Abstract
The mandibular condylar cartilage (MCC) is an essential component of the temporomandibular joint, which orchestrates the vertical growth of the mandibular ramus through endochondral ossification with distinctive modes of cell differentiation. Parathyroid hormone–related protein (PTHrP) is a master regulator of chondrogenesis; in the long bone epiphyseal growth plate, PTHrP expressed by resting zone chondrocytes promotes chondrocyte proliferation in the adjacent layer. However, how PTHrP regulates chondrogenesis in the MCC remains largely unclear. In this study, we used a Pthrp-mCherry knock-in reporter strain to map the localization of PTHrP+ cells in the MCC and define the function of PTHrP in the growing mandibular condyle. In the postnatal MCC of Pthrp mCherry/+ mice, PTHrP-mCherry was specifically expressed by cells in the superficial layer immediately adjacent to RUNX2-expressing cells in the polymorphic layer. PTHrP ligands diffused across the polymorphic and chondrocyte layers where its cognate receptor PTH1R was abundantly expressed. We further analyzed the mandibular condyle of Pthrp mCherry/mCherry mice lacking functional PTHrP protein (PTHrP-KO). At embryonic day (E) 18.5, the condylar process and MCC were significantly truncated in the PTHrP-KO mandible, which was associated with a significant reduction in cell proliferation across the polymorphic layer and a loss of SOX9+ cells in the chondrocyte layers. The PTHrP-KO MCC showed a transient increase in the number of Col10a1+ hypertrophic chondrocytes at E15.5, followed by a significant loss of these cells at E18.5, indicating that superficial layer–derived PTHrP prevents premature chondrocyte exhaustion in the MCC. The expression of Runx2, but not Sp7, was significantly reduced in the polymorphic layer of the PTHrP-KO MCC. Therefore, PTHrP released from cells in the superficial layer directly acts on cells in the polymorphic layer to promote proliferation of chondrocyte precursor cells and prevent their premature differentiation by maintaining Runx2 expression, revealing a unique PTHrP gradient-directed mechanism that regulates MCC chondrogenesis.
Keywords
Introduction
The mandibular condylar cartilage (MCC) is primarily responsible for the vertical growth of the mandibular ramus and constitutes an essential component of the temporomandibular joint (TMJ) (Mizoguchi et al. 2013; Bender et al. 2018). The MCC undergoes endochondral ossification through distinctive modes of cell differentiation, which is often described as a secondary cartilage that entails chondrogenic conversion of the periosteum (Robinson et al. 2015; Bechtold et al. 2019). Functionally, the MCC adapts to the midface growth and masticatory forces (Hinton 2014). The MCC is composed of 4 distinct layers, including the superficial, polymorphic, flattened chondrocyte, and hypertrophic chondrocyte layer (Shibata et al. 1997; Bechtold et al. 2019). Among these layers, the polymorphic layer houses chondrocyte precursor cells that differentiate into hypertrophic chondrocytes in the deeper layers (Rabie and Hägg 2002; Rabie et al. 2003; Bechtold et al. 2019). Consequently, proliferation of chondrocyte precursor cells controls the rate of generating hypertrophic chondrocytes (Kantomaa et al. 1994; Robinson et al. 2015). This process of cellular differentiation makes the polymorphic layer pivotal to the growth of the mandibular condylar process and endows the MCC with substantial functional adaptability.
The parathyroid hormone–related protein (PTHrP) is a master regulator of chondrogenesis that promotes proliferation of chondrocytes and inhibits their hypertrophic differentiation (Schipani et al. 1997). PTHrP binds to its cognate receptor, the PTH/PTHrP receptor (PTH1R), which activates its downstream G protein–coupled receptor signaling (Kronenberg 2006). A previous study reports that Pthrp messenger RNA (mRNA) is present in the polymorphic layer of the MCC (Shibukawa et al. 2007) and regulates chondrocytes in the flattened layer. The early study demonstrates that chondrocyte proliferation is reduced in the MCC of PTHrP-deficient mice (Suda et al. 1999). In the epiphyseal growth plate, resting zone chondrocytes generate clones of columnar chondrocytes (Mizuhashi et al. 2018). In contrast, chondrocytes in the flattened layer differentiate rapidly into hypertrophic chondrocytes (Shibata et al. 2006) without generating distinct columns of chondrocytes, indicating that a unique mechanism of chondrocyte proliferation and differentiation exists in the MCC. However, the precise source of PTHrP and how PTHrP promotes chondrogenesis in the MCC remains largely undefined. In addition, the target gene of PTHrP in the polymorphic layer has not been identified.
In this study, we aimed to determine the identity of cells expressing PTHrP in the MCC and define how PTHrP regulates chondrogenesis in the MCC. Our findings demonstrate that PTHrP derived from the superficial layer promotes MCC chondrogenesis by regulating Runx2 expression in the polymorphic layer, revealing a unique PTHrP gradient-directed mechanism of chondrogenesis in the MCC.
Materials and Methods
Mouse Strains
Pthrp-mCherry knock-in reporter mice have been previously described (Mizuhashi et al. 2018). Briefly, the native start codon in exon 2 of the Pthrp gene was replaced with a Kozak-mCherry-bGHpA cassette using CRISPR/Cas9 genome editing. All mice were housed in the animal facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, located in the Behavioral and Biological Sciences Building of the University of Texas Health Science Center at Houston. All procedures were conducted in compliance with the Guide for the Care and Use of Laboratory Animals and approved by the University of Texas Health Science Center at Houston’s Animal Welfare Committee (AWC), protocol AWC-21-0070. We have complied with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines. More detailed protocols for mouse management are available in the Appendix Materials and Methods.
For postnatal experiments, male Pthrp mCherry/+ mice were mated to female C57BL/6 mice and their pups were sacrificed at postnatal day (P) 3. For prenatal experiments, male Pthrp mCherry/+ mice were mated to female Pthrp mCherry/+ mice and the vaginal plug was checked in the morning. Pregnant mice were sacrificed at embryonic day (E) 15.5 or E18.5. Pups and fetuses were used for analysis regardless of the sex. A total of 16 pregnant female mice and 72 fetuses were used for the experiments. No statistical method was used to predetermine sample size. Mice were euthanized by overdosage of inhalation anesthesia in a drop jar (Fluriso, Isoflurane USP; VetOne) followed by decapitation.
Histology
Samples were fixed in 4% paraformaldehyde for 24 h at 4°C. P3 samples were decalcified in 15% EDTA for 3 h. Samples were cryoprotected in 30% sucrose/phosphate-buffered saline (PBS) solutions and then in 30% sucrose/PBS:optimal cutting temperature (OCT) compound (1:1) solutions, each at least overnight at 4°C. Samples were embedded in an OCT compound (Tissue-Tek; Sakura). Frozen sections at 12 µm thickness were prepared using a cryostat (Leica CM1860) and adhered on positively charged glass slides (Fisherbrand ColorFrost Plus).
RNAscope In Situ Hybridization and Immunohistochemistry
In situ hybridization was performed with an RNAscope Multiplex Fluorescent Detection Reagents v2 kit (Advanced Cell Diagnostics 323110) using the following probes: Col10a1 (426181), Runx2 (414021), Pth1r (426191), Ihh (413091), and Sp7 (403401). After RNAscope, sections were incubated in blocking buffer (3% bovine serum albumin/Tris-buffered saline with Tween (TBST)) for 30 min at room temperature (RT), and subsequently stained with SOX9 polyclonal antibody (1:500; R&D systems, AF3075), RUNX2 polyclonal antibody (1:100; Novus Biological, NBP1-89104), PTHrP polyclonal antibody (1:100; Invitrogen, PA5-57493), PTH1R monoclonal antibody (3D1.1) (1:200; Novus Biological, NBP1-40067), DsRed Polyclonal Antibody (Living Colors) (1:700; Takara Bio, 632496), or mCherry monoclonal antibody (16D7) (1:400; Invitrogen, M11217), overnight at 4°C. Sections were subsequently stained with appropriate Alexa Fluor–conjugated secondary antibodies for 3 h at 4°C, followed by DAPI (4′,6-diamidino-2-phenylindole, 5 µg/mL; Invitrogen D1306) staining. More detailed experimental procedures are available in the Appendix Materials and Methods.
Signal Quantification for RNAscope on Images
Quantification of RNAscope fluorescent signals on sections was performed using Fiji software (version 2.9.0). A 100-µm × 100-µm square was set on the polymorphic layer in microscopic images, and the expression level of a gene was calculated within the square in pixels. The expression level was represented as pixels per µm2.
Statistical Analysis
Results are presented as mean values ± SD. Statistical evaluation was conducted using the unpaired t test or 1-way analysis of variance (ANOVA) followed by Tukey’s honestly significant difference multiple comparison test. A P value of <0.05 was considered significant.
Results
PTHrP Is Expressed in the Superficial Layer Adjoining RUNX2+SOX9neg Cells in the Polymorphic Layer
First, we mapped the localization of PTHrP+ cells across the postnatal mandible using Pthrp-mCherry knock-in reporter mice (Mizuhashi et al. 2018). In the Pthrp-mCherry allele, red fluorescent protein mCherry is expressed instead of a functional PTHrP protein, constituting a null reporter allele (Fig. 1A). Pthrp mCherry/+ mice carry 1 functional copy of PTHrP and 1 copy of mCherry and are phenotypically indistinguishable from wild-type mice. Importantly, PTHrP-mCherry recapitulates the endogenous expression pattern of Pthrp mRNA, providing a sensitive and convenient readout of Pthrp gene expression. (Fig. 1B, Appendix Fig. 1A).
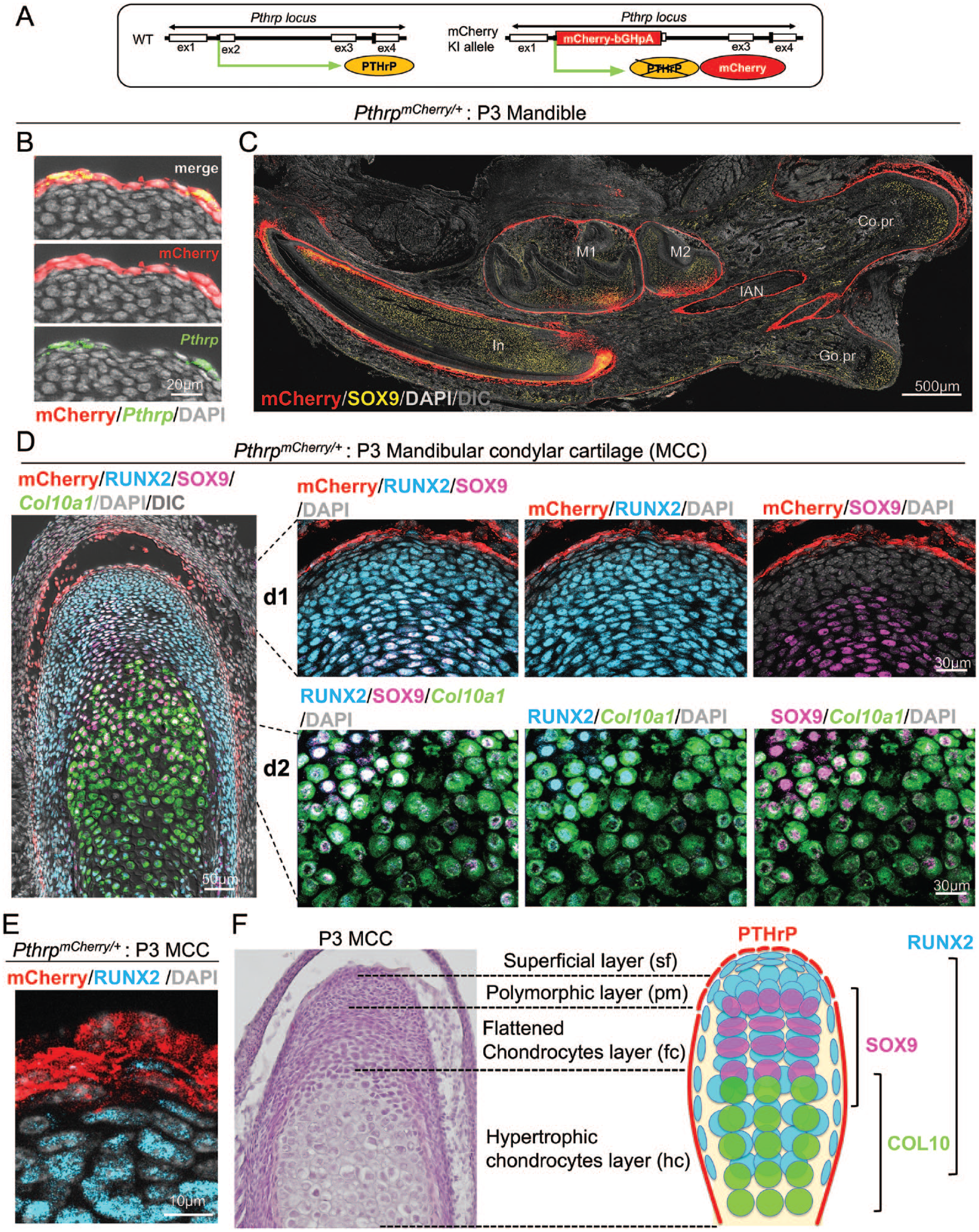
Parathyroid hormone–related protein (PTHrP) is expressed in the superficial layer adjoining RUNX2+SOX9neg cells in the polymorphic layer. (
At P3, PTHrP-mCherry+ cells were preferentially localized in a manner surrounding SOX9+ cells in the mandible (Fig. 1C). Particularly in the dentoalveolar compartment, PTHrP-mCherry+ cells occupied the dental follicle surrounding growing incisor and molars, as well as the dental epithelium in the labial cervical loop of the incisor, as reported previously (Sharir et al. 2019; Takahashi et al. 2019; Nagata et al. 2021). Interestingly in the posterior compartment, PTHrP-mCherry+ cells occupied the entire surface of the condylar process but not the gonial process (Fig. 1C). SOX9+ cells in these processes were chondrocytes, as they were embedded in Safranin O+ cartilaginous matrices (Appendix Fig. 1B).
We closely examined the precise location of PTHrP-mCherry+ cells in the MCC. PTHrP-mCherry+ cells were localized to the superficial layer, occupying 2 to 3 cell layers on the surface of the MCC (Fig. 1D, E), in a more restricted pattern than what was previously reported using radioactive in situ hybridization (Shibukawa et al. 2007; Bechtold et al. 2019). No Pthrp mRNA RNAscope signal was observed in the deeper layers (Fig. 1B, Appendix Fig. 1A). PTHrP-mCherry+ cells were also localized to the forming temporomandibular joint disc as well as the fibrous region of the forming glenoid fossa (Fig. 1C). The PTHrP-mCherry+ cells in the superficial layer adjoined RUNX2+SOX9neg cells in the polymorphic layer (Fig.1DK–d1, E). Cells in the polymorphic layer underwent robust proliferation, as indicated by the presence of EdU+ cells in this layer (Appendix Fig. 1C). Importantly, essentially all SOX9+ cells in the flattened chondrocyte layer coexpressed RUNX2 (Fig. 1D –d1), denoting the unique characteristics of MCC chondrocytes. Col10a1+ cells in the upper hypertrophic layer were RUNX2+SOX9+, while those in the lower hypertrophic layer were negative for both RUNX2 and SOX9 (Fig. 1D–d2). This difference indicates that MCC chondrocytes lose RUNX2 and SOX9 expression during terminal stages of differentiation.
We next asked if PTHrP ligands released from the superficial layer act on the polymorphic and chondrocyte layers of the MCC. For this purpose, we performed PTHrP immunohistostaining on PTHrP-mCherry+ sections, which gives a simultaneous readout of Pthrp mRNA by PTHrP-mCherry and PTHrP protein (Appendix Fig. 1D). Robust PTHrP immunohistochemical signal was observed in the polymorphic and flattened chondrocyte layers but not in the hypertrophic chondrocyte layer (Appendix Fig. 1D, left panel). Interestingly, PTHrP was predominantly detected on the cell surface in the polymorphic layer, whereas it was also detected intracellularly in the flattened chondrocyte layer (Appendix Fig. 1E), indicating an organized diffusion of PTHrP ligands. Immunostaining revealed that PTH1R was abundantly expressed in the polymorphic and flattened chondrocyte layers by RUNX2+ cells (Appendix Fig. 1D, right panel, Appendix Fig. 1F). Therefore, these data indicate that PTHrP released from the superficial layer diffuses across the MCC and binds to its cognate receptor PTH1R on RUNX2+ and SOX9+ cells in the polymorphic and flattened chondrocyte layer. This diffusion process emanates biological actions by activating PTHrP-PTH1R signaling in cells in the polymorphic and chondrocyte layers, supporting the model that PTHrP stands atop the hierarchical structure of the MCC (Fig. 1F). PTHrP produced by the superficial layer of the lateral side also likely plays an important role in regulating the polymorphic layer.
PTHrP Promotes MCC Chondrogenesis and Mandibular Condyle Formation
We next set out to determine the functional significance of superficial layer–derived PTHrP in the mandibular condylar morphogenesis and MCC chondrogenesis. To this end, we analyzed littermates of Pthrp mCherry/+ (Control) and Pthrp mCherry/mCherry (PTHrP-KO) mice at E18.5. Of note, Pthrp mCherry/mCherry mice lacked functional PTHrP protein but expressed 2 copies of mCherry reporter proteins in cells that were supposed to express Pthrp mRNA (Fig. 2A). Importantly, PTHrP immunoreactivity was completely absent in Pthrp mCherry/mCherry mice (Fig. 2B, left panel), confirming the loss of functional PTHrP protein in PTHrP-KO MCC. There was no notable alternation in the expression pattern of PTH1R in PTHrP-KO MCC (Fig. 2B right panel). Therefore, we postulated that an organized diffusion of PTHrP ligands is essential for promoting MCC chondrogenesis, consistent with the concept that morphogen gradient regulates organogenesis (Fig. 2C) (Gurdon and Bourillot 2001).
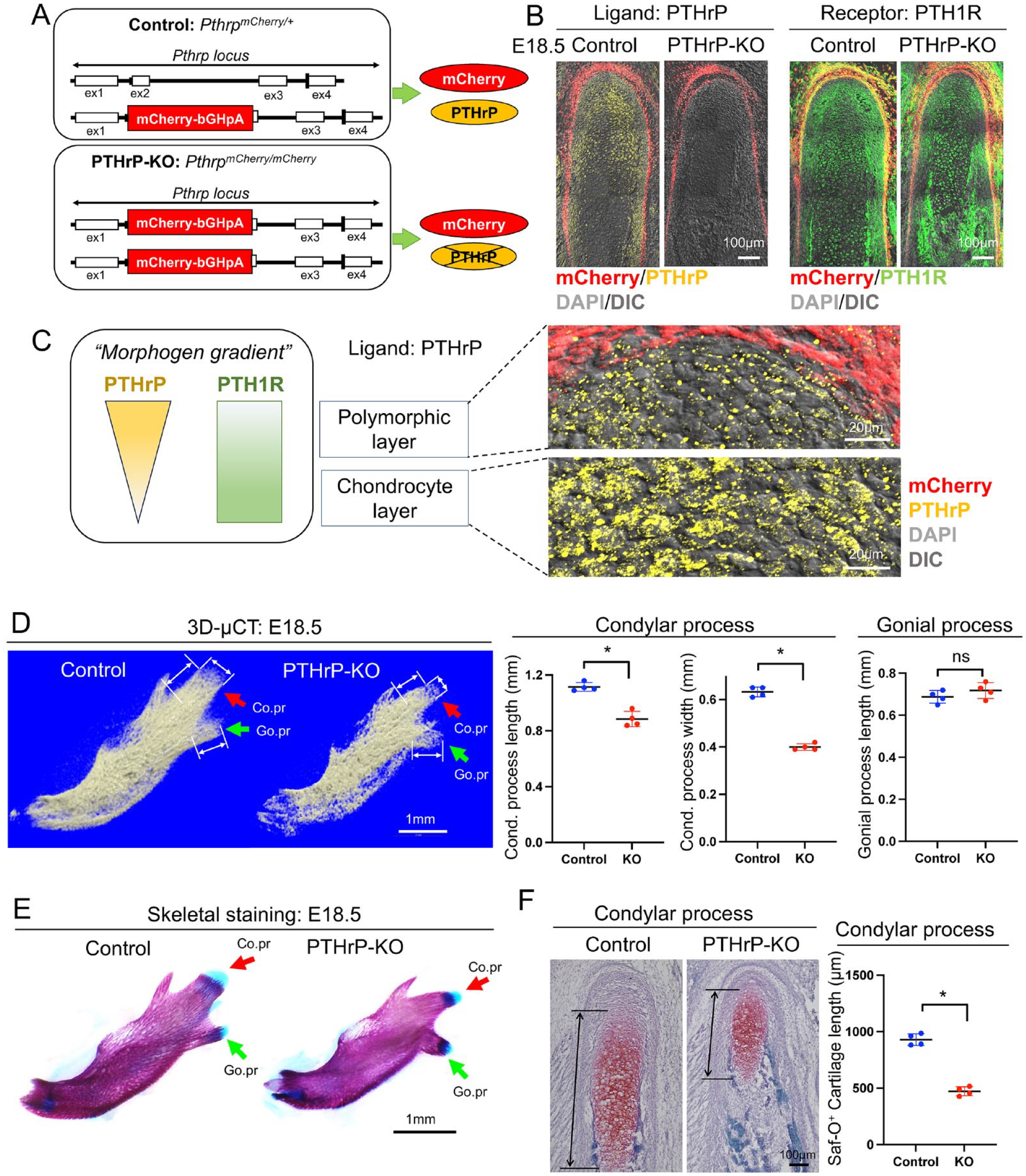
Parathyroid hormone–related protein (PTHrP) promotes mandibular condylar cartilage (MCC) chondrogenesis and mandibular condyle formation. (
We performed 3-dimensional micro–computed tomography (3D-µCT) analysis of Control and PTHrP-KO mandibles at E18.5 (Fig. 2D, left panel). Importantly, the length and width of the condylar process were significantly reduced in the PTHrP-KO mandible (Fig. 2D, red arrows in left and middle panels). In contrast, the length of the gonial process was not altered in the PTHrP-KO mandible (Fig. 2D, green arrows in left and right panels). Therefore, PTHrP specifically promotes mandibular condylar morphogenesis, which corroborates our finding that PTHrP+ cells are present on the surface of the condylar process but not on the gonial process.
We further performed skeletal staining of Control and PTHrP-KO mandibles. Alcian Blue+ cartilaginous tissues were substantially diminished in the condylar process but not in the gonial process of the PTHrP-KO mandible (Fig. 2E, left panel). Additionally, Safranin O staining on sections revealed that the cartilage length was significantly reduced in the condylar process of the PTHrP-KO mandible (Fig. 2F). Therefore, PTHrP plays major functional roles in promoting MCC chondrogenesis and the formation of the mandibular condyle.
PTHrP Promotes Cell Proliferation in the Polymorphic Layer and Formation of SOX9+ Chondrocytes
To identify the cause of the significant defects observed in the PTHrP-KO mandibular condyle, we next evaluated cell proliferation of the polymorphic layer at 2 distinct stages of condylar morphogenesis—one stage at E15.5 when the MCC is specified, as well as another stage at E18.5 when the MCC growth is most prominent (see Fig. 3A for hematoxylin and eosin [H&E] staining). Cell proliferation defined by EdU was significantly reduced in the polymorphic layer of the PTHrP-KO MCC at both E15.5 and E18.5 (Fig. 3B). At higher magnification, most of the reduction in EdU+ cells within the PTHrP-KO MCC occurred in the polymorphic layer occupied by RUNX2+ cells; the reduction in EdU+ cells was more prominent at E18.5 (Fig. 3C). Therefore, PTHrP derived from the superficial layer preferentially regulates proliferation of RUNX2+ cells in the polymorphic layer.
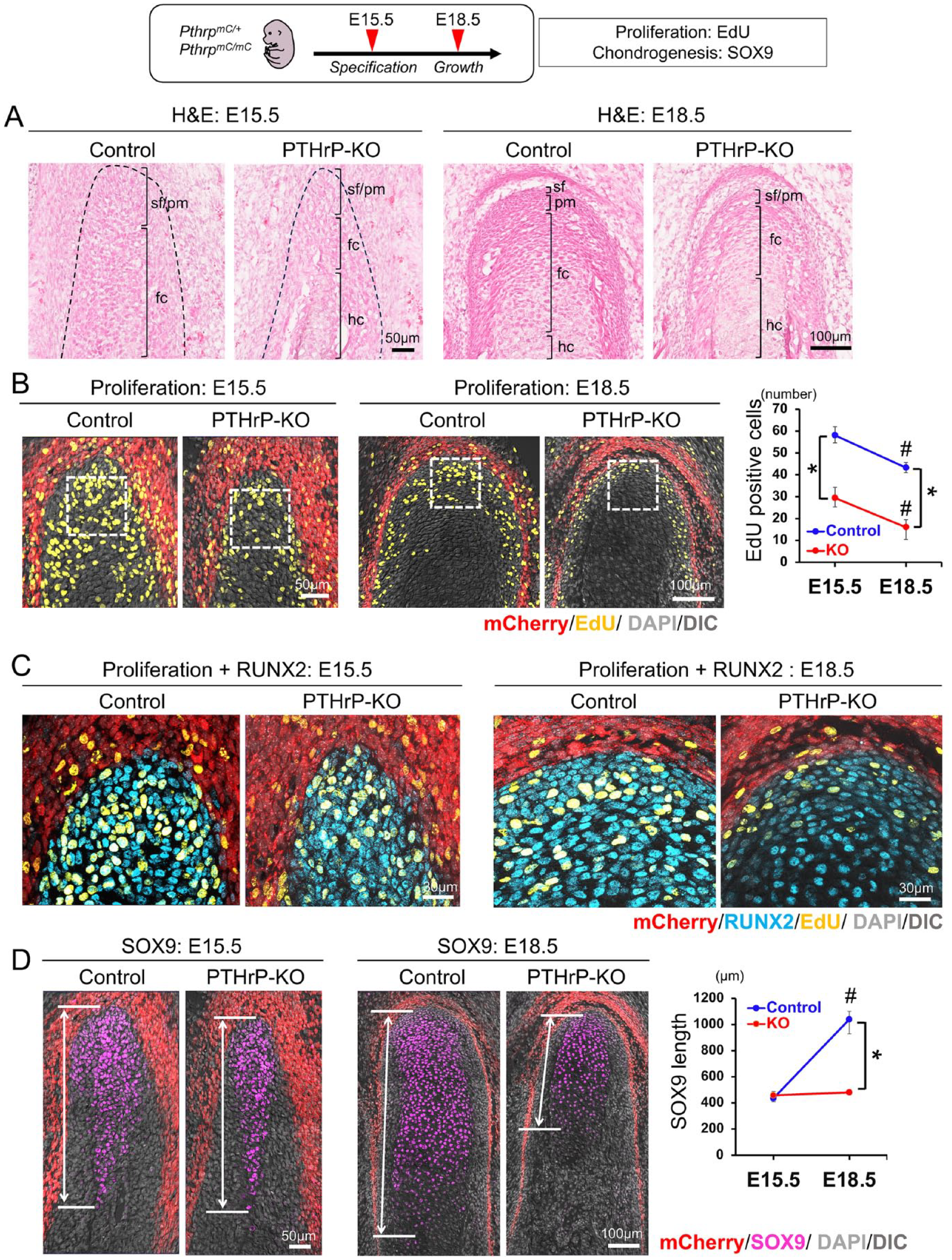
Parathyroid hormone–related protein (PTHrP) promotes cell proliferation in the polymorphic layer and formation of SOX9+ chondrocytes. (
We also quantified the length of SOX9+ domains in the PTHrP-KO mandibular condyle. Interestingly, no difference was observed at E15.5; however, there was a significant reduction in the SOX9+ domain in the PTHrP-KO MCC at E18.5 (Fig. 3D). Therefore, PTHrP does not regulate the initial specification of chondrocyte precursors in the early MCC but promotes continuous growth of the MCC in later stages, presumably through maintaining proliferation of chondrocyte precursor cells in the polymorphic layer.
PTHrP Prevents Premature Chondrocyte Differentiation and Exhaustion
The significant loss of SOX9+ chondrocytes observed in the E18.5 PTHrP-KO MCC promoted us to investigate the late-stage markers of chondrocyte differentiation. To this end, we first examined the expression of a canonical hypertrophic chondrocyte marker, Col10a1. While no difference in the SOX9+ domain between the Control and PTHrP-KO was noted at E15.5 (Fig. 3C), the Col10a1+ domain was significantly expanded in the PTHrP-KO at the same stage (Fig. 4A, left panel), indicating that MCC chondrocytes undergo premature hypertrophy in the absence of PTHrP. In contrast, the Col10a1+ domain was significantly reduced in the PTHrP-KO at E18.5, indicating that a lack of PTHrP depletes chondrocyte precursor cells in the polymorphic layer and subsequently reduces hypertrophic chondrocytes.
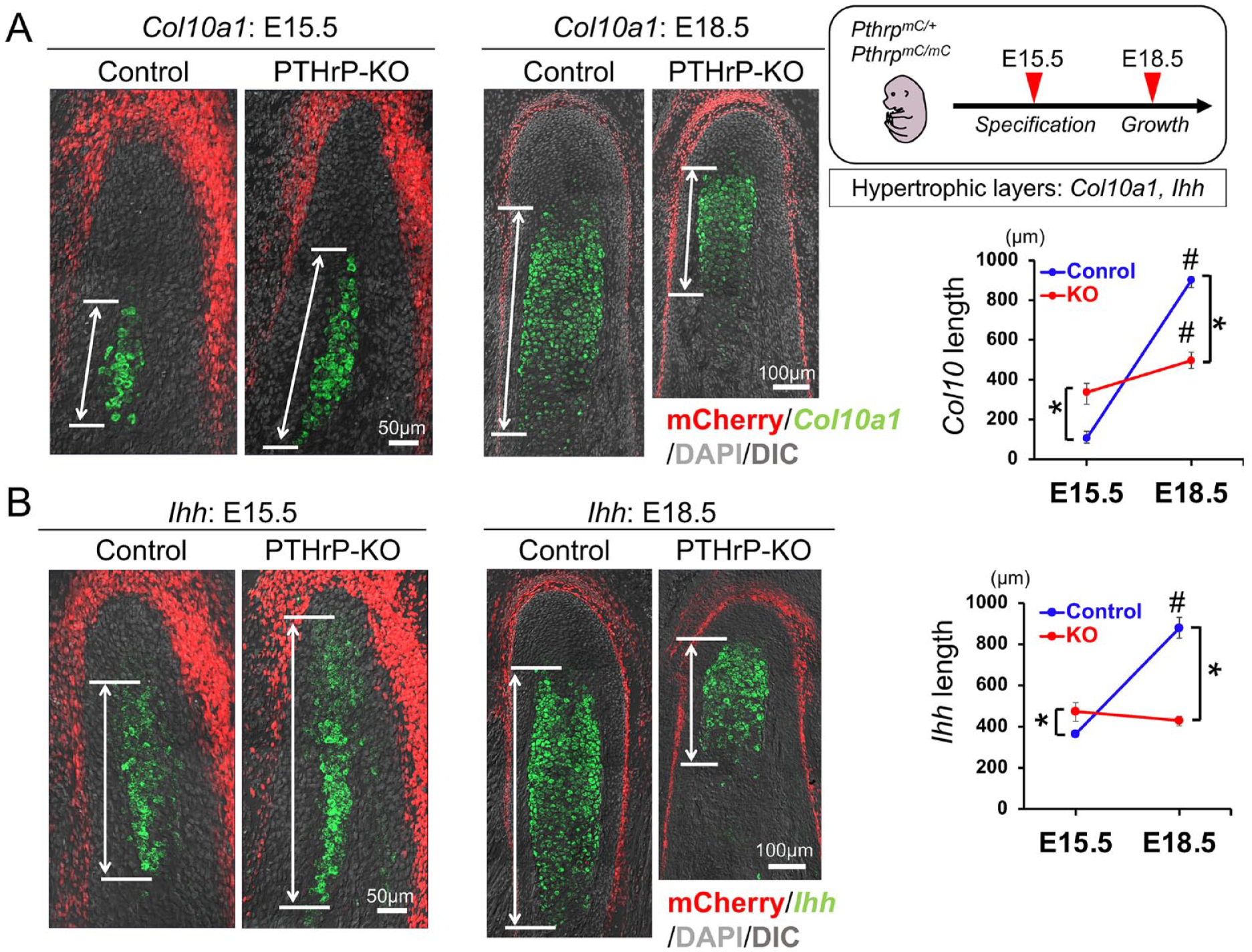
Parathyroid hormone–related protein (PTHrP) prevents premature chondrocyte differentiation and exhaustion. (
We also analyzed the expression of Ihh, which is a classical target gene of PTHrP in the growth plate. In early development of the long bone growth plate, PTHrP released from the periarticular layer inhibits Ihh expression in the prehypertrophic layer (Karaplis et al. 1994; St-Jacques et al. 1999). In line with this, the Ihh+ domain was expanded in the PTHrP-KO MCC compared to the Control at E15.5 (Fig. 4B, left panel). Interestingly, however, the Ihh+ domain was markedly diminished in the PTHrP-KO MCC at E18.5 (Fig. 4B, right panel), indicating that superficial layer–derived PTHrP is required to maintain Ihh expression in MCC chondrocytes. Therefore, PTHrP prevents premature differentiation of SOX9+ chondrocytes to Col10a1+ hypertrophic chondrocytes during active MCC growth, while also positively regulating Ihh expression from prehypertrophic chondrocytes, highlighting a unique mechanism of MCC chondrogenesis.
PTHrP Preferentially Promotes Runx2 Expression in the Polymorphic Layer
The polymorphic layer of the MCC develops from the periosteum of the mandibular ramus during early condylar morphogenesis and maintains its contiguity with the periosteum in the postnatal mandibular condylar process (Shibata et al. 2013; Kurio et al. 2018). Therefore, we examined the expression of transcription factors Runx2 and Sp7 (encoding Osterix) in the polymorphic layer, which are essential for osteoblast differentiation. Importantly, both Runx2 and Sp7 were abundantly expressed in the polymorphic layer at both E15.5 and 18.5 (Fig. 5A–a1,2). In the PTHrP-KO MCC, Runx2 expression was significantly reduced at both E15.5 and 18.5 (Fig. 5A–a1), while Sp7 expression was not altered (Fig. 5A–a2). These findings support the notion that Runx2 in the adjacent polymorphic layer may be an immediate target of PTHrP derived from the superficial layer of the MCC.
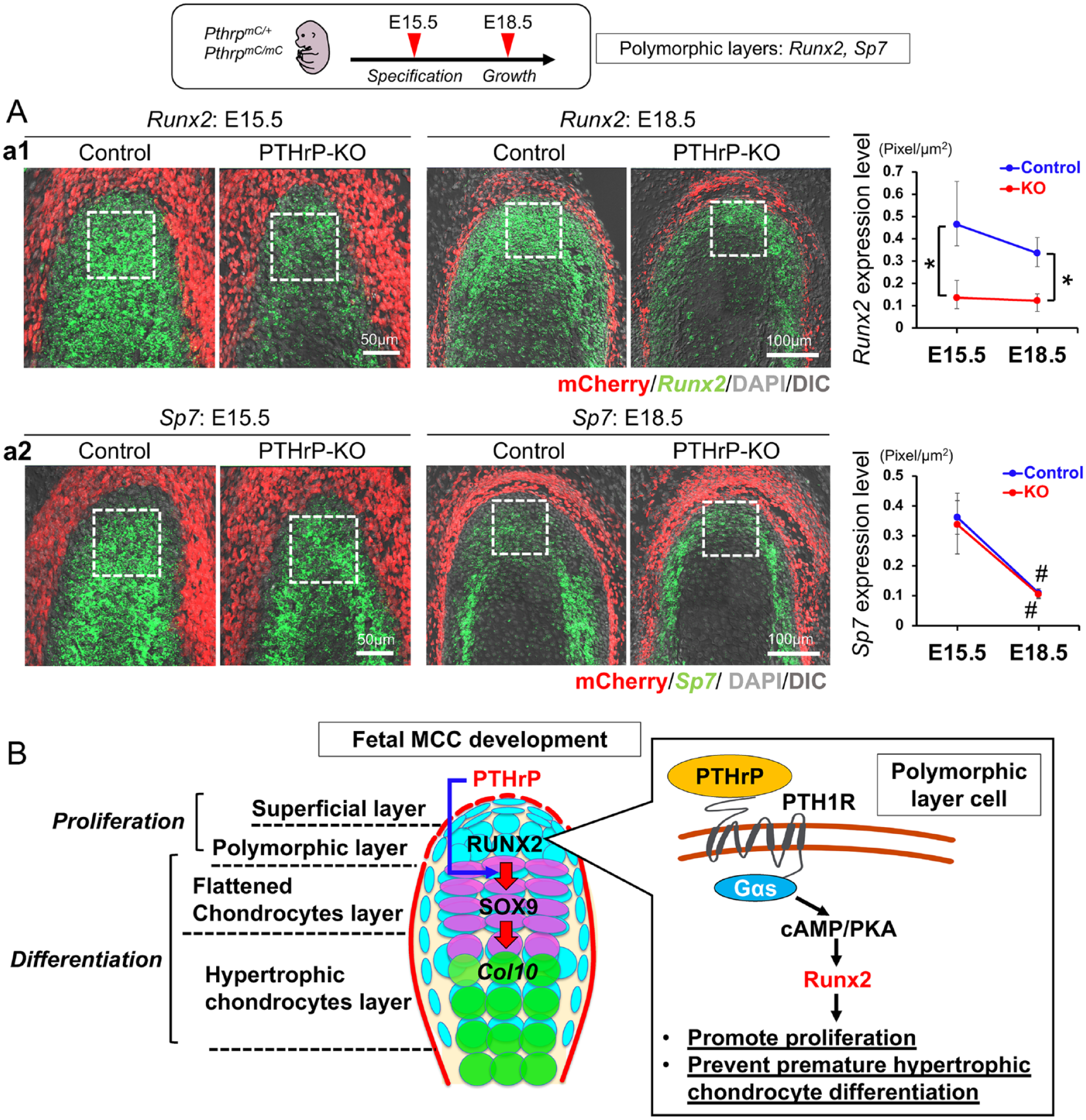
Parathyroid hormone–related protein (PTHrP) preferentially promotes Runx2 expression in the polymorphic layer. (
Taken together, our findings support the model that, in the MCC, PTHrP released from the superficial layer of the MCC acts on cells in the polymorphic layer, promotes proliferation of chondrocyte precursor cells in the polymorphic layer, and prevents their premature differentiation by stimulating Runx2 expression through the PTHrP-PTH1R signaling pathway (Fig. 5B). Of note, Runx2 has been shown to be required for chondrocyte proliferation in the postnatal MCC (Liao et al. 2019). A quantitative reduction in the RUNX2 functional activity causes cleidocranial dysplasia-like bone phenotype in mice (Lou et al. 2009) and may also induce the impairment in MCC chondrogenesis.
Discussion
In this study, we report that PTHrP is expressed in the superficial layer of the MCC and regulates the expression of Runx2 in the adjacent polymorphic layer through an organized diffusion. PTHrP released from the superficial layer acts on RUNX2+ chondrocyte precursor cells in the adjacent polymorphic layer and promotes proliferation and differentiation of RUNX2+ precursor cells into SOX9+ chondrocytes (see Fig. 5B for the model of fetal MCC development). Consistent with the concept that morphogen gradient regulates organogenesis (Gurdon and Bourillot 2001), PTHrP ligands diffuse in an organized manner, localized predominantly on the cell surface in the polymorphic layer to bind to its cognate receptor PTH1R, and subsequently localize intracellularly in the chondrocyte layer, presumably through PTH1R internalization. Lack of PTHrP reduces Runx2 expression in the MCC and inhibits proliferation of precursor cells in the polymorphic layer and accelerates differentiation of chondrocyte precursor cells to hypertrophic chondrocytes. As a result, lack of PTHrP causes defective endochondral bone formation of the mandibular condyle, associated with an altered zonal structure and the overall reduced size in the PTHrP-KO MCC. In other words, PTHrP-responsive Runx2 maintains cell proliferation of precursor cells in the polymorphic layer and prevents their premature terminal differentiation, which ultimately determines cartilage extension in the MCC. It has been previously reported that PTHrP-deficient mice show a reduction in thickness of all layers of the MCC (Suda et al. 1999). Although this observation is consistent with our present histologic examination, we have further clarified the mechanisms underlying this phenotype from 2 perspectives; first, PTHrP is expressed by the superficial layer and diffuses across the polymorphic and chondrocyte layers, and second, the reduction in the MCC thickness is due to accelerated differentiation and inhibition of cell proliferation, which is associated with reduced Runx2 expression.
In long bones, the resting zone of the epiphyseal growth plate plays central roles in the proper development and growth of endochondral bones by housing a population of chondrocytes expressing PTHrP (Mizuhashi et al. 2018). The population is recognized as skeletal stem cells, defined by their ability to undergo self-renewal and clonally give rise to columnar chondrocytes in the postnatal growth plate. These chondrocytes also possess the ability to differentiate into a multitude of cell types, including osteoblasts and bone marrow stromal cells, during skeletal development. It is not clear from the current study whether PTHrP+ cells in the superficial layer contribute to cells in other layers, including the polymorphic and chondrocyte layers of MCC. Unraveling the cell fate of PTHrP+ superficial layer cells in the MCC will facilitate our mechanistic understanding of MCC formation and maintenance.
In our study, although the detailed actions of PTHrP signaling in the MCC were not studied, we revealed the causal relationship between PTHrP and Runx2 but not PTHrP and Sp7 (Osterix). In the long bone, Runx2 promotes chondrocyte hypertrophy, whereas PTHrP delays this process (Li et al. 2004; Yoshida et al. 2004; van der Horst et al. 2005; Zhang et al. 2009). It has been reported that PTHrP downregulates Runx2 expression through the PKA signaling pathway in chick upper sternal chondrocytes (Li et al. 2004). PTHrP also prevents chondrocyte premature hypertrophy by inducing RUNX2 degradation in long bones in mice (van der Horst et al. 2005; Zhang et al. 2009). In other words, the action of Runx2 on chondrocyte differentiation in the MCC appears to be opposite to that of PTHrP in the long bone growth plate. Our results indicate that both PTHrP and Runx2 have prochondrogenic functions, suggesting that there is a unique mechanism of chondrocyte proliferation and differentiation in the MCC.
In conclusion, our study identifies a unique mechanism by which PTHrP stimulates MCC formation by maintaining Runx2 expression in chondrocyte precursor cells in the polymorphic layer through an organized diffusion. Modulating a PTHrP gradient within the MCC may provide a viable venue to intervene with the continual and adaptive growth of the MCC.
Author Contributions
C. Tsutsumi-Arai, N. Ono, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; Y. Arai, contributed to conception, data acquisition and analysis, drafted and critically revised the manuscript; A. Tran, M. Salinas, contributed to data analysis, drafted and critically revised the manuscript; Y. Nakai, S. Orikasa, contributed to data acquisition, drafted and critically revised the manuscript; W. Ono, contributed to conception, design, data interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231208175 – Supplemental material for A PTHrP Gradient Drives Mandibular Condylar Chondrogenesis via Runx2
Supplemental material, sj-docx-1-jdr-10.1177_00220345231208175 for A PTHrP Gradient Drives Mandibular Condylar Chondrogenesis via Runx2 by C. Tsutsumi-Arai, Y. Arai, A. Tran, M. Salinas, Y. Nakai, S. Orikasa, W. Ono and N. Ono in Journal of Dental Research
Supplemental Material
sj-pdf-2-jdr-10.1177_00220345231208175 – Supplemental material for A PTHrP Gradient Drives Mandibular Condylar Chondrogenesis via Runx2
Supplemental material, sj-pdf-2-jdr-10.1177_00220345231208175 for A PTHrP Gradient Drives Mandibular Condylar Chondrogenesis via Runx2 by C. Tsutsumi-Arai, Y. Arai, A. Tran, M. Salinas, Y. Nakai, S. Orikasa, W. Ono and N. Ono in Journal of Dental Research
Footnotes
Acknowledgements
We thank Dr. Catherine G. Ambrose (Department of Orthopedic Surgery at the University of Texas Health Science Center at Houston McGovern Medical School) for assistance in 3D-microCT scanning and Drs. Yoshihiro Komatsu and Hiroyuki Yamaguchi (Department of Pediatrics at the University of Texas Health Science Center at Houston McGovern Medical School) for assistance in skeletal staining.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institutes of Health (R01DE030630, R01DE026666 to N. Ono and R01DE029181, R01DE030416 to W. Ono). C. Tsutsumi-Arai was supported by the Japan Society for the Promotion of Science Fellowship for Research Abroad.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
