Abstract
Odontoblasts are terminally differentiated cells that exhibit mechanosensitivity and mineralization capacity. Mechanosensitive ion channels such as Piezo1 are present in odontoblasts and are associated with their physiological functions via Ca2+ signaling. Both Ca2+ signals via Ca2+ influx from mechanosensitive ion channels and Ca2+ release from Ca2+ stores function as secondary messenger systems for various biological phenomena. The endoplasmic reticulum (ER) serves as an intracellular Ca2+ store that mobilizes intracellular Ca2+. Changes in Ca2+ concentration inside the ER are among the factors that cause ER stress. Perivascular cells are located around odontoblasts in the dental pulp. Although such formation indicates that perivascular cells interact with odontoblasts, their detailed profiles under developmental and pathological conditions remain unclear. In this study, we revealed that pericyte marker, neural/glial antigen 2 (NG2)–positive cells, in cell-rich zones (CZs) can differentiate into Piezo1-positive odontoblasts following genetic odontoblast depletion in mice, and modeled as odontoblast death after severe dentin injury and as reparative dentin formation. NG2-positive pericytes differentiated into odontoblasts faster than glial cells. To determine how NG2-positive cells differentiate into Piezo1-positive odontoblasts, we focused on the ER-stress sensor protein, activating transcription factor 6a (ATF6a). After genetic odontoblast depletion, NG2-positive cells regenerated in the odontoblast layer and were capable of acting as functional odontoblasts. In the presence of extracellular Ca2+, the application of a sarco/ER Ca2+-ATPase (SERCA) inhibitor, thapsigargin, known as an ER-stress inducer, increased the intracellular Ca2+ concentration in the odontoblast lineage cells (OLCs). The increase was significantly inhibited by the application of a pharmacologic Piezo1 inhibitor, indicating that ER stress by SERCA inhibition augmented Piezo1-induced responses in odontoblast progenitor cells. However, the physiological activation of Gq-coupled receptors by adenosine diphosphate did not induce Piezo1 activation. Gene silencing of ATF6a and/or NG2 impaired the mineralization of OLCs. Overall, ATF6a orchestrates the differentiation of NG2-positive pericytes into functional odontoblasts that act as sensory receptor cells and dentin-forming cells.
Highlights
Pericytes located just beneath the odontoblast layer differentiate into functional odontoblasts.
ATF6a regulates mechanosensitivity and dentin mineralization in odontoblasts.
Pericytes are cell sources that contribute to odontoblast differentiation after odontoblast injuries.
Introduction
Odontoblasts are sensory receptor cells involved in sensory transduction, as an odontoblast hydrodynamic/mechanosensory receptor model (Shibukawa et al. 2015; Sato et al. 2018; Ohyama et al. 2022), and are essential for driving dentin mineralization (Goldberg and Smith 2004; Charadram et al. 2012; Neves and Sharpe 2018). Dentin mineralization is activated not only by physiological and developmental processes but also by mechanical, temperature-related, osmotic, and/or chemical stimulation to the dentin surface. In addition, dentin production by odontoblasts is a key process in developmental tooth formation, and understanding its detailed mechanism is important in generating drugs for dentin regeneration. Odontoblasts are well-polarized columnar cells that are aligned at the periphery of the dental pulp. To participate in dentin formation, they synthesize and secrete collagenous and noncollagenous matrix proteins as well as transcellular transportation of accumulated intracellular Ca2+ to the mineralizing front (Linde and Lundgren 1995). During dentinogenesis, transcellular Ca2+ transporting mechanisms are mediated by Ca2+ influx, mobilization, and extrusion (Linde and Lundgren 1995). These mechanisms include Ca2+ influx from the extracellular medium via various plasma membrane Ca2+ channels and Ca2+ release from intracellular inositol 1,4,5-trisphosphate (IP3)- or ryanodine-sensitive Ca2+ stores (Lundgren and Linde 1997; Shibukawa and Suzuki 1997; Shibukawa and Suzuki 2003). An increased concentration of intracellular Ca2+ in odontoblasts is extruded by plasma membrane Na+-Ca2+ exchanger (NCX) (Lundgren and Linde 1988; Tsumura et al. 2010) and plasma membrane Ca2+-ATPase (PMCA) activity (Kimura et al. 2021).
Mouse incisors are model systems used to study the molecular mechanisms in the local niche environment and process of cell differentiation (Zhao et al. 2014). The neural/glial antigen 2 (NG2)–positive (NG2+) pericytes display its position around vessels inside the odontoblast layer (OB) (Khatibi Shahidi et al. 2015). During steady state in the developmental process, odontoblasts originate from glial cells (Kaukua et al. 2014), while in injury repair for the dentin/pulp complex, odontoblasts are thought to originate from NG2+ pericytes (Feng et al. 2011; Kaukua et al. 2014; Zhao et al. 2014). Single-cell RNA sequencing revealed that pericytes could be classified into 2 groups: positive for Nestin and NG2 (Nestin+ NG2+) and negative for Nestin and positive for NG2 (Nestin− NG2+) (Gomes et al. 2022). Nestin is an odontoblastic marker protein (Nakatomi et al. 2018). When NG2+ cells isolated from human dental pulp are treated with lipopolysaccharides, they exhibit stronger proliferation, migration, and odontoblastic differentiation, indicating their important role in rapid repair after injury of dentin/pulp complex (Yang et al. 2019). These results imply that NG2+ pericytes are a potent candidate of cell source for odontoblast development.
Voltage-dependent L-type Ca2+ channels are expressed by NG2+-pericyte lineage cells after dental pulp injury (Fu et al. 2023), while we have previously demonstrated that odontoblasts functionally express the mechanosensitive ion channel, Piezo1, and mechanical stimulation–induced intercellular Ca2+ signaling modulates mechanosensory transduction and mineralization of dentin (Matsunaga et al. 2021; Ohyama et al. 2022). Piezo1 channels in odontoblasts predominantly contribute to the detection of cellular deformation, as mechanical stimulation, induced by dentinal fluid movements in dentinal tubules during various stimuli applied to the exposed dentin surface. Activation of the Piezo1 channel and subsequent Ca2+ influx by Piezo1 negatively regulates reactionary dentinogenesis (Matsunaga et al. 2021). Owing to its essential properties, Piezo1 is useful odontoblast marker protein directly related to its cellular functions, dentin sensation, and dentinogenesis.
The endoplasmic reticulum (ER) serves as an intracellular Ca2+ store that releases and mobilizes intracellular Ca2+. Factors that cause ER stress include changes and an imbalance in Ca2+ concentration inside the ER by releasing Ca2+ via IP3 and/or ryanodine receptors from the store and by uptaking Ca2+ via sarco/ER Ca2+-ATPase (SERCA) into the store (Ron and Walter 2007; Kim et al. 2008). The accumulation of unfolded proteins in the ER represents a cellular stress induced by multiple stimuli and pathological conditions. Although perivascular NG2+ pericytes interact with odontoblasts for several reasons, their detailed developmental, physiological, and pathological profiles in association with the Ca2+ signals and subsequent ER stress remain unclear. Dental pulp is exposed to various stresses during the wound-healing process following dentin/pulp complex injury, and ER stress is assumed to occur via gene regulation of dental pulp cell properties (Walter and Ron 2011; Kim et al. 2014; Aryal et al. 2020; Li et al. 2022). Therefore, the purpose of this study was 2-fold. First, we elucidated the dynamic properties and characteristics of NG2+ pericytes during functional odontoblast regeneration. Second, we examined the expression changes and regulatory mechanisms of ER-stress proteins during local differentiation of NG2+ pericytes to odontoblasts.
Materials and Methods
The study was approved by our institute according to several guidelines described in the appendix. Other information is also described in the appendix.
Results
Expansion of NG2+ Pericytes in Cell-Rich Zones after Odontoblast Death
To evaluate the microenvironment after odontoblast death, we administered diphtheria toxin (DT) every 24 h for 1 wk to iDTR and Col1(2.3)Cre;iDTR mice and sacrificed them on the eighth day (Fig. 1A). Morphological observations of the hematoxylin-eosin–stained dental pulp from iDTR- and Col1(2.3)Cre;iDTR mice incisors revealed highly polarized odontoblasts arranged at the dentin pulp border in iDTR mice (as a control) (OB; Fig. 1B1–B3) but not in Col1(2.3)Cre;iDTR mice (OB; Fig. 1C1–C3), enabling somatic odontoblast-specific depletion via DT administration. The expression of the odontoblast markers, Nestin and dentin sialophosphoprotein (DSPP), was observed in the OBs of iDTR mice (OB; Fig. 1D1–D3), but their levels were downregulated in Col1(2.3)Cre;iDTR mice (OB; Fig. 1E1–E3). The number of cells positive for Nestin and DSPP (Nestin+ DSPP+) in the OB was significantly lower in the odontoblast depletion model (Col1(2.3)Cre;iDTR mice) as compared with control (iDTR mice) (Fig. 1F). In wild-type control mice (8 wk old), glial fibrillary acidic protein (GFAP)–positive (GFAP+) glial cells were observed internally in the dental pulp of the incisors compared with where NG2+ pericytes were observed (Fig. 1G1 and G2). When we observed mice for 14 d after the last injection of DT (Fig. 1H), NG2+ cells were found to reach the OB earlier than GFAP+-glial cells for their compensation as odontoblasts in Col1(2.3)Cre;iDTR mice incisors, indicating that NG2+ pericytes, not glial cells, are local odontoblast progenitor cells (Fig. 1I1–J3). In the negative controls for immunofluorescence staining, we used samples short chased after odontoblast depletion models (Fig. 1K). The negative control by omitting the first antibodies showed no fluorescence detection (Fig. 1L1–M2). Thus, odontoblast depletion can trigger for NG2+ pericytes migrating into the OB.
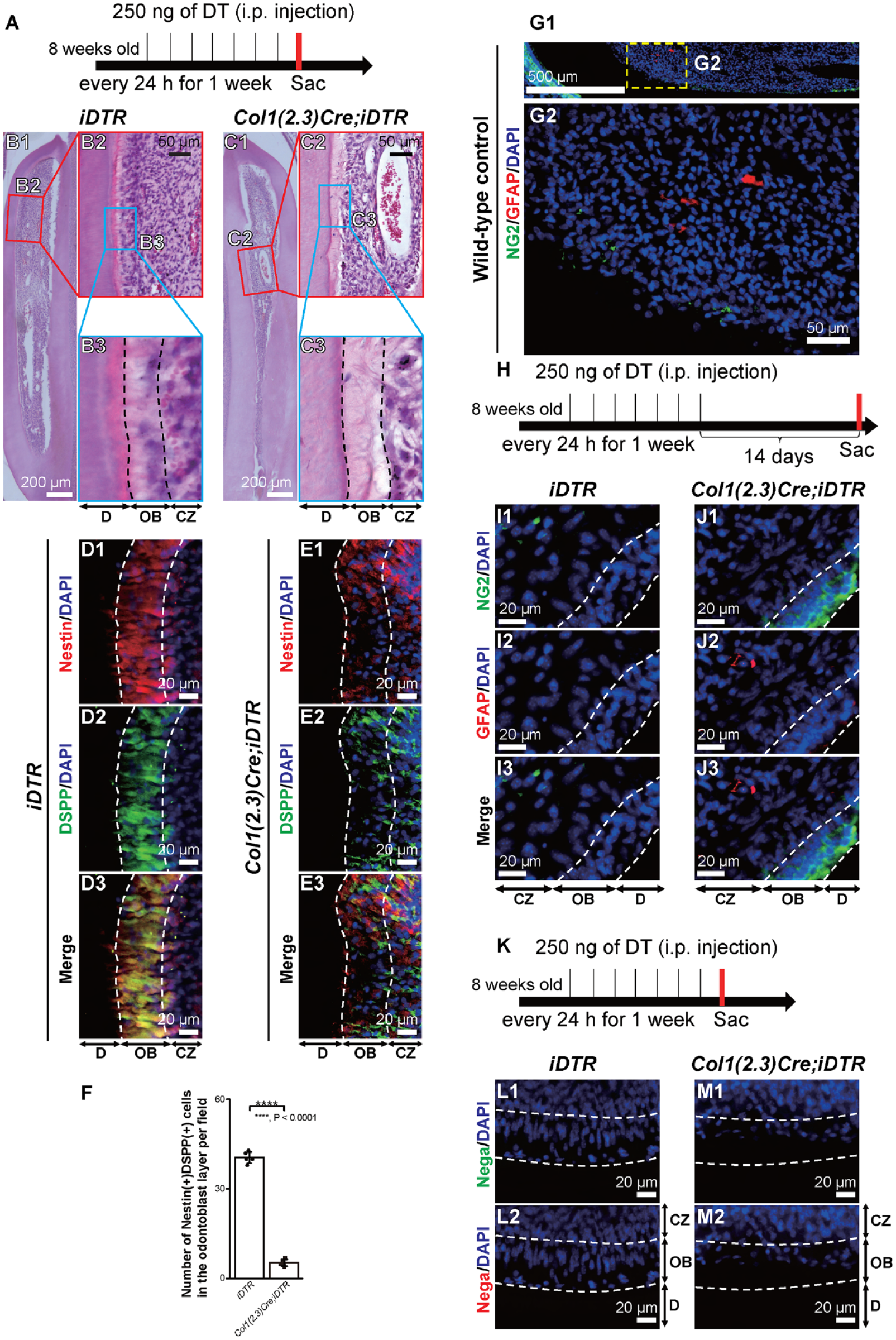
Cre recombination in Col1(2.3)Cre;iDTR mice drives odontoblast depletion. (
To identify the distribution patterns of pericytes, we conducted immunofluorescence staining using mandibular incisors. The number of NG2+ cells was lower (Fig. 2A1, A2) than that of other pericyte markers such as platelet-derived growth factor receptor beta (PDGFRb) (cluster of differentiation 140b; CD140b) (Fig. 2B1, B2), melanoma cell adhesion molecule (MCAM) (CD146) (Fig. 2C1, C2), and actin alpha 2 (Acta2) (alpha-smooth muscle actin; aSMA) (Fig. 2D1, D2). Due to the small number of NG2+ pericytes in dental pulp, we hypothesized that they may exist as a minor population of stem cells or progenitor cells in dental pulp. Next, we performed immunofluorescence staining to determine how NG2+ pericytes could differentiate into odontoblasts. Mandibular incisors were dissected from sacrificed mice after 7 consecutive days of DT administration (Fig. 2E). After DT administration, the number of NG2+ pericytes increased in the cell-rich zone (CZ) and expressed Nestin (Fig. 2G1-H3), DSPP (Fig. 2K1-L3), and Piezo1 (Fig. 2O1-P3) in Col1(2.3)Cre;iDTR mice, while the small number of NG2+ pericytes did not change obviously in iDTR mice (Fig. 2F1–F3, J1–J3, N1–N3). Cell shape of NG2+ cells in CZ in Col1(2.3)Cre;iDTR mice showed several patterns due to the fact that pericytes are generally classified by their morphologies. In the CZ, the number of cells that were double-positive for NG2 and each odontoblast marker, Nestin, DSPP, and Piezo1, was dominant in Col1(2.3)Cre;iDTR mice compared with iDTR mice. The number of NG2+ Nestin+ cells in the CZ significantly increased by 7-fold in Col1(2.3)Cre;iDTR mice compared with iDTR mice (Fig. 2I). The number of NG2+ DSPP+ cells in the CZ also significantly increased by 6- to 7-fold in Col1(2.3)Cre;iDTR mice (Fig. 2M). NG2+ Piezo1+ cells in the CZ were dominant in Col1(2.3)Cre;iDTR mice (5-fold increase) relative to iDTR mice (Fig. 2Q).
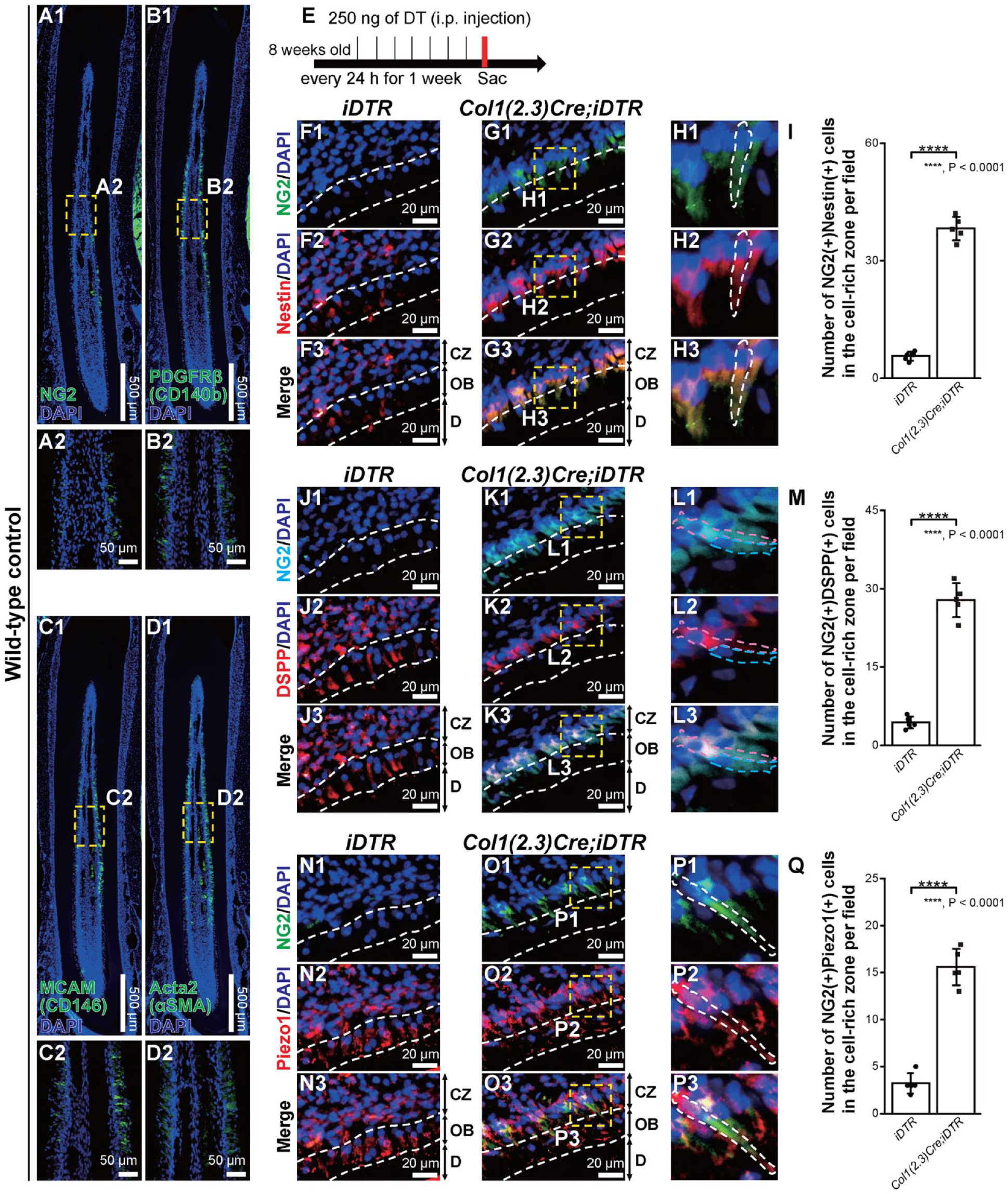
Expansion of neural/glial antigen 2–positive (NG2+) cells in the cell-rich zone after odontoblast depletion. (
Expression of ER-Stress Sensor Proteins after Odontoblast Death
We hypothesized that ER stress is involved in odontoblast differentiation under pathological conditions by odontoblast death (Fig. 3A). Antibodies against activating transcription factor 6a (ATF6a), inositol-requiring enzyme 1 a (IRE1a), and protein kinase RNA-like endoplasmic reticulum kinase (PERK), which are ER-stress sensor proteins, were used to evaluate their immunoreactivities. Piezo1-positive (Piezo1+) odontoblasts expressed ATF6a and PERK but not IRE1a under physiological conditions in iDTR mice (Fig. 3B1–B3, E1–E3, H1–H3). In the CZ in iDTR mice, we observed the small number of ATF6a-positive (ATF6a+) cells, IRE1a-positive (IRE1a+) cells, and PERK-positive (PERK+) cells. After odontoblast depletion mimicking dental pulp/odontoblast injury, ATF6a+ Piezo1+ cells were abundantly observed in the CZ (4- to 5-fold change) (Fig. 3C1–C3, D), whereas these changes could not be observed for IRE1a+ Piezo1+ cells and PERK+ Piezo1+ cells (Fig. 3F1–F3, G, I1–I3, J). These data indicate that ER-stress sensor proteins, especially ATF6a, may regulate the differentiation from pericytes into odontoblasts.
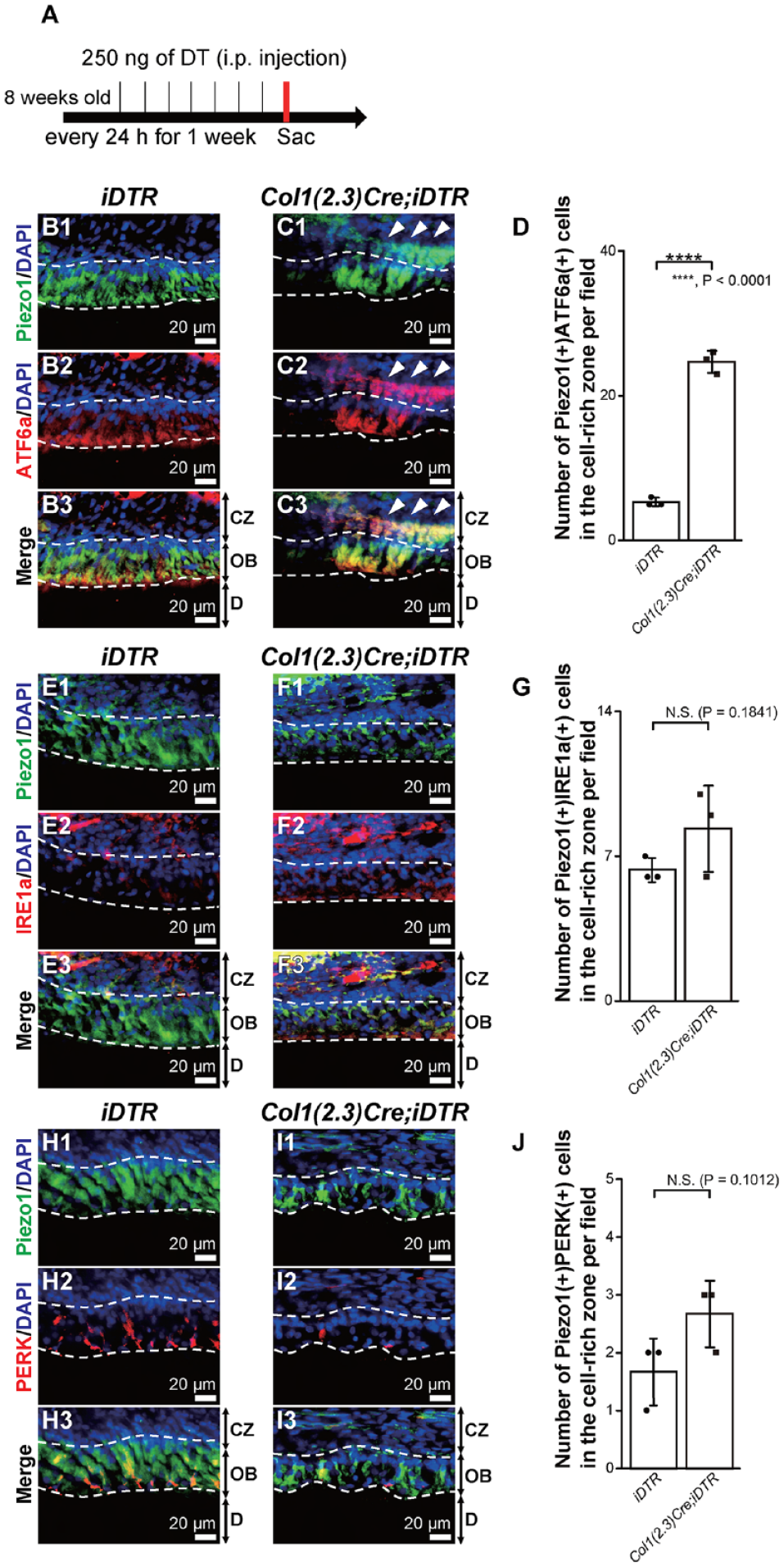
Expression of the endoplasmic reticulum stress marker, activating transcription factor 6a (ATF6a), in the cell-rich zone (CZ) after odontoblast depletion. (
Differentiation of NG2+ Pericytes into Odontoblasts as Sensory Receptor Cells
To trace the differentiation of NG2+ pericytes into odontoblasts, we analyzed the NG2CreERT2;tdTomato mice. After tamoxifen was administered at postnatal day 3 of NG2CreERT2;tdTomato mice, incisor dental pulp cells were isolated and primary cultured in 1-mo-old mice (Appendix Fig. 1A). Cultured whole NG2-tdTomato–positive (NG2-tdTomato+) cells were immunopositive for Nestin (Appendix Fig. 1B1–B3), DSPP (Appendix Fig. 1C1–C3), Piezo1 (Appendix Fig. 1D1–D3), and runt-related transcription factor 2 (Runx2) (Appendix Fig. 1E1–E3). These results indicate that NG2+ pericytes have the potential to differentiate into odontoblast lineage cells.
Mouse incisors grow throughout their lives, whereas molars, like human teeth, have limited growth. Next, we used molar teeth and observed Col1(2.3)Cre;iDTR mice longer to see whether NG2+ cells in CZ differentiate into odontoblasts under the ATF6a regulatory mechanisms. We sacrificed Col1(2.3)Cre;iDTR mice 14 d after the last injection of DT (Fig. 4A). Regenerated NG2+ ATF6a+ cells after odontoblast death were located in the OBs of the molar tooth in Col1(2.3)Cre;iDTR mice (Fig. 4C1–C4), while NG2− ATF6a+ cells were observed as steady state in OBs in iDTR mice (Fig. 4B1–B4). The number of NG2+ ATF6a+ cells in the CZ and the OB was significantly more dominant in Col1(2.3)Cre;iDTR mice than that in iDTR mice 14 d after odontoblast depletion (in CZ; 5-fold change/in OB; 4- to 5-fold change) (Fig. 4D, E). Because ATF6a activates by its nuclear translocation as an ER-stress response, we analyzed the ATF6a expression in nuclei in NG2− ATF6a+ and NG2+ ATF6a+ cells. The data revealed greater observation of nuclear translocation of ATF6a in NG2+ cells than in NG2− cells (3-fold change) (Fig. 4F, G).
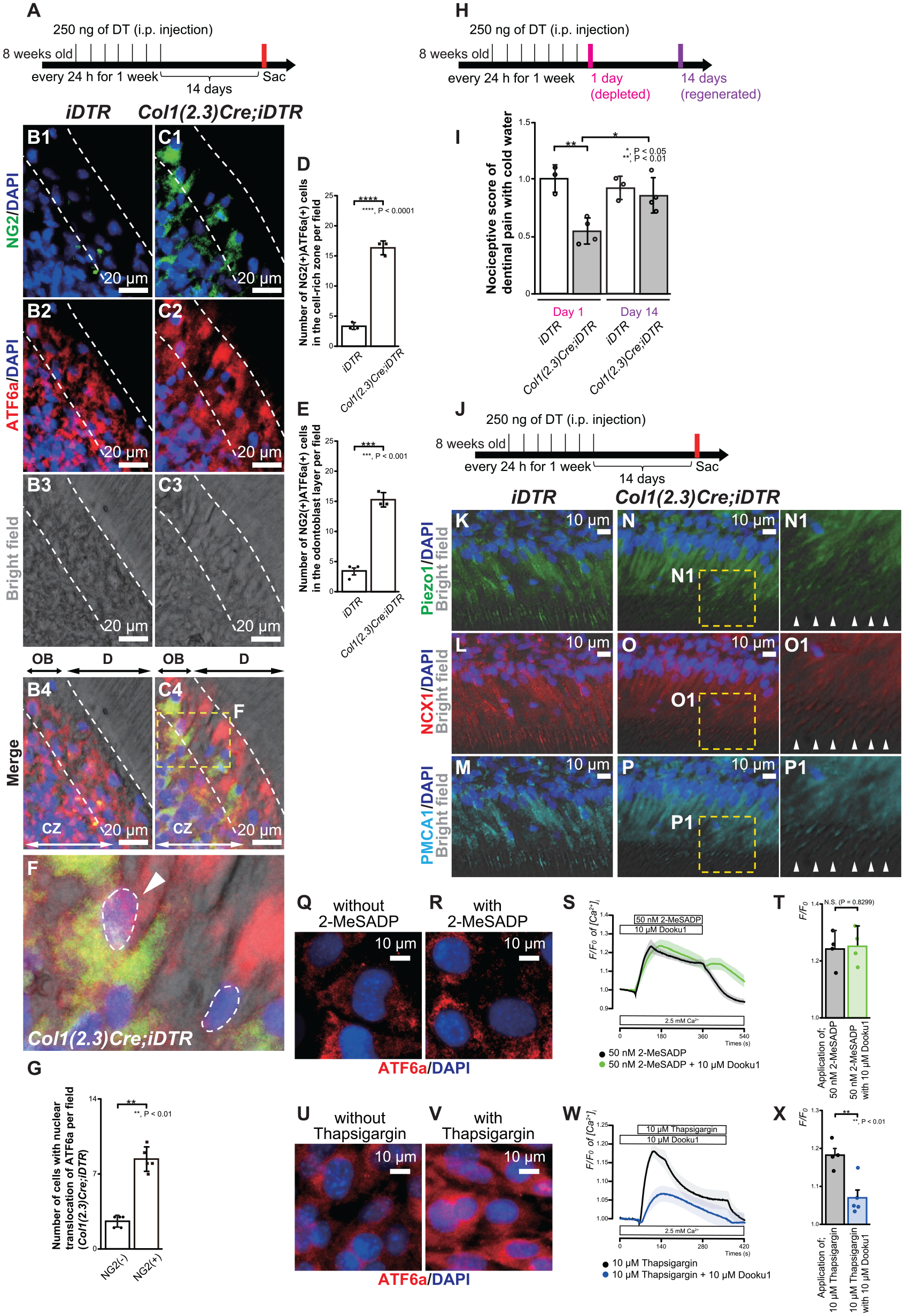
Neural/glial antigen 2–positive (NG2+) pericyte–derived odontoblasts exhibited a sensory receptor cell property. (
Next, we analyzed nociceptive scores after application of cold water to exposed dentin in iDTR and Col1(2.3)Cre;iDTR mice. We chose 2 time points: (1) when odontoblasts were depleted (as day 1; 1 d after last DT administration) and (2) when pericyte-derived odontoblasts regenerated (as day 14; 14 d after last DT administration) (Fig. 4H). Nociceptive score data revealed that dentin mechanosensitivity of Col1(2.3)Cre;iDTR mice was recovered at the same level of iDTR mice, showing that NG2+ pericyte–derived odontoblasts gained a sensory receptor cell property (Fig. 4I). Immunofluorescent staining of regenerated odontoblasts, which were seen at 14 d after the last DT administration (Fig. 4J) in Col1(2.3)Cre;iDTR mice showed that they were immunopositive for Piezo1, similar to that in iDTR mice (Fig. 4K, N, N1). In addition, regenerated odontoblasts in Col1(2.3)Cre;iDTR mice expressed NCX1 and PMCA1, similar to that in iDTR mice (Fig. 4L, O, O1, M, P, P1), both of which have essential roles in maintaining Ca2+ homeostasis.
Change and imbalance of the internal ER Ca2+ concentration ([Ca2+]ER) and intracellular Ca2+ concentration ([Ca2+]i) can induce ER stress. We next measured [Ca2+]i by applying 2-methylthio-ADP (2-MeSADP; an agonist of P2Y1, 12, 13 receptors) and the ER-stress inducer thapsigargin (a potent and noncompetitive inhibitor of SERCA) in mouse odontoblast lineage cells (OLCs). OLCs expressed Nestin, DSPP, and Piezo1 (Appendix Fig. 2A–C). OLCs were also immunopositive for P2Y1 receptor (P2Y1R) as well as Gnaq, which encodes the heterotrimeric G-protein a-subunit Gaq (Appendix Fig. 2D–F). Data showed that 1-h application of 10 mM thapsigargin (Fig. 4U, V) but not 50 nM 2-MeSADP (Fig. 4Q, R) induced nuclear translocation of ATF6a, confirming that the ER-stress response was induced by pathological SERCA regulation. Furthermore, we examined the effect of 10 mM Dooku1, a pharmacologic Piezo1 inhibitor, on 50 nM 2-MeSADP– or 10 mM thapsigargin-induced [Ca2+]i increases in OLCs. In the presence of extracellular Ca2+ (2.5 mM), a 5-min application of 2-MeSADP or thapsigargin increased [Ca2+]i in OLCs. The application of Dooku1 significantly inhibited the increase of [Ca2+]i by thapsigargin but not the increase of [Ca2+]i by 2-MeSADP, indicating that ER stress by SERCA inhibition augmented Piezo1-induced responses in OLCs (Fig. 4S, T, W, X).
Thus, we qualified NG2+ pericyte–derived odontoblasts functioning as sensory receptor cells via Piezo1. To further determine the function of regenerated odontoblasts, we next investigated the dentin mineralization ability of NG2+ pericyte–derived odontoblasts in molar teeth.
ATF6a, a Key Driver of Dentinogenesis Driven by NG2+ Pericytes
We analyzed whether NG2+ pericytes are cell sources for odontoblasts in molars in physiological condition. We produced genetically NG2 reporter mice (NG2CreERT2;tdTomato mice) and traced NG2+ pericytes and their descendants. One day after tamoxifen injection (Fig. 5A), NG2-tdTomato–positive (NG2-tdTomato+) cells, expressing the other pericyte marker PDGFRb and the preodontoblast marker Nestin, were located around blood vessels (Fig. 5B1–B6). Fourteen days after tamoxifen injection (Fig. 5C), NG2-tdTomato+ cells were located around the odontoblast niche in the molar (Fig. 5D1–E4). In addition, in different mammal species, NG2 was expressed in cells during odontoblast differentiation (Fig. 5F, G). These data indicate that NG2+ pericytes are local cell sources for odontoblasts.
We further examined whether mechanical injury to the molar dentin can induce regenerative dentinogenesis via odontoblasts originating from NG2+ pericytes. Injury models in the first molar of NG2CreERT2;tdTomato mice were established to observe the potential mineralization role of NG2+ pericytes and their descendants (Fig. 5H). Calcein labeling in dentin–pulp borders in both control and injury model mice revealed that NG2-tdTomato+ cells were located beneath the calcein-labeled mineralizing region in both conditions, and some NG2-tdTomato+ cell populations were embedded in the mineralization front (Fig. 5I1–N3). The injured group had a higher number of embedded NG2-tdTomato+ cells into calcein labeling than that of intact group (7-fold change) (Fig. 5O). These data suggest that NG2+ pericytes are localized near CZ in steady-state condition in mouse molars, and NG2+ pericytes drive dentin mineralization immediately after injury.
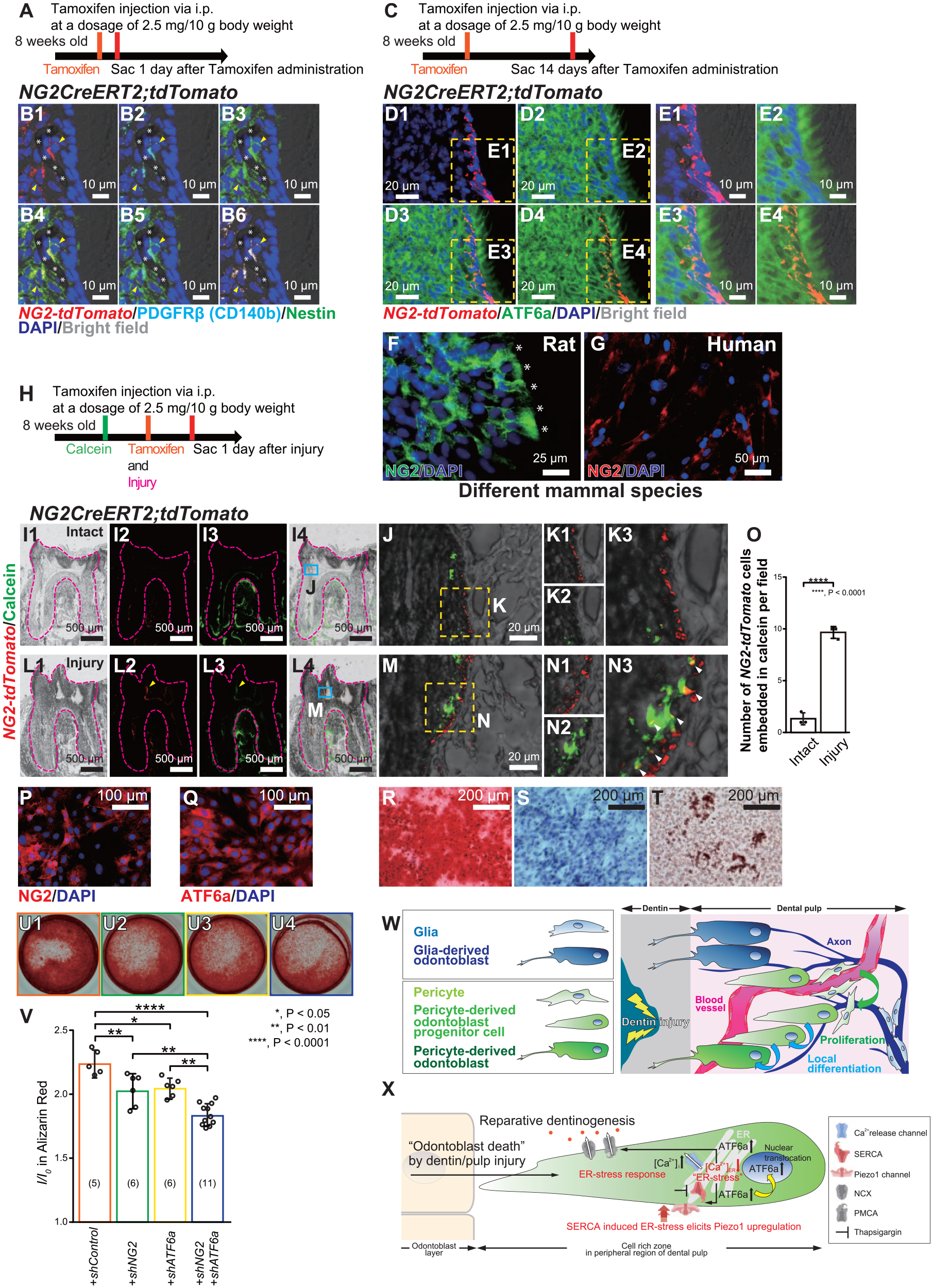
Activating transcription factor 6a (ATF6a) regulates the mineralization driven by neural/glial antigen 2–positive (NG2+) pericytes. (
To evaluate the stem cell properties of NG2+ pericytes, we demonstrated the differentiation assays of OLCs. The OLCs were immunopositive for NG2 and ATF6a antibodies (Fig. 5P, Q). OLCs also expressed mesenchymal stem cell (MSC) markers, CD73, CD90, and CD105 (Appendix Fig. 2G-I) and had the potential to differentiate into mesenchymal lineages such as osteocyte, chondrocyte, and adipocyte (Fig. 5R–T), confirming that NG2+ ATF6a+ OLCs matched with the definition of MSCs (Pittenger et al. 1999). To evaluate NG2 and ATF6a regulation during mineralization, we performed gene silencing using shRNA and measured mineralization efficacies by Alizarin red staining. Genetic knockdown of NG2 and/or ATF6a negatively regulated mineralization (Fig. 5U1-U4, V), suggesting that the specific odontoblast population participating in dentin mineralization has NG2+ pericytes as their ancestor under the regulation of ATF6a (Fig. 5W, X).
Discussion
In the CZ adjacent to the OB, odontoblast progenitor cells were analyzed by Nestin reporter mice. Both Nestin+ and Nestin− cells localized in the CZ proliferate and differentiate into odontoblast-like cells in response to odontoblastic depletion (Zhao et al. 2021). In the present study, we further showed that NG2+ Nestin+ cells regenerated in CZ after odontoblast depletion and expressed odontoblast markers. We also showed that NG2+ cells are localized near CZ in steady-state condition and NG2+ cells drive dentin mineralization after injury. These findings indicate that NG2+ pericytes are a local cellular source of odontoblasts, as odontoblast progenitor cells that immediately differentiate into odontoblasts after severe dental pulp injury.
NG2 and Nestin are markers of MSCs (Méndez-Ferrer et al. 2010; Kunisaki et al. 2013; Ouchi et al. 2018). Recently, scRNA-seq revealed that Nestin is both an odontoblast progenitor cell marker and a pericyte marker (Gomes et al. 2022). Moreover, intrapulpal NG2+ cells are not derived from glial cells but from MSCs (Kaukua et al. 2014; Zhao et al. 2014). The results of this study support that the NG2+ Nestin+ pericytes are odontogenic mesenchymal progenitor cells localized in the periphery but not deep inside of the dental pulp and are capable of differentiating into functional odontoblasts.
ER stress is a state in which proteins with abnormal higher-order structures or proteins that have not undergone normal modification accumulate in the lumen of the ER. Such proteins are called “unfolded proteins” and are affected by various physiological stresses such as calcium depletion in the ER. Because ER stress damages cells, cells are equipped with a system to avoid this, called the ER-stress response (unfolded protein response; UPR). The accumulation of unfolded proteins in the ER is sensed by ER-stress sensor proteins (Ron and Walter 2007; Kim et al. 2008; Walter and Ron 2011).
Our present study showed that odontoblasts in steady-state condition expressed ATF6a, indicating that ATF6a in odontoblasts may physiologically sense the ER stress and function in the UPR process. After genetically somatic depletion of odontoblasts, NG2+ cells in the CZ expressed Nestin and nuclear-translocated ATF6a. Thapsigargin, known as an ER-stress inducer, suppresses SERCA, resulting in the inhibition of Ca2+ uptake into the ER (Shaban et al. 2022). In the present study, thapsigargin induced nuclear translocation of ATF6a as the ER-stress response in OLCs. Our data revealed that thapsigargin increased [Ca2+]i. The increase was significantly inhibited by the application of the Piezo1 inhibitor. However, Piezo1 inhibitor did not inhibit the [Ca2+]i increase by the activation of P2Y1R. In addition, we could not observe nuclear translocation of ATF6a by activation of P2Y1R. Thus, ER stress by the [Ca2+]ER change via direct SERCA regulation augmented Piezo1-induced responses in odontoblast progenitor cells. PLC-coupled P2Y1R modulates the change in cell shape (Jin et al. 1998). The [Ca2+]ER changes via P2Y1R activation may physiologically modulate cellular functions, such as cytoskeletal changes, through odontoblast development from the progenitor cells. ER is located between dentin and nuclei in odontoblasts (Liang et al. 2023). The presence of ER at the distal side within the odontoblasts may lead to odontoblasts effectively acting as sensory receptor cells and dentin-forming cells. Our data in the measurement of [Ca2+]i showed the immediate response of Piezo1 inhibitor on the increase in thapsigargin-induced [Ca2+]i, suggesting that Piezo1 may directly interact with SERCA in odontoblasts at close range. Further in vivo studies to clarify how SERCA is inhibited in NG2+ pericytes in CZ will lead to the development of regenerative therapy and preventive dentistry.
NG2+ pericytes drove dentin mineralization after injury. When ATF6a and/or NG2 were genetically downregulated by shRNA, mineralization was impaired. Several reports have suggested that Piezo1 negatively (Xu et al. 2024) or positively (Huang et al. 2024) regulates dentinogenesis. Under the process of dentinogenesis, reactionary dentinogenesis is driven by developmental and physiological intact odontoblasts, while reparative dentinogenesis is mediated by differentiated odontoblasts derived from dental pulp cells, especially as it was revealed that NG2+ pericytes are the potential origin based on the present study. Thus, dentin regeneration is achieved through different origins. Based on previous reports and the present study, in the process of the formation of new reparative dentin by regenerated Piezo1+ odontoblasts derived from NG2+ pericytes, mineralization might be achieved by other drivers except for Piezo1. Although further study is needed, promotion of the acquisition of odontoblast characteristics originating from different types of cells will be a promising regenerative dentin therapy in the future.
In conclusion, the results of the present study showed that NG2+ Nestin+ cells beneath the odontoblasts are odontoblast progenitor cells. NG2+ Nestin+ cells that creep up from the CZ to OB are also involved in mechanosensitivity and dentin mineralization. ATF6a plays important roles in the differentiation of NG2+ pericytes in the CZ into functional odontoblasts that act as sensory receptor cells and dentin-forming cells.
Author Contributions
T. Ouchi, M. Ando, R. Kurashima, M. Kimura, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; N. Saito, A. Iwasaki, H. Sekiya, K. Nakajima, T. Hasegawa, T. Mizoguchi, Y. Shibukawa, contributed to conception and design, data interpretation, drafted and critically revised the manuscript. All authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241307944 – Supplemental material for Pericytes Are Odontoblast Progenitor Cells Depending on ER Stress
Supplemental material, sj-docx-1-jdr-10.1177_00220345241307944 for Pericytes Are Odontoblast Progenitor Cells Depending on ER Stress by T. Ouchi, M. Ando, R. Kurashima, M. Kimura, N. Saito, A. Iwasaki, H. Sekiya, K. Nakajima, T. Hasegawa, T. Mizoguchi and Y. Shibukawa in Journal of Dental Research
Footnotes
Correction (March 2025):
Article updated to correct figure 5X.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Japan Society for the Promotion of Science KAKENHI (grant Nos. 22K17025 [TO], 19K10117, 22K09972 [MK], 19H03833, 24K12953 [YS]), a Research Fund from the Nakatomi Foundation, a Research Fund from Nishiyama Dental Academy, the Tokyo Dental College Research Branding Project (Multidisciplinary Research Center for Jaw Disease [MRCJD]: Achieving Longevity and Sustainability by Comprehensive Reconstruction of Oral and Maxillofacial Functions), Tokyo Dental College Research Grant (Well-being Project), and Tokyo Dental College Dean’s Encouragement Research Grant.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
