Abstract
Regeneration of periodontal tissue, particularly the cementum–periodontal ligament (PDL)–bone complex, has long been challenging because the differentiation kinetics of cells and the molecular pathways contributing to the regeneration process are largely unknown. We aimed to evaluate the cell behavior and molecular pathways that contribute to periodontal tissue regeneration in vivo. We analyzed the process of periodontal tissue regeneration through subrenal capsule transplantation of immediately extracted molars in mice. We showed that the regenerated periodontal tissue in the subrenal capsule was morphologically comparable to the intact periodontal tissue, with increased cellular cementum thickness in the apical region. Cell tracing analysis revealed that the cells comprising the regenerated periodontal tissue were derived from transplanted teeth and were indispensable for periodontal tissue regeneration, whereas recipient mouse-derived cells partly contributed to angiogenesis. Bioinformatics analysis based on the gene expression profile in the transplanted teeth indicated that Wnt/β-catenin signaling is involved in periodontal tissue regeneration, which was further confirmed through β-catenin immunohistochemistry. Moreover, the constitutive activation of β-catenin in the cells of transplanted teeth was found to promote accelerated cellular cementum apposition, while the conditional knockout of β-catenin in the cells of transplanted teeth suppressed cellular cementum apposition. Notably, the manipulation of Wnt/β-catenin signaling did not interfere with the bone–PDL–cementum complex, while endogenous osteoclast activity was affected in bone. Our results demonstrated the essential roles of endogenous PDL cells in periodontal tissue regeneration and that Wnt/β-catenin signaling is involved in this process, particularly cellular cementum apposition. Hence, controlling this pathway could promote cementum regeneration, which is a critical process for the regeneration of the cementum–PDL–bone complex. This study provides novel insights into cell behavior and signaling pathways that will advance practical periodontal tissue regeneration.
Keywords
Introduction
The periodontal ligament (PDL) is a specialized fibrous tissue that connects the cementum to the alveolar bone. It plays important roles in oral functions, including the dissipation of masticatory force, tooth eruption, and neurologic control of mastication; therefore, it is an essential component of the periodontal tissue in natural teeth (Beertsen et al. 1997; Kaku and Yamauchi 2014). PDL functions rely on the anchorage of its principal fibers to the bone and cementum proper, comprising the tooth–bone interface; thus, the PDL needs to be considered a cementum–PDL–bone complex (PDL complex). Owing to its layered structure, the PDL harbors various cell types, such as fibroblasts, osteoblasts, cementoblasts, osteoclasts, endothelial cells, and peripheral nerve cells (Beertsen et al. 1997). To maintain such cell diversity, the PDL is abundant in tissue stem cells (Seo et al. 2004; Bartold and Gronthos 2017). PDL harbors mesenchymal stem cell marker-expressing cells, which exhibit perivascular localization and contribute to tissue remodeling (Seo et al. 2004; Roguljic et al. 2013). These observations are consistent with the recruitment of bone marrow–derived cells to the PDL through the bloodstream (Tomida et al. 2013; Kimura et al. 2014; Kaku et al. 2017) and stem cells in the PDL possessing pericyte-like characteristics (Iwasaki et al. 2013).
Owing to the vital role of the PDL in tooth support and masticatory function, the PDL complex has been a primary target for periodontal tissue regeneration. Combining the root apical papilla and PDL-derived cells generates a root/periodontal complex that supports a porcelain crown and results in normal tooth function (Sonoyama et al. 2006). Reconstructing the epithelial and mesenchymal cells of the tooth germ, which mimics the multicellular assembly conditions in tooth morphogenesis, has also been found to result in whole-tooth regeneration, including that of the PDL complex (Ikeda et al. 2009). Applying the embryonic dental follicle tissue around a hydroxyapatite-coated titanium implant forms a cementum-like structure at the implant surface and establishes a PDL-like fibrous connection that is functionally comparable with that observed in natural teeth (Oshima et al. 2014). A titanium implant combined with PDL-derived cell sheets can induce cementum-like tissue on the titanium surface with perpendicularly oriented PDL-like tissue (Washio et al. 2018). Although successful regeneration of the PDL complex using stem cells has been demonstrated, cell-based periodontal tissue regeneration remains impractical because the differentiation kinetics of stem cells during this process are still poorly understood.
In this study, we analyzed periodontal tissue regeneration using subrenal capsule transplantation of immediately extracted molars from mice to understand the molecular mechanisms underlying periodontal tissue regeneration. Through bioinformatic analyses, we identified the association between the Wnt/β-catenin signaling pathway and periodontal tissue regeneration. Further, we investigated the in vivo cell behaviors and the impact of the Wnt/β-catenin signaling pathway in periodontal tissue regeneration upon subrenal capsule tooth transplantation.
Methods
Ethics Statement
The use of animals and all animal procedures were approved by the Niigata University Animal Experiment Ethics Committee (approval No. SA00532), and the experiments were performed in accordance with the Guidelines for the Proper Conduct of Animal Experiments specified by the Science Council of Japan. All animal handling and experiments were performed in accordance with the ARRIVE Guidelines for Animal Research Reporting of In Vivo Experiments.
Animals
Wild-type (WT) mice (C57BL/6J, male) were purchased from Charles River Laboratories (Yokohama, Japan). R26GRR mice (RBRC04874, C57BL/6N-Gt(ROSA)26Sor
Transplantation of Extracted Teeth into the Subrenal Capsule
The upper first molars were extracted from 6-wk-old mice and transplanted into the subrenal capsules of the same mice or their littermates. Details are described in the Supplementary Materials and Methods.
Preparation of Decellularized Teeth
The extracted molars were rinsed once with phosphate-buffered saline and treated with 2% sodium deoxycholate and 0.5% Triton-X100 at 4 °C for 3 d for decellularization (Ida et al. 2018; Son et al. 2019).
Histology and Immunohistochemistry
Histology and immunohistochemistry were performed as described previously (Mizukoshi et al. 2021; Thant et al. 2022; Iwama et al. 2024). Details are described in the Supplementary Materials and Methods.
Transplantation of Wnt/β-catenin Signaling-Modified Teeth
Two days and 1 day before transplantation, tamoxifen (T5648; Sigma-Aldrich) was injected intraperitoneally (75 mg/kg body weight) into the Ctnnb1CA/UBC-CreERT2 or Ctnnb1cKO/UBC-CreERT2 mice. The upper first molars of tamoxifen-injected mice were extracted and transplanted into the subrenal capsules of the control littermates. The transplanted tissue was harvested 1, 2, and 4 wk after transplantation.
Comprehensive Gene Expression Analysis and Bioinformatics
We have described the comprehensive gene expression analysis and bioinformatics in detail in the Supplementary Materials and Methods.
Histomorphometry and Statistical Analysis
Histomorphometric measurements were performed using Fiji/ImageJ (National Institutes of Health). Distal side of the interradicular septum was used as the region of interest. An unpaired t test with Welch correction or ordinary One-way analysis of variance was performed using GraphPad Prism 9 (GraphPad Software), and P values less than 0.05 were considered significant.
Results
Transplantation of Extracted Teeth into the Subrenal Capsule Results in Successful Periodontal Tissue Regeneration
To analyze periodontal tissue regeneration, we extracted the upper first molars from WT mice and transplanted them into their subrenal capsules of the same mice (Fig. 1A). Histological samples were prepared 1, 2, and 4 wk after transplantation (Fig. 1B). When the extracted teeth were histologically analyzed before transplantation, only a small fraction of the PDL was retained on the tooth side, whereas it was largely attached to the bone side (Supplementary Fig. S1). Four weeks posttransplantation, regeneration of the PDL complex was observed in the interradicular septum (Fig. 1C), displaying a morphology comparable with that of intact periodontal tissue (Cont). At high magnification, increased cellular cementum was evident, particularly in the apical region (Fig. 1D). Notably, the PDL-like fibrous tissue was maintained between the regenerated alveolar bone and cementum. High alkaline phosphate (ALP) activity was observed near the cementum surface 1 wk after transplantation (Fig. 1E). At weeks 2 and 4, high ALP activity was sustained in the PDL and interstitial space of the regenerated bone, and tartrate-resistant acid phosphatase (TRAP)–positive cells were present on the regenerated bone surface. Immunohistochemistry confirmed that cells embedded in the regenerated alveolar bone and cementum were positive for Dmp1, a well-characterized osteocyte/cementocyte marker (Fig. 1F). The PDL-like tissue between the cementum and bone was periostin positive, further confirming regeneration of the PDL complex (Fig. 1G). Picrosirius red staining of the regenerated PDL demonstrated that extrinsic fibers were evident in the superficial layer of the regenerated cellular cementum, although the organization of the collagenous matrix in the regenerated PDL was not as mature as that of the control (Fig. 1H). Immunological rejection of the transplanted teeth was not observed in the autologous transplantation experiment. The success rates of the transplantation experiments are summarized in Supplementary Figure S2. These results demonstrate that the transplantation of extracted mouse molars into the subrenal capsule successfully induces periodontal tissue regeneration.
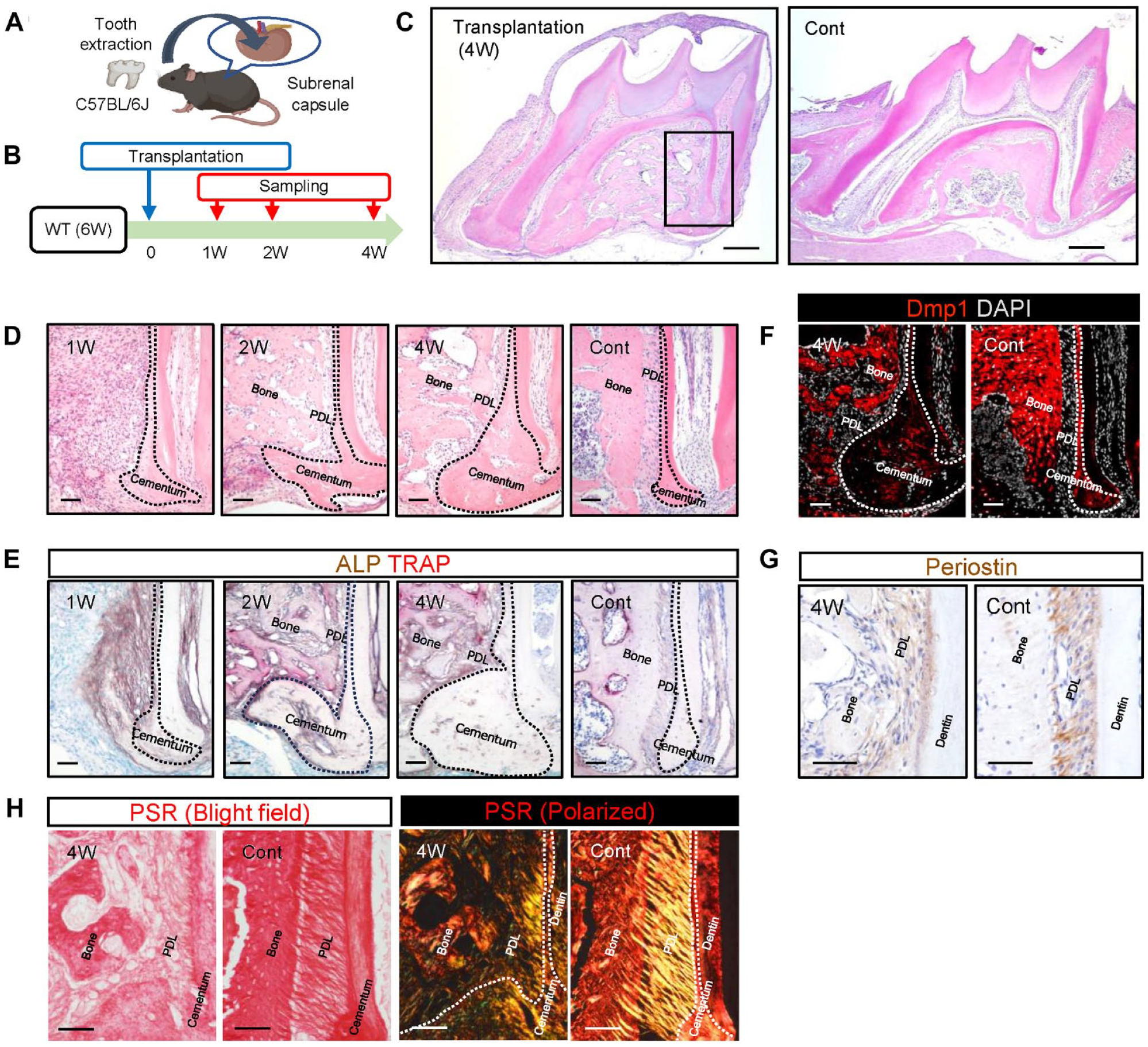
Transplantation of extracted teeth into the subrenal capsule of mice. (
Contribution of Transplanted Tooth–Derived Cells to Periodontal Tissue Regeneration
To analyze the origin of the cells contributing to periodontal tissue regeneration, the upper first molars of GFP-expressing mice were transplanted into the subrenal capsules of their WT littermates (Fig. 2A). Histological analysis 4 wk posttransplantation (Fig. 2B) revealed GFP-positive cells in the regenerated periodontal tissue (Fig. 2C). The proportions of GFP-positive cells in the PDL, bone, and cementum of the regenerated tissue were 90.3%, 82.8%, and 88.3%, respectively (Fig. 2D). Parts of the endomucin-positive endothelial cells were negative for GFP-positive cells, indicating that these cells originated from the host tissue (Fig. 2E). To confirm this observation, we transplanted extracted teeth from WT mice into GFP-expressing littermates. Only a small proportion of cells were GFP positive in the regenerated periodontal tissue, and most GFP-positive cells were endomucin-positive endothelial cells (Fig. 2F).
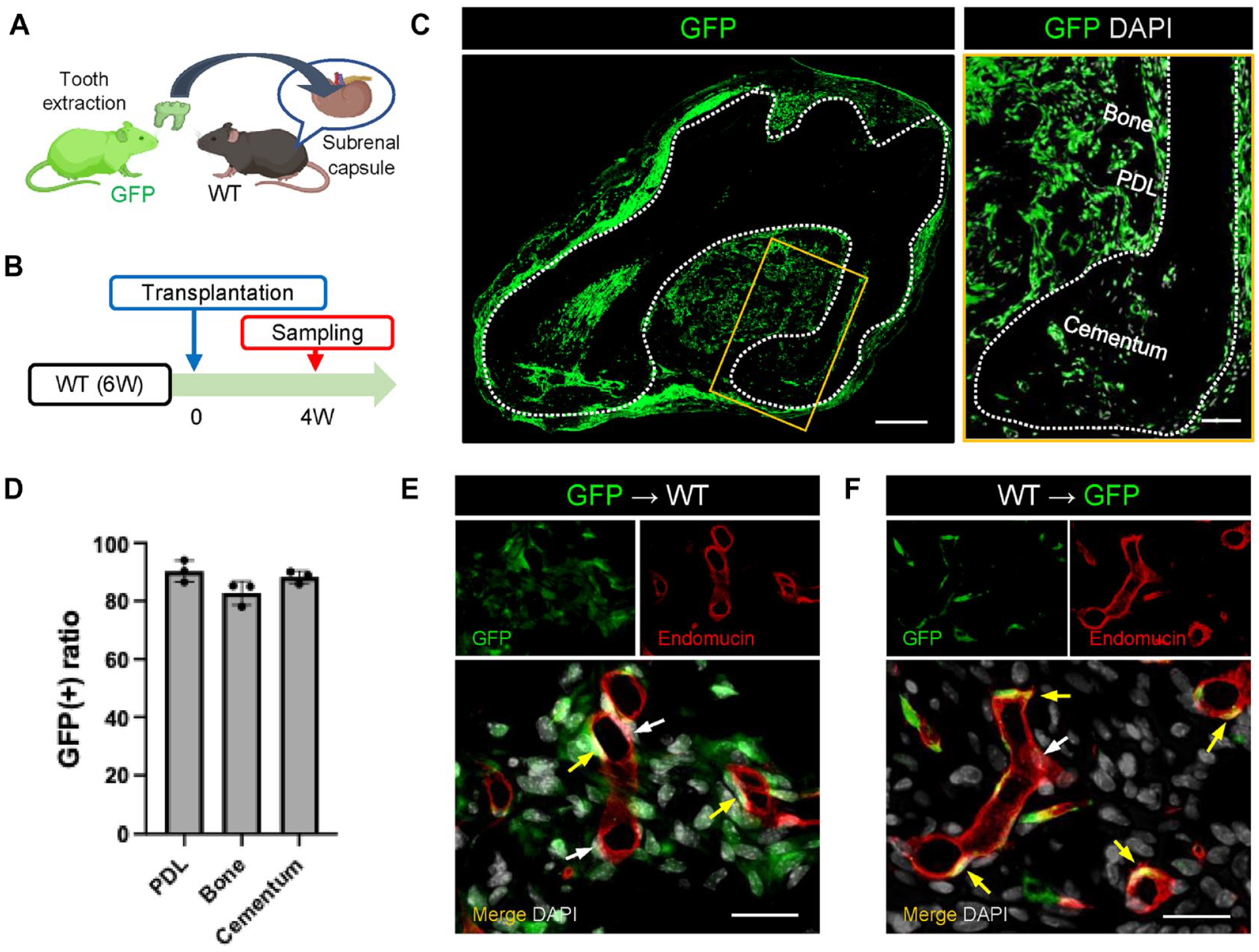
Fate of transplanted tooth–derived cells during periodontal tissue regeneration. (
Decellularization of Transplanted Teeth Interferes with Periodontal Tissue Regeneration
Next, to elucidate the requirement of cells from the transplanted teeth for periodontal tissue regeneration, we decellularized the extracted teeth, transplanted them into the subrenal capsule, and analyzed them after 4 wk (Fig. 3A, B). After decellularization, the complete removal of cells from the cementum surface PDL was confirmed (Fig. 3C). Periodontal tissue regeneration was not observed in decellularized teeth 4 wk after transplantation (Fig. 3D). Immunohistochemistry using an anti-DMP1-antibody confirmed that bone regeneration and apposition of the cellular cementum, which was remarkable when using teeth transplanted with cells (Fig. 1D), was not observed (Fig. 3E). ALP activity (Fig. 3F) and cells positive for osterix (Fig. 3G), which is an osteoblast/cementoblast differentiation marker, were not detected. We also observed that the periostin-positive PDL area did not regenerate (Fig. 3H). These results indicate that transplanted tooth cells are essential for periodontal tissue regeneration in the subrenal capsule model.
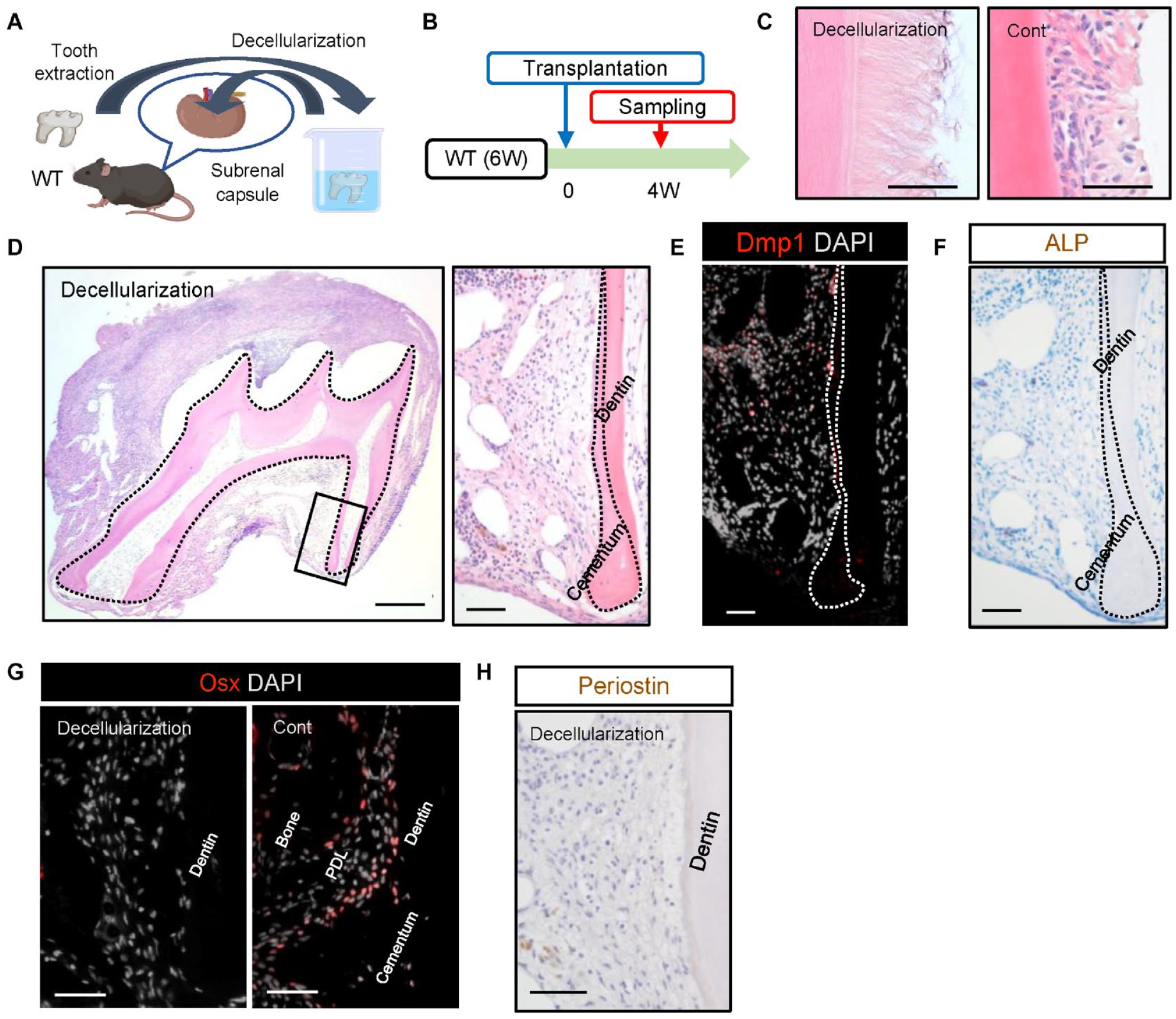
Effects of decellularization on periodontal tissue regeneration. (
Enrichment of Pathways Associated with the Regulation of Periodontal Tissue Regeneration
Comprehensive gene expression analysis was performed using a bioinformatics approach to identify the possible molecular mechanisms regulating periodontal tissue regeneration. Total RNA was isolated from the transplanted teeth at 1 and 2 wk after transplantation, when active tissue regeneration occurred (Fig. 1D). Total RNA isolated from immediately extracted molars was used as a control. Principal component analysis revealed that each sample group differed from the others (Supplementary Fig. S3a, b). The enriched terms associated with Wnt/β-catenin signaling were found to be the third significant in the comparison between the 2 wk and Cont groups (Wnt ligand biogenesis and trafficking; R-MMU-3238698, pAdj = −26.6) (Fig. 4A). The Wnt/β-catenin signaling was also found to be significant in the comparison between the 1 wk and Cont groups (Supplementary Fig. S3c). To confirm the activation of Wnt/β-catenin signaling during the regeneration of periodontal tissue, we performed immunohistochemistry using an anti-β-catenin antibody (Fig. 4B). At 1 wk, β-catenin–positive cells had spread near the cementum surface. At 2 and 4 wk, β-catenin–positive cells were found at the bone and cementum surfaces, with a stronger signal intensity than that in the control. These results strongly indicated that Wnt/β-catenin signaling is involved in the regeneration of periodontal tissue.
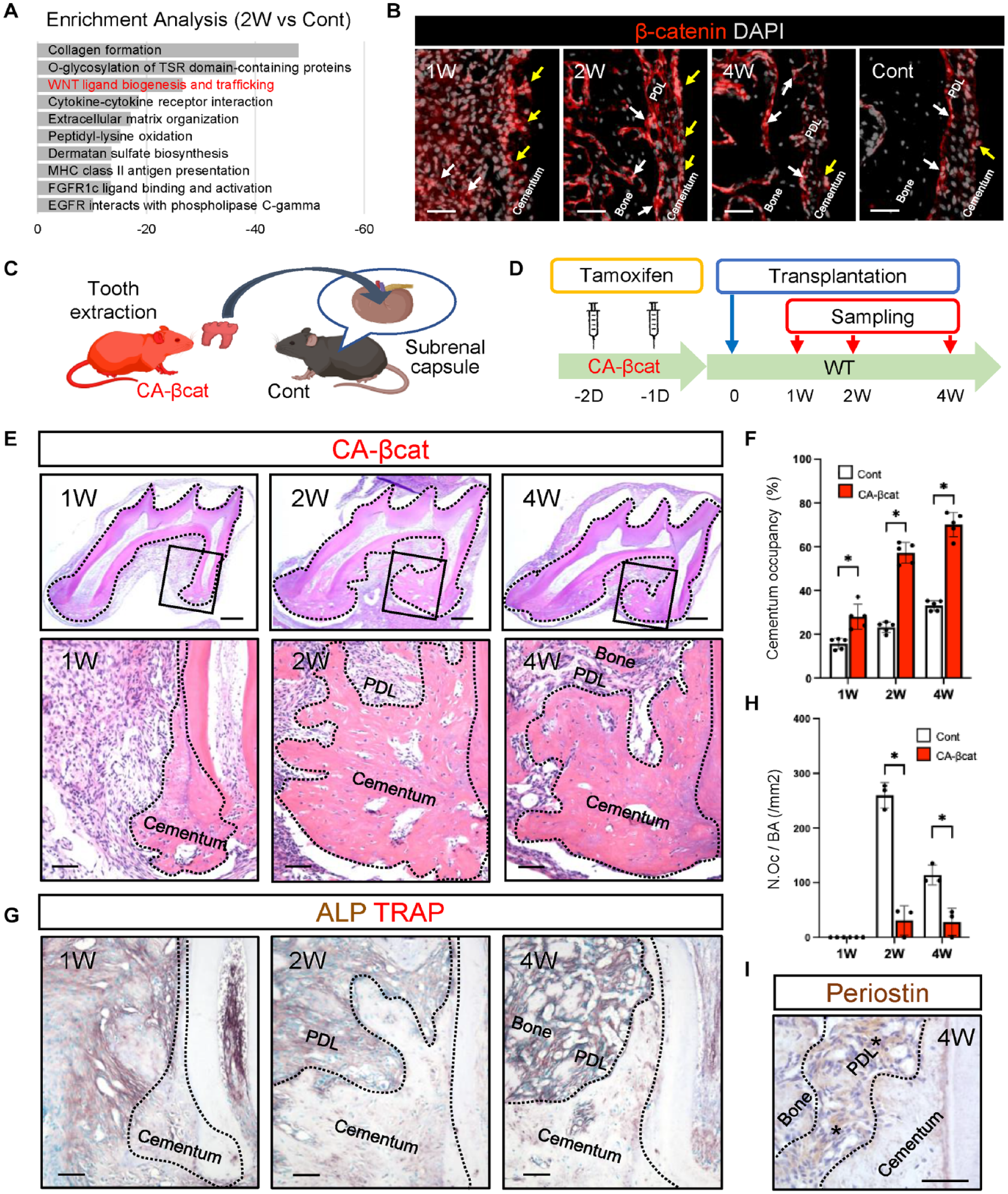
Effects of constitutively active β-catenin on periodontal tissue regeneration. (
Constitutive Activation of β-catenin in Cells of Transplanted Teeth Accelerates Cellular Cementum Apposition
To investigate the effect of Wnt/β-catenin signaling on periodontal tissue regeneration, we generated an inducible gain-of-function model using constitutively active β-catenin (CA-βcat)–expressing mice crossed with inducible Cre under the control of a ubiquitous promoter (Ctnnb1CA/UBC-CreERT2). The upper first molars of CA-βcat mice were extracted and transplanted into the subrenal capsule of control littermates (Fig. 4C). The CA-βcat was established via tamoxifen injection for 2 consecutive days before tooth transplantation (Fig. 4D). The enhancement of Wnt/β-catenin signaling on the transplanted tooth from CA-βcat mice was confirmed by the β-catenin immunostaining (Supplementary Fig. S4a). Marked apposition of the cellular cementum was observed as early as 1 wk after transplantation (Fig. 4E). The DMP1-positive cementum area on the transplanted teeth of CA-βcat mice was significantly larger than that in WT (Fig. 4F, Supplementary Fig. S4b). Conversely, the regenerated bone area decreased owing to the excessive formation of cellular cementum in the interradicular septum (Supplementary Fig. S4c). ALP activity was detected at the cementum and regenerated bone surface of CA-βcat transplanted teeth, similar to that in WT counterparts (Fig. 4G). However, at the surface of the regenerated bone, a smaller number of TRAP-positive osteoclasts was observed compared with the control (Fig. 4G, H). Immunohistochemistry showed that the PDL-like tissue on the cementum surface was periostin positive, confirming the establishment of PDL regeneration (Fig. 4I). These results suggest that the enhancement of Wnt/β-catenin signaling in the cells of transplanted teeth accelerated cellular cementum apposition without compromising the regeneration of the PDL complex.
Conditional Knockout of β-catenin in Cells of Transplanted Teeth Prevents Cellular Cementum Apposition
To further investigate the role of Wnt/β-catenin signaling in periodontal tissue regeneration, we generated an inducible loss-of-function model using conditional β-catenin-knockout (cKO-βcat) mice crossed with inducible Cre under the control of a ubiquitous promoter (Ctnnb1cKO/UBC-CreERT2). Tooth transplantation from the cKO-βcat mice was performed similarly to that for the CA-βcat mice (Fig. 5A, B). The decreased activity of Wnt/β-catenin signaling on the transplanted tooth from cKO-βcat mice was confirmed by β-catenin immunostaining (Supplementary Fig. S5a). While the PDL and alveolar bone regeneration occurred as seen in WT mice, apposition of the cellular cementum at the apical region was significantly smaller in the transplanted teeth of cKO-βcat mice (Fig. 5C, D, Supplementary Fig. S5b). High ALP activity was observed 1 and 2 wk after transplantation; however, it returned to a level comparable with that in the controls at 4 wk (Fig. 5E). At the surface of the regenerated bone, more TRAP-positive osteoclasts were observed in the transplanted teeth of cKO-βcat mice compared with that in the control mice (Fig. 5E, F). Immunohistochemistry revealed that periostin-positive PDL-like tissues were established on the cementum surface (Fig. 5G). These results suggest that the attenuation of Wnt/β-catenin signaling in the cells of transplanted teeth suppresses cementum apposition without affecting regeneration of the PDL complex.
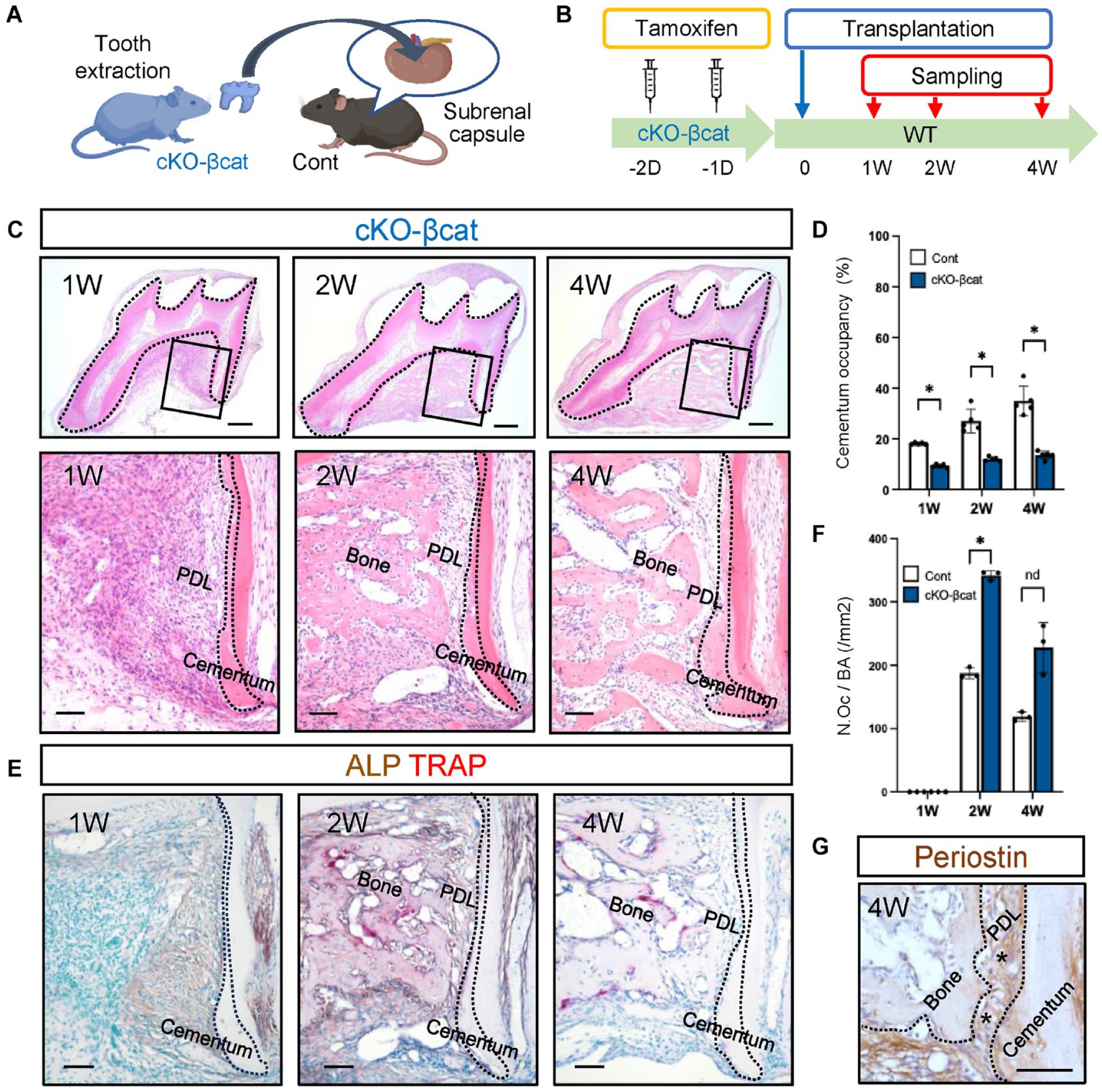
Effects of conditional knockout of β-catenin on periodontal tissue regeneration. (
Discussion
The regeneration of periodontal tissue, particularly the PDL complex, has long been challenging because of its complex and layered structure, which harbors various cell types. Several methods have been applied to analyze the process of periodontal tissue regeneration, but each has biological complications and limitations (Sculean et al. 2015), such as the occurrence of root resorption and ankylosis (Lee et al. 2020). It has been reported that subrenal transplantation of tooth germ has a superior regeneration capacity compared with subcutaneous transplantation (Higuchi et al. 2008). In this study, we used a rigorous mouse model of periodontal tissue regeneration, specifically the subrenal capsule transplantation of immediately extracted teeth. The regenerated periodontal tissue of the transplanted teeth was morphologically comparable with that of the intact periodontal tissue; thus, we analyzed the process of periodontal tissue regeneration in a well-established environment.
We conducted allogeneic tooth transplantation to evaluate the cell origin and achieve the transplanted cell-specific manipulation of Wnt/β-catenin signaling. Immunological complications are a concern in allogeneic tooth transplantation. Immunological rejection can occur after allogeneic tooth transplantation in outbred mouse strains but never occurs in inbred mouse strains, such as B6 mice (Mutoh et al. 2011) or between littermates (Takamori et al. 2008). Therefore, we performed allogeneic tooth transplantation using littermates of C57BL/6J mice and did not observe morphological disturbances or signs of immunological rejection during periodontal tissue regeneration after subrenal capsule tooth transplantation (Supplementary Fig. S2).
Our bioinformatics approach indicated that Wnt/β-catenin signaling is involved in periodontal tissue regeneration, and immunohistochemistry for β-catenin further confirmed this finding. Moreover, manipulation of Wnt/β-catenin signaling affects the cellular cementum apposition. In agreement with our observation, the CA of β-catenin in Wnt-responsive Gli1- or Axin2-lineage cells results in cementum hyperplasia (Xie et al. 2019; Xie et al. 2021). Conversely, the cKO of β-catenin or depletion of these cells attenuates cementum apposition (Xie et al. 2021; Ma et al. 2023). Therefore, Wnt/β-catenin signaling is most likely a common pathway promoting cementogenesis during the development, remodeling, and regeneration processes.
Our immunohistochemistry for β-catenin indicated that Wnt/β-catenin signaling was enhanced both at the cementum and bone surface during periodontal tissue regeneration. However, in the subrenal transplantation of extracted teeth from cKO mice, cellular cementum apposition was completely inhibited, whereas bone regeneration was not impaired with an increased number of osteoclasts. These results are consistent with the manipulation of Wnt/β-catenin signaling affecting the remodeling stage rather than the developmental stage in bone (Glass et al. 2005; Lim et al. 2014; Wu et al. 2019). This indicates that the Wnt/β-catenin signaling does not significantly affect the bone-forming property of osteoblasts; it affects the osteoclast activity through the RANK/RANKL/OPG axis (Lim et al. 2014).
The expression of CA-βcat in Dmp1-expressing osteocytes and cementocytes results in excess cellular cementum and alveolar bone, eventually leading to ankylosis, subjecting teeth to occlusal force (Wu et al. 2019). In contrast, in our study, in which CA-βcat expression was induced in all the cells of transplanted teeth, the periostin-positive PDL layer was maintained at the surface of the transplanted teeth, and ankylosis did not occur. This may be because the transplanted teeth in the subrenal capsule were free of occlusal forces. Conversely, the fiber organization of the regenerated PDL in the subrenal capsule is less mature than that in the intact PDL, indicating the necessity of occlusal loading for proper matrix organization in the PDL (Kaku and Yamauchi 2014; Kaku et al. 2016). Taken together, these results highlight that temporal management of occlusal force along with Wnt/β-catenin signaling is crucial for successfully regenerating the PDL complex in a clinical setting.
Our cell tracing analysis revealed that most cells in the regenerated periodontal tissue were derived from transplanted teeth and that the transplantation of decellularized teeth completely interfered with periodontal tissue regeneration, indicating the requirement for endogenous PDL cells in transplants in our experimental setting. This suggests the unique characteristics of cells in the PDL, particularly PDL stem cells (Seo et al. 2004; Bartold and Gronthos 2017). Although endogenous cells from the transplanted teeth were essential for periodontal regeneration in our subrenal transplantation model, circulating bone marrow–derived cells may colonize the regeneration milieu under certain conditions (Tomida et al. 2013; Kimuraet al. 2014; Kaku et al. 2017). Our data demonstrated that some of the endothelial cells in the regenerated tissue originated from the host tissue and were recruited via the bloodstream. Supplementation with stem cells or management of the extracellular environment can help achieve periodontal tissue regeneration on the surface of decellularized teeth, which can be considered a severely damaged tooth root analog in a clinical setting (Kaku et al. 2015).
The cementum apposition in the transplanted teeth and affected by the manipulation of Wnt/β-catenin signaling in this study specifically refers to the cellular cementum. The cellular cementum of the apical region in rodents has been a concern because of its differences from the human cementum; however, we recently reported that extrinsic fibers are invariably present at the surface of the apical cementum in mice, indicating that the cementum of the apical region contributes to tooth support, similar to humans (Iwama et al. 2024). Consistent with this observation, we found extrinsic fibers in the superficial layer of the regenerated cellular cementum. Although further assessments are required, the cementum apposition controlled by the Wnt/β-catenin signaling can apply to human cementum regeneration.
To elucidate the detailed mechanism of periodontal tissue regeneration, we analyzed cell behavior using a novel periodontal tissue regeneration model. Our findings revealed that cells of the transplanted teeth are crucial for periodontal tissue regeneration, whereas endothelial cells are partly recruited from the host tissue. Bioinformatics analysis of regenerated tissue indicated the association with Wnt/β-catenin signaling in the periodontal tissue regeneration process. Furthermore, the manipulation of Wnt/β-catenin signaling in the cells of transplanted teeth affects cellular cementum apposition without affecting the regeneration of the PDL complex. Our results demonstrate that Wnt/β-catenin signaling is involved in cellular cementum apposition, and controlling this pathway could promote cementum regeneration, a critical process for periodontal tissue regeneration. This study provides novel insights into cell behavior and signaling pathways that will advance practical periodontal tissue regeneration.
Author Contributions
Y. Ono, M. Kaku, conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; L. Thant, H. Iwama, data acquisition and analysis, critically revised the manuscript; M. Arai, M. Mizukoshi, A. Dobashi, M. Kitami, M.M. Taketo, data analysis, critically revised the manuscript; A. Ohazama, I. Saito, K. Uoshima, data interpretation, critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241286490 – Supplemental material for Wnt/β-catenin Promotes Cementum Apposition in Periodontal Regeneration
Supplemental material, sj-docx-1-jdr-10.1177_00220345241286490 for Wnt/β-catenin Promotes Cementum Apposition in Periodontal Regeneration by Y. Ono, M. Kaku, L. Thant, H. Iwama, M. Arai, M. Mizukoshi, A. Dobashi, M. Kitami, M.M. Taketo, A. Ohazama, I. Saito and K. Uoshima in Journal of Dental Research
Footnotes
Acknowledgements
We acknowledge that part of the illustration in this work was created with the assistance of BioRender.com.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI (grant No. JP21H03127, JP21K19895, and JP24K02630 to M.K).
Data Availability Statement
The datasets generated and analyzed in the current study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
