Abstract
Genetic dental disorders (GDDs) can occur either isolated or as part of syndromes. Clinically, deviations in tooth shape, size, or structure, as well as the absence of multiple teeth, lead to severe dysfunction and a reduced quality of life, requiring lifelong preventive, conservative, and prosthodontic dental care. The dental management of prevalent dental diseases, such as caries or periodontitis, has been based on decades of research, whereas scientific data on the dental management of GDDs are scarce. This lack of data is challenging for dental practitioners, who must primarily rely on empirical knowledge only. Therefore, a systematic literature search and review were conducted on the dental management of common GDDs, such as ectodermal dysplasia, amelogenesis imperfecta, dentinogenesis imperfecta, periodontitis as a manifestation of rare systemic diseases, and X-linked hypophosphatemia and hypophosphatasia. The review revealed that 468 of the 9,115 retrieved publications met the inclusion criteria, with most being case reports or case series, highlighting a lack of robust clinical trials. This critical review provides a brief summary of the genetic background, key clinical signs, and treatment options for these conditions. The dominance of case reports emphasizes the need for improved reporting standards and long-term follow-up to support comprehensive data synthesis and meta-analyses. In addition, the uneven global distribution of publications suggests disparities in access to advanced dental care for GDDs. Efforts to standardize reporting and improve treatment documentation globally are crucial to addressing these challenges. In this way, information on GDD management can be improved, and statistical analyses of the data can be performed.
Keywords
Introduction
Genetic dental disorders (GDDs) either occur as isolated dental diseases or can be part of a symptom complex in syndromic diseases (de La Dure-Molla et al. 2019). GDDs are often associated with severe functional disorders, an atypical clinical presentation, and a reduced oral health–related quality of life (Friedlander et al. 2019). Although the genotype can usually be determined precisely, the phenotype often shows a heterogeneous appearance among individuals with the same genetic diagnosis. Decisive indications of a GDD include generalized symptom constellations, which may manifest as deviations in tooth number, shape, size, or surface structure, often affecting both primary and permanent dentition, although the degree of involvement may vary (Albandar et al. 2018; James and Roudsari 2019; Bloch-Zupan et al. 2023). In some cases, multiple primary and permanent teeth may be missing (Bergendal 2010), and early periodontal destruction may occur during childhood (Albandar et al. 2018). Furthermore, there may be a multisystemic or syndromic clinical presentation, along with familial clustering of comparable symptoms (Prasad et al. 2016; Bloch-Zupan et al. 2023).
There are currently more than 400 known GDDs, with the exact genetic changes known for ~79% of these diseases (de La Dure-Molla et al. 2019). It is also assumed that there are considerably more GDDs (Hennekam et al. 2010; Bloch-Zupan et al. 2012). Furthermore, new mutations can occur in genes relevant for tooth development and thus lead to deviating signs or clinical disease patterns that have not yet been described. Although countless epidemiological data have been published for common dental diseases such as caries and periodontitis (e.g., Bernabe et al. 2020; Trindade et al. 2023), for GDDs, only estimates of prevalence are available (Orphanet 2024). In principle, rare diseases are defined in Europe as those that affect no more than 1 person in every 2,000 people (European Parliament and Council 2019).
While modern preventive and operative caries and periodontitis management approaches are based on decades of research effort and a large number of scientific studies and recommendations (e.g., Schwendicke et al. 2016; Duncan et al. 2019; Sanz et al. 2020; Herrera et al. 2022; Kühnisch et al. 2024), both clinical studies and systematically developed recommendations for the dental management of GDDs are lacking. Dental professionals, individuals affected by these conditions, and all those involved in their medical and/or dental care face the challenge of making decisions on the basis of insufficient data and evidence.
Aim of the Critical Review
To address the gap in the international literature regarding dental health care and the extensive and lifelong therapeutic needs of individuals with GDDs, the aim of this critical review was to assess the existing body of knowledge on selected GDDs through a systematic literature search. These include ectodermal dysplasia (ED); amelogenesis imperfecta (AI); dentinogenesis imperfecta (DI); periodontal disease as a manifestation of rare systemic diseases (RSD), such as primary immunodeficiencies, Papillon-Lefèvre syndrome, Ehlers-Danlos syndrome, and plasminogen deficiency (Albandar et al. 2018); X-linked hypophosphatemia (XLH); and hypophosphatasia (HPP) (Fig. 1). The selection of these specific GDDs is based on an empirical survey within the multicenter group of authors, with priority given to diseases with key manifestations such as tooth structure disorders in both dentitions, dental agenesis, and atypical tooth loss. This review also provides an overview of specific genotypes, key clinical manifestations, and reported dental interventions while identifying knowledge gaps.
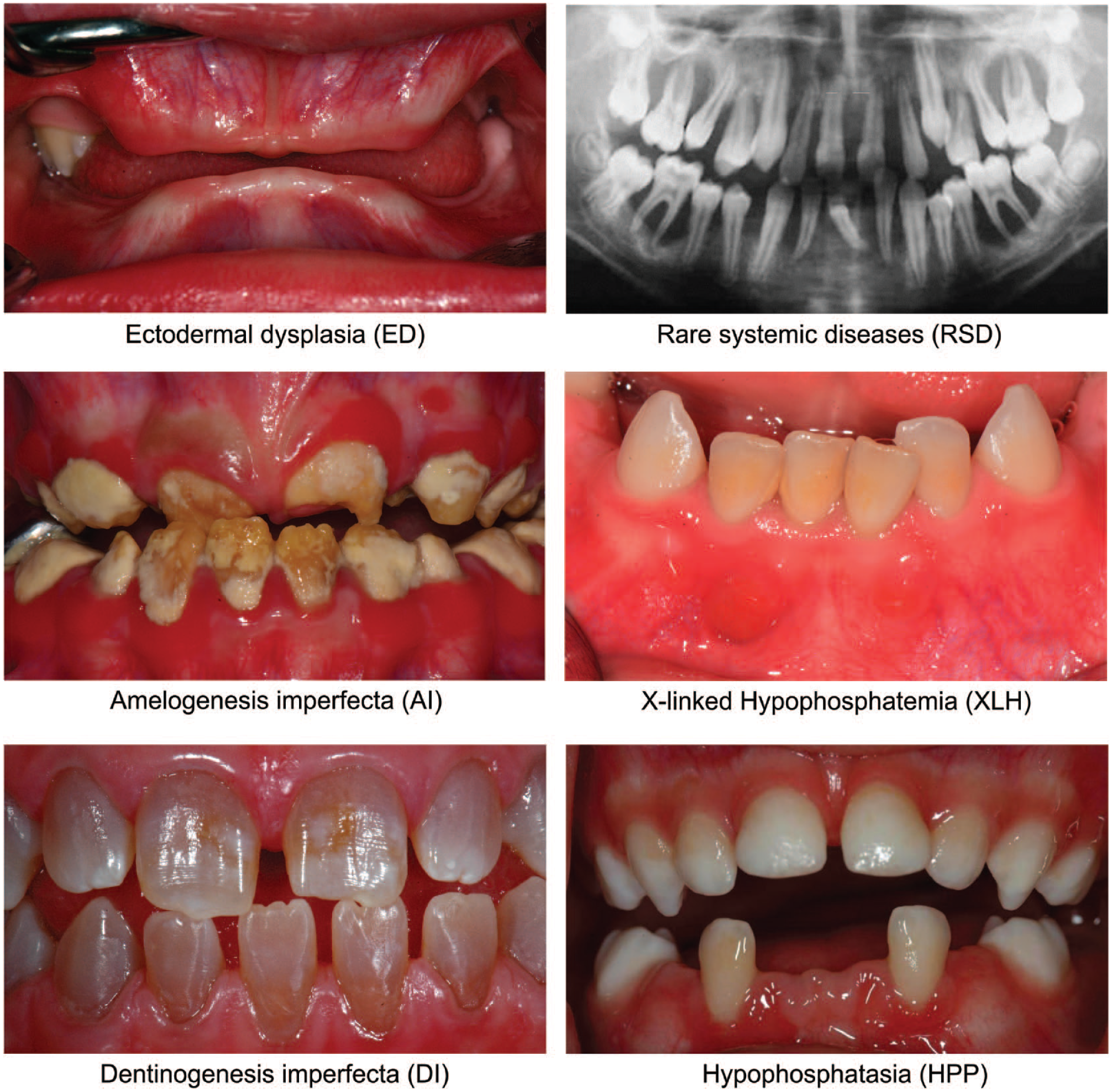
Overview of the clinical and radiologic appearance of selected genetic dental disorders. Key clinical signs include missing teeth, irregular eruption patterns, and peg-shaped teeth in ectodermal dysplasia; varying enamel hardness and tooth discoloration in amelogenesis imperfecta; and enamel loss and amber to bluish-gray teeth in dentinogenesis imperfecta. Periodontitis as a manifestation of rare systemic diseases is characterized by early bone and tooth loss, whereas spontaneous tooth avitality due to altered enamel and dentin density typically occurs in X-linked hypophosphatemia. Premature tooth loss is a key sign of hypophosphatasia.
Systematic Review of the Literature
For the systematic literature search, we applied the well-proven PICOS methodology to compile disease-specific search terms. The PICOS questions focused on identifying relevant studies involving patients with various GDDs (patient); examining therapeutic dental care interventions such as preventive care, restorative measures, and treatment of periodontitis (intervention); noting that direct comparative studies are rare owing to the low prevalence of these conditions (comparison); and assessing outcomes related to dental function, esthetics, quality of life, and tooth survival (outcome). We included all available study types to cover the available evidence in this area (study type). The respective search terms can be found in the supplementary material (Appendix Tables 1–5). Searches were conducted in MEDLINE and Embase between January 2000 and December 2022. The search was limited to articles published in English or German. In addition, a manual search was conducted in dental journals published in German. As part of the first systematic literature screening, all electronic search results (N = 9,115) were transferred to the web-based app Rayyan (Ouzzani et al. 2016), and duplicates were removed. In the second step, a systematic title and abstract-screening process was performed so that each reference could be assessed individually before being included or excluded. If no clear decision on inclusion or exclusion could be made, the reference was initially included, and the decision was made later on the basis of the retrieved full text. All identified full texts were subjected to a systematic and detailed review and evaluation. Next, each reference was subjected to a qualitative assessment. For this purpose, the methodological questions formulated by Moga et al. (2012) for case reports (CRs) and case series (CSs) were adapted and used for the specific context of dental care for GDDs. In detail, information on the hypothesis posed, dental and other medical interventions, reporting before and after dental intervention, follow-ups or adverse events during the dental treatment, and so forth were recorded. Furthermore, the recommendations of the Scottish Intercollegiate Guidelines Network (SIGN 2021) were used for the qualitative evaluation of clinical studies. The subsequent evaluation of these data was used to assess the risk of bias. All collected data were recorded in a database (Epidata software, version 4.6.0.6, Christiansen and Lauritsen 2010) so that all the information was available digitally and could later be exported to an Excel spreadsheet (Excel 2019, Microsoft Corporation, Redmond, WA, USA).
Available Publications on Selected GDDs
The systematic literature search identified a total of 468 publications on the dental management of GDDs that were eligible for inclusion (Fig. 2). The number of included publications varied remarkably between the selected GDDs: ED (n = 175), AI (n = 160), DI (n = 55), RSD (n = 38), XLH (n = 20), and HPP (n = 20). These numbers indicate that the overall number of available publications on the dental management of GDDs is very low. The number of publications may indirectly represent the occurrence of GDDs in the population. Thus, it may be assumed that AI and ED are among the most frequent entities in the GDD group. The systematic literature search results clearly revealed that experiences and findings concerning the dental treatment of individuals affected by GDDs are extremely rare compared with those concerning caries or periodontitis management. Thus, efforts should be made to improve scientific and practical knowledge and provide more information in all dental disciplines.
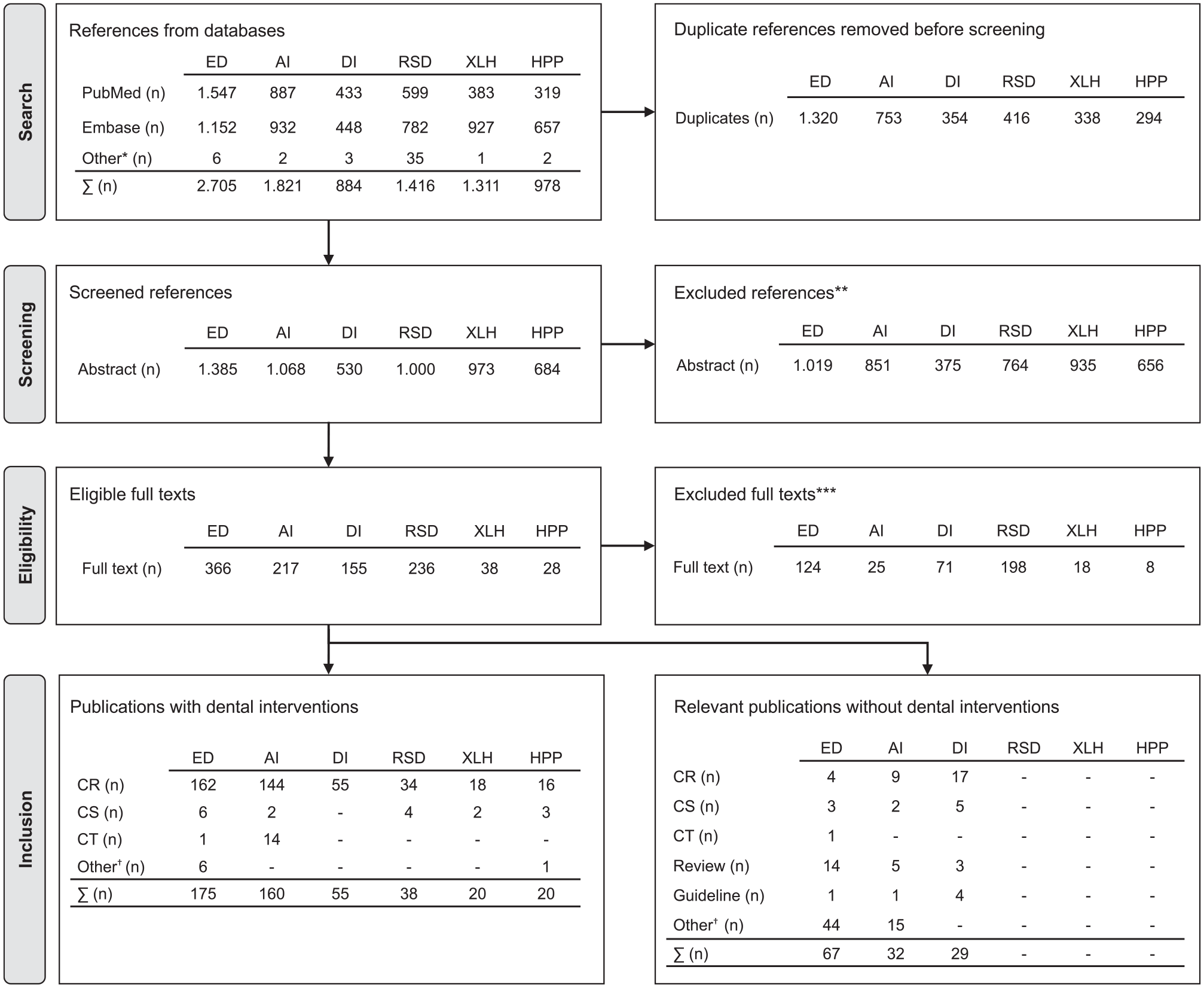
The flowchart illustrates the systematic literature search, title, abstract, and full-text screening for ectodermal dysplasia (ED), amelogenesis imperfecta (AI), dentinogenesis imperfecta (DI), periodontal disease as a manifestation of rare systemic diseases (RSD), X-linked hypophosphatemia (XLH), and hypophosphatasia (HPP) (Page et al. (2021) adapted from the PRISMA 2020 flow diagram). CR, case report; CS, case series; CT, clinical trial. *References retrieved through hand search. **Exclusion criteria: foreign language, abstract unavailable, wrong year, wrong disease, wrong publication type, wrong population, animal study, in vitro, incorrect result, background article, missing dental intervention, wrong study design, genetic study. ***Exclusion criteria: no full-text publication, full text unavailable, other language, no reference to the topic, no human study, incorrect diagnosis, no dental diagnosis or intervention reported. †Different types of studies, that is, quality-of-life studies, genetic studies, and diagnostic studies.
Genes Associated with Selected GDDs
As part of the systematic review, we documented the genetic background of the selected entities where available. Many of these disorders are caused by pathogenic variants in different genes, leading to diverse phenotypes. For example, ED may be caused by variants in more than 30 genes associated with different clinical manifestations. Notable examples include hypohydrotic/anhydrotic ED (e.g., EDA, EDAR, and EDARADD), TP63-associated forms (e.g., TP63 and KDF1), and WNT10A-associated forms (e.g., WNT10A and KREMEN1). Regarding the AI spectrum, we identified at least 26 major causative genes. Isolated forms of AI, such as those associated with FAM83H and MMP20, are more common than syndromic forms, such as enamel–renal syndrome (FAM20A), Kohlschütter–Tönz syndrome (ROGDI), and junctional epidermolysis bullosa (JEB) (LAMA3). Bloch-Zupan et al. (2023) reported that more than 70 genes are associated with AI, indicating a broader range of genetic findings. However, only a minority of the included studies provided genotypic information, which may explain the difference in the number of identified genes. For DI, isolated cases have been linked only to pathogenic variants in the DSPP gene, whereas DI in osteogenesis imperfecta is caused by alterations in the COL1A1 and COL1A2 genes. In the RSD group, notable conditions included primary immunodeficiencies such as severe congenital or cyclic neutropenia (e.g., ELANE, HAX1, and G6PC3) and Papillon–Lefèvre syndrome (CTSC). In XLH, more than 200 mutations in the PHEX gene have been identified (Ruppe et al. 2011). These mutations lead to disrupted phosphate regulation, resulting in hypophosphatemia and mineralization defects in the dentition and skeleton. HPP, caused by pathogenic variants in the ALPL gene, for which more than 400 mutations have been identified (Farman et al. 2024), contributes to the wide clinical heterogeneity of the condition, affecting multiple tissues. Among these, cementum is the most severely affected, impairing the attachment of the periodontal ligament and leading to weakened periodontal support and early tooth loss, particularly in the primary dentition.
In summary, numerous genes influence tooth development, and pathogenic alterations in these genes can have severe functional and esthetic consequences (Appendix Tables 6–11). If GDDs are suspected, molecular genetic analyses guided by clinical characteristics should be performed to confirm or exclude the suspected diagnosis (Bloch-Zupan et al. 2023). A precise genetic diagnosis provides essential information about the prognosis of the disease, prevents misdiagnosis, and reveals possible syndromic involvement of other organ systems.
Clinical Signs of GDDs
Compared with healthy dentition, GDDs are often associated with serious dental functional limitations and esthetic impairments. Furthermore, they might be linked to additional medical conditions. ED forms are frequently associated with oligodontia (more than 6 missing teeth) or, in rare cases, with anodontia (all teeth are missing) (Bergendal 2010). Key clinical signs include more missing teeth in the lower jaw than in the upper jaw and hypoplastic bone in the edentulous region. In addition, reduced salivary flow may be observed, potentially leading to fragile oral mucosa (Bergendal et al. 2006; Bergendal 2010). Consequently, functional impairments are associated with ED, affecting mastication and speech development as well as reduced esthetics due to tooth spacing and peg-shaped teeth. Furthermore, general symptoms such as hypohidrosis, hypotrichosis, or onchodysplasia may be associated with ED. In the case of AI, the primary clinical forms include hypomineralized, hypomature, and hypoplastic enamel. Hypomineralized and hypomature forms are associated with severe hypersensitivity or toothache, which can severely restrict physiological use and regular dental care (Parekh et al. 2014; Quandalle et al. 2020). In addition, teeth may show noticeable deviations in color and shape (Poulsen et al. 2008). The main sign of DI is rapid and continuous tooth wear on the incisal and occlusal surfaces (de La Dure-Molla et al. 2015). In RSD, early, generalized, and abnormal gingival/periodontal inflammatory tendencies, tooth loosening, and/or tooth loss are common clinical signs. Oral mucosal manifestations associated with RSDs include ligneous gingivitis in plasminogen deficiency (Gunhan et al. 2012) and aphthous stomatitis, ulcers, and candidiasis in primary immunodeficiencies (Szczawinska-Poplonyk et al. 2009). In XLH, the main dental concern includes spontaneous avitalities or odontogenic infections (Linglart et al. 2014). The main clinical sign of HPP is premature tooth loss, which can occur just a few years after tooth eruption (Hughes et al. 2017).
Although the primary signs of these conditions are easily detectable during clinical examination, additional diagnostic tools, such as radiologic, microbiologic, and human genetic diagnostic tests, are often recommended to confirm the diagnosis and identify treatment needs. Notably, the CRs included in this review presented heterogeneous clinical presentations. Therefore, future studies should correlate clinical symptoms and management strategies with specific genotypes, leading to a better assessment of treatment outcomes.
Reported Dental Interventions for Individuals with GDDs
In terms of the generalized manifestation and severity of symptoms, dental treatment of GDDs requires preventive, conservative, orthodontic, and/or prosthodontic measures and, depending on the underlying disease and clinical findings, periodontal therapy beginning early in childhood and continuing throughout life. This usually extends far beyond the usual treatment spectrum and requires interdisciplinary/interprofessional collaboration. For example, in the case of HPP, enzyme replacement therapy has shifted care from purely symptomatic management to treatment that significantly benefits teeth still undergoing mineralization, improving their density and structure (Schroth et al. 2021; Smart et al. 2023). The dental management of GDDs should combine individual care with a strong emphasis on secondary and tertiary prevention adjusted to the age-related needs of those affected (Table 1). The main treatment goals are pain reduction and the restoration of function and esthetics, which directly affect the oral health–related quality of life (Pousette Lundgren et al. 2015). Managing the oral mucosa is particularly important in conditions such as JEB and AI, where mucosal lesions and ulcers require careful treatment to avoid further trauma during dental procedures (Krämer et al. 2020). Although GDDs may also affect oral tissues such as the mucosa, this review focuses on dental and periodontal manifestations, which are central to management strategies. Notably, most of these correlations are empirically documented and have not been quantified.
Overview of the Described Types of Treatment, Specific Dental Interventions, and Number of patients with Selected Genetic Dental Disorders. a
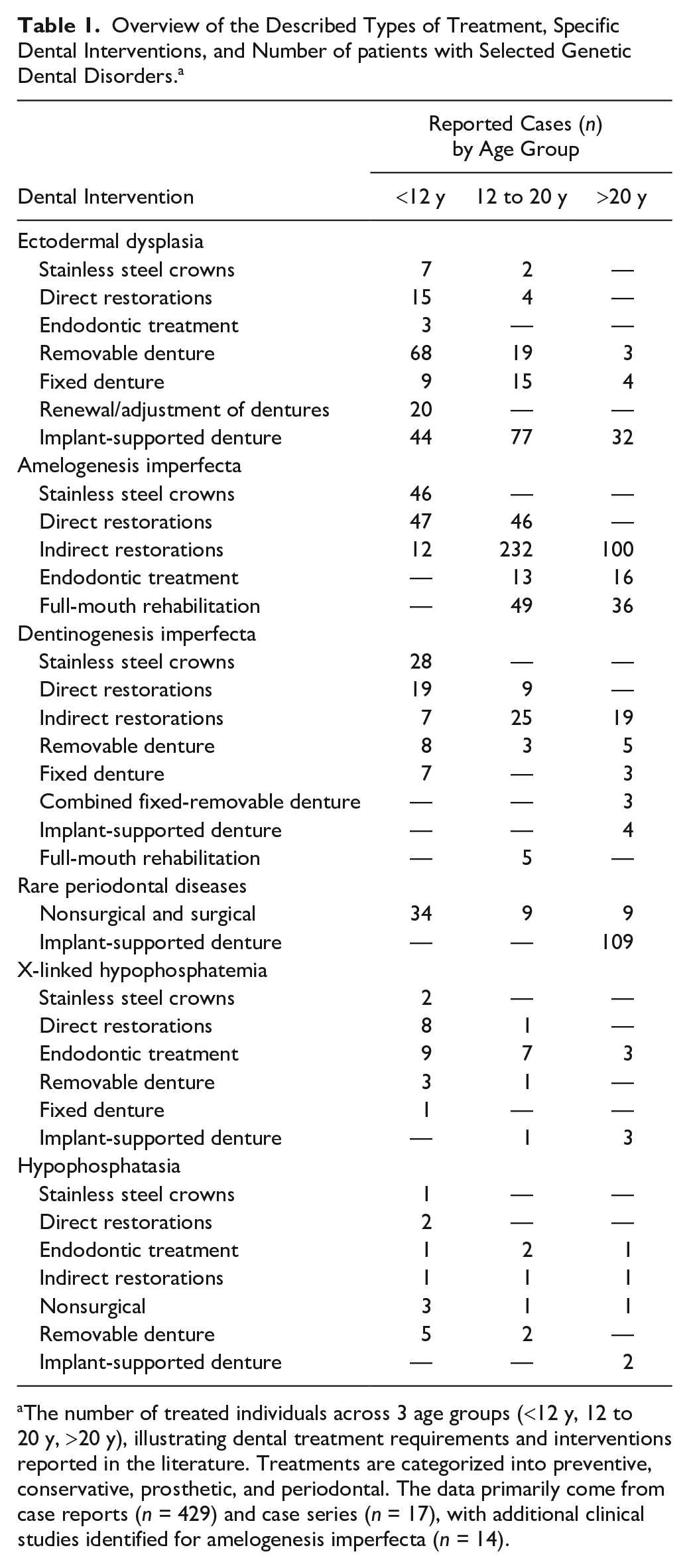
The number of treated individuals across 3 age groups (<12 y, 12 to 20 y, >20 y), illustrating dental treatment requirements and interventions reported in the literature. Treatments are categorized into preventive, conservative, prosthetic, and periodontal. The data primarily come from case reports (n = 429) and case series (n = 17), with additional clinical studies identified for amelogenesis imperfecta (n = 14).
Although the primary prevention of GDDs cannot be achieved in principle, the avoidance of other accompanying pathologies (e.g., caries, erosion, gingivitis, or periodontitis) should be given high priority so that secondary diseases do not further impair the primary burden of disease caused by the disadvantaged dental situation. This includes dietary advice aimed at reducing sugar consumption, safeguarding optimal oral hygiene, professional dental cleaning, and adequate fluoride supply and use.
For GDDs at high risk for tooth wear and posteruptive breakdown, preventive measures to protect the tooth structure are crucial. Thus, early restorative measures could simultaneously have a preventive role and limit further tooth wear. For children and adolescents, stainless steel crowns are a simple treatment option for the comprehensive restoration of primary teeth (Table 1). In addition, the clinical use of indirect restorations, especially crown restorations, is indicated to replace the lost tooth structure of extensively destroyed teeth and to restore functionality. Interestingly, studies in patients with AI have shown that the early use of indirect restorations in the second decade of life is associated with positive effects (e.g., Pousette Lundgren et al. 2015; Klink et al. 2018; Lundgren et al. 2018) (Table 1). Endodontic strategies are key to the dental management of tooth preservation; this is particularly indicated for GDDs characterized by frequent pulp infections, such as XLH (Table 1). RSDs require periodontal treatment according to clinical practice guidelines (Sanz et al. 2020; Herrera et al. 2022) and must adhere to strict biofilm management. In the case of missing or lost teeth, removable, fixed, or implant-supported dentures can be used. For most people affected by GDDs, full-mouth rehabilitation should be an integral component of the lifelong dental treatment strategy, as disease-related findings usually affect both dentitions equally. This management approach includes consideration of the primary and permanent teeth that should be preserved as well as the (re)establishment of function, occlusion, and esthetics. This concept is pursued for all age groups with the aid of direct, indirect, (temporary) removable, and implant-supported prosthodontic restorations. Individuals affected by GDDs also appear to be much more frequently and extensively affected by tooth loss, as the survival rate of teeth appears to be reduced in a number of GDDs. To minimize the functional and esthetic consequences of early tooth loss, the necessity and importance of early dental prosthodontic planning should be emphasized.
Available Study Types
The literature on the dental management of GDDs is almost exclusively dominated by CRs (n = 429; 91.7%) and CSs (n = 17; 3.6%) (Fig. 2). Only 15 clinical trials (3.2%) were identified for the clinical management of GDDs, with most of these trials (n = 14) involving AI patients (Fig. 2). The focus of the clinical studies was exclusively based on the comparison of different forms of restoration (e.g., Lundgren et al. 2018; Ohrvik and Hjortsjö 2020). Only 1 study compared treatment success in individuals with AI with that of an independent control group (Klink et al. 2018).
As a consequence, the dominance of CR and the low number of high-quality clinical trials are accompanied by a lack of evidence analyses or meta-analyses for GDDs. The scarcity of clinical trials for GDDs is related both to the rarity of the disease in the population and the fact that only a very small number of individuals with the same diagnosis are treated in the same care settings (e.g., general dental practices, university dental clinics, or dental schools). This can further vary according to genotype, phenotype, or age. Consequently, both mono- and multicenter study designs are unlikely to be feasible for a reasonable recruitment time; this finding most likely explains the lack of literature on the dental management of GDDs. Therefore, it is essential for dentists to appreciate the importance of CR, to pragmatically evaluate this form of publication as the only possible publication, and to extend the literature as much as possible in the future. In addition, CR can describe new diagnostic and dental interventions as well as unusual forms of known diseases and highlight differences in international medical/dental care (Gagnier et al. 2013; Nissen and Wynn 2014).
Global Perspective
A heterogeneous global distribution of available publications was observed on the basis of the data collected (Fig. 3). Most of the included studies were published in Europe (n = 204), followed by Asia (n = 147), North America (n = 55), South America (n = 44), Africa (n = 10) and Australia (n = 8). However, this uneven distribution should not be misinterpreted to suggest that the GDDs in question are not present in regions with few or no publications. Moreover, it is important to interpret the global distribution of publication numbers with caution, as our search was limited to studies published in English and German, which may have influenced the number of retrieved articles. The implementation of complex therapies, as is required in the case of GDDs, demands specialized dental knowledge, technical skills, and financial resources, which vary globally. Therefore, regional differences in health care resources and the financial capacities of affected individuals may affect access to dental care for GDDs, potentially contributing to the uneven distribution of publications. Nevertheless, although GDDs are rare, their dental management deserves broader recognition.
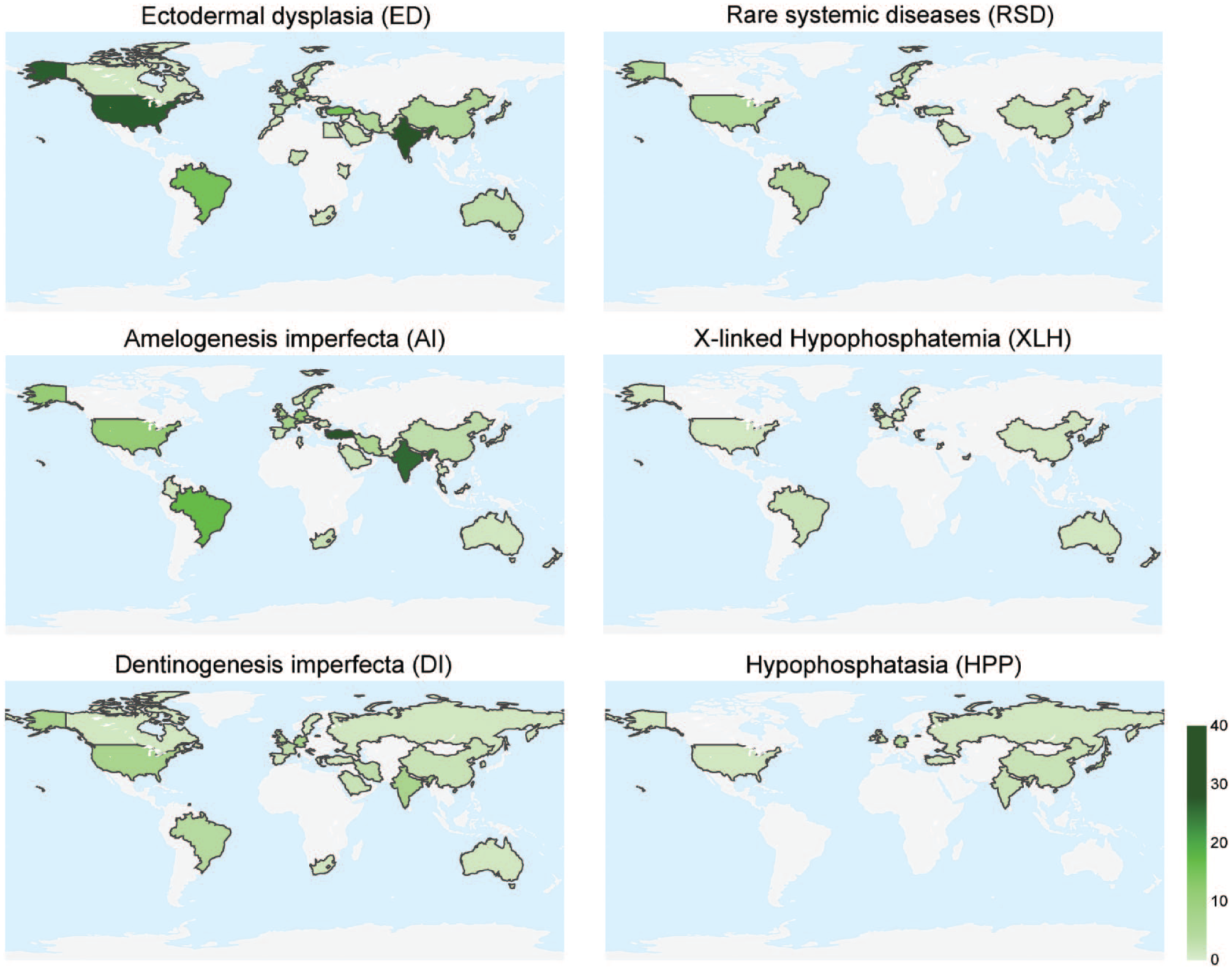
Overview of the regional origin of the included case reports, case series, and clinical trials with dental interventions for genetic dental disorders.
Quality of the Available Literature
The quality of the available literature was analyzed using the criteria proposed by Moga et al. (2012). For the 18 defined quality domains, Figure 4 shows moderate publication quality, with relevant information often missing or remaining unclear, indicating that reporting in CRs and CSs must be improved to reduce the heterogeneity between the reports (Gagnier et al. 2013). Heterogeneity is already high due to individual case characteristics (e.g., the genotype/phenotype of the GDDs, age, other medical conditions, cooperation status, and required treatment setting) and is further increased by suboptimal reporting. First, it is important to address the differences in long-term follow-up. The spectrum ranges from reporting on the achievement of dental treatment without further monitoring to less frequent reports with more than a decade of monitoring (e.g., Bergendal et al. 2015; Fernandes et al. 2016; James and Roudsari 2019). Although long-term recall allows conclusions to be drawn about the durability of the selected treatment option, the same is not possible in the case of short follow-up periods. In this context, a minimum observation period (e.g., 2 y) would be preferable to increase the comparability of CR. It would also be important to report the failure of restorations and the associated refurbishments, repairs, or renewals. This further requires objective outcome reporting following treatment to obtain information on relevant clinical outcomes (e.g., pain reduction, functional and esthetic improvement, tooth or restoration survival) (Hickel et al. 2023), periodontal status (Papapanou et al. 2018), or oral health–related quality of life. Furthermore, possible adverse events during and after treatment should be documented and reported. An additional challenge with CRs and CSs is that the exact number of treated teeth or the number of restorations are often not reported. However, such information would be valuable for obtaining information on restoration survival, which could be summarized in reviews and statistically explored. One additional limitation to be mentioned is the framework of national health care and reimbursement systems, which may limit the choice of treatment options. The fact that full-mouth rehabilitation is complex and cost intensive appears to be another major barrier. As a result, restorative therapies are often unaffordable for those affected and ultimately cannot be performed. These economic aspects may have led to further undetected bias in reporting. In conclusion, CRs and CSs should follow well-established reporting protocols to improve the consistency and quality of reporting in the field (Gagnier et al. 2013).
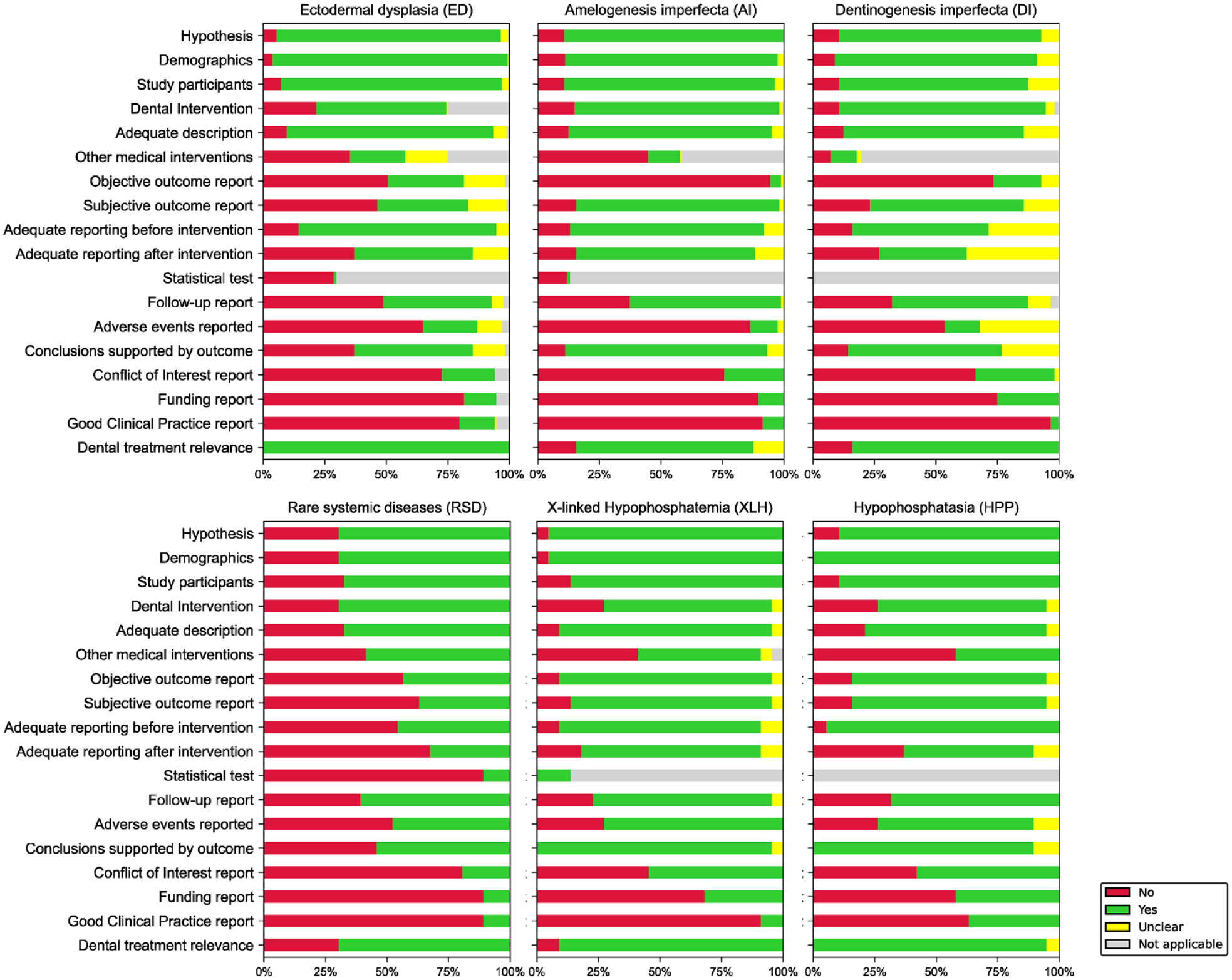
Overview of the qualitative assessment of the included case reports and case series on genetic dental disorders in a total of 18 quality domains, modified according to Moga et al. (2012).
Knowledge Gaps
This systematic review of the available literature on GDDs has uncovered knowledge gaps. Here, the diagnosis of GDDs should be noted. Phenotype assessment has been traditionally used exclusively for classification; from today’s perspective, human genetic diagnostics should be regarded as the diagnostic “gold standard.” However, this was not performed in most of the studied CRs. Furthermore, genetic testing is by no means established in daily dental practice. Given the importance of human genetic diagnostics for those affected by GDDs, for example, for exact diagnosis, the differentiation between “isolated” and “syndromic” forms, and prognosis information, such information should not be neglected in the context of dental care.
Although the spectrum of key clinical signs is widely known, there is a lack of detailed analyses of genotype-phenotype associations for the GDDs under consideration. Hypothetically, successful linking of genetic and clinical information would allow existing classification systems (e.g., Shields et al. 1973; Witkop 1988) to be further developed or refined (e.g., de La Dure-Molla et al. 2015; Simmer et al. 2022; Bloch-Zupan et al. 2023; Farman et al. 2024). However, this requires consistent implementation of human genetic testing among individuals affected by GDDs.
Although the disease- and age-specific characteristics of dental treatment for the selected diseases cannot be discussed in detail at this point, this review revealed heterogeneous treatment strategies documented in the CR, very few in vitro studies (e.g., Yaman et al. 2014; Epasinghe and Yiu 2018; Massé et al. 2021), a widespread lack of clinical trials on adhesive bonding to structurally altered enamel and dentin, and significant information gaps in the use of digital manufacturing processes. The latter aspect appears to be of particular importance, as new digital technologies enable the planning and fabrication of customised restorations in complex situations more quickly, efficiently, and predictably. This is noteworthy, as the dental management of caries, enamel hypomineralisation, or tooth loss using modern, mostly computer-aided design/computer-aided manufacturing processes, has already become part of everyday dental practice. To date, this trend has been only partially recognized in the treatment of GDDs (e.g., Sarapultseva et al. 2020), representing another knowledge gap.
Another serious barrier for those affected and their families is the need for extensive and lifelong dental treatment coupled with high financial costs. It is likely that many people affected globally lack the financial means to afford dental treatment; research on this topic is completely lacking. Moreover, the economic aspects of the dental management of GDDs and the expected costs for those affected have been addressed in only a few studies (Murdock et al. 2005; Incici et al. 2009; Pousette Lundgren et al. 2021). Documenting this—very likely—undertreatment would help to describe the global inequality and barriers in providing dental care for this disadvantaged group of individuals. Ideally, all countries would follow the United Nations Convention on the Rights of Persons with Disabilities and grant those affected by GDDs the highest attainable standard of health and unrestricted access to health rehabilitation (United Nations 2008).
In view of the described limitations and knowledge gaps associated with the diagnosis and treatment of patients with GDDs, conducting clinical trials and achieving more evidence represent important challenges. It can therefore be pragmatically concluded that this situation can be accepted only as it is and that extensive efforts should be made to improve the reporting quality of CR and CS. Ideally, the genotype, phenotype, age-related restorative treatments, survival data, failures and their causes, as well as adverse events, should be published, potentially enabling data to be systematically extracted from CRs and CSs and methodically presented as a meta-analysis (Nissen and Wynn 2014; Murad et al. 2018). However, this requires a standardized, clinical procedure and reporting (Gagnier et al. 2013). In addition to high-end solutions, low-budget approaches and interim therapeutic interventions should also be published to reflect the worldwide health care alternatives of individuals affected by GDDs.
Conclusions
This critical review highlights multiple challenges and knowledge gaps in the treatment of individuals affected by GDDs and emphasizes the importance of CRs and CSs as reporting instruments. Future dental treatments should be documented on a long-term follow-up basis, and professionals should be encouraged to publish clinical experiences to obtain broader evidence on the dental management of GDDs. Moreover, the reporting quality should be improved to enable statistical analysis of CRs and CSs.
Author Contributions
H. Dujic, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; K. Bücher, I.M. Schüler, P. Schmidt, S. Hertel, J. Timpel, A. Jablonski-Momeni, R. Schilke, I. Kapferer-Seebacher, J. Zschocke, A. Liebermann, J.F. Güth, D. Edelhoff, contributed to conception, design, data acquisition, analysis, and interpretation and critically revised the manuscript; R. Heinrich-Weltzien, J. Kühnisch, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241305330 – Supplemental material for Dental Management of Genetic Dental Disorders: A Critical Review
Supplemental material, sj-docx-1-jdr-10.1177_00220345241305330 for Dental Management of Genetic Dental Disorders: A Critical Review by H. Dujic, K. Bücher, I.M. Schüler, P. Schmidt, S. Hertel, J. Timpel, A. Jablonski-Momeni, R. Schilke, I. Kapferer-Seebacher, J. Zschocke, A. Liebermann, J.F. Güth, D. Edelhoff, R. Heinrich-Weltzien and J. Kühnisch in Journal of Dental Research
Footnotes
Acknowledgements
We would like to express our gratitude to Dr. Monika Nothacker for providing invaluable independent methodological advice and training throughout the course of AWMF guideline project No. 083-043. Her expertise and guidance were instrumental in the development of the guidelines and, subsequently, this review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is funded by the German Innovation Fund with funding number 01VSF21007.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
