Abstract
Most of the elderly population is afflicted by periodontal diseases, creating a health burden worldwide. Cellular senescence is one of the hallmarks of aging and associated with several chronic comorbidities. Senescent cells produce a variety of deleterious secretions, collectively termed the
Keywords
Introduction
Periodontitis is a chronic disease triggered by deregulated inflammatory response–induced microbial dysbiosis that gradually deteriorates the tooth-supporting structures (Sahingur 2020). The prevalence of periodontal diseases increases with age, exceeding 60% among those older than 65 y (Eke et al. 2020). This rise creates a major health and socioeconomic burden worldwide. Nevertheless, there is still limited information about the molecular signatures of aging periodontal tissues (Aquino-Martinez 2023).
Cellular senescence is one of the chief hallmarks of aging characterized by irreversible cell cycle arrest, resistance to apoptosis, and pronounced morphological and metabolic alterations, including enlarged cells, DNA damage, and lysosomal and mitochondrial changes (Chaib et al. 2022; Huang et al. 2022). Senescent cells remain metabolically active and secrete a variety of cytokines, chemokines, and tissue-destructive enzymes, collectively termed
Chronic inflammation and oxidative stress as observed in periodontitis can trigger senescence (Albuquerque-Souza et al. 2022). Remarkably, the pathogenesis of periodontal diseases and associated comorbidities share several common features with senescence (Hajishengallis 2022). Growing evidence suggests that the periodontal tissue microenvironment can be conducive for premature senescence, which can increase susceptibility to the disease even in younger populations (Aquino-Martinez 2023). However, there are still no well-defined clinical studies that have assessed the molecular signatures of senescence in the oral mucosa. We hypothesized that diseased gingival tissues display increased features of senescence and SASP. Our goal was to assess the levels of key senescence markers including p16, lipofuscin, and β-galactosidase and inflammatory mediators associated with SASP such as interleukin (IL)–1β, IL-6, IL-8, tumor necrosis factor (TNF)–α, and matrix metalloproteinases (MMPs) in gingival tissues of periodontitis and healthy subjects and define the cells sensitive to senescence.
Materials and Methods
Study Participants
A total of 126 subjects with a noncontributory medical history were recruited from the Graduate Periodontics Clinic, School of Dental Medicine, University of Pennsylvania (Penn), and Virginia Commonwealth University (VCU). The study was approved by Penn (protocol 844212) and VCU (protocol 11025) and conducted in accordance with the Declaration of Helsinki of the World Medical Association. This case-control study followed the STROBE guidelines (Appendix). Periodontal examination was performed by using a calibrated University of North Carolina periodontal probe (Hu-Friedy). Probing depth (PD), recession, bleeding on probing, and clinical attachment levels were recorded. The criteria for periodontitis diagnosis were PD ≥ 5 mm as defined by stages II and III according to periodontal classification from the World Workshop 2017 (Papapanou et al. 2018). Healthy periodontium was defined as PD ≤ 4 mm and obtained from periodontally healthy patients who have been scheduled for crown-lengthening procedures. Exclusion criteria were as follows: (1) <18 y of age, (2) smoker, (3) concomitant systemic disease (e.g., diabetes, HIV/AIDS), and (4) pregnant or lactating. The gingival biopsies were harvested including both epithelial and connective tissues from approximately 1 to 3 mm beyond the submarginal tissue as indicated by the treatment plan (Newman 2023). Subgroup analyses included those less than 55 y old (younger) and subjects who were 55 y and older (older) (Albuquerque-Souza et al. 2022). Details are given in the Appendix.
Quantitative Real-Time Polymerase Chain Reaction
Total RNA was isolated from gingival tissue specimens using the RNeasy Plus Mini kit (Qiagen), and quantitative real-time polymerase chain reaction was determined using specific primers (Appendix Table 1) for SYBR Green Master Mix (SABiosciences) in the StepOne PlusSystem (Applied Biosystems) following established protocols (Mooney et al. 2021).
Histological Specimens
Human periodontal tissue biopsies obtained from the diseased and healthy sites were fixed in 4% paraformaldehyde. For cryosection, the tissues were fixed and dehydrated with 30% sucrose solution before embedding using optimal cutting temperature (OCT) for cryosection. The samples were cut at a thickness of 5 µm at −20 °C by cryostat (Jannone et al. 2020; Mooney et al. 2021).
Lipofuscin Staining
Lipofuscin was stained with Sudan Black B (SBB) solution as described (Georgakopoulou et al. 2013). The images were acquired at 40× magnification with light microscopy (Leica). The region of interest (ROI) was outlined with the same tissue architecture including both epithelial and connective tissue layers from each sample. The lipofuscin-positive cells were determined by perinuclear accumulation aggregation stained with SBB. Random ROI per sample was evaluated, and the positive cells were counted by a blinded examiner. Details are in the Appendix.
SA-β-Galactosidase Activity
The samples were stained with SA-β-galactosidase staining solution (Cell Signaling 9860) overnight at 37 °C in a dry incubator following instructions. Randomly selected fields per sample were captured at a magnification of 20× resolution using a microscope (Leica) in a blinded manner. The SA-β-galactosidase–positive areas were quantified using ImageJ Fiji software by automated image acquisition. The percentage of the positive areas was calculated as a stained area/total tissue area × 100 (Jannone et al. 2020). Details are in the Appendix.
Immunofluorescence
Immunofluorescence staining for p16, β-galactosidase, TE7, and CD68 was performed on gingival tissue sections as described previously (Albuquerque-Souza et al. 2022). Three randomly selected images per sample were captured at 40× magnification. The relative fluorescence intensity and the percentage of colocalization were calculated using ImageJ Fiji software. Details are in the Appendix.
Statistical Analysis
The recruitment of subjects in the study was powered by considering pocket depth as the primary outcome (details are given in the Appendix). GraphPad Prism software version 10 was used for statistical analyses. Categorical variables were tested across groups using chi-square tests. Continuous variables were initially tested for normality assumptions via the Shapiro-Wilk test with Lilliefors correction. The difference (of the continuous variables) between the 2 groups was statistically assessed by unpaired, 2-sided
Results
Study Subjects
A total of 126 eligible subjects, 44 healthy and 82 with periodontitis, were recruited. The participants were nonsmokers with a noncontributory medical history. All patients with periodontitis were diagnosed with stage II and III periodontitis with a mean PD of 7.07 mm and clinical attachment loss of 7.51 mm. The demographics of the study participants and assessments of related group differences corresponding to demographics (age and gender), periodontal parameters (clinical attachment loss and PD), and regions of tissue harvest (maxillary and mandibular) are presented in Tables 3 to 6 in the Appendix. In Appendix Table 3, comparisons corresponding to subject demographics and periodontal parameters reveal significant differences between the healthy and periodontitis groups. For subgroup analyses, subjects were further allocated according to their age and clinical diagnoses as follows: 28 young healthy, 33 young periodontitis, 16 aged healthy, and 49 aged periodontitis. The mean age of the subjects was 35.25 y for young healthy, 41.67 y for young periodontitis, 67.75 y for aged healthy, and 66.20 y for aged periodontitis. In Appendix Table 4, significant differences in age (as expected) were observed when comparing young versus aged subgroups within the healthy and periodontitis groups, whereas significant differences in PD were observed only within the healthy group. Furthermore, comparisons corresponding to demographics and periodontal parameters also revealed significant differences between the 4 subgroups (see Appendix Table 5), with the adjusted post hoc
Periodontitis Lesions Present Increased Levels of Markers Associated with Senescence
The extent of senescence in the gingival tissues was examined first through monitoring the levels of p16 and lipofuscin. Increased gingival p16 gene expression was noted in patients compared with healthy subjects, suggesting enhanced activity of cell cycle arrest pathways during periodontitis (Fig. 1A). Following age-based classification, at the gene level, p16 expression was significantly elevated in both young and aged subjects with periodontitis compared with the healthy groups as well as in older patients with periodontitis compared with the younger diseased group (Fig. 1B). Further analysis at the protein level using immunofluorescence confirmed the increased number of p16-positive cells both in connective and epithelial tissues. displaying more abundance within the epithelium (Fig. 1C, D).
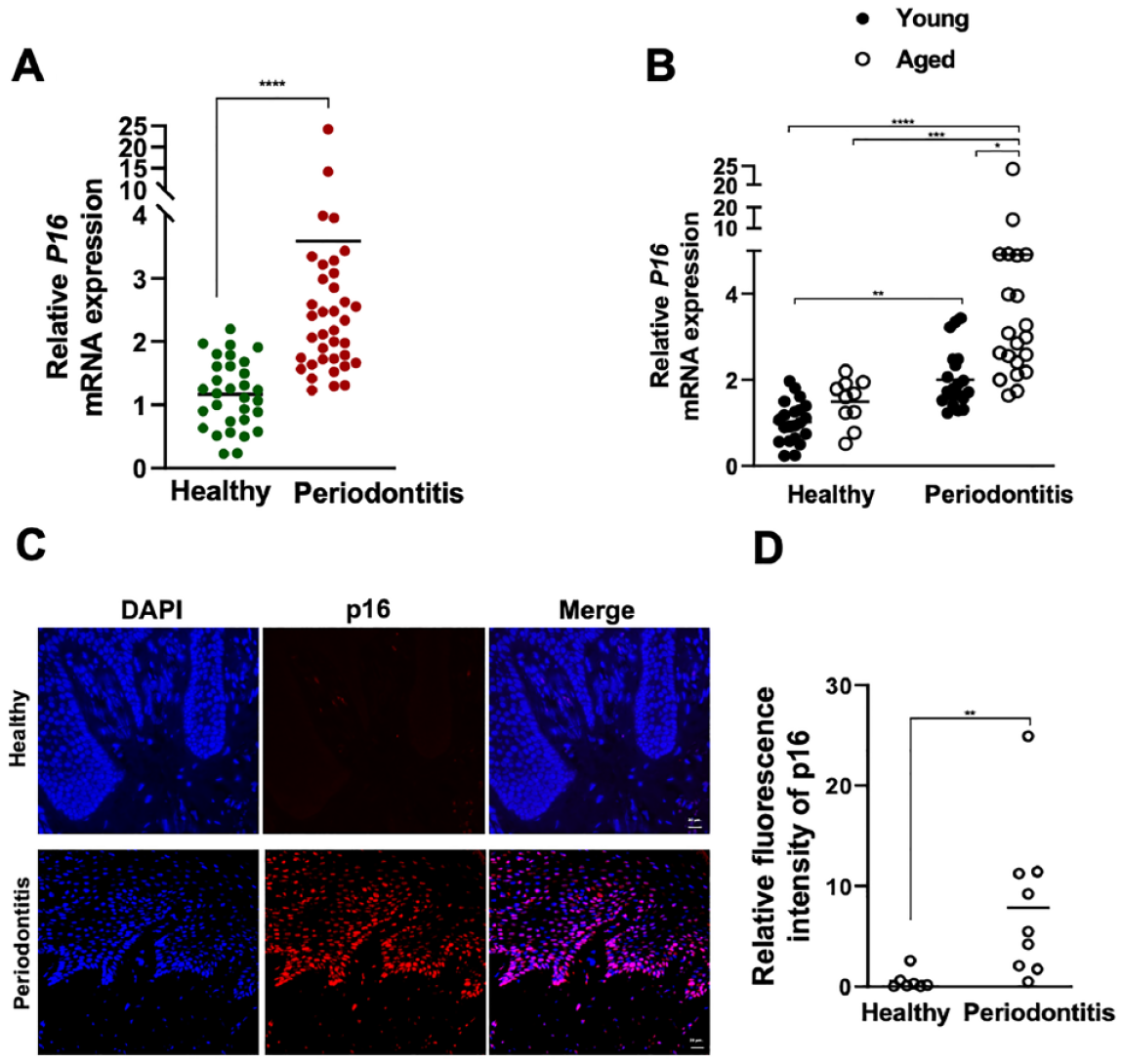
P16 levels in the gingival tissues of healthy and periodontitis subjects. (
Similarly, there was a significant increase in the accumulation of lipofuscin in periodontitis lesions compared with healthy tissues, primarily concentrated in the connective tissue region (Fig. 2A). Further analyses across different age groups revealed notably elevated gingival lipofuscin in both young and older periodontitis patients, in which a significant buildup was detected in older periodontitis lesions compared with the young healthy group (Fig. 2B, C). These findings support the emerging concept that various stressors can induce premature senescence, potentially leading to accelerated aging in younger populations. This deleterious effect gradually accumulates over time in the oral mucosa, likely contributing to and exacerbating the development of periodontal diseases.
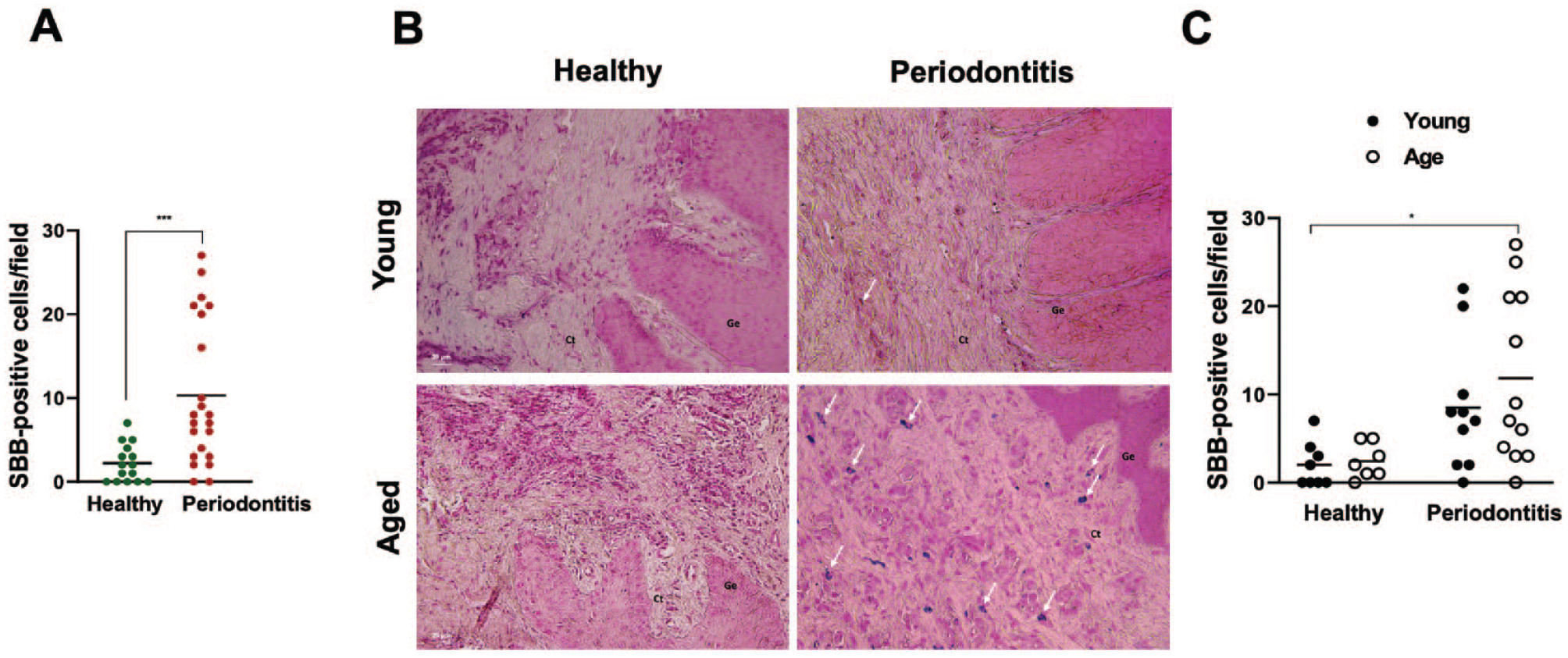
Lipofuscin deposition in the gingival tissues of healthy and periodontitis subjects. (
SA-β-Galactosidase Is Heightened in Periodontitis Lesions, Unveiling a Distinct Colocalization with Gingival Macrophages and Fibroblasts
Another well-recognized biological marker of senescence is SA-β-galactosidase, which signifies distinctive metabolic alterations and a conspicuous augmentation in lysosomal mass within senescent cells (González-Gualda et al. 2021; Huang et al. 2022). Consistently, gingival tissue SA-β-galactosidase activity was also significantly elevated in the periodontitis lesions of both age groups compared with the age-matched healthy sites, validating elevated senescence features in the course of periodontitis (Fig. 3A–C). To determine the cells that are sensitive to senescence within the connective tissue, we conducted immunofluorescence staining with β-galactosidase, macrophage-(CD68), and fibroblast-specific (TE7) antibodies and noted significant enrichment of β-galactosidase in macrophages (Fig. 3D, E) and fibroblasts (Fig. 3F, G) within the periodontitis lesions compared with healthy samples.
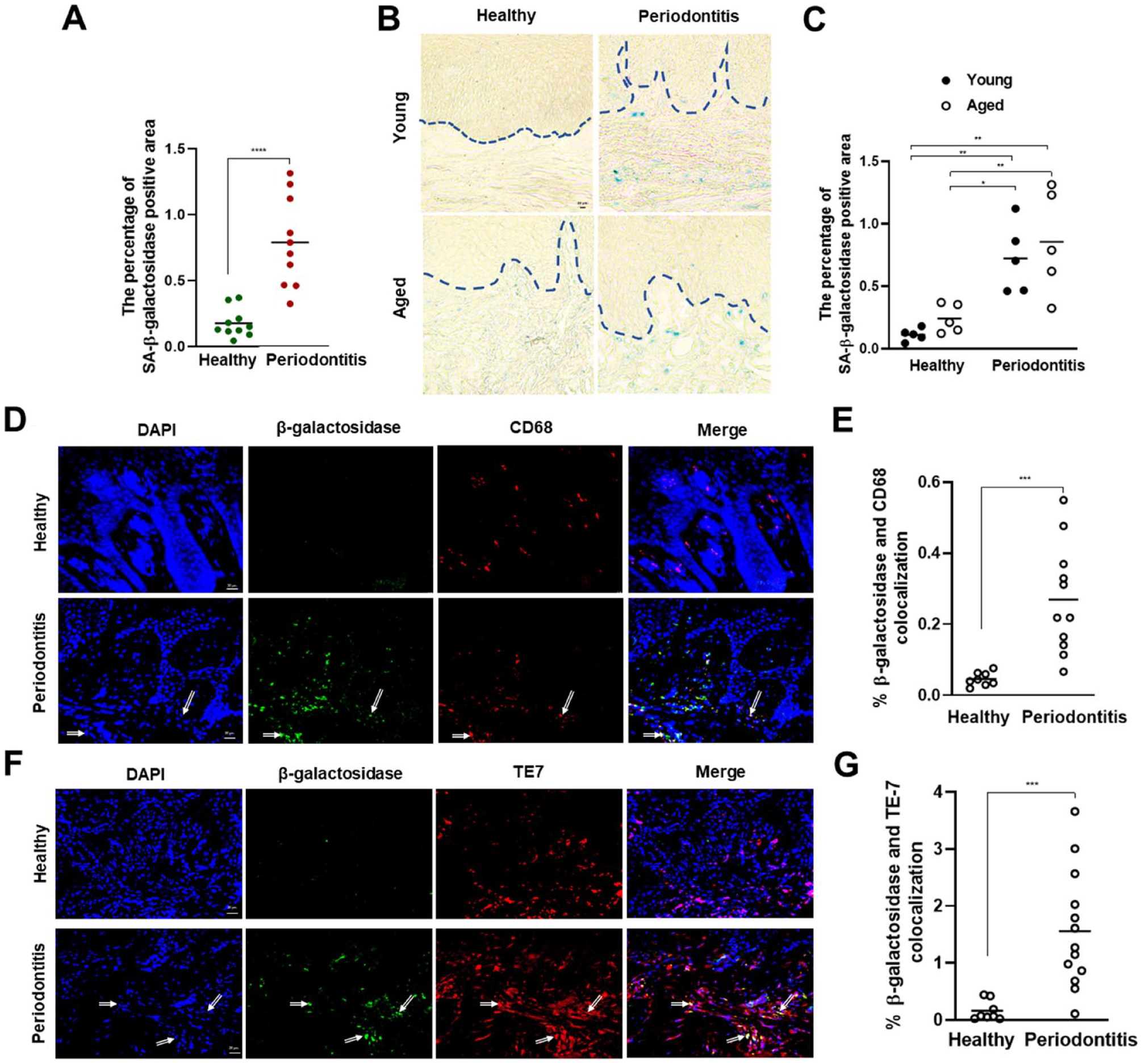
SA-β-galactosidase levels in the gingival tissues of healthy and periodontitis subjects. (
Periodontitis Lesions Present Elevated Inflammatory Milieu Consistent with SASP
The pathophysiology of periodontitis shares common molecular characteristics with SASP and inflammaging (Ebersole and Gonzalez 2020; Albuquerque-Souza et al. 2022). To further characterize senescence-like features in the course of periodontitis, we assessed the expression of common SASP mediators, including IL-1β, IL-6, IL-8, MMP-1, MMP-3, and TNF-α. As anticipated, the mRNA levels of all mediators were elevated in periodontitis lesions compared with healthy tissues (Fig. 4A–L). While we observed a trend toward increased SASP factors in older tissues with periodontitis, the levels remain comparable in young and aged groups. Notably, the gingival expression of inflammatory mediators consistent with SASP remained at baseline among healthy groups, with the exception of IL-8, in which healthy aged tissues showed significantly elevated expression of this chemokine when compared with younger healthy counterparts, implying a unique role for this chemokine in aging gingiva (Fig. 4F). Overall, our findings suggest that a gingival tissue microenvironment inflicted with inflammation can elicit senescence-associated cellular and tissue alterations, which can be the drivers of the transition from health to disease as illustrated (Fig. 5).
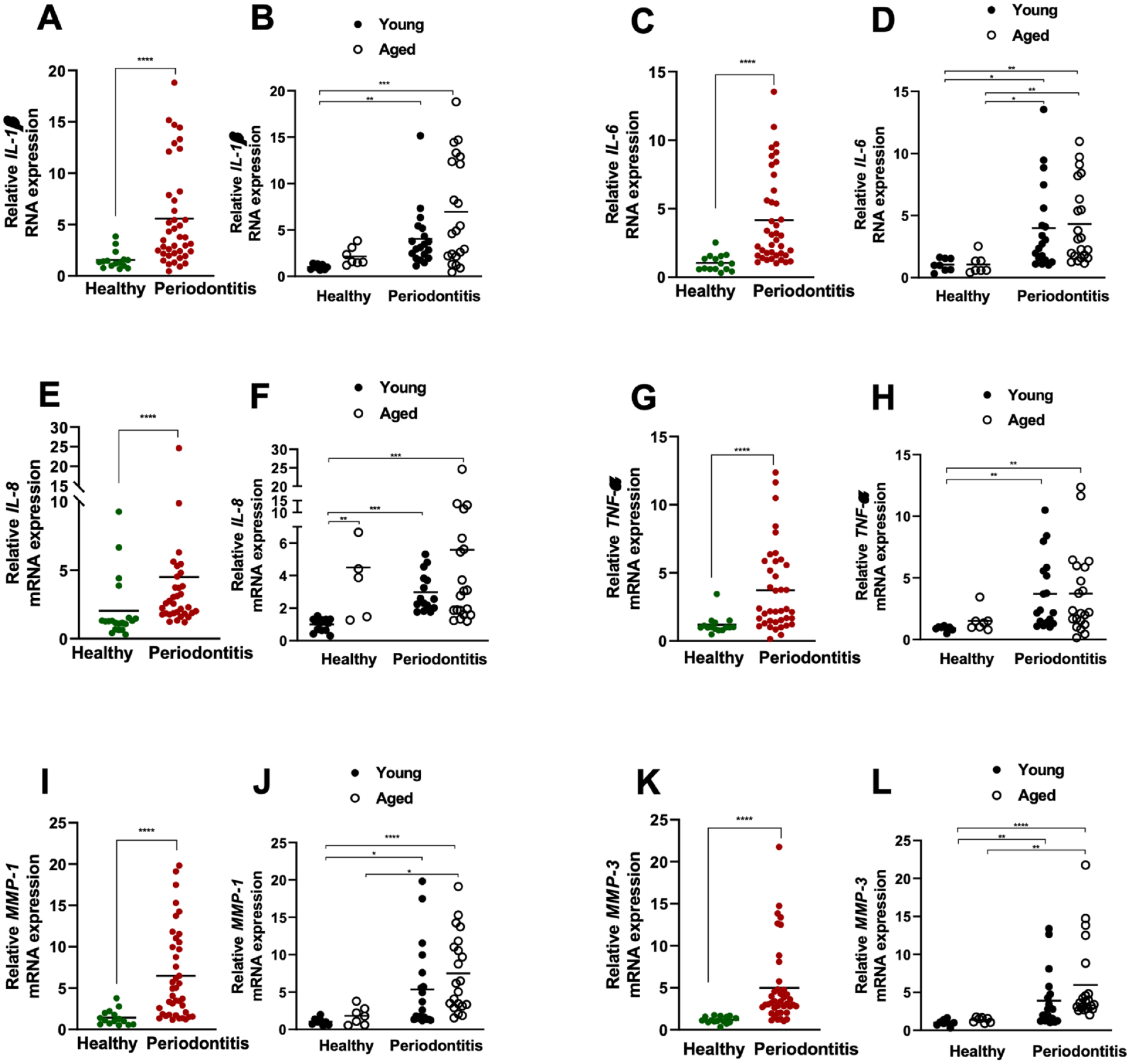
Inflammatory mediators consistent with SASP in the gingival tissues of healthy and periodontitis subjects. Relative mRNA expression of
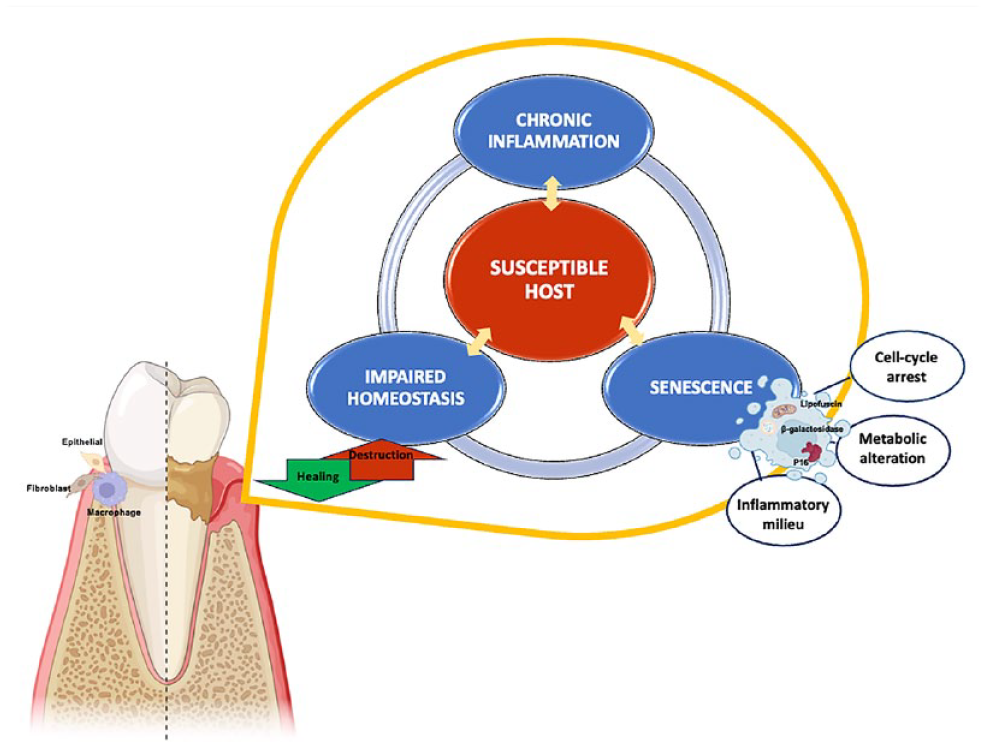
Conceptual model illustrating the impact of cellular senescence on periodontal health. The interplay between chronic inflammation, senescence, and host susceptibility can collectively establish a damaging cycle, disrupting tissue homeostasis. Elevated p16 expression signals a tendency toward cell cycle arrest, hindering cellular function and regenerative capabilities. Increased levels of β-galactosidase and lipofuscin deposition, coupled with heightened inflammation, imply metabolic and lysosomal alterations. As a result, cellular senescence likely plays a substantial role in the pathophysiology of periodontal diseases.
Discussion
Senescent cells enter a state of cellular growth arrest, making them resistant to apoptosis and compromising cellular and tissue repair mechanisms (Zhu et al. 2024). Despite their lower numbers, they release inflammatory mediators and proteases as well as significantly transform neighboring cells, eliciting substantial metabolic and functional alterations, inflammation, and tissue damage. To our knowledge, this is the first clinical study in a well-defined population that concurrently assessed multiple molecular signatures of senescence in the periodontal tissues following recent consensus (Gil 2023). Our results showed increased senescence phenotype in the gingival tissues of periodontitis patients compared with healthy subjects and revealed evidence of senescence-related alterations in younger tissues affected by periodontitis. We also uncovered insights about the specific cell types that are sensitive to senescence within the periodontium. These findings provide critical information to fill the knowledge gap between preclinical and translational studies.
Senescent cells, specifically p16high cells, have emerged as contributors to the pathophysiology of chronic disorders (Huang et al. 2022). The selective elimination of p16-expressing cells expands the life span and improves clinical outcomes in several conditions such as pulmonary diseases, neuropathy, and osteoarthritis (Baker et al. 2016; Jeon et al. 2017; Acklin et al. 2020; Chaib et al. 2022; Kaur et al. 2023). Here, we showed that gingival tissues obtained from periodontitis patients exhibited higher levels of p16, which is an observation consistent with prior preclinical studies indicating that, similar to other tissues, cell cycle arrest occurring in p16-expressing cells can potentially impair cellular function and tissue response and foster disease in the oral cavity as well (Zhang et al. 2019; Albuquerque-Souza et al. 2022). Further supporting this notion, prolonged exposure to
Another novel finding of our study was the detection of significantly elevated lipofuscin deposition in periodontitis lesions, indicative of oxidative stress and lysosomal dysfunction (Skoczyńska et al. 2017; Kakimoto et al. 2019). Elevated lipofuscin levels in younger patients likely stem from subtoxic oxidative stress–induced cellular damage, leading to premature senescence. This heightened oxidative nature of lipofuscin poses a threat to cellular integrity and may contribute to periodontal disease pathogenesis. Dysregulated inflammation, along with senescent cells and lipofuscinogenesis, can perpetuate a vicious cycle in oral mucosa pathology. Similar mechanisms are observed in various age-related conditions across different organs (Skoczyńska et al. 2017; Moreno-García et al. 2018; Kakimoto et al. 2019; Blasiak 2020; Keshavjee et al. 2022). Animal models demonstrate how stress-induced liver disease and traumatic injury to the brain trigger premature senescence, accentuated by lipofuscin accumulation (Ritzel et al. 2019; Keshavjee et al. 2022). Thus, understanding the interplay between senescence, lysosomal dysfunction, and lipofuscin deposition is crucial for elucidating oral cavity disease mechanisms as well.
Senescence-associated changes in lysosomal function can also be detected through SA-β-galactosidase activity, marked by an insoluble, blue stain at pH 6.0, distinct from the pH 4.0 activity seen in nonsenescent cells (González-Gualda et al. 2021). The coexistence of lipofuscin and SA-β-galactosidase in tissues has significant implications for senescence, with SA-β-galactosidase activity being more prominent and lipofuscin accumulation progressing gradually (Georgakopoulou et al. 2013). Further verifying the senescence-like features in the milieu of clinical periodontitis, elevated SA-β-galactosidase was observed in the gingival tissues of periodontitis patients that were enriched in macrophages and fibroblasts. Studies in aged mice and in vitro models confirm increased SA-β-galactosidase in gingival fibroblasts, particularly under oxidative stress (Furukawa et al. 2022). Fibroblasts have been identified as one of the most vulnerable cell types to senescence across many other tissues as well (Furukawa et al. 2022; Huang et al. 2022; Cohn et al. 2023). Macrophages also exhibit senescence-like characteristics, showing differences in gene expression of proinflammatory genes along with dysregulation of other immune-related genes between old and young mice (Aquino-Martinez et al. 2020; Clark et al. 2020). Similar outcomes are observed in other periodontal tissue cells, such as senescent osteocytes disrupting alveolar bone formation by impeding the migration of osteoprogenitor cells in response to lipopolysaccharide (Aquino-Martinez et al. 2020).
Senescent cells are metabolically active and vigorous sources of inflammation and tissue-destructive enzymes themselves and can drive tissue and organ dysfunction through interfering with the function of neighboring cells, thereby establishing a damaging cycle (Fafián-Labora and O’Loghlen 2020; Chaib et al. 2022; Huang et al. 2022). Increased expression of inflammatory mediators consistent with SASP was observed in periodontitis lesions compared with healthy tissues. Notably, IL-8 expression was significantly higher in aged tissues compared with younger counterparts among healthy groups, suggesting a unique role for IL-8 in mediating senescence-related events in gingival tissues. However, SASP factors alone do not suffice to define a senescent state as they can originate from diverse cell types. For instance, it has been shown that p16INK4a could induce senescence without eliciting SASP (Coppé et al. 2011). Similarly, gingival fibroblasts undergoing replicative senescence do not exhibit SASP (Páez et al. 2020). In addition, the quantity and characteristics of SASP vary depending on the cell type and inducer of senescence. Various stressors, such as hyperglycemia, can activate senescence pathways and SASP, involving inflammatory signaling pathways such as NF-κB, which is implicated in periodontitis pathogenesis (Li et al. 2019; Zhang et al. 2019; Mooney et al. 2021; Aquino-Martinez 2023). Thus, understanding the interactions between SASP mediators, NF-κB, and other signaling pathways in different cells could lead to novel strategies to maintain oral health. While the primary objective of this study was to examine gingival tissue senescence in periodontitis, further investigations into SASP factors in gingival crevicular fluid samples and senescence response during gingivitis are warranted to complement our findings.
In conclusion, our clinical findings substantiate the preclinical investigations and reveal that gingival tissues can be sensitive to senescence and senescence-related metabolic alterations, which can affect the trajectory of periodontal health (Fig. 5). Numerous studies have reported that eliminating senescent cells can alleviate disease in aging-related comorbidities (Chaib et al. 2022). Since persistent inflammation and senescence mutually reinforce each other, reducing senescent cells and their secretome holds promise in potentially alleviating periodontal diseases as well (Fig. 5). Similarly, successful management of periodontal diseases may improve senescence-related alterations in the oral mucosa, ultimately fostering healthy aging, thus warranting further investigation. Finally, while preclinical studies have indicated a sensitivity to DNA damage and senescence in females, there is still a significant lack of clinical research regarding the influence of biological sex on the senescence response (Olivieri et al. 2023). Variations in gingival tissue biology between sexes have been observed, suggesting that gender itself may influence susceptibility to periodontitis (Ebersole et al. 2023). Hence, future studies are warranted ideally involving population-based cohorts and more comprehensive tools and global data sets to thoroughly characterize cell- and tissue-specific interactions across diverse sexes and demographics and ascertain whether targeting senescence and the senescence-associated secretome could present a novel strategy for preserving periodontal health (Zhu et al. 2024).
Author Contributions
K. Rattanaprukskul, S.E. Sahingur, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; X.-J. Xia, M. Jiang, E. Albuquerque-Souza, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; D. Bandyopadhyay, contributed to design, data analysis and interpretation, critically revised the manuscript. All authors gave their final approval and agree to be accountable for all aspects of work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345241255325 – Supplemental material for Molecular Signatures of Senescence in Periodontitis: Clinical Insights
Supplemental material, sj-docx-1-jdr-10.1177_00220345241255325 for Molecular Signatures of Senescence in Periodontitis: Clinical Insights by K. Rattanaprukskul, X.-J. Xia, M. Jiang, E. Albuquerque-Souza, D. Bandyopadhyay and S.E. Sahingur in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by US Public Health Service grants R01DE025037 and R01DE027374 to S.E. Sahingur from National Institute of Dental and Craniofacial Research/National Institute of Health.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
