Abstract
Mesenchymal stem cells (MSCs), characterized by their undifferentiated and multipotent nature, can be derived from various sources, including bone marrow, adipose, and dental tissues. Among these, dental MSCs (DSCs) exhibit universal MSC characteristics and are attracting considerable attention for regenerating oral and craniofacial tissues. This review provides a contemporary overview of recently published clinical studies using DSCs for various orodental and maxillofacial regenerative applications, including bone, periodontal, and endodontic regeneration. It also explores the utilization of DSCs in treating systemic conditions, exemplified by their application in managing conditions such as COVID-19 and osteoarthritis. The available evidence underscores the potential of DSCs and their secretome as efficacious tools in regenerative medicine for both dental and nondental clinical applications, supporting the continued promise of stem cell–based therapies. It is nevertheless evident that there are a number of important challenges that restrict the widespread utilization of DSCs, namely, difficulty in standardizing autologous preparations, insufficient cell surface marker characterization, high production costs, and regulatory compliance requirements. Further, the unique requirements of dental applications, especially complex structures such as the periodontium, where temporospatial control over the healing process is required, necessitate the combination of stem cells with appropriate scaffolds according to the principles of tissue engineering. There is currently insufficient evidence to support the clinical translation of DSCs into clinical practice, and phase 3 clinical trials with standardized protocols for cell sourcing, propagation, dosing, and delivery are required to move the field forward. In summary, this review provides a contemporary overview of the evolving landscape of stem cell therapy, offering insights into the latest developments and trends as well as the challenges that need to be addressed for the widespread application of DSC-based cell therapies.
Keywords
Introduction
Mesenchymal stem cells (MSCs) are undifferentiated self-renewing multipotent cell populations that can be sourced from various tissues, including bone marrow, adipose, neonatal birth–associated, and dental tissues. They are characterized by the ability to differentiate into multiple tissues of mesenchymal origin, including osteoblasts, adipocytes, and chondrocytes. MSCs, with their multipotent differentiation and immunomodulatory capabilities, have gained significant attention in regenerative medicine (Samsonraj et al. 2017). Thus far, several commercialized MSC therapies have received global regulatory approval, mostly sourced from bone marrow or adipose tissues (Levy et al. 2020). Given the scarcity of readily harvestable MSCs in adult humans, there is a continual emphasis on identifying alternative sources, with dental tissues recognized for more than 20 y as a readily available source (Gronthos et al. 2000).
Dental tissue–derived MSCs (DSCs) share similar traits with other MSCs, namely, self-renewal and multipotency (Huang et al. 2009). Since the initial discovery in 2000 of dental pulp stem cells (DPSCs) (Gronthos et al. 2000), further niches have been identified in deciduous or permanent teeth (Fig. 1A), including periodontal ligament stem cells, stem cells from apical papilla, stem cells from exfoliated deciduous teeth (SHED), alveolar bone marrow–derived MSCs (BMMSCs), gingiva-derived MSCs, dental follicle stem/progenitor cells, and buccal fat pad–derived MSCs (BFP-MSCs) (Ghaderi et al. 2018).
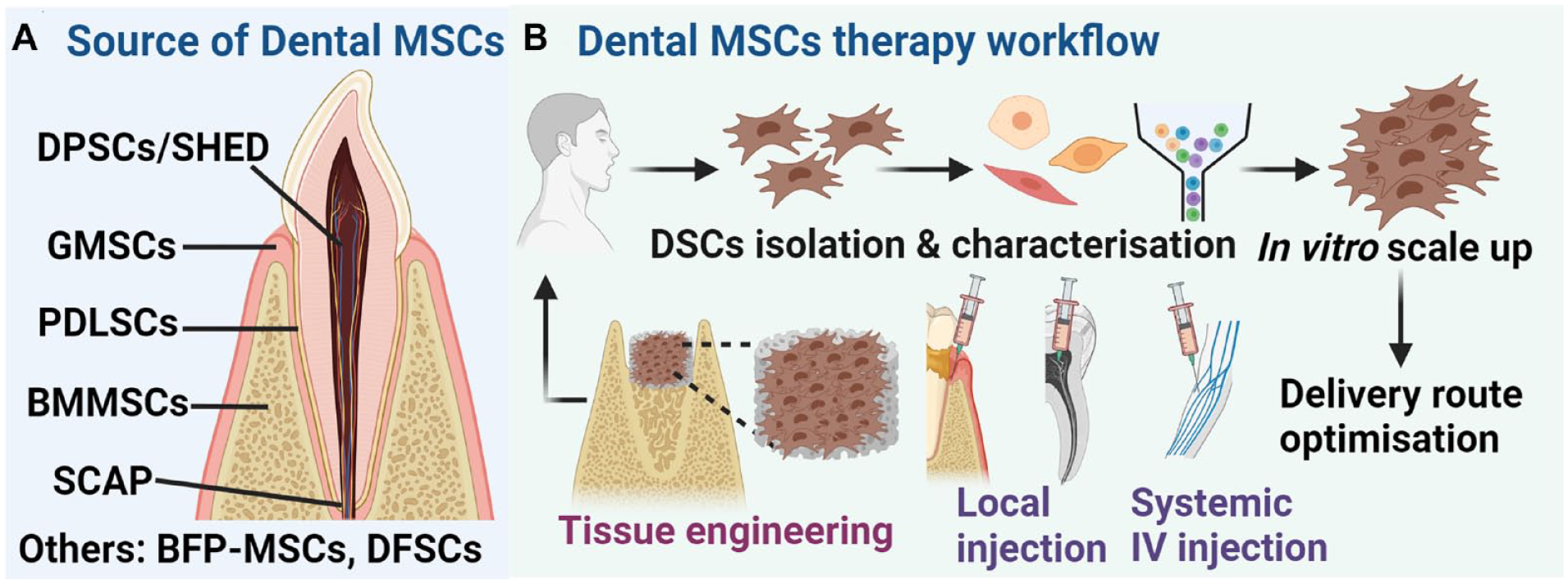
Origins of dental mesenchymal stem cells (DSCs) and the workflow for autologous DSCs cell therapy. (
A general overview of the workflow for the clinical utilization of autologous DSCs is shown in Figure 1B. As recommended by the International Society for Cell and Gene Therapy (ISCT) (Lovell-Badge et al. 2021), MSCs, including DSCs, must be characterized by viability, by fluorescence-activated cell sorting (FACS) for surface marker expression (≥95% positive of CD73, CD105, and CD90 and ≤2% negative for CD45, CD34, CD14, CD11b, CD19, and HLA-DR; along with more putative positive markers, i.e., CD29, CD44, CD71, CD106, CD166, STRO-1, and CD146) and by trilineage differentiation (osteogenic, chondrogenic, and adipogenic) potential. In vitro cultivation is usually required to achieve sufficient cell numbers, followed by the determination of the optimal delivery route, either systemic intravenous (IV) injection or local injections. Tissue-engineering approaches, involving the use of biomaterials scaffolds, offer another avenue for site-specific cell delivery of DSCs to defect sites (Mao et al. 2006; Ivanovski et al. 2014). Through optimized cell dosage and delivery routes, these cells can be retransplanted to the patient, completing the DSCs cell therapy cycle. DSCs should undergo thorough surface marker characterization before and after isolation, including monitoring maintenance of surface markers during in vitro expansion. The significant number of published and/or registered clinical studies highlights the widespread interest and potential for the use of DSCs in dental applications as well as various systemic disease therapies. Both dental and systemic applications of DSCs across 10 countries will be discussed in this review, with an emphasis on the most recent published studies in the field (since 2020) (Tables 1 and 2).
Selected Clinical Studies Published since 2020 Involving DSCs in Bone, Periodontal, and Endodontic Regeneration.
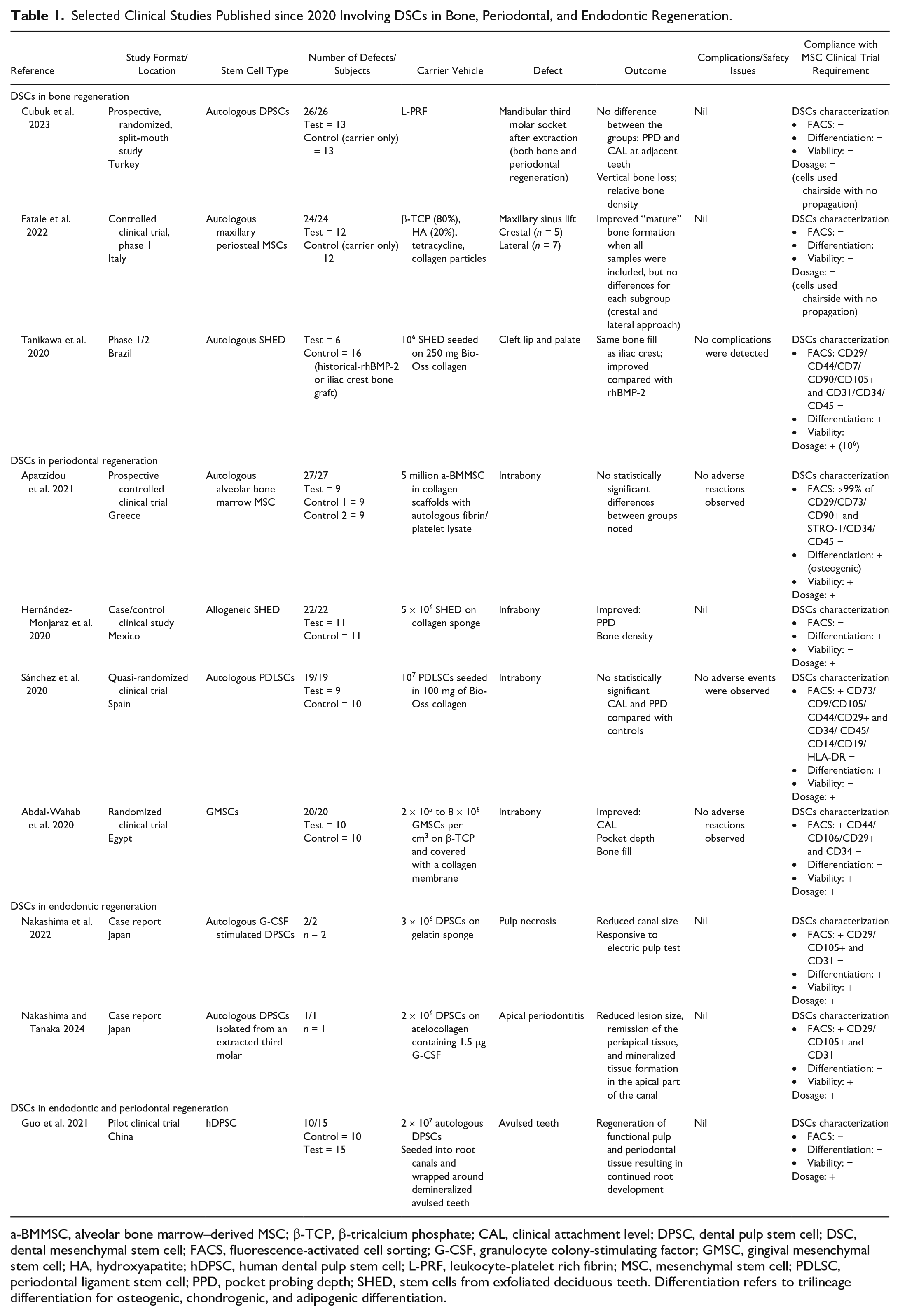
a-BMMSC, alveolar bone marrow–derived MSC; β-TCP, β-tricalcium phosphate; CAL, clinical attachment level; DPSC, dental pulp stem cell; DSC, dental mesenchymal stem cell; FACS, fluorescence-activated cell sorting; G-CSF, granulocyte colony-stimulating factor; GMSC, gingival mesenchymal stem cell; HA, hydroxyapatite; hDPSC, human dental pulp stem cell; L-PRF, leukocyte-platelet rich fibrin; MSC, mesenchymal stem cell; PDLSC, periodontal ligament stem cell; PPD, pocket probing depth; SHED, stem cells from exfoliated deciduous teeth. Differentiation refers to trilineage differentiation for osteogenic, chondrogenic, and adipogenic differentiation.
Recently Published Clinical Trials Involving DSCs in Systemic Diseases.
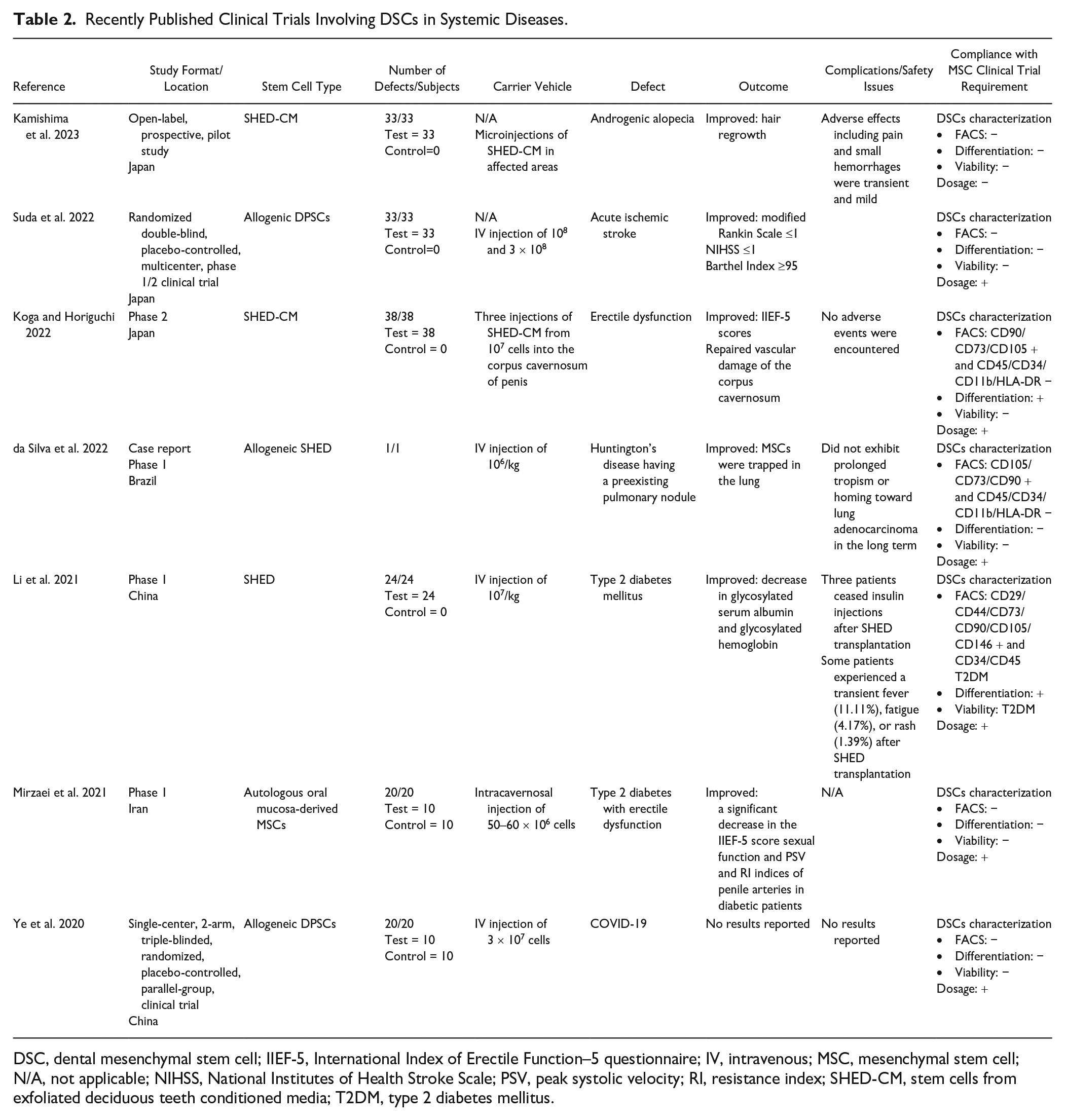
DSC, dental mesenchymal stem cell; IIEF-5, International Index of Erectile Function–5 questionnaire; IV, intravenous; MSC, mesenchymal stem cell; N/A, not applicable; NIHSS, National Institutes of Health Stroke Scale; PSV, peak systolic velocity; RI, resistance index; SHED-CM, stem cells from exfoliated deciduous teeth conditioned media; T2DM, type 2 diabetes mellitus.
Use of MSCs in Oral and Craniofacial Applications
Bone Regeneration
Clinical studies have demonstrated the regenerative potential of MSCs in treating both intraoral and extraoral bone defects, as reviewed by Padial-Molina et al. (2015). Noteworthy pioneering examples involve the use of BMMSCs for maxillary sinus (Kaigler et al. 2013) and alveolar ridge augmentation (Kaigler et al. 2015; Gjerde et al. 2018), which demonstrated safe and effective bone formation after a 1-y follow-up. While early studies predominantly used BMMSCs, DSCs have also been used to assess their effectiveness in addressing intraoral bone defects. Reviews of clinical studies published up to 2020 (Sybil et al. 2020; Yamada et al. 2020) demonstrated that the majority of published papers are case reports or series that report bone formation following DSC transplantation, albeit without a comparison group. Significantly, in all studies, biomaterials are used as carriers for cells, including bovine and porcine xenografts, collagen sponge, β-tricalcium phosphate (β-TCP), platelet-rich plasma, and platelet-rich fibrin (L-PRF).
Table 1 documents the 3 recently published phase 1 clinical studies (2020 to 2023), using dental MSCs for orofacial bone regeneration (Tanikawa et al. 2020; Fatale et al. 2022; Cubuk et al. 2023). Various DSC/carrier combinations were used, and all 3 studies demonstrated bone formation with no adverse effect detected, albeit on relatively small patient numbers (5 to 13 patients in the test groups). Notably, only 1 clinical study showed improved bone formation with DSCs treatment compared with the carrier-only control (Fatale et al. 2022). In contrast, Tanikawa et al. (2020) showed that propagated DPSCs combined with collagenated xenograft hydroxyapatite to treat cleft palate defects yielded similar results to a historical comparison group of autologous iliac bone graft (Fig. 2). Similarly, Cubuk et al. (2023) found no additional benefit of adding DPSCs to L-PRF to improve bone and adjacent tooth periodontal healing following third molar extraction. The inconsistent findings using DPSCs in bone regeneration are mirrored in more recent studies using nonoral-sourced MSCs, that is, BMMSCs (Asahina et al. 2021; Kadry et al. 2021).
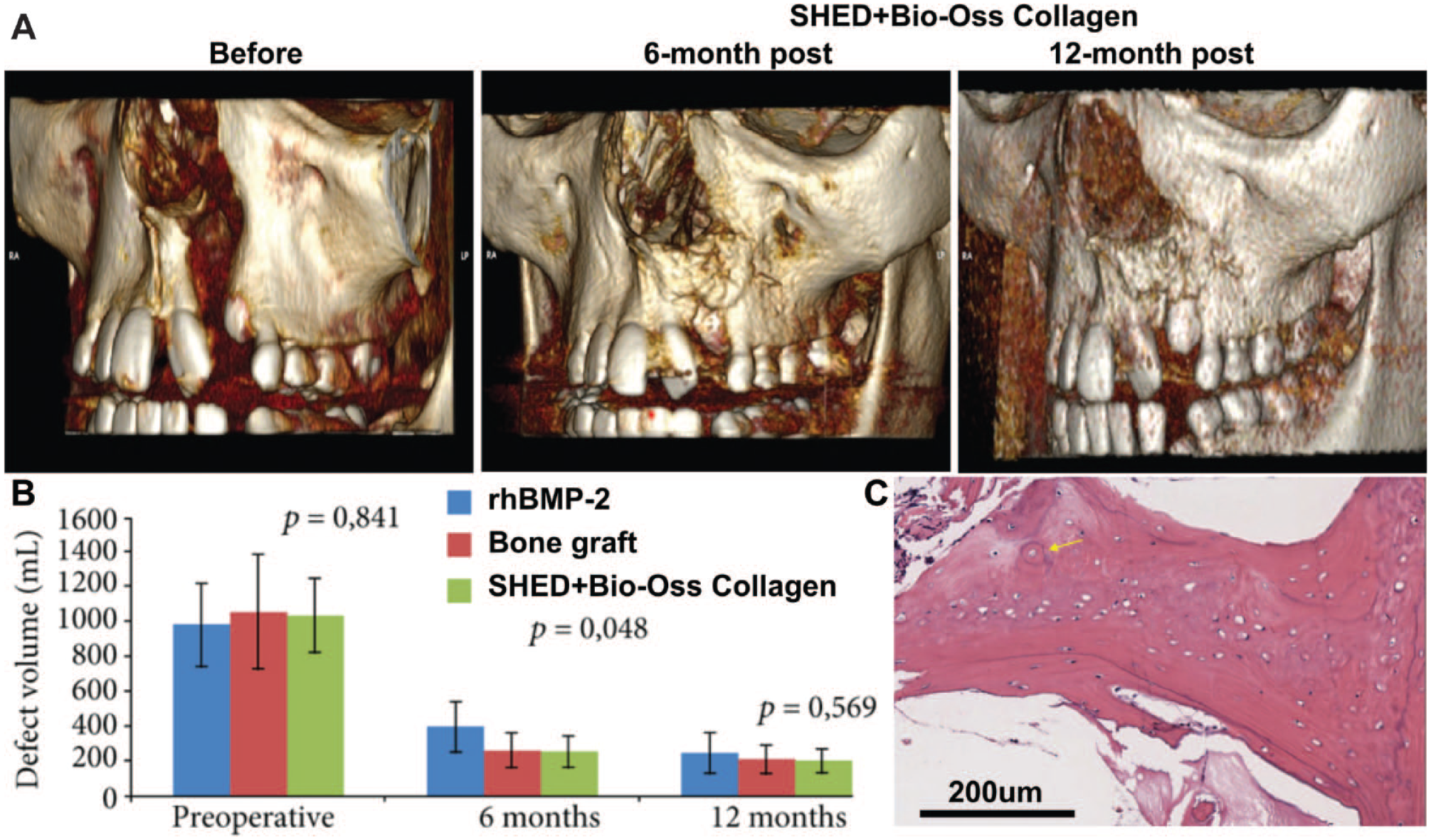
Clinical application of stem cells from exfoliated deciduous teeth (SHED) in the regeneration of alveolar bone, as adapted from Tanikawa et al. (2020). (
Notably, as shown in Table 1, there is a lack of full clarity regarding the source, dosage, and characterization of DSCs before their transplantation into bone defects, with only 1 of the studies (Tanikawa et al. 2020) reporting on these parameters. Indeed, the other 2 studies simply used bedside methods to “isolate” DSCs without any propagation, which is unlikely to result in sufficient cell numbers for therapeutic effectiveness. These challenges are consistent with previously reported MSC clinical trials for orodental bone regeneration (Sybil et al. 2020; Yamada et al. 2020). Thus, it is essential to highlight that adherence to international stem cell therapy guidelines is strongly recommended in all stem cell research (Lovell-Badge et al. 2021).
A review of ClinicalTrials.gov (https://clinicaltrials.gov/) and the International Clinical Trials Registry Platform (ICTRP; https://trialsearch.who.int/), Appendix Table 1 identifies 11 registered clinical trials, using BMMSCs, SHED, alveolar bone marrow–derived MSCs, or BFP-MSCs for the treatment of alveolar bone, cleft lip and palate, and craniomaxillofacial defects (ChiCTR2000039119). The list includes 3 current phase 3 clinical trials (NCT04297813, NCT03766217, ChiCTR2000039119), while the remainder are phase 1/2 trials. Notably, the 2 largest registered phase 3 trials are follow-up investigations of 2 of the better-documented published phase 1/2 trials (Tanikawa et al. 2020), which use adequate numbers of propagated MSCs. It is therefore possible that these trials will provide more definitive evidence on the potential of DSCs to enhance intraoral and maxillofacial bone regeneration.
Summary and perspective
Most published clinical studies show that the use of DSCs is associated with successful bone regeneration with no reported side effects, although the evidence for additional efficacy beyond that achieved by controls is not unequivocal. Further, these studies are phase 1/2 safety trials with limited patient numbers and often inadequate characterization of the implanted stem cell populations, and hence, the outcome of registered phase 3 trials will be essential in demonstrating the effectiveness of stem cell therapies for orofacial bone regeneration. It should also be noted that an inherent requirement for bone regeneration is the structural integrity of the implanted material; hence, the cell delivery method is an important consideration. In this context, the ideal carrier for various applications needs to be determined, and the ability of DSCs to achieve superior performance compared with the carrier, ideally in a cost-effective manner, is required to make DSC-based therapies a viable clinical reality. The future may lie in combining stem cell therapies with personalized biodegradable scaffolds tailored to individual defects, using advanced manufacturing, such as custom 3-dimensional (3D) printing based on computed tomography scans and computer-aided design (Bartnikowski et al. 2020; Ivanovski et al. 2023).
Periodontal Regeneration
Identification and characterization of the MSC population within the periodontium was a significant advance in the field of periodontal cell biology using STRO-1+/MCAM+ MSCs population (Seo et al. 2004). Early preclinical studies in animal models demonstrated the potential of MSCs in periodontal regeneration (Bright et al. 2015). Hence, human clinical studies commenced to determine the viability and efficacy of this treatment modality (Bartold and Ivanovski 2022). To date, there have been 26 human clinical studies reporting the use of MSCs from a variety of intra- and extraoral sources for periodontal regeneration across 10 countries (Appendix Table 2), making periodontal regeneration by far the most studied application in the oral and maxillofacial field. The studies have ranged from case reports, case series, randomized clinical trials as well as phase 1/phase 2 clinical trials. Cell sources have included periodontal ligament (8 studies), BMMSCs (8), dental pulp (5), deciduous tooth pulp (2), gingiva (1), umbilical cord (1), and adipose tissue (1) (Bartold and Ivanovski 2022).
As the number of human studies has increased, reviews have attempted to distill the information gleaned from these early clinical studies (Hynes et al. 2012). Systematic reviews considering the outcomes of the clinical application of MSCs for periodontal regeneration reported evidence of improved clinical outcomes such as clinical attachment levels (CALs), probing pocket depths (PPDs), and bone fill in the test groups (Novello et al. 2020). However, these reviews noted that the evidence is still low level due to the limited number of studies, levels of heterogeneity between studies, and risk of bias, and hence, any definitive conclusions were not possible.
Table 1 outlines the most recent studies (2020 to 2023), consisting of 2 randomized clinical trials (Abdal-Wahab et al. 2020; Sánchez et al. 2020), 1 a prospective clinical trial (Apatzidou et al. 2021) and 1 case-control quasi-experimental clinical trial (Hernández-Monjaraz et al. 2020). The number of subjects in all of these studies was low (fewer than 30 subjects), meaning that any results obtained must be viewed with some caution. One of these 4 studies reported improvements in CAL, PPD, and bone fill that were statistically significant compared with the controls (Abdal-Wahab et al. 2020), while another reported a “borderline” statistically significant amount of bone fill (P = 0.09) with the addition of cells. However, while another 2 of these studies reported some improvements in similar clinical outcome measures, they were not significantly different compared with controls (Fig. 3) (Sánchez et al. 2020; Apatzidou et al. 2021). The stem cell sources in these 4 studies were well characterized with well-defined dosages of propagated cells. However, it should be noted that, as was the case with bone regeneration, the delivery vehicles for these studies have generally incorporated some type of carrier scaffold (e.g., collagen, β-TCP), which imparts a level of heterogeneity that makes it difficult to compare outcomes across different studies.
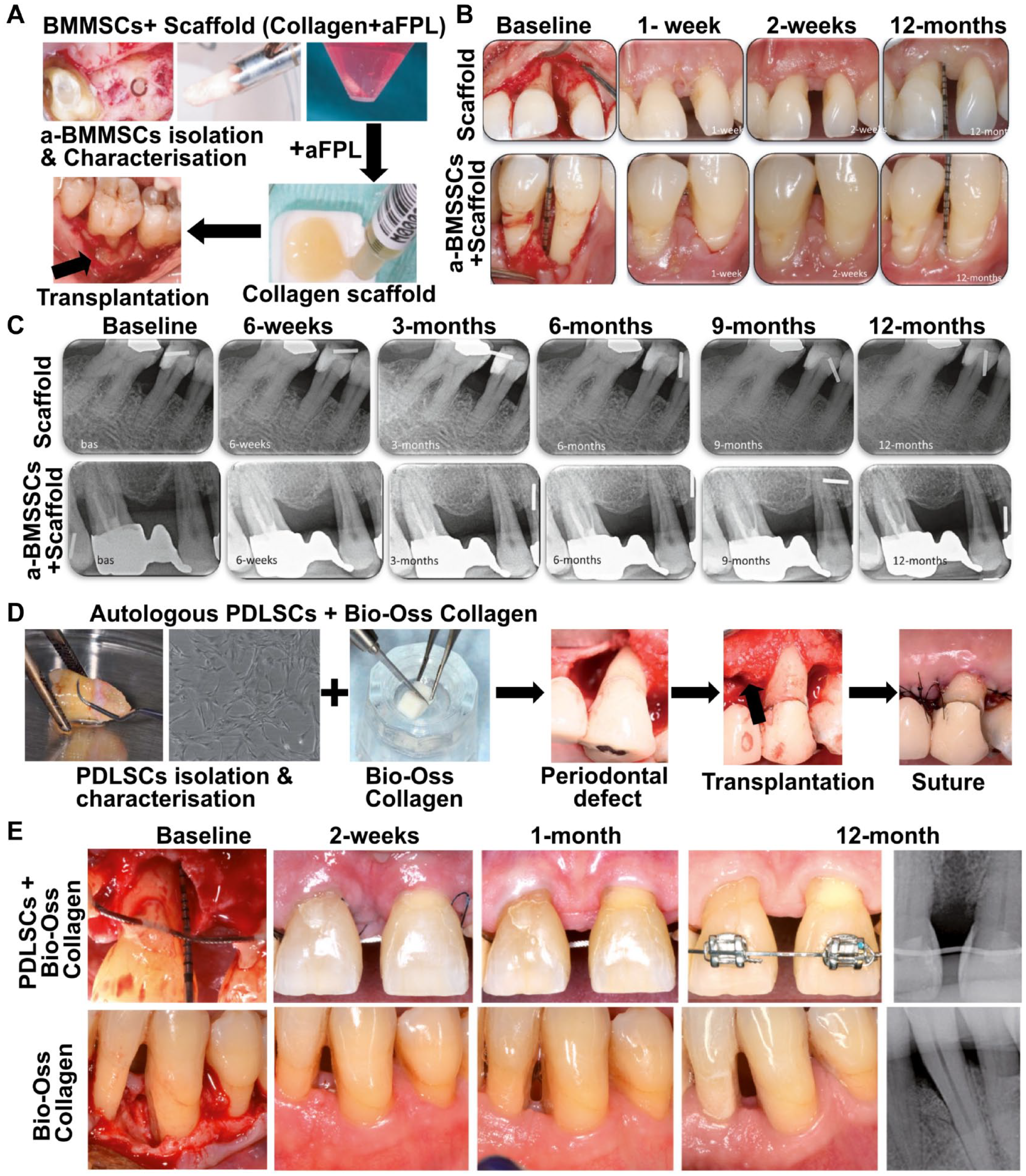
Two recently published clinical studies using DSCs in periodontal regeneration, with a 12-mo postoperation follow-up. (
In terms of registered clinical trials that are yet to be published (Appendix Table 3), they are almost entirely phase 1/2 trials. Interestingly, one of the largest trials (ChiCTR210 0051466; N = 96) did not target periodontal regeneration but rather the treatment of chronic periodontitis, using the immunomodulatory properties of DSCs, which have gained greater attention in recent years.
Summary and perspectives
Since the first human studies investigating MSC-based approaches for periodontal regeneration were published in 2005, the ensuing 18 y have seen a steady increase in the number of human clinical studies. While the general findings are encouraging, the clinical outcomes have often been only minimally better than if no cells were used. This could be due to the use of impure DSC populations, as none of the published studies used CD73/CD90/CD105+ DSCs as per ISCT gudelines, with the use of heterogeneous cell populations likely to lead to varied clinical outcomes. As the most recent systematic reviews have noted, there is a need for further larger and better-controlled studies before any definitive conclusions can be made, and phase 3 trials are required, which do not appear to be in the pipeline. Even if these studies are carried out, one final hurdle to overcome in the field of cell-based regenerative technologies will be to demonstrate that these procedures have a clear cost/benefit outcome for patients and can enhance tooth retention over and above what current approaches can provide. A further consideration for periodontal regeneration is the complex structure of the periodontium and the requirement for transmucosal healing. The use of stem cells alone is unlikely to address these challenges, and the combination with biomaterials that can impart control over spatiotemporal healing and antimicrobial properties may be required to maximize the benefits of DSCs in periodontal regeneration (Ivanovski et al. 2014; Vaquette et al. 2018).
Endodontic Regeneration
Endodontic therapies, specifically vital pulp therapy and apexification/apexogenesis (Rafter 2005), demonstrate clinical and radiographic evidence of hard-tissue deposition (Fig. 1). Moreover, case reports (Banchs and Trope 2004) confirm the potential for tissue ingrowth into pulpless root canal spaces filled with a blood clot (Fig. 4A, B). Yet, it is now well established that the current clinical approach does not result in the regeneration of a functional dentin–pulp complex (Martin et al. 2013), and it has therefore been suggested to use the term guided endodontic repair.
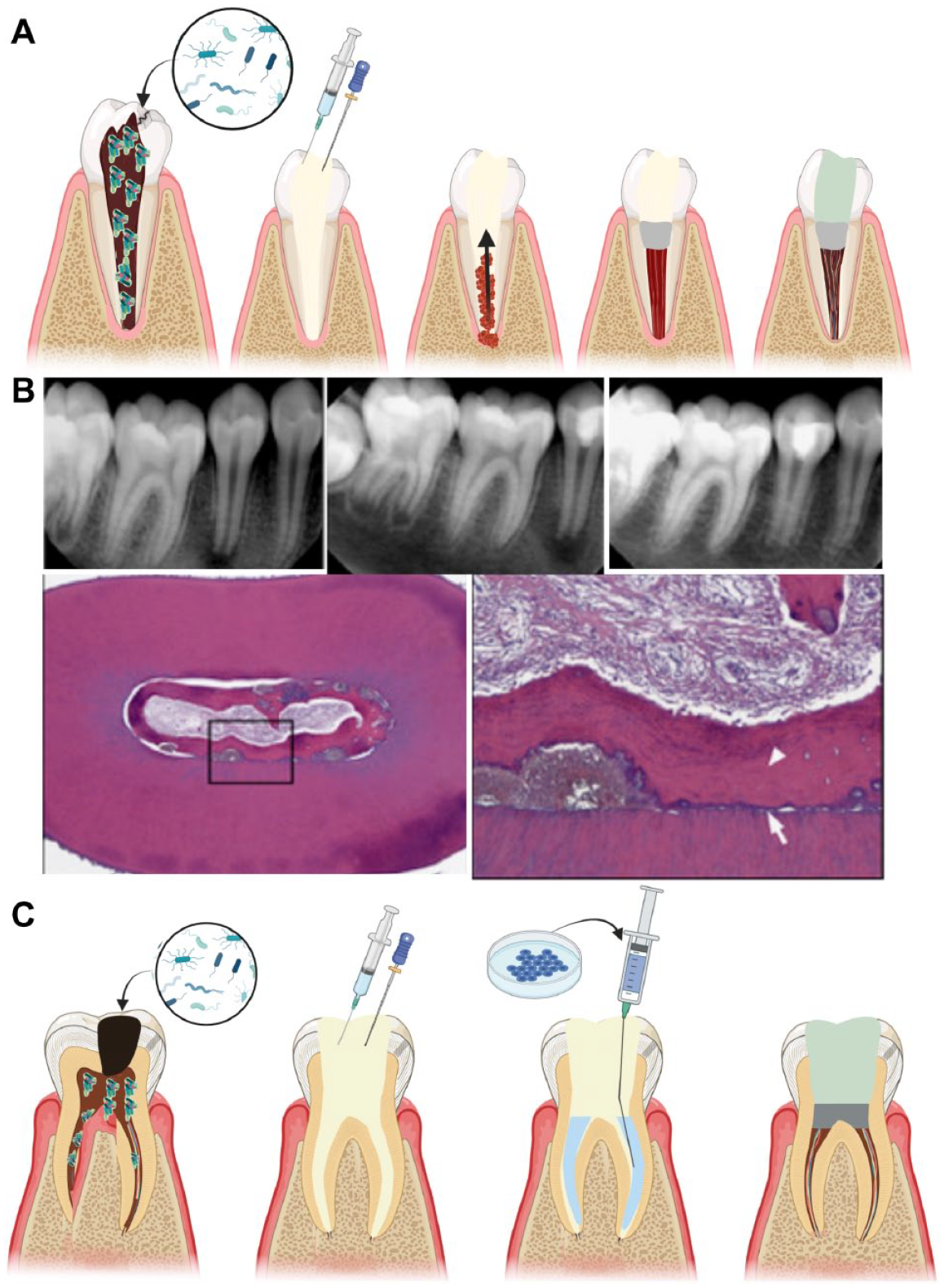
Different regenerative approaches to the management of pulpless teeth. (
Despite a clear rationale to support the goal of dentin–pulp complex regeneration, the latest clinical trials using DSCs in this area are very sparse. There are only 6 published studies using MSCs for endodontic regeneration (Table 1; Appendix Table 4), with only 1 pilot clinical trial and 2 case series using DSPCs (Nakashima et al. 2022; Nakashima and Tanaka 2024) published in the period 2020 to 2024 (Table 1). Furthermore, there is only 1 active currently registered clinical trial investigating the use of SHED for pulp regeneration (Appendix Table 5).
The most notable study demonstrating the successful use of DSCs for pulpal regeneration was a prospective randomized controlled trial with 40 patients (Xuan et al. 2018). SHED cells were expanded for 1 mo prior to implantation into instrumented canals. At 24-mo follow-up of 30 available patients, teeth in the SHED intervention group demonstrated sensation to electric stimulus, an increase in vascular formation, increased root length, and decreased apical foramen width. Meanwhile, controls receiving traditional apexification showed no response to pulp tests or root growth.
Case reports provide further evidence of viable tissues developed in pulpless teeth or in cases diagnosed with irreversible pulpitis following the use of BMMSCs (Gomez-Sosa et al. 2022). A prospective trial (Brizuela et al. 2020) used banked umbilical cord cells that were expanded and encapsulated in platelet-poor plasma and grafted into instrumented root canals, while control subjects received conventional root canal treatment. All 30 subjects who were available for recall (20 experimental, 10 controls) had the treated teeth present, and all were clinically asymptomatic. About half of the stem cell–grafted cases responded positively to pulp testing and showed a slight increase in blood flow in the grafted cases.
A notable recent clinical investigation described 15 patients with avulsed teeth (15 test teeth, 10 controls treated conventionally) treated with autologous DPSCs for both pulp and periodontal regeneration (Guo et al. 2021). At 6 and 12 mo, there were significant differences in several pulpal and periodontal parameters, including continued root development, positive pulp test, and significantly increased vascular flow, indicating favorable healing in the experimental group compared with controls.
Two case reports provide further evidence of viable tissues developed in pulpless teeth or in cases diagnosed with irreversible pulpitis following the use of DPSCs (Nakashima et al. 2022; Nakashima and Tanaka 2024). Nakashima et al. (2022) described the delivery of expanded autologous granulocyte colony-stimulating factor–stimulated DPSCs that had been harvested from third molars into shaped and medicated canals of maxillary molar teeth (Fig. 4C), resulting in magnetic resonance images that were similar to intact pulps (Table 1). The same group reported a case that used 2 × 106 DPSCs on atelocollagen scaffolds placed into an optimally disinfected root canal system, resulting in a decrease in lesion size, alleviation of periapical tissue inflammation, and the formation of mineralized tissue in the apical canal segment (Nakashima and Tanaka 2024).
It should be cautioned that the outcome measures applied in the various published studies (positive pulp test, magnetic resonance imaging, Doppler flowmetry) merely indicate the presence of viable tissue; they do not confirm regeneration of a functional dentin–pulp complex.
Summary and perspectives
There are several reasons for the scarcity of clinical evidence beyond the work of Xuan et al. (2018) for effective cell-mediated dental pulp regeneration. For one, there is well-described evidence for effective alternative options, namely, vital pulp therapy and root canal treatment in cases of painful vital pulps and pulp necrosis with apical periodontitis, respectively (Duncan et al. 2023), while good manufacturing practices for cell-based procedures may be prohibitively costly (Huang and Garcia-Godoy 2014). Second, established biofilm in cases of pulp necrosis is unlikely to be completely eradicated (Nosrat et al. 2015); the presence of microbiota and their by-products is a significant impediment to regeneration. Third, the spatial constraints of the root canal space present unique difficulties with angiogenesis for cell/scaffold constructs beyond a certain dimension (Huang and Garcia-Godoy 2014).
On the other hand, contemporary clinical approaches in cases of pulp necrosis rely on intracanal medication, subsequently stimulating bleeding into the canals and instream of MSCs from the periapical tissues. These so-called cell-free or cell-homing methods (Nosrat et al. 2015) have clinical utility; however, the resulting tissues are not indicative of a regenerated dentin pulp complex (Fig. 4A, B). Taken together, current clinical data do not demonstrate cell-based regeneration in cases of an infected root canal space. Regenerative approaches to extend the spectrum of vital pulp therapy are more promising.
Use of Oral Tissue–Derived Stem Cells in Nonoral/Craniofacial Regenerative Medicine
Extensive research showed that DSCs have potential applications in systemic conditions, demonstrating their efficacy not only in addressing craniofacial conditions but also in serving as a valuable resource for treating systemic diseases via their immunomodulatory and regenerative properties (Li et al. 2022; Li et al. 2023).
Seven clinical studies were published after 2020 using DSCs for the treatment of various systemic diseases in phase 1/2 clinical trials (Table 2). In terms of DSCs source, SHED, SHED-CM, DPSCs, and oral mucosa MSCs were used in these clinical studies for various conditions, including COVID-19, erectile dysfunction both with and without type 2 diabetes, acute ischemic stroke, Huntington’s disease, and hair loss (Ye et al. 2020; Mirzaei et al. 2021; da Silva et al. 2022; Koga and Horiguchi 2022; Kamishima et al. 2023), with generally positive results (Table 2). For example, studies indicated that SHED-CM or SHED can significantly improve erectile function, with or without type 2 diabetes mellitus (T2DM), as demonstrated by enhanced International Index of Erectile Function–5 scores from questionnaire assessments (Mirzaei et al. 2021; Koga and Horiguchi 2022).
In a published clinical trial (Li et al. 2021), 24 insulin-treated T2DM patients received 3 IV infusions of SHED at 1 × 106 cells/kg over 6 wk and were monitored for 12 mo. At the end of follow-up, SHED transplantation led to a significant reduction in glycosylated serum albumin and hemoglobin levels, although 3 patients ceased insulin injections posttransplant. Transient fever (11.11%), fatigue (4.17%), and rash (1.39%) occurred posttransplantation but were easily resolved. This study demonstrated that SHED infusion is a safe and effective therapy for improving glucose metabolism and islet function in T2DM patients.
In addition, a search of trial registries (ClinicalTrials.gov and ICTRP) identified 14 clinical trials using DSCs for systemic applications that have either been registered and/or completed since 2018 but are yet to be published (Appendix Table 6). In these trials, DSCs have been investigated across a range of systemic diseases, including COVID-19, diabetes, antiaging applications, severe lung contusion patients, spinal cord injury, cerebral palsy, liver cirrhosis, and osteoarthritis. In the context of the 5 COVID-19 trials, 4 used DPSCs and 1 used MSCs derived from oral mucosa tissue. Except for an unclear statement regarding the dosage, delivery route, and frequency in 1 trial (NCT04382547), the remaining 4 trials chose systemic IV injection or infusion. Dosage and injection numbers varied, ranging from 3 IV administrations of 3 × 107 cells (n = 10; N CT04336254), 3 IV infusions of 1.0 × 106 cells/kg (n = 24, NCT04302519), 4 IV infusions of 0.5 × 106 cells/kg (n = 24, RBR-65trt53), to 1 IV injection of 4 × 107 cells (n = 120, IRCT20140911019125N8). Nevertheless, the quality of reporting of the studies using DSCs for treating systemic conditions, including cell characterization and protocol standardization with clear reporting of cell numbers and method of delivery, is generally more robust compared with oral/maxillofacial regenerative applications.
The broadly accepted dosage for IV injection of MSCs in human patients typically ranges from 1 × 106 to 2 × 106 cells per kilogram (Galipeau and Sensébé 2018). Because paracrine factors released by MSCs depend on cell number and cell-to-cell contact (Dunn et al. 2021; Krampera and Le Blanc 2021), variations in dosing can substantially affect therapeutic outcomes, and hence, future studies should consider applying this standard dosage per kilogram.
Current Status and Future Outlook
A significant number of studies report on the utilization of DSCs for periodontal regeneration, a moderate number for bone regeneration, and a few for endodontic regeneration (Table 1). Despite stem cell therapies being used for more than a decade in regenerative dentistry, the available evidence consists of case reports and phase 1/2 clinical trials. While stem cell use has generally been shown to be safe, the efficacy compared with controls has not always been evident. Indeed, many of the studies suffer from incomplete reporting, with a lack of clarity and standardization of cell source, dosage, and characterization and better adherence to stem cell therapy guidelines (Lovell-Badge et al. 2021). There is also increasing interest in the use of the regenerative and immunomodulatory properties of DSCs for systemic diseases (Table 2), where the documentation of the stem cell therapy protocols tends to be more robust, although the evidence for effectiveness across a variety of clinical applications is still developing.
Key Challenges
An underlying premise of any stem cell therapy is that the cell source is well characterized, uniform, phenotypically stable, and able to be sourced via a reproducible method. Hence, identifying specific markers for DSCs is imperative due to their variability from other skeletal MSCs cell types (Chan et al. 2018). However, DSCs, which are sourced from various dental tissues, tend to be poorly characterized in terms of highly specific and distinct cell markers, which in itself is likely a key contributor to the isolation and utilization of heterogeneous populations that results in variable outcomes. In the absence of specific markers for various DSC populations, the generic ISCT guidelines should at least be used. However, it is noted that only 3 of 7 studies used CD73/CD90/CD105+ DSCs for systemic disease treatment as per ISCT guidelines, with no study using CD73/CD90/CD105+ DSCs population for oral/craniofacial purposes. It is noted that among the 17 included studies, 52% (n = 9) used FACS to monitor surface marker expression after in vitro expansion. This raises concerns about the quality of the research exploring the use of MSCs in various applications, as pure MSC populations and surface marker monitoring before and after cell isolation, and during in vitro expansion, are crucial for optimal clinical results.
Apart from surface markers, cell viability, trilineage differentiation, and microbial contamination assessment are crucial for obtaining pure and appropriate DSC cell populations. Variations in cell delivery routes, dosage, and frequency in current published studies may also contribute to the minimal effectiveness of DSC transplantation compared with conventional clinical treatments. Hence, it is worthwhile to carefully consider all these factors both before and after transplantation to ensure optimal clinical outcomes. In the case of non–life-threatening conditions, such as dental and orofacial disorders, cost-effectiveness and regulatory compliance are major impediments to the widespread utilization of cell-based therapies. For commercialization of stem cell–based therapies, compliance with international regulatory standards such as the US Food and Drug Administration and European Medicines Agency is essential, supported by substantial evidence from well-controlled randomized clinical trials. In addition, in addressing bone and periodontal defects, of robust cell delivery carriers and/or 3D scaffolds used for space maintenance are important considerations for effective outcomes (Ivanovski et al. 2014; Chang et al. 2021) but can themselves have a significant impact on cell behavior, which needs to be fully elucidated (Yin et al. 2019).
Cell-free approaches
There is growing evidence that implanted cells do not directly contribute to tissue regeneration but instead act by creating a favorable environment to foster the inherent endogenous healing potential of native tissues (Mattei et al. 2021). Combined with the regulatory and cost-effectiveness challenges of cell-based therapies, this has given impetus to the search for cell-free therapies. An emerging approach to using the well-established regenerative and immunomodulatory properties of MSCs via a cell-free approach is to use their secreted extracellular vesicles (EVs).
EVs are cell-derived membrane-bound vesicles that carry bioactive molecules from the parent cell that can be delivered to recipient cells with reduced immunogenicity (Cheng and Hill 2022). EVs from DSCs or oral tissues show potential for regeneration and immunomodulation (Li et al. 2022), including in vivo evidence that DPSCs-EVs promote jawbone regeneration in rats (Lee et al. 2023).
At present, 3 human clinical trials have been registered investigating the use of DPSCs-EVs, adipose-derived MSCs-EVs, and umbilical cord MSCs-EVs for the treatment of periodontitis and irreversible pulpitis (ChiCTR1900027140, IRCT20230513058168N1, and NCT04270006). However, using MSCs-EVs for therapeutic purposes faces several challenges, including issues related to standardization, clinical-grade EV characterization, cell source, and efficient delivery of EVs to disease sites (Cheng and Hill 2022). Presently, lyophilization of cell line–derived EVs is facilitated through established commercial companies such as Abcam’s ExoStd lyophilized exosome standards and Lonza’s HansaBioMed Exosome Standards. This implies that the future application of DSCs-EVs has the potential to serve as readily available cell-free products, addressing the hurdles encountered in the use of their parent stem cells. Nevertheless, the clinical development of DSCs-EVs is in its nascent phase requiring further preclinical investigations and the standardization of DSCs-EVs before human clinical trials.
In conclusion, the past few years (2020 to 2023) have not brought significant new evidence that has advanced the clinical use of DSCs in regenerative dentistry, and well-controlled phase 3 clinical trials with clearly standardized protocols for cell sourcing, propagation, dosing, and delivery that follow the most recent guidelines (Lovell-Badge et al. 2021) are needed to move the field forward. The use of DSCs for treating systemic diseases and conditions appears to be gaining momentum, with a number of registered studies harnessing their immunomodulatory and regenerative properties.
Author Contributions
S. Ivanovski, P. Han, M. Sanz, P.M. Bartold, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; O.A. Peters, contributed to conception, data acquisition, analysis, and interpretation, drafted the manuscript. All authors gave their final approval and agree to be accountable for all aspects of work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241261900 – Supplemental material for The Therapeutic Use of Dental Mesenchymal Stem Cells in Human Clinical Trials
Supplemental material, sj-docx-1-jdr-10.1177_00220345241261900 for The Therapeutic Use of Dental Mesenchymal Stem Cells in Human Clinical Trials by S. Ivanovski, P. Han, O.A. Peters, M. Sanz and P.M. Bartold in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
A supplemental appendix to this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
