 Wnt1Tg, while Sost was not affected. Besides, 5 Wnt ligands were significantly downregulated (Wnt3a, Wnt5a, Wnt7a, Wnt9a, and Wnt10b); meanwhile, Wnt1 was 5-fold upregulated (Appendix Fig. 15D–G and Appendix Fig. 16A). Additionally, the osteoclast differentiation pathway in P28 Wnt1Tg versus controls was more activated compared with P14 Wnt1Tg. This activation was manifested by increased expression of secreted M-CSF, Rankl along with its antagonist Opg and its receptor Rank, Nfatc1, and osteoclast-specific proteins such as Ctsk and Trap (Appendix Fig. 16B).
Wnt1 Inhibits Osteoclastogenesis in a Dose-Dependent Manner
To investigate whether the observed discrepancy in osteoclastogenesis between P0 and P14/P28 pups may stem from a dose-dependent influence, we conducted an in vitro osteoclastogenesis assay by differentiating bone marrow into osteoclasts. Bone marrow isolated from wild-type mice was stimulated with varying concentrations of Wnt1/sFRP1, alongside sFRP1 as control, assuming 1:1 complex formation ratio of Wnt1 and sFRP1. Our findings revealed a significant inhibition of osteoclast formation with increasing Wnt1 concentration (Fig. 5A and B). In addition, mRNA expression of Rank was significantly downregulated in higher Wnt1 concentrations compared with the sFRP1 control (Fig. 5C).
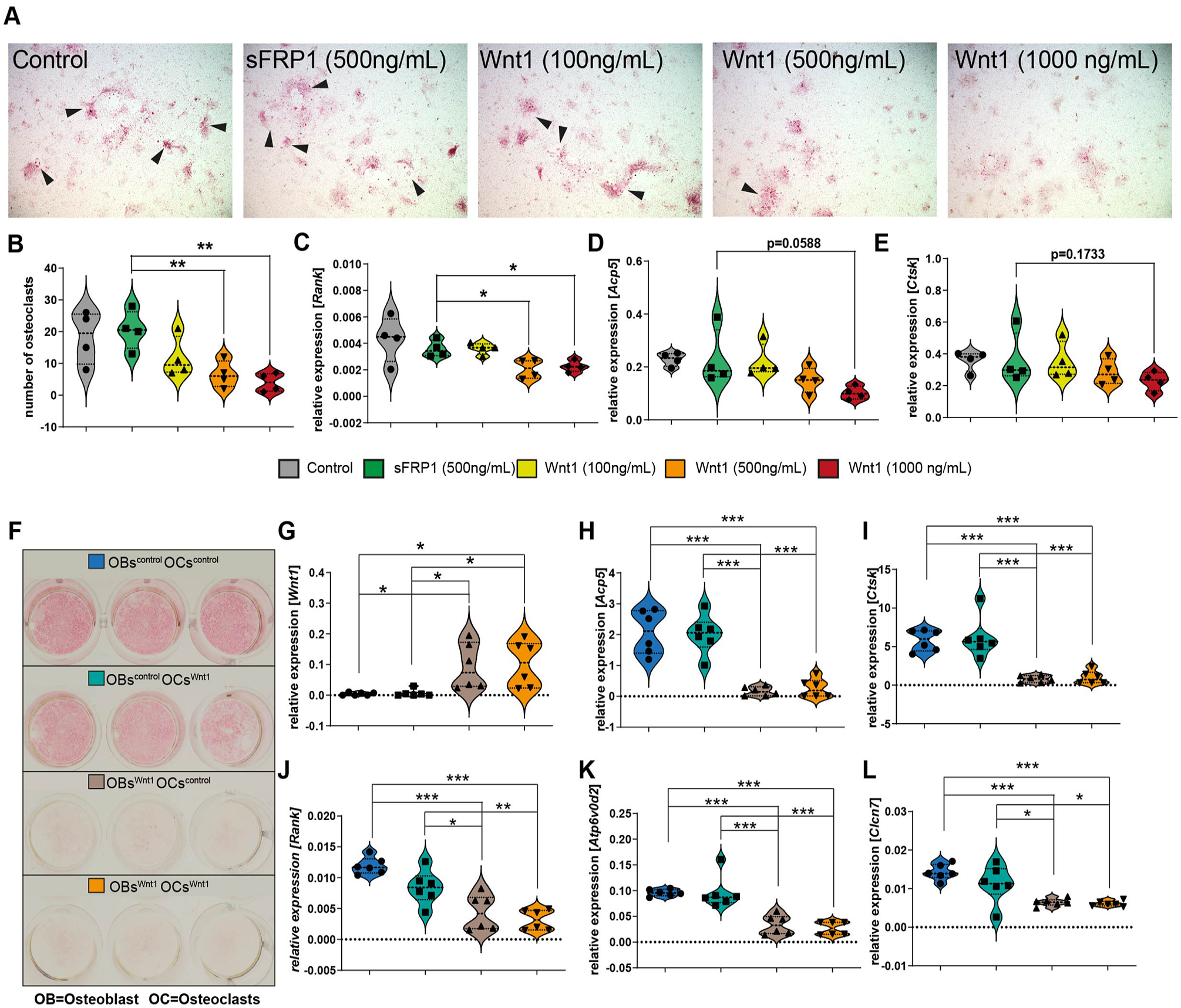
Effect of Wnt1 on osteoclast differentiation and gene expression in bone marrow cells and osteoblast/osteoclast co-cultures. (
Furthermore, to test whether Wnt1-expressing osteoblasts can inhibit osteoclasts, we conducted co-culture experiments to investigate this interaction. Bone marrow cells isolated from femora, tibiae, and pelvis of Wnt1Tg and control mice were plated in 24-well plates. Osteoblast differentiation was induced at confluency using osteogenic medium (details are given in the appendix). After 7 d of differentiation, freshly isolated bone marrow cells from Wnt1Tg and control mice were seeded on top of the differentiated osteoblasts. Osteoclast differentiation was then induced for 9 d (details are in the appendix). Cultures were analyzed via TRAP staining, which revealed a significant reduction in TRAP-positive multinucleated cells when Wnt1Tg osteoblasts were co-cultured with osteoclast precursors compared with controls (Fig. 5F–L). These findings demonstrate that Wnt1-expressing osteoblasts inhibit osteoclast formation, further supporting our hypothesis that Wnt1 indirectly suppresses osteoclastogenesis through its effects on osteoblasts.
Discussion
Using a transgenic mouse model, we investigated the effects of Wnt1 induction at different developmental stages, employing various techniques to assess phenotypic changes. Our results indicate that Wnt1 activation during early embryonic development (E10.5 to E15.5) did not disrupt tooth bell formation but significantly affected mandibular bone development. This finding is of interest given previous studies highlighting Wnt/β-catenin signaling’s role in tooth morphogenesis (Chen et al. 2009). Our findings show that Wnt1 induction in Col1a1-expressing cells does not affect early tooth development but affects subsequent craniofacial bone formation.
Bone Origin Determines Osteoanabolic Effects
Intriguingly, the phenotype of P0 pups differed drastically from P14 pups and revealed an osteopetrosis-like pathology characterized by a massive increase in bone volume, a severe decrease in osteoclasts, shortened teeth, and abnormal odontoblast formation. The observed tooth and enamel phenotype is likely secondary to the extensive bone growth, similar to the findings previously published on odontomas in osteopetrotic Src-/- mice (Koehne et al. 2013). Detailed investigation of the affected bones revealed that while the mandible, maxilla, and frontal bone responded strongly to Wnt1 induction, other cranial bones remained relatively unaffected. Here, we noticed that the strongly affected bones ossify intramembranously (Gilbert 2000). In intramembranous bone, many osteoblasts are found early (Lana-Elola et al. 2007). Thus, it likely contributes to the constitutive activation of Wnt/β-catenin signaling and amplifies this effect through a positive feedback loop. This mechanism may explain the substantial increase in bone mass, with osteoblasts populating the endochondral-formed bone at a later stage (Ortega et al. 2004). However, this approach alone fails to account for the varying osteoanabolic effects seen in different bones. Especially, considering the parietal bone, an intramembranous ossification example, shows no response to Wnt1 stimulation (Fig. 2, Appendix Fig. 8, and Fig. 3). This discrepancy may stem from differing embryonic origins of osteoblasts. While the osteoblasts of the mandible, maxilla, nasal bone, and frontal bone originate from the neural crest, those in the parietal bone derive from the mesoderm (Jin et al. 2016). This distinction significantly influences their osteogenic potential; neural crest–derived osteoblasts form bone more rapidly but exhibit lower differentiation propensity compared with mesoderm-derived parietal osteoblasts (Xu et al. 2007). Here, it would be of interest to investigate how these differences contribute to the varying osteoanabolic response to Wnt1 stimulation.
Wnt1 Suppresses Osteoclastogenesis in a Dose-Dependent Manner
Another interesting finding of our study was the complete inhibition of osteoclastogenesis in Wnt1-induced P0 mandibles accompanied by high bone formation, whereas P14 pups (activated Wnt1 for 14 d) or P28 pups (activated Wnt1 for 28 d) displayed an elevated bone phenotype, without an observed effect on osteoclasts. These findings align with the existing literature documenting the varied effect of Wnt signaling on osteoclastogenesis.
For example, activation of Wnt4 under the control of the 2.3kb Col1a1 promoter led to osteoclast inhibition and prevented estrogen deficiency–induced osteoporosis in vivo (Yu et al. 2014). Furthermore, constant activation of β-catenin in differentiated osteoblasts also under the control of the Col1a1 promoter inhibited osteoclastogenesis and increased bone formation (Glass et al. 2005). On the other hand, activation of Wnt1 under the control of 2.3kb Col1a1 promoter for 9 wk in adult mice resulted in no significant reduction in osteoclasts (Luther et al. 2018). Moreover, activating β-catenin in Dmp1-expressing osteoblasts and alveolar bone osteocytes resulted in significantly higher osteoclast and osteoblast activity compared with controls (Wu et al. 2019).
By using an in vitro bone marrow osteoclast differentiation assay, we revealed that Wnt1 exhibits a dose-dependent inhibition effect on osteoclastogenesis. This aligns with our in vivo observations, which indicate a robust induction of Wnt1 during early embryonic development, whereas the initiation of Wnt1 postnatally is comparatively weaker. This dose-dependent effect can also explain the diverse and in part contradictory observations in the literature regarding the Wnt signaling impact on osteoclastogenesis, as previous studies have not investigated the extent of Wnt signaling activation. Furthermore, using the co-culture experiment, we identified that osteoblasts can influence osteoclastogenesis through secretion of Wnt ligands, thereby regulating Wnt signaling in osteoclasts. This aligns well with a study that showed how Wnt1 signaling regulates osteoclast differentiation in a juxtacrine manner (Wang et al. 2019).
In conclusion, our finding indicates that prolonged activation of Wnt1 during development has detrimental effects on tooth morphogenesis. Furthermore, our study provides valuable insights into the dose-dependent and cell-specific effects of Wnt1 signaling on craniofacial bone and tooth development. The findings contribute to our understanding of the intricate regulatory mechanisms involved in these processes. A limitation of the used Tet-off system is the potential gene expression during doxycycline administration. However, adult mice with continuous doxycycline exposure (Nottmeier et al. 2021) showed no bone or tooth phenotypes, minimizing concerns about unintended gene expression. Another limitation is a nonlinear dose response after doxycycline removal. It was therefore important to perform RNA sequencing of each analyzed stage and to interpret the results according to the respective Wnt1 expression. Further research, including investigating the downstream targets and signaling interactions of Wnt1, will provide a more comprehensive understanding of the molecular mechanisms governing craniofacial development. Since the stimulatory effect of Wnt1 on osteoblasts at all stages is undisputed, a focused pharmacologic strategy to activate osteoblasts while concurrently avoiding influence on osteoclasts holds significant promise as a therapeutic avenue for addressing bone metabolic disorders such as osteoporosis. For regenerative dentistry, in addition to the osteoanabolic effect, the positive effect on cementum formation in later developments as shown in Nottmeier et al. (2021) is also important, which makes Wnt1 an interesting target for periodontology in the future. Therefore, deciphering the exact mechanisms behind the Wnt1 signaling is essential and should remain the subject of future research to develop specifically effective pharmaceuticals. Furthermore, β-catenin can initiate signalling through its transcription-independent domain, independent of LEF/TCF interactions, suggesting a broader and more complex role in cellular processes beyond its traditional function in the canonical Wnt pathway (Doumpas et al. 2019).
Lastly, β-catenin–activating drugs, such as BC21 (Kahn 2014), may significantly affect fetuses in pregnant patients, requiring careful consideration of their use during pregnancy.
Author Contributions
R. Mahmoud, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; A. Simon, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; J. Luther, J. Pothe, Y. Du, C. Nottmeier, E. Okine, S. Knauth, M.G. Lopez, contributed to data acquisition and analysis, critically revised the manuscript; E. Bockamp, J. Krivanek, A. LeBlanc, J. Helms, M. Amling, M. Kaucka, T. Schinke, contributed to data interpretation, critically revised the manuscript; T. Koehne, J. Petersen, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251336191 – Supplemental material for Wnt1’s Differential Effects on Craniofacial Bone and Tooth Development
Supplemental material, sj-docx-1-jdr-10.1177_00220345251336191 for Wnt1’s Differential Effects on Craniofacial Bone and Tooth Development by R. Mahmoud, A. Simon, J. Luther, J. Pothe, Y. Du, C. Nottmeier, E. Okine, S. Knauth, M.G. Lopez, E. Bockamp, J. Krivanek, A. LeBlanc, J. Helms, M. Amling, M. Kaucka, T. Schinke, T. Koehne and J. Petersen in Journal of Dental Research
Footnotes
Acknowledgements
Computations for this work were done (in part) using resources of the Leipzig University Computing Center.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research of J.K. was supported by Czech Science Foundation grant No. 23-06160S. T.S. was supported by the DFG grant SCHI 504/19-1, T.K. was supported by the DFG grant KO5420/1-1 and KO5420/1-2
Declaration of AI and AI-Assisted Technologies
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
