Abstract
Childhood is considered crucial in the establishment of future oral microbiota. However, the precise period of oral microbiota development remains unclear. This study aimed to identify the progression of oral microbiota formation in children. We longitudinally investigated the salivary microbiota of 54 children across 13 time points from 1 wk to 60 mo (5 y) old and their parents at 2 time points as a representative sample of the adult microbiota. Using next-generation sequencing, we obtained 10,000 gene sequences of the 16s rRNA V1-V2 region for each sample. The detection rate in children of 110 operational taxonomic units commonly detected in more than 85% of mothers and fathers, defined as the main constituent bacteria, was 25% at 1 wk old, increased to 80% between 6 and 18 mo old, and reached approximately 90% by 36 mo old. Early main constituent bacteria detected at 1 wk old were limited to Streptococcus, Rothia, and Gemella. At 6 to 18 mo old, the detection rates of various main constituent bacteria, including Neisseria, Haemophilus, and Fusobacterium, increased. UniFrac distance analysis showed that the oral microbiota of children approached that of adults at 6 to 18 mo old. In the weighted UniFrac distance index, unlike the unweighted index, there were no significant changes in children between 36 and 60 mo old from adults, and microbiota formation at 60 mo old was sufficiently advanced to be included within the range of adult individual differences. Our findings suggest that the initial 36 mo, particularly the period from 6 to 18 mo old, consists of a time window for oral microbiota maturation. In addition, the development of microbiota during this period may be critical for future oral disease prevention.
Introduction
Recent studies have shed light on the critical role of the oral microbiota in the onset and progression of oral diseases, such as dental caries and periodontal disease, and its implications for systemic health and diseases (Kilian et al. 2016; Gao et al. 2018; Lamont et al. 2018). The early microbial community is believed to contribute to the establishment and maturation of the oral microbiota (Sampaio-Maia and Monteiro-Silva 2014), emphasizing the importance of understanding its postnatal formation process.
After birth, the newborn’s oral cavity is exposed to various microorganisms, with the oral microbiota developing as the newborn grows (Dzidic et al. 2018; Kahharova et al. 2020). Tooth eruption and dietary changes are considered primary influencers of oral cavity alterations during this period (Mason et al. 2018; Sulyanto et al. 2019; Könönen 2000; Xiao et al. 2020). Infant saliva and tongue microbiota resemble those of adults by about 2 y of age. (Kageyama et al. 2019; Kennedy et al. 2019). However, there remains a research gap regarding investigations spanning from 6 mo (when teeth erupt and solid foods are introduced) to approximately 36 mo (when all primary teeth have erupted).
The human oral microbiota comprises numerous bacterial species (Aas et al. 2005), with major species commonly shared among individuals, constituting a significantly high proportion of oral microbiota (Huse et al. 2012; Li et al. 2013; De Filippis et al. 2014; Takeshita et al. 2016). Understanding the colonization timeline of these major constituent bacteria in the oral microbiota is crucial to comprehend the foundational processes shaping future oral microbiota. Evidence suggests that differences in the proportion of these major bacteria are associated with the absence of decayed teeth and gingival bleeding (Takeshita et al. 2016), underscoring the potential importance of the timing of their colonization in influencing the future state of the oral microbiota.
This study aimed to identify the progression of oral microbiota formation in children by investigating periods when the main constituent bacteria of the oral microbiota become detectable and when the composition of the oral microbiota resembles that of adults.
Patients and Methods
Participants
This study included 54 children (27 boys and 27 girls, born between June 2015 and January 2017) and their parents (mean ± SD age at birth of the children, father: 33.0 ± 4.5 y; mother: 31.6 ± 3.7 y). The study was performed in accordance with the Declaration of Helsinki and the Ethical Guidelines for Epidemiological Research and was approved by the Ethics Committee of the Japanese Society for Oral Health, Tokyo, Japan (issuing No. 26-5). Participants were employees of Lion Corporation or family members of employees. All parents of participants provided written informed consent.
Sample Collection
Saliva samples from children were collected at various time points: 1 wk, 1, 3, 6, 9, 12, 18, 24, 30, 36, 42, 48, and 60 mo using SalivaBio Infant’s Swab (Salimetrics). In addition, mouth-rinsed water was collected from children at 60 mo. Mouth-rinsed water was collected by vigorously rinsing the mouth with 3 mL sterile water (Otsuka Distilled Water, Otsuka Pharmaceutical Co.) for 10 s. Previous reports have shown that the microbiota of saliva collected by mouth-rinsed water can be considered similar to that of stimulated or resting saliva (Jo et al. 2019). Parents’ samples were collected through mouth-rinsed water when their child was 18 and 36 mo old as representative adult samples. Participants were instructed not to brush their teeth after their last meal until collection and were prohibited from eating or drinking for at least 1 h precollection and from taking antibiotics within 24 h precollection. Samples were stored in Styrofoam boxes with an ice pack and were processed within 30 h. Sample swabs were immersed in sterile physiological saline (Otsuka Normal Saline, Otsuka Pharmaceutical Co.) and vortexed to disperse saliva. Subsequently, the sample was centrifuged at 16,400×g for 5 min to pellet the bacteria. Mouth-rinsed water was centrifuged at 16,400×g for 5 min to collect bacteria. The resulting bacterial pellet was stored at −80 °C until DNA extraction.
Generation of Microbiota Data
Detailed methods for DNA extraction, polymerase chain reaction, and sequencing are provided in the Appendix Methods. The 16S rRNA gene V1-V2 region was sequenced using universal primers (27Fmod and 338R) as previously described (Jo et al. 2021; Yama et al. 2023). From the obtained sequences, 10,000 reads per sample were randomly selected to create operational classification units (OTUs) with an identity threshold of 97% after excluding sequences with an average quality value <25, sequences without primer sequences at both ends, and sequences considered chimeric. Similarity searches were conducted for each OTU in 4 public databases: RDP, CORE, NCBI FTP site, and HOMD. Details are described in the Appendix Methods.
Participant Information Collection
Parents completed a comprehensive questionnaire on diet and oral health while saliva was collected from their children; data included baby food initiation and powdered milk consumption up to 6 mo old and erupted teeth count up to 42 mo old. In addition, the number of children diagnosed with dental caries among those who visited the dental clinic (Appendix Table 1), the number of parents who had subjective symptoms and dental caries among those who visited the dental clinic (Appendix Tables 2 and 3), and the number of subjects who took antibiotics in the period prior to each sampling were determined (Appendix Tables 4–6).
Data Analysis
To compare microbiota, we calculated the UniFrac distance (Ihaka and Gentleman 1996) with OTUs and their representative sequences created with 97% homology using 10,000 reads per sample. The Steel–Dwass test was performed using NSM3 package version 1.16 in R software version 4.04 (Lozupone and Knight 2005). P < 0.05 was considered significant.
Reporting Guidelines
The study was conducted according to STROBE guidelines, ensuring adherence to established standards in research methodology and reporting practices.
Results
Extraction of the Main Constituent Bacteria Forming Oral Microbiota
We obtained high-quality gene sequences of 10,000 reads for each sample, except for five 1-wk-old participants, and gene sequence data for bacteria constituting the oral microbiota. The results of the negative control sample measurements, shown in Appendix Results 1, confirmed that the data were properly acquired. Based on gene sequence data, we determined that genes sharing 97% similarity in sequence indicated the same bacterial species. Subsequently, sequences exhibiting 97% similarity were grouped as an OTU. Data from parents were considered representative of the adult population. OTUs consistently detected in >85% of maternal and paternal samples across 2 time points were identified as highly prevalent oral bacteria (HPOB), signifying primary adult oral microbiota constituents. Appendix Table 7 lists HPOB, encompassing 110 OTUs spanning 6 phyla. Collectively, all HPOB contribute approximately 60% of the total abundance rate in the parents (Fig. 1).
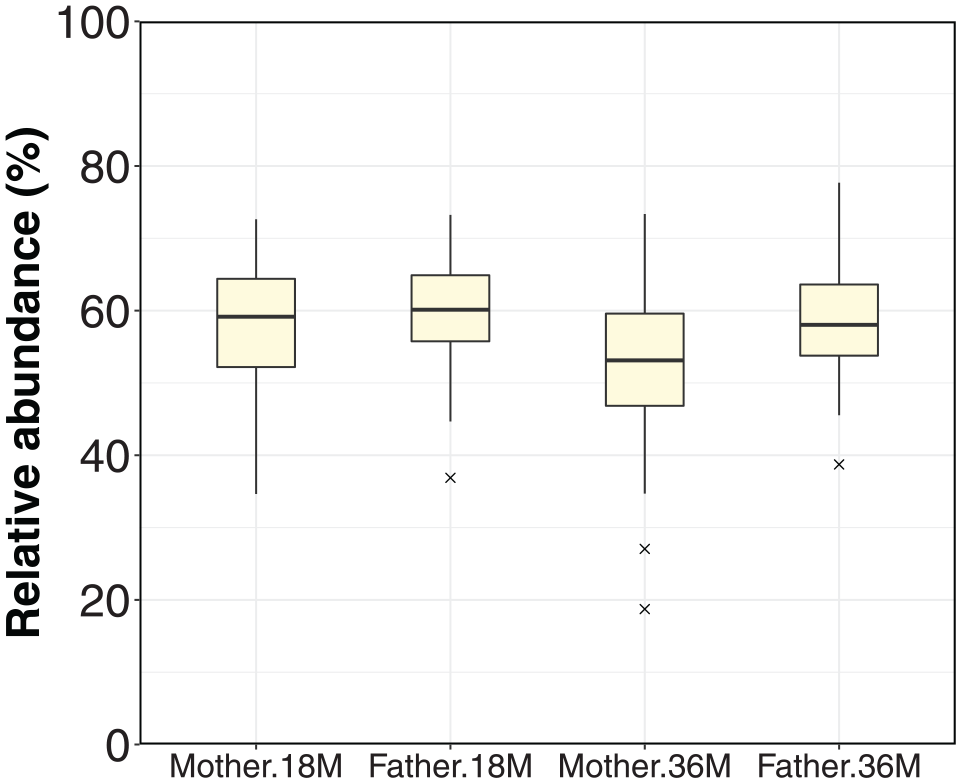
Rate of highly prevalent oral bacteria (HPOB) in the composition of the salivary microbiota of mothers and fathers. “18M” and “36M” indicate samples collected when their children were 18 and 36 mo old, respectively. HPOB, highly prevalent oral bacteria.
Trends in HPOB Detection Rate at Each Age
To elucidate the timeline of HPOB detection, we performed a trend analysis of the proportion of 110 identified HPOBs in children each week/month old. Figure 2A illustrates the dynamics of HPOB detection rates alongside the number of erupted teeth. Initially, the detection rate in child saliva collected with a swab was 25% at 1 wk old, with a notable increase at approximately 6 mo old, coinciding with the onset of tooth eruption, reaching approximately 80% at 18 mo old before all teeth erupted and peaking at 90% at 36 mo old, when all teeth erupted. To explore the relationship between HPOB detection rates, timing of baby food initiation, and number of erupted teeth at 6 mo old, a scatter plot was generated (Fig. 2B). Three children were confirmed to have an HPOB detection rate of greater than 50%, even though they had not yet started weaning and their teeth had not yet erupted.
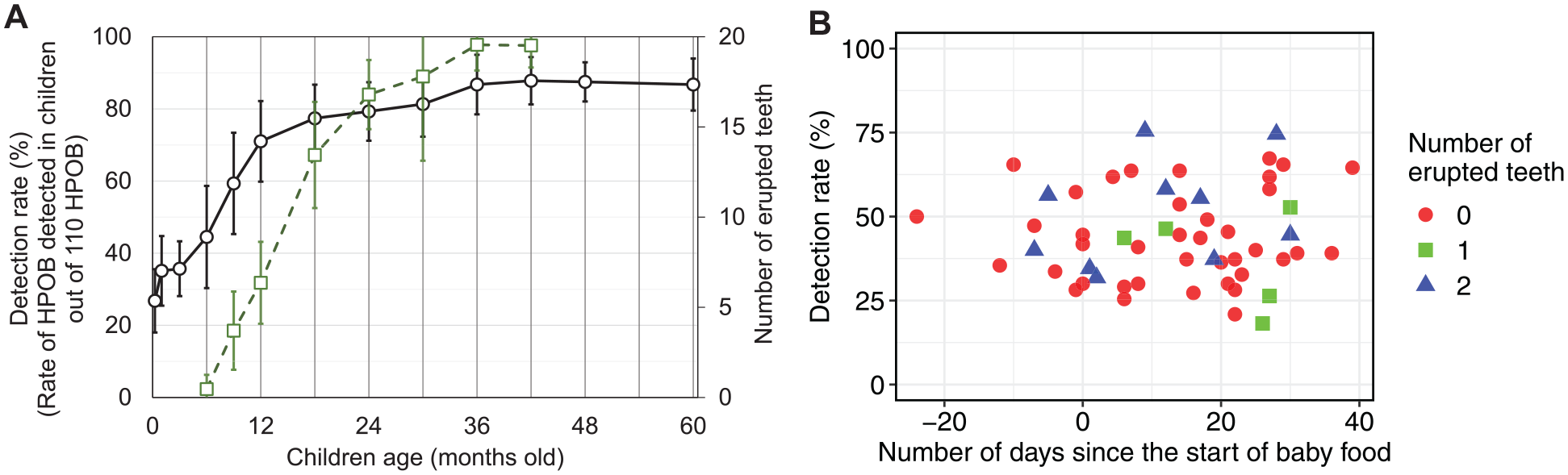
(
Trends in HPOB Detection Rate
To investigate differences in detection timing by bacterial species, Figure 3 illustrates shifts in detection rates among children for each HPOB. Certain facultative anaerobic bacteria, such as Rothia mucilaginosa, Gemella haemolysans, Gemella morbillorum, and Streptococcus spp., exhibited high detection rates shortly after birth, at 1 wk old. Furthermore, an increase in the detection rate was confirmed in children for other HPOB at approximately 6 mo old. Among these, a substantial number exhibited detection rates near 100% from birth to 18 mo, including nitrate-reducing bacteria, such as the Neisseria flavescens, Haemophilus parainfluenzae, and Fusobacterium nucleatum. However, some HPOBs showed a slower increase in detection rates.
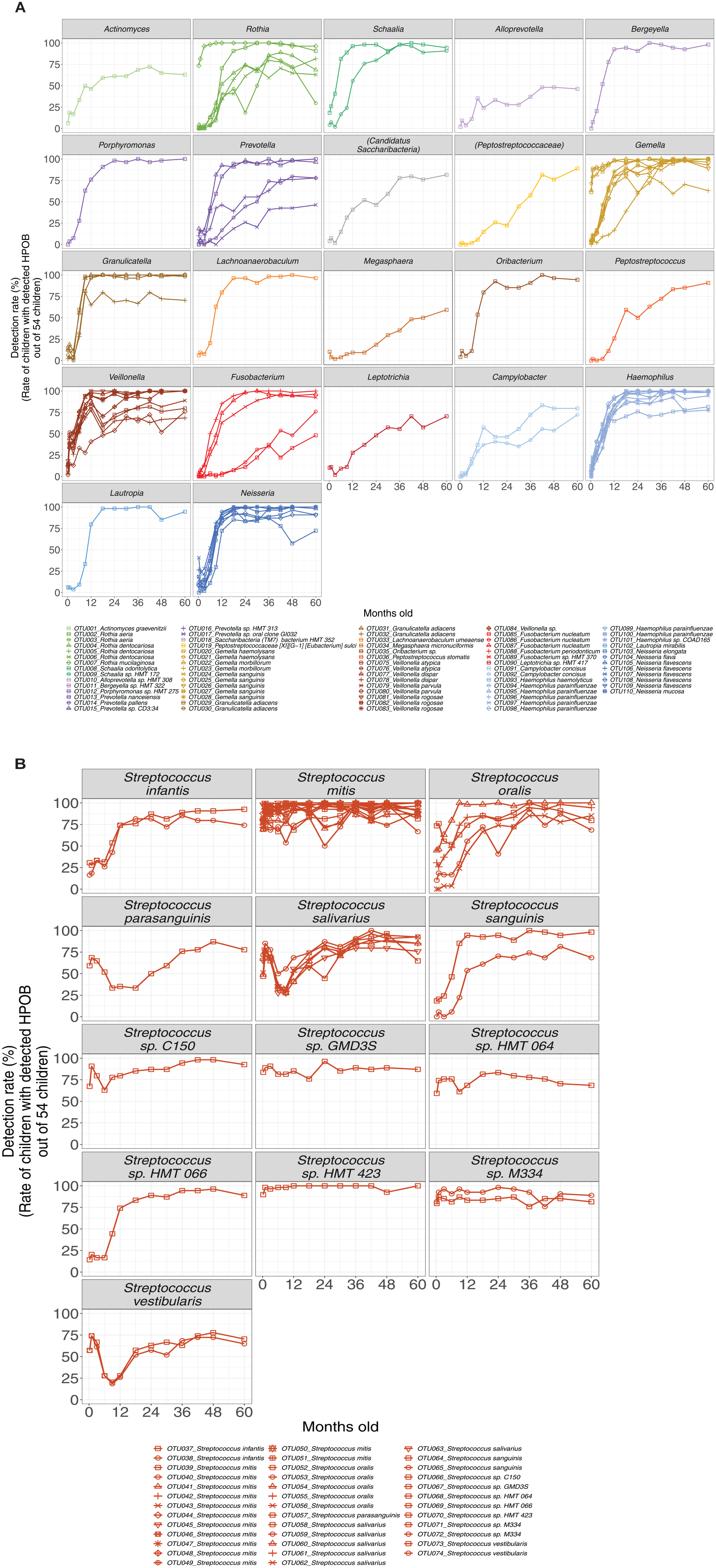
Line graphs showing the changes in each HPOB on the trend in the rate of children with detected HPOB. The horizontal axis represents the child’s age in months, and the vertical axis displays the detection rate. The legend provides the OTU number and the name of the assigned bacterial species. (
Trends in Similarities in Oral Microbiota between Adults and Children
We investigated the period when the microbiota composition approaches that in adults. We used the UniFrac distance index, a measure of microbiota dissimilarity (Lozupone and Knight 2005), to evaluate the degree of dissimilarity between children’s and adults’ microbiota. The parents’ microbiota was used as a representative sample of the adult microbiota; we plotted the change in UniFrac distance between microbiota from each week-/month-old children and 216 adult data (54 pairs of parents including kinship × two time points; Fig. 4A, B). We also conducted a significant difference test (Steel–Dwass test) between each time point for each index. Our analysis of changes in the weighted UniFrac distance revealed substantial fluctuations from 1 wk to 18 mo old. Steel–Dwass test results indicated a significant difference (P < 0.01) in comparisons from 1 wk to 18 mo old, with trends differing from 18 mo onward, including no significant difference observed between 18- and 30-mo-olds; also, no significant changes were observed between 36- and 60-mo-olds. Exploring shifts in unweighted UniFrac distance, we observed an increase in dissimilarity between adults and 30- and 36-mo-olds, but thereafter, it significantly approached adult levels with age. To further gauge the proximity of the children’s microbiota to that of adults during this period, we compared UniFrac distances between adults and between 60-mo-old children and adults using the mouth-rinsed collection method to eliminate the influence of the saliva-collection method. Figure 4C and D present the UniFrac distance comparison; the weighted UniFrac distance index was significantly closer between 60-mo-old children and adults compared with between adults’ microbiota (adult individual differences). Conversely, the unweighted UniFrac distance index was significantly closer in adult individual differences than between 60-mo-old children and adults.
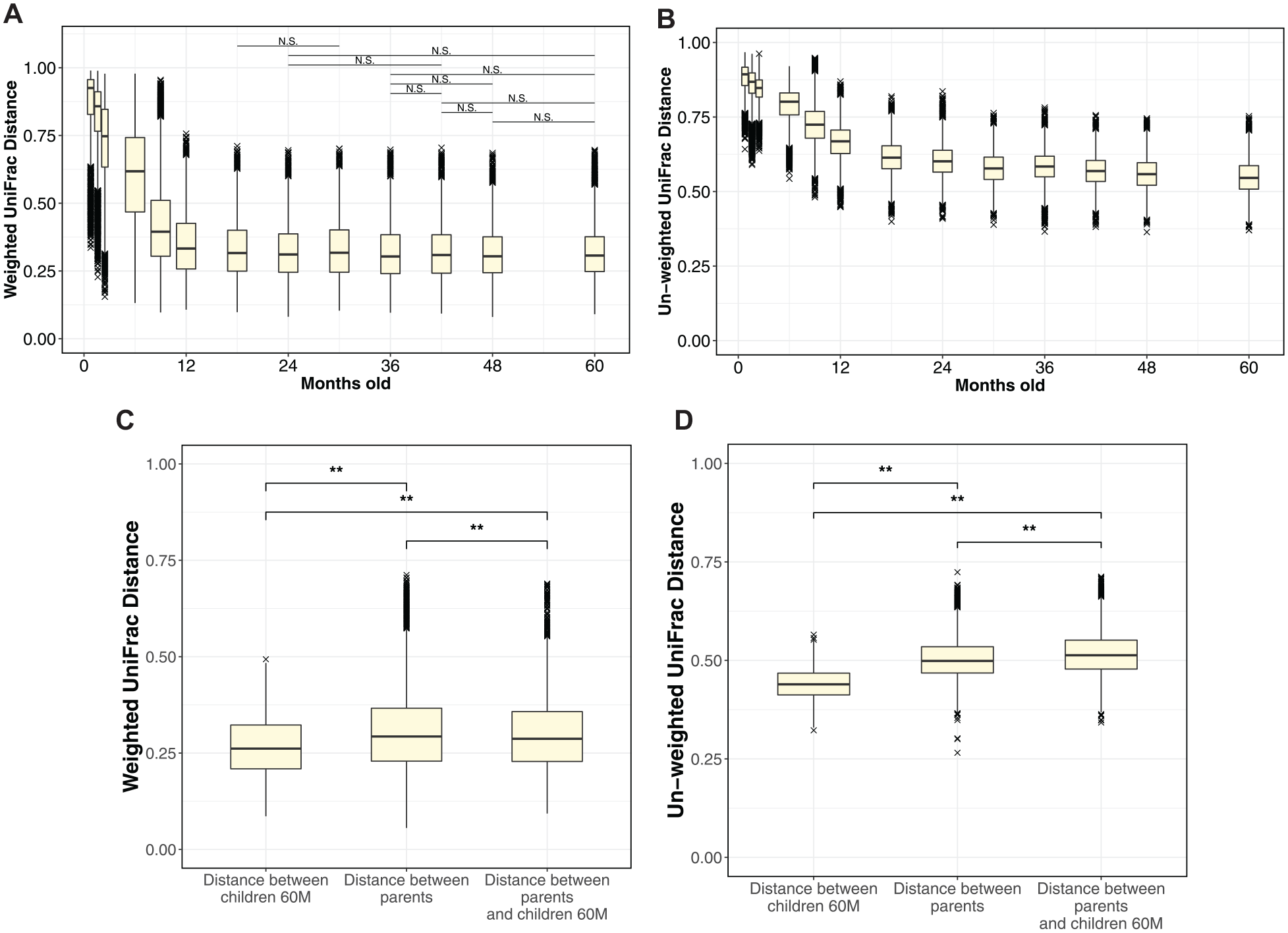
(
Discussion
In this study of oral microbiota spanning 13 time points from 1 wk to 60 mo old—specifically focusing on the critical period from 6 to 36 mo old when baby teeth erupt—we elucidated the detection timeline of key oral microbiota constituents and when their composition approximates that of adult microbiota. Key oral microbiota components assigned to various bacterial species began to be detected at about 6 mo old, with 80% detected by 18 mo old. An analysis of UniFrac distance showed that the microbiota resembled that of adults by 18 mo old. In addition, the HPOB detection rate reached 90% at 36 mo old, and no significant changes were observed in the weighted UniFrac distance between 36- and 60-mo-olds and adults. These results underscore the pivotal role of the initial 36 mo, particularly from 6 to 18 mo old, in the dynamic formation of oral microbiota. Herein, we discuss factors driving HPOB detection rates from 6 mo old, explore the significance of this period for bacterial types with increased detection rates, and assess microbiota formation during this critical phase for future oral-bacteria development. Furthermore, we note the limitations and conclusions of these findings.
At 6 mo old, the detection rate of HPOB begins to increase, indicating the tooth eruption period (Fig. 2A). Previous research has suggested that the appearance of new bacterial attachment sites, such as teeth, contributes to new bacterial colonization (Mason et al. 2018; Könönen 2000); in this study, bacterial species exhibiting high detection rates before 6 mo old predominantly comprise bacteria concurrently identified in the oral mucosa, suggesting that tooth eruption contributes to the colonization of diverse bacterial species. Simultaneously, the 6-mo mark aligns with the introduction of solid foods; prior research indicates that the ingestion of new nutrients through complementary feeding can influence microbiota diversity (Sulyanto et al. 2019). However, our study identified 3 exceptional children in whom HPOB detection rates exceeded 50% (50%, 57%, and 65%, respectively) despite the absence of erupted teeth and commencement of solid food intake at 6 mo old (Fig. 2B). Our assessment of the degree of bacterial colonization of the oral microbiota, considering the first 6 mo of life as an important period and focusing on HPOB, indicated that factors beyond tooth eruption and new nutritional intake are involved in oral microbiota formation. A previous report suggested that infants fed formula milk, compared with breast milk, exhibit earlier oral microbiota maturation (Kaan and Zaura 2022). However, parental questionnaires confirmed that the 3 cases predominantly relied on breastfeeding, with low percentages of formula milk in their total milk intake (3%, 6%, and 15%, respectively). The questionnaire confirmed that these 3 children did not take antibiotics until 6 mo old. The 6-mo postnatal period represents a transition phase for immune function, characterized by a decline in maternally derived IgG concentration and the establishment of the child’s independent immune defenses (Braibant and Barin 2013), indicating that other factors, such as changes in immune function, may contribute to the colonization of foreign bacteria.
From about 6 mo old (Fig. 3), HPOBs assigned to nitrate-reducing bacteria of the genera Neisseria and Haemophilus, which are effective in preventing dental caries and periodontal disease (Doel et al. 2004; Rosier et al. 2022), showed increased detection. In addition, F. nucleatum, which is associated with periodontal disease and bad breath (Chen et al. 2022), colonized the oral cavity during the initial 18 mo. Previous studies have reported that the proportion of these nitrate-reducing bacteria and F. nucleatum in the oral microbiota differed between the healthy oral cavity group and the dental caries/periodontal disease group (Shi et al. 2021; Chen et al. 2022; Rosier et al. 2022; Yama et al. 2023). Since bacteria assume prominence in the oral microbiota during this developmental window, our findings could indicate its critical relevance to the future risk of oral diseases. Strategic oral care interventions and guidance during this period could shape oral microbiota in the future, mirroring the composition observed in individuals with normal oral health.
Advancement in oral microbiota formation was notably evident not only in HPOB-focused analyses but also in UniFrac distance index assessments. The unweighted UniFrac distance index illustrated a continuous convergence of children’s microbiota toward that of adults from 36 mo onward (Fig. 4B). This unweighted distance index, uninfluenced by the abundance of individual bacteria, indicates that there are still differences between 60-mo-old children and adults (Fig. 4D). These findings, along with the fact that observed OTUs in children at 60 mo old were lower than those in adults (Appendix Fig. 3), suggest the possibility that bacterial species with a low abundance (low contribution based on weighted distance index) among those that constitute the oral microbiota become the constituent bacteria of the oral microbiota even after 60 mo old. Meanwhile, no significant changes from 36 mo to 60 mo old were observed in the weighted distance index (Fig. 4A), and the distance between children and adults narrowed considerably when the children were 60 mo old (Fig. 4C). This suggests that even in terms of the weighted UniFrac distance index, the period up to 36 mo old is crucial in establishing the foundation for the microbiota.
This study has 3 main limitations. First, we used swabs to collect samples for the trend analysis in children, which differs from the method used for adults. Previous studies suggested that the detection results and relative abundance of certain bacteria are different when using swab-collected microbiota data compared with data derived from at-rest saliva (Kaan et al. 2022). This study also observed that the OTUs of the microbiota collected by swabs were lower than those of the microbiota collected by mouth rinsing (Appendix Fig. 4). Nevertheless, all HPOBs that were the focus of this study were detected from microbiota data collected by swabs. Detection outcomes of HPOB in both swab-collected and mouth rinsed–collected microbiota were nearly identical (90%, Appendix Fig. 5) for the same 60-mo-old child. This suggests that the variance in collection methods had minimal impact on the investigation of HPOB detection rates in our study, ensuring the accuracy of the detection rate trend analysis. In addition, the methodological distinction for the UniFrac distance was confirmed to have a substantially smaller effect than temporal changes within the same individual (Appendix Fig. 6). The difference in the collection method had a negligible impact on the analyses using the UniFrac distance, signifying the precision of the microbiota trend analysis. Second, the subjects’ history of antibiotic intake and oral health conditions was not considered. Since significant changes in oral microbiota due to antibiotics are not identified after 2 mo (Zaura et al. 2015) and the oral microbiota is dependent on oral health conditions (Yama et al. 2023), sampling should ideally be performed at least 2 mo after antibiotic administration and account for oral conditions. However, we could not adhere to this requirement as samples were collected from children at short intervals after they were born. Although antibiotic use within 2 mo of sampling was identified (Appendix Tables 4–6), we confirmed that most subjects did not take antibiotics within 2 mo of sampling, and similar results were obtained when the analysis was repeated excluding those who had taken antibiotics in the past 2 wk (Appendix Figs. 7–10). In addition, the results of the diagnostic information on oral conditions (Appendix Tables 1–3) confirmed that most subjects did not have oral health conditions. Notably, our results capture the average characteristics of the 54 children, which suggests that the effects of antibiotics and oral health conditions are masked. Third, the study focused on Japanese children. However, we believe that our results capture a general trend, as similar trends have been observed elsewhere, including a Swedish study in which many bacterial genera, such as Neisseria, became established after the age of 1 to 2 y (Dzidic et al. 2018) and an American study in which microbial diversity and caregiver similarity increased most between 1 and 2.5 y (Kahharova et al. 2020).
Our findings suggest the importance of oral hygiene from the early stages of tooth eruption from the perspective establishing oral microbiota. As the microbiota of 36-mo-olds reaches adult levels in the weighted UniFrac index, it is reasonable to consider it as adult microbiota, especially after all baby teeth have erupted. Furthermore, F. nucleatum, which participates in human teeth plaque formation and plays a central role as a physical bridge mediating bacterial coaggregation (Kolenbrander 2000; Chen et al. 2022), is an oral-microbiota component in 6 to 18 mo old, suggesting that plaque accumulation should be prevented by proper oral hygiene before 36 mo old when the infant diet differs from an adult diet and teeth have not yet erupted.
In conclusion, most of the main constituent bacteria in the oral microbiota colonized between 6 and 36 mo old, emphasizing the importance of the 36 mo following birth as a pivotal period to establish the foundation for future oral microbiota formation. The period from 6 mo to 18 mo old exhibited considerable progress in microbiota formation and is when Neisseria and Haemophilus, which suppress the onset of dental caries and periodontal disease, and F. nucleatum, which is suggested to be related to periodontal disease and bad breath, become the constituent bacteria of the oral microbiota. Therefore, the development of microbiota during this period may be critical for future oral disease prevention.
Author Contributions
K. Yama, contributed to data acquisition, analysis, and interpretation, drafted the manuscript; S. Morishima, K. Tsutsumi, R. Jo, contributed to conception and design, data acquisition and interpretation, critically revised the manuscript; Y. Aita, T. Inokuchi, T. Okuda, D. Watai, K. Ohara, contributed to data acquisition and interpretation, critically revised the manuscript; M. Maruyama, contributed to conception and design, data acquisition and interpretation, critically revised the manuscript; T. Chikazawa, T. Iwamoto, Y. Kakizawa, contributed to data interpretation, critically revised the manuscript; T. Oniki, contributed to data acquisition and analysis, critically revised the manuscript. All authors have provided their final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241272011 – Supplemental material for Oral Microbiota Development in the First 60 Months: A Longitudinal Study
Supplemental material, sj-docx-1-jdr-10.1177_00220345241272011 for Oral Microbiota Development in the First 60 Months: A Longitudinal Study by K. Yama, S. Morishima, K. Tsutsumi, R. Jo, Y. Aita, T. Inokuchi, T. Okuda, D. Watai, K. Ohara, M. Maruyama, T. Chikazawa, T. Iwamoto, Y. Kakizawa and T. Oniki in Journal of Dental Research
Footnotes
Acknowledgements
The authors would like to thank the participants for their continued participation in this long-term clinical study. We would also like to thank Kaori Takeda, Chika Sekine, Kaori Nohara, Chikako Ishihara, Yuna Takimoto, Takayuki Ebi, and Hisashi Kawasaki for their assistance in sample collection from participants in the past. We also thank Editage (![]() ) for English editing.
) for English editing.
Correction (December 2024):
Article updated to correct “4 mo old” to “42 mo old” in the Patients and Methods section, under “Participant Information Collection.”
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K. Yama, K. Tsutsumi, R. Jo, Y. Aita, T. Inokuchi, T. Okuda, D. Watai, K. Ohara, M. Maruyama, T. Chikazawa, T. Iwamoto, and Y. Kakizawa work at Lion Corporation. The other authors declare no competing interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
A supplemental appendix to this article is available online.
Data Availability
Microbiota analysis data (16S rRNA gene sequences) are deposited in the DNA Data Bank of Japan (accession No. PRJDB10059, PRJDB17623).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
