Abstract
Recapitulation of the natural healing process is receiving increasing recognition as a strategy to induce robust tissue regeneration. Endochondral ossification has been recognized as an essential reparative approach in natural jawbone defect healing. However, such an approach has been overlooked in the recent development of cell-based therapeutics for jawbone repair. Therefore, this study aimed to explore a bioinspired stem cell–based strategy for jawbone repair by mimicking the mesenchymal condensation of progenitor cells during the early endochondral ossification process. For this purpose, passage 3 of jawbone periosteum-derived cells (jb-PDCs) was cultured in our previously reported nonadherent microwells (200 µm in diameter, 148 µm in depth, and 100 µm space in between) and self-assembled into spheroids with a diameter of 96.4 ± 5.8 µm after 48 h. Compared to monolayer culture, the jb-PDC spheroids showed a significant reduction of stemness marker expression evidenced by flow cytometry. Furthermore, a significant upregulation of chondrogenic transcription factor SOX9 in both gene and protein levels was observed in the jb-PDC spheroids after 48 h of chondrogenic induction. RNA sequencing and Western blotting analysis further suggested that the enhanced SOX9-mediated chondrogenic differentiation in jb-PDC spheroids was attributed to the activation of the p38 MAPK pathway. Impressively, inhibition of p38 kinase activity significantly attenuated chondrogenic differentiation jb-PDC spheroids, evidenced by a significant decline of SOX9 in both gene and protein levels. Strikingly, the jb-PDC spheroids implanted in 6- to 8-wk-old male C57BL/6 mice with critical-size jawbone defects (1.8 mm in diameter) showed an evident contribution to cartilaginous callus formation after 1 wk, evidenced by histological analysis. Furthermore, micro–computed tomography analysis showed that the jb-PDC spheroids significantly accelerated bone healing after 2 wk in the absence of exogenous growth factors. In sum, the presented findings represent the successful development of cell-based therapeutics to reengineer the endochondral bone repair process and illustrate the potential application to improve bone repair and regeneration in the craniofacial skeleton.
Keywords
Introduction
Jawbone defects caused by trauma and tumorectomy possess high morbidity but poor prognosis (Allareddy et al. 2011; Kumar et al. 2016). Over 400,000 people have jawbone defects each year in the United States, with young adults being the most prevalent population (Allareddy et al. 2011). Currently, the gold standard for treatment is autogenous bone grafts. However, this approach is often restricted by the quantity of bone tissue at the donor site and grafting complications (Momoh et al. 2011; Marcucio et al. 2023). Therefore, developing cell-based strategies may provide significant advantages to critical-size bone defects, particularly in compromised conditions (Ji et al. 2017). To date, extensive research for jawbone repair focuses on enhancing intramembranous repair (Tan et al. 2021). However, very few of them have provided sufficient in vivo efficacy guarantees and thus support the clinical translation, evidenced by the nonequivalence between murine outcomes and human phase III studies (Galipeau and Sensébé 2018). Researchers attribute such inconsistent in vivo robustness to the discrepancy between current cell-based therapies and the endogenous mechanism of jawbone repair (Marcucio et al. 2023).
In the jawbone, both intramembranous and endochondral ossifications exist in development (Yuan and Chai 2019; Wong et al. 2021). Clinically, most fractures heal through a combination of endochondral and intramembranous ossification, which is directed by the mechanical environment (Marcucio et al. 2023). Specifically, a segmental jawbone defect has been primarily healed via endochondral ossification, in which a cartilaginous “soft callus” occurs in the center of the defect and is eventually replaced by bone (Wong et al. 2021; Marcucio et al. 2023). A previous study has shown that most cells forming the “soft callus” during mandibular fracture healing are derived from the periosteum (Leucht et al. 2008), and the latter represents promising skeletal stem cell sources to repair bone defects (Ding et al. 2022). Hence, developing a jawbone periosteum-derived cell (jb-PDC)–based strategy using “endochondral ossification” represents a promising therapeutic modality to repair jawbone defects (Kruijt Spanjer et al. 2017; Marcucio et al. 2023).
Endochondral ossification initiates with mesenchymal stem cell (MSC) aggregation, which is the prerequisite event for the progression of chondrogenic differentiation (Vinatier et al. 2009). To recapitulate this critical event, several cellular spheroid culture approaches, including centrifugation, low-adherence substrate, and hanging drop culture, have been proposed (Bhattacharjee et al. 2015). However, the aforementioned approaches are not only laborious but also difficult for in vivo applications, due to scalability challenges and uncontrolled complexity in 3-dimensional cell culture formats (Atala et al. 2012). Furthermore, despite the increasing popularity of using spheroids as building blocks to regenerate skeletal tissues (Laschke and Menger 2017; Lee and Lee 2022), the mechanisms behind their regenerative potential are still poorly understood.
Therefore, this study aimed to develop a biomimetic endochondral ossification strategy to repair critical-size jawbone defects based on the high-throughput formation of jb-PDC spheroids. Herein, the high-throughput production of chondrogenic spheroids could initiate the process of endochondral ossification upon implantation. The in vitro chondrogenic potentials of jb-PDC spheroids and the underlying mechanisms were explored. The in vivo endochondral repair efficacy of the jb-PDC spheroids was assessed orthotopically in mouse critical-size mandibular defects.
Materials and Methods
Animals
Six- to 9-wk-old C57BL/6J mice (The Jackson Laboratory, #000664) and Rosa26-mTmG mice (Muzumdar et al. 2007) (The Jackson Laboratory, #007676) were used. All the animal experiments were conducted in compliance with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines and with approval by the Animal Ethics Committee of the School of Stomatology, Wuhan University (No. S07921060A).
Isolation, Expansion, and Characterization of Mouse jb-PDCs
The primary jb-PDCs from 6- to 8-wk-old mouse mandibles were pooled from at least 3 animals and expanded until passage 3. For in vitro studies, triplicates (n = 3) were used in each condition, and all the in vitro experiments were repeated for 2 runs. Flow cytometry was performed to characterize the MSC stemness markers according to a previous report (Baustian et al. 2015). A trilineage differentiation assay was performed as previously described (Duchamp de Lageneste et al. 2018).
High-Throughput Formation of jb-PDC Spheroids
The expanded jb-PDCs were seeded in our previously reported agarose 24-well inserts containing ≈2,000 microwells (Leijten et al. 2016) at a concentration of 400,000 cells/well.
Animal Surgery
A circular penetrating mandibular critical-size defect (1.8 mm in diameter) was made as previously described (Wong et al. 2021). Spheroids or monolayer cells of 50,000 cells encapsulated in 5 µL collagen I gel were transplanted. Defects with gel alone or left empty were used as controls.
Online Content
See Appendix for the detailed materials and methods.
Results
Characterization of Mouse jb-PDCs
In vitro expanded mouse jb-PDCs (passage 3) displayed 76.8%, 97.3%, and 99.5% positivity for CD105, CD44, and CD29, respectively, whereas 99.5% were detected negative for endothelial and hematopoietic markers CD31 and CD45 (Appendix Fig. 1A–D). The osteogenic, chondrogenic, and adipogenic differentiation capacities of the expanded jb-PDCs were further confirmed by positive staining of Alizarin red S, Alcian blue, and oil red O, respectively (Appendix Fig. 1E).
High-Throughput Formation of jb-PDC Spheroids
To recapitulate aggregation that initiates chondrogenesis, the jb-PDCs were seeded onto a 24-well plate containing microwells 200 µm in diameter, 148 µm in depth, and a 100-µm space in between (Fig. 1A–C). After 3 h, 200 cells were self-assembled in each microwell and formed a spheroid with a diameter of 152.3 ± 15.0 µm (Fig. 1C–E). The obtained spheroids remained stable during 48-h in vitro culture, and the average diameter decreased to 96.4 ± 5.8 µm after 48 h (Fig. 1D, E). Compared to monolayer cells, the jb-PDC spheroids demonstrated distinct cytoskeleton reorganization over 48-h in vitro culture (Fig. 1F). Furthermore, the majority of cells within spheroids remained viable after 48 h (Fig. 1G).
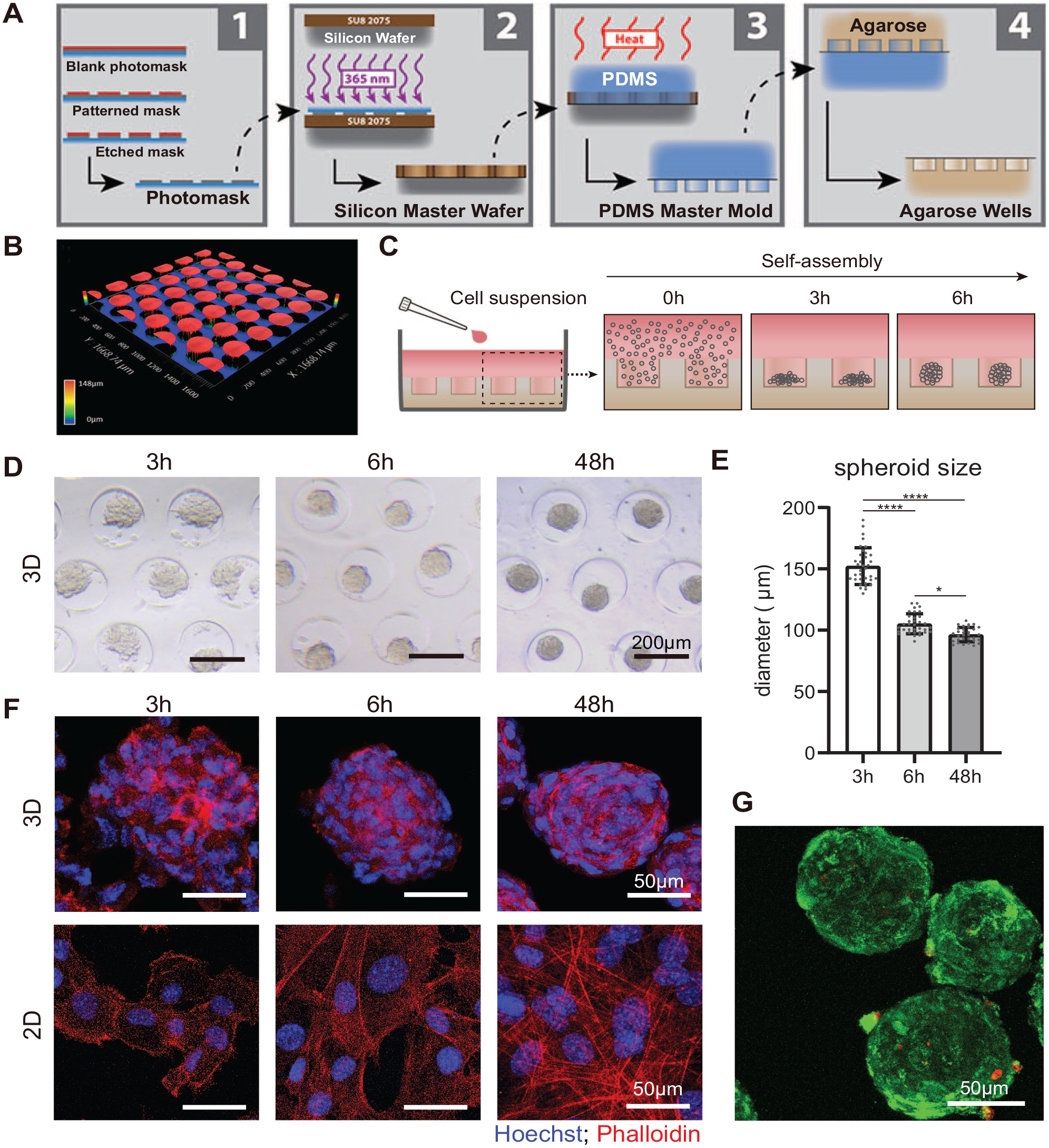
The high-throughput generation of jawbone periosteum-derived cell (jb-PDC) spheroids. (
The jb-PDC Spheroids Induced Early Chondrogenic Commitment
A key feature of chondrogenic commitment of skeletal stem cells is a decrease in their stemness markers. Therefore, we analyzed the expression of stemness markers using flow cytometry. Indeed, compared to the monolayer culture, the self-assembly of jb-PDCs into spheroids decreased cell surface expression of CD140a and CD105 by 1.34-fold and 1.20-fold, respectively (Appendix Fig. 2). This phenomenon resulted in a significant reduction of the skeletal stem cell population (CD140a+CD105+CD45−CD31−) from 51.17% in the monolayer to 29.83% in spheroids (Fig. 2A, B).
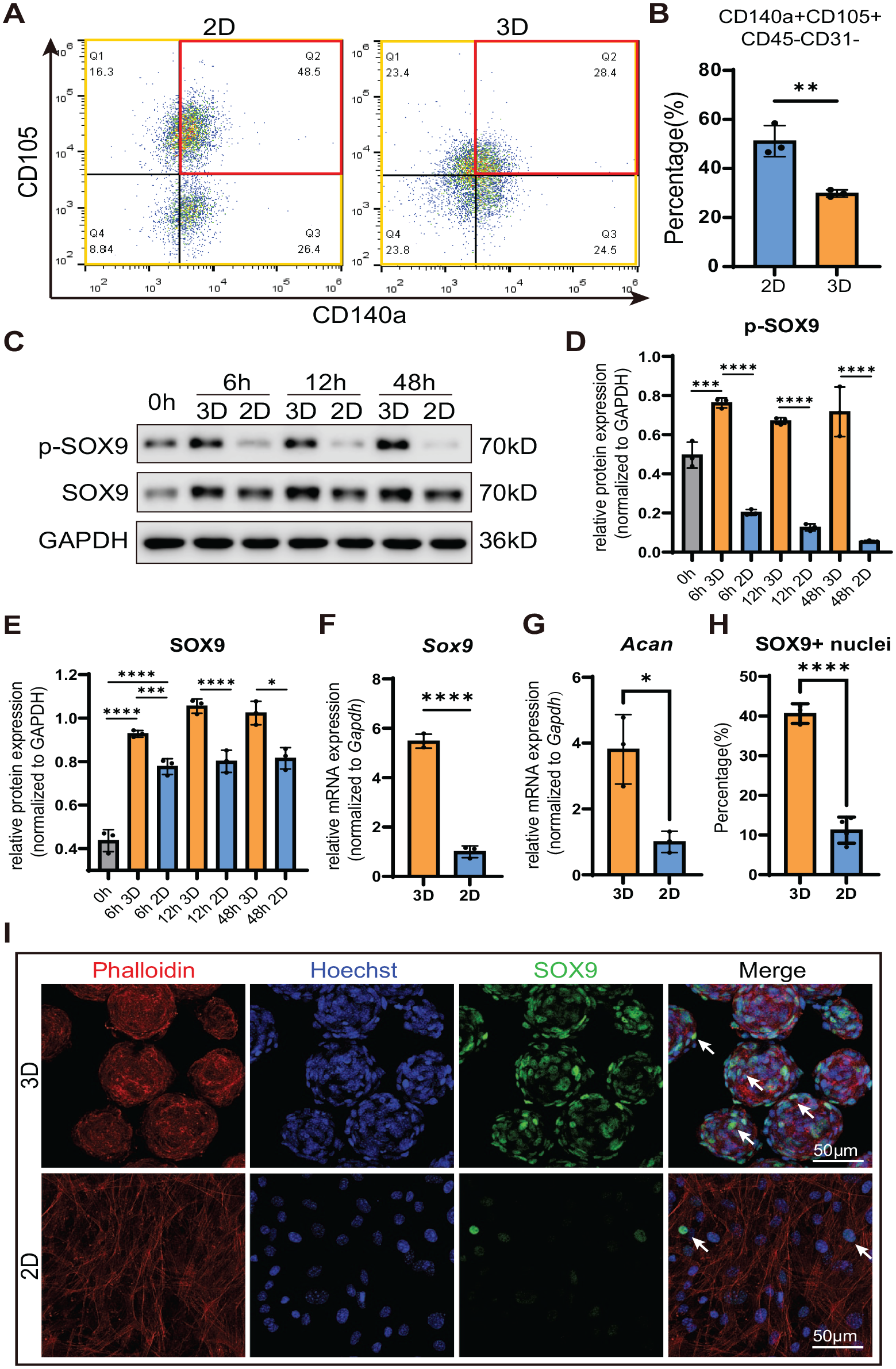
The jawbone periosteum-derived cell (jb-PDC) spheroids induced early chondrogenic commitment. (
Furthermore, upon chondrogenic induction, spheroids showed a significantly higher level of SOX9 and phosphorylated SOX9 after 6 h of self-assembling (Fig. 2C–E). Consistently, the messenger RNA (mRNA) levels of Sox9 (5-fold) and Acan (3-fold) were also significantly higher compared to the monolayer culture (Fig. 2F, G).
A SOX9-mediated transcription is a key event in chondrogenic commitment. To compare the SOX9 nuclear translocation in spheroids to those of monolayer culture, we imaged the jb-PDC spheroids using fluorescent confocal microscopy (Appendix Fig. 3) and represented them as stacked images (Fig. 2I). The jb-PDC spheroids showed an average of 40.6% SOX9+ nuclei, which was significantly higher than those of monolayer culture (Fig. 2H).
The jb-PDC Spheroids Enhanced the Noncanonical TGFβ p38 MAPK Pathway
To better explore the mechanism underlying the enhanced chondrogenic differentiation of jb-PDC spheroids, we conducted RNA sequencing (RNA-seq) analysis of monolayer cultured (2-dimensional [2D]) and spheroids (3-dimensional [3D]) of jb-PDCs. Compared to the monolayer (2D), jb-PDC spheroids showed a total of 1,255 upregulated genes (log2 FC(3D/2D) > 1, Q < 0.05) and 562 downregulated genes (log2 FC(3D/2D) < −1, Q < 0.05), suggesting the dramatic changes occurred within 48 h. Specifically, the chondrogenesis-related genes, including Sox9, Acan, Mef2c, Tgfb1, and Postn, were significantly upregulated in jb-PDC spheroids (Fig. 3A). Kyoto Encyclopedia of Genes and Genomes pathway and Gene Ontology enrichment analysis with upregulated genes revealed significant enrichment in TGFβ and MAPK signaling (Fig. 3B–D, Appendix Fig. 4A). Quantitative polymerase chain reaction (PCR) data further confirmed that jb-PDC spheroids showed significant upregulation of the TGFβ signaling downstream transcription factors Id1, Id2, and Id3 (Fig. 3E), as well as the p38 MAPK downstream targets Map3k5 (Ask1), Mef2c, Fos, and Cdc25b (Fig. 3F).
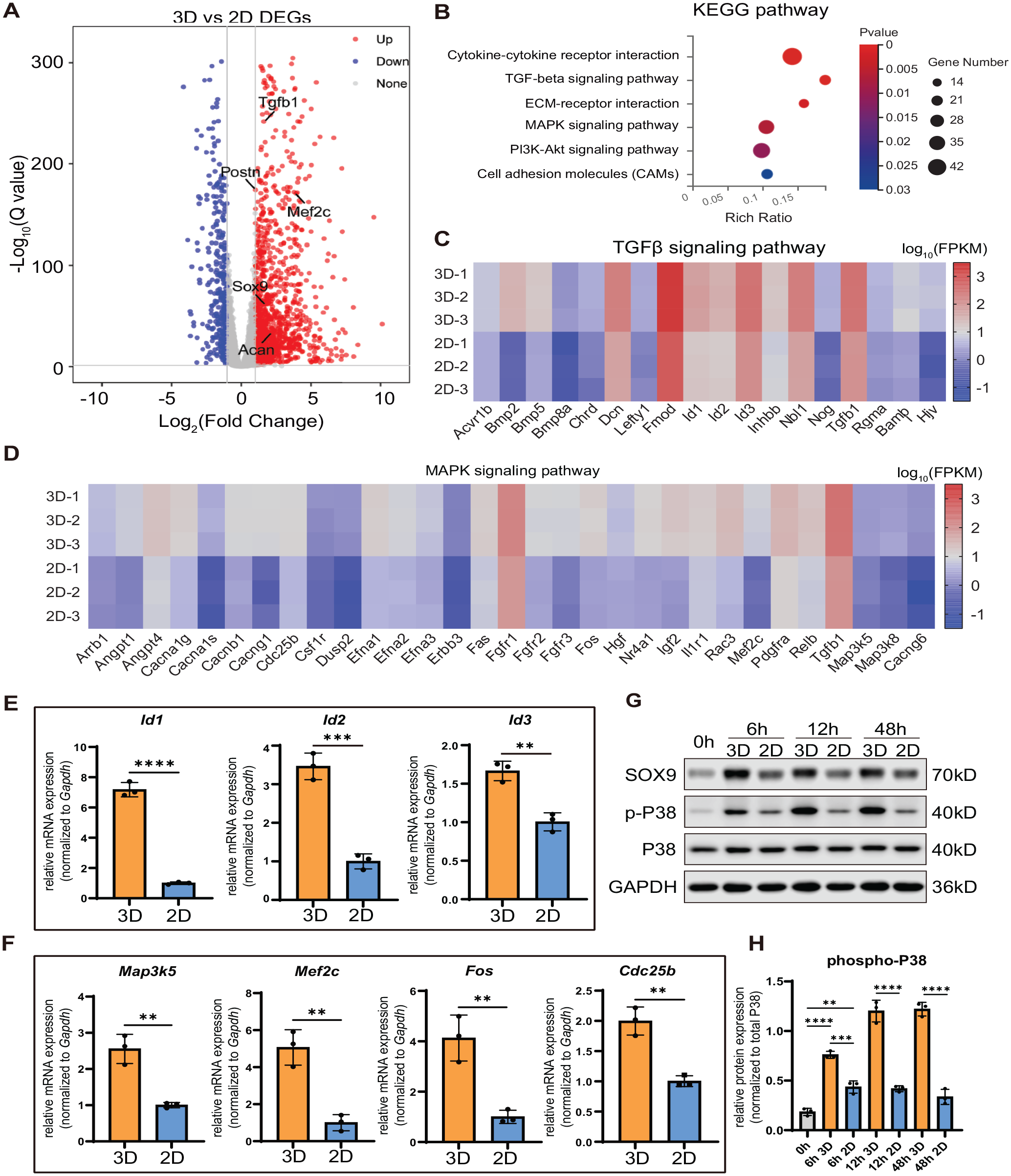
The jawbone periosteum-derived cell (jb-PDC) spheroids enhanced the noncanonical TGFβ p38 MAPK pathway. (
To verify the activation of TGFβ and p38 MAPK signaling pathway in jb-PDC spheroids, we performed Western blotting of jb-PDC spheroids and monolayer cells over 48-h in vitro chondrogenic induction. After 6 h, elevation of phosphorylated SMAD2/3 was observed for spheroids (3D) and monolayer cells (2D), indicating the activation of the canonical TGFβ signaling. However, the phosphorylation of SMAD2/3 in spheroids significantly decayed after 12 and 48 h of chondrogenic induction (Appendix Fig. 4B, C). In contrast, the spheroids (3D) showed significantly higher expression of phosphorylated P38 (p-P38) than monolayer-cultured PDCs (2D) over 48 h (Fig. 3G, H). Furthermore, such upregulation of p-P38 in spheroids (3D) was in accordance with the upregulation of SOX9 (Appendix Fig. 4D). Taken together, these results indicated that SOX9 expression was potentially regulated by activation of the p38 MAPK pathway.
P38 MAPK Pathway Modulates Chondrogenic Differentiation in jb-PDC Spheroids
Next, we conducted p38 kinase activity inhibition experiments to verify whether the p38 MAPK pathway played a key role in driving the SOX9-mediated chondrogenic commitments in jb-PDC spheroids. To this end, jb-PDCs in spheroids (3D) and monolayer (2D) were cultured for 48 h in chondrogenic media treated with or without different p38 kinase inhibitors, SB203580 (10 µM) or SB202190 (10 µM). Upon inhibition, jb-PDC spheroids showed a significant decrease of p38 MAPK downstream transcription factors Mef2c and Fos (Fig. 4A, B). Consistently, Sox9 and Acan, the chondrogenic marker genes, were also evidently downregulated in jb-PDC spheroids upon inhibition of p38 kinase activity (Fig. 4C, D).
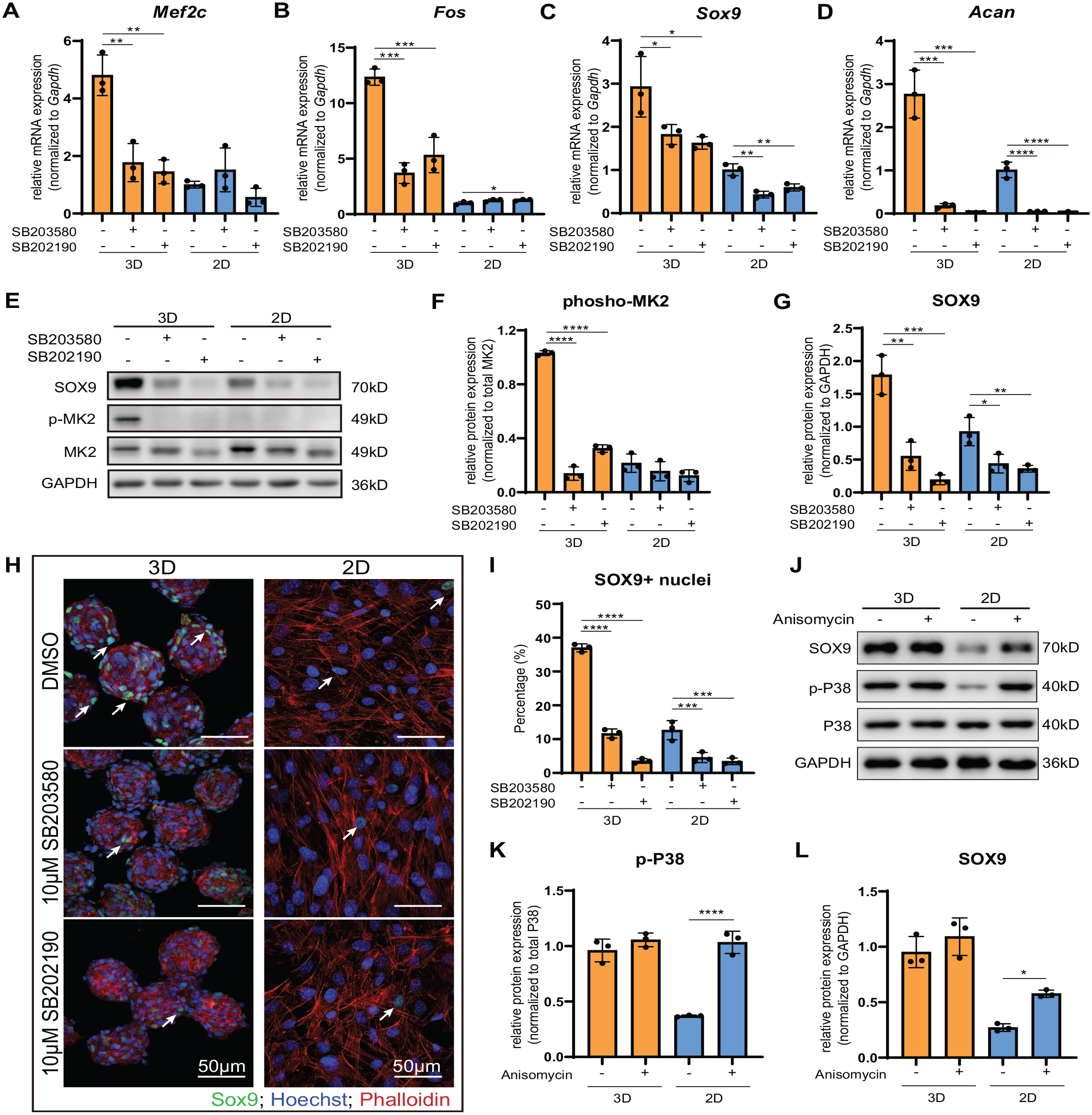
The p38 MAPK pathway modulates chondrogenic differentiation in jawbone periosteum-derived cell (jb-PDC) spheroids. (
Western blotting was then performed to testify the protein expression upon inhibition of p38 kinase activity (Fig. 4E–G). The jb-PDC spheroids treated with p38 kinase inhibitor showed a significant decrease of phosphorylated p38 MAPK downstream kinase MAPKAPK2 (p-MK2) (Fig. 4E, F). Consistently, a significant decline of SOX9 protein (2-fold) was also observed in jb-PDC spheroids treated with p38 kinase inhibitor (Fig. 4E, G). Furthermore, confocal microscopy demonstrated a significant reduction of the SOX9+ nucleus (white arrow) in the spheroids treated with 10 μM SB203580 or SB202190 (Fig. 4H, I).
Similarly, knocking down of p38 MAPK using small interfering RNAs also resulted in decreased SOX9 expression (Appendix Fig. 5). In addition, the role of p38 MAPK was furthered verified using p38 MAPK activator anisomycin (2.5 µM). An increase of SOX9 was observed after a transient stimulus (3 h) with anisomycin (Fig. 4J–L). These results suggest that the enhanced chondrogenic differentiation in spheroids was attributed to the upregulation of p38 MAPK signaling.
The jb-PDC Spheroids Repair Critical-Size Mandibular Defects via Endochondral Ossification
To assess whether jb-PDC spheroids are superior in bone healing, we transplanted jb-PDC spheroids (3D) or single-cell suspension (2D) encapsulated in rat-tail collagen I hydrogel laden into a mouse mandibular penetrating circular defect at the ramus (1.8 mm in diameter) (Fig. 5A). Fluorescence microscopy illustrated a comparable existence of the implanted mTomato+ cells in the callus after 3, 7, and 14 d in both groups (Appendix Fig. 6), suggesting the active integration of the donor cells in the defect. Notably, in both groups, the mTomato+ cells significantly decreased at 14 d, and the remnant mTomato signals were mainly observed within the newly formed trabeculae in the center of the callus (Appendix Fig. 6).
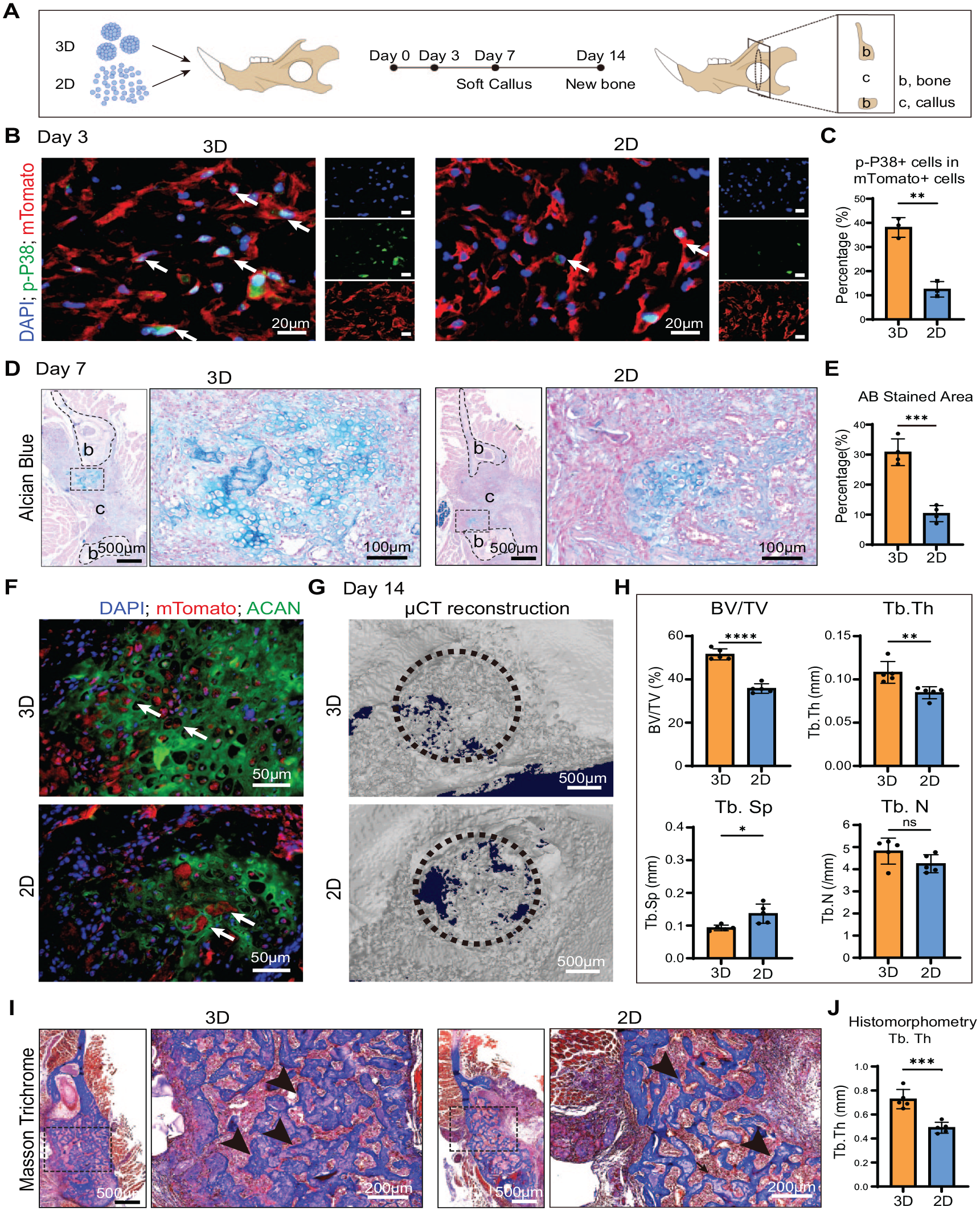
The jawbone periosteum-derived cell (jb-PDC) spheroids repaired critical-size mandibular defects via endochondral ossification. (
After 3 d, we performed immunofluorescent staining of p-P38 on the in vivo implanted mTomato+ cells to verify the difference in cell signaling activation between spheroids (3D) and single-cell suspension (2D). Our data showed that in the spheroids (3D) group, about 40% of mTomato+ cells expressed p-P38 (arrows), which was significantly more than that of single-cell suspension (2D) (Fig. 5B, C).
After 7 d, hematoxylin and eosin (HE) and Alcian blue staining revealed that the empty defects (blank group) and the defects filled with bare gel (scaffold group) showed only fibrous tissue infiltration without a characteristic cartilaginous callus structure (Appendix Figs. 7 and 8). In contrast, the defects implanted with jb-PDC spheroids (3D) and monolayer cultured cell suspension (2D) showed obvious cartilaginous callus composed of lacuna structure and proteoglycan matrix deposition in addition to fibrous tissue infiltration (Appendix Fig. 7, Fig. 5D). Furthermore, histomorphometry of Alcian blue staining revealed that the defects filled with jb-PDC spheroids (3D) showed significantly more cartilaginous callus formation compared to the defects filled with single-cell suspension (Fig. 5D, E). We further costained the cartilage-specific proteoglycan core protein (ACAN) on the mTomato+ samples at 7 d. We observed the existence of mTomato+ cells within the cartilage lacuna and ACAN+ area in both the spheroid and the monolayer cell groups (Fig. 5F), indicating the direct contributions of the donor cells to the cartilaginous callus formation.
After 14 d of healing, micro-CT analysis and HE staining revealed that the vast area of empty defects and the defects filled with bare collagen gel remained unhealed. However, a small amount of new bone formation occurred on the border of the defect site (Appendix Figs. 9 and 10). In contrast, the defects filled with cells, regardless of spheroids or monolayer cell suspension, led to evident bone bridging (Fig. 5G), suggesting the enhanced regenerative capacity of the implanted cells. The quantitative analysis of micro-CT images further revealed that the jb-PDC spheroid (3D) treated samples showed significantly higher bone volume/tissue volume (BV/TV) and trabecular thickness (Tb. Th) than monolayer cell suspension (2D) treated groups (Fig. 5H). Consistently, HE and Masson trichrome staining illustrated prominent newly formed trabecular structures in the defect area filled with jb-PDC spheroids (3D) or single-cell suspension (2D) (Appendix Figs. 10, 11, Fig. 5I). Notably, the mTomato+ cells were observed within the newly formed trabeculae, indicating the active contribution of implanted cells to the new bone formation (Appendix Fig. 12). Moreover, histomorphometry of trabecular thickness in Masson trichrome staining suggested significantly thicker trabeculae in the 3D group compared to the 2D group (Fig. 5J), which is consistent from quantitative micro-CT analysis.
Discussion
The conceptual and technical merging of biology and engineering principles to recapitulate the native defect-healing process is receiving increasing recognition within regenerative medicine. In this work, we developed a biomimetic bioengineering approach to generate jb-PDC spheroids with chondrogenic potentials in a high-throughput manner. Furthermore, the jb-PDC spheroids were able to repair critical-size mandibular defects via the endochondral ossification pathway, which mimics the native healing process.
Periosteum-derived cells have recently been shown to contain a unique skeletal stem cell population with distinct functions during endogenous bone repair (Jeffery et al. 2022). Our recent meta-analysis revealed that cells from periosteum possess equivalent in vivo bone-forming capacity as bone marrow mesenchymal cells (Zhu et al. 2023). However, the jawbone periosteal cells currently are understudied. It was recently reported that human jawbone periosteum can be easily accessed during maxillofacial surgeries, providing convenience for cell isolation (Ding et al. 2022). Furthermore, periosteal cells from jawbone possess specific progenitor subsets active in jawbone development and defect healing (Ding et al. 2022). Specifically, the mandible-derived periosteal cells were shown excellent potentials in both in vitro and in vivo chondrogenic and osteogenic differentiation potential (Groeneveldt et al. 2020). Hence, this less explored progenitor cell source possesses critical advantages in the development of novel cell therapies to treat large jawbone defects.
Clinically, segmental jawbone defects heal primarily via endochondral ossification with a small amount of direct bone formation, and cells from the periosteum are the main contributors to the cartilaginous callus (Wong et al. 2021; Marcucio et al. 2023). During this process, cell condensation is the prerequisite event for the following cartilaginous callus and bone formation. To date, in vitro cell aggregation was frequently obtained through the use of pellets containing a large number of cells (>2 × 105 cells). However, the use of such methods is laborious and has resulted in diffusion-related challenges that hinder the in vitro differentiation and in vivo tissue maturation to their final phenotype upon implantation (Nilsson Hall et al. 2019). To overcome such limitations, we demonstrated in this study a high-throughput approach to generate stable jb-PDC spheroids using a nonadherent microwell array prepared by our previously described soft lithographic technique (Leijten et al. 2016). The submicron size of the obtained jb-PDC spheroids is indeed ideal for the nutrients’ diffusion (Langan et al. 2016), which overcomes the critical limiting factor of cell viability and homogeneous differentiation in the conventional approaches (Mueller and Tuan 2008; Markway et al. 2010; Lyashenko et al. 2011; Watts et al. 2013).
In this study, the jb-PDC spheroids induced early chondrogenic commitment early in the 48-h in vitro culture, evidenced by the upregulation of SOX9, the master regulatory factor for early chondrogenesis (Hata et al. 2017). Our flow cytometry data showed that the skeletal stem cell population (CD140a+ CD105+CD31−CD45−) (Ortinau et al. 2019) had rapidly reduced within the spheroids, suggesting the initiation of lineage-specific differentiation. Furthermore, we observed that jb-PDC spheroids intensified SOX9 nuclear staining early in 48 h in vitro and correlated with increased expression of genes driven by SOX9-mediated transcription. We also explored the mechanism driving such enhanced SOX9-mediated chondrogenesis. Our data revealed that the elevated SOX9 expression in jb-PDC spheroids correlates highly with the elevated p38 MAPK signaling rather than SMAD signaling, although both pathways can regulate SOX9 expression (Coricor and Serra 2016). The exact mechanism driving the elevation of p38 MAPK signaling still needs further investigation. One possible explanation is that the mechanical cues within cell–cell and cell–matrix interaction thus subsequently activate the p38 MAPK pathway (Xia et al. 2015). Notably, inhibition of the p38 activity significantly attenuated the SOX9-mediated transcriptions in both gene and protein levels. Taken together, our data for the first time revealed that spheroid formation elevated p38 MAPK, the noncanonical TGFβ signaling (Zhang 2009), hence enhancing the SOX9-mediated chondrogenic differentiation.
As proof of concept, the in vivo endochondral potentials of jb-PDC spheroids were further validated in mouse critical-size jaw bone defects. For this purpose, we created a circular (1.8 mm in diameter) penetrating defect at the ramus. The empty defect showed less than 20% spontaneous healing over 2 wk. Such findings confirmed the validity of the defect as “critical size,” which does not heal over a specified period (Shah et al. 2016). Importantly, compared to the bare scaffolds group, the cell-laden groups showed active integration in the cartilaginous callus formation on day 7 followed by superior bone formation over 2 wk, indicating the significant implications of cell therapies in the reconstruction of critical-size defects. Notably, compared to the single-cell suspension (2D), the implanted mTomato+ jb-PDC spheroids showed a significant increase of p-P38 staining on day 3, which correlated with significantly more cartilaginous callus formation on day 7 and a greater amount of new bone formation on day 14. Such observations suggest that the jb-PDC spheroids maintained their chondrogenic potentials driven by activation of p38 MAPK signaling and actively contributed to the endochondral repair of the critical-size jawbone defects. Indeed, we observed a significant decrease of mTomato+ cells within the newly formed bone callus on day 14. This could be attributed to cell apoptosis following chondrocyte hypertrophy or active bone remodeling (Yang et al. 2014; Bolamperti et al. 2022). Further studies with advanced lineage tracing and imaging techniques are expected to elucidate the cell fate change during defect healing (Chen et al. 2018; Jing et al. 2019).
In conclusion, this study reports an endochondral ossification strategy to repair critical-size jawbone defects using chondrogenic jb-PDC spheroids generated through a high-throughput manner. The self-aggregation of jb-PDCs into spheroids induced early SOX9-mediated chondrogenic commitment via activating the p38 MAPK, the noncanonical TGFβ signaling. Upon in vivo implantation, the jb-PDC spheroids further accelerated endochondral ossification in jawbone defects, mimicking the natural defect healing. The presented findings represent the successful development of cell-based therapeutics to reengineer the endochondral bone repair process and illustrate the potential mitigation of currently unmet clinical needs to improve bone repair and regeneration in the craniofacial skeleton.
Author Contributions
J. Zhu, contributed to data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; S. Zhang, S. Jin, C. Huang, contributed to data acquisition and interpretation, critically revised the manuscript; B. Shi, Z. Chen, contributed to data interpretation, critically revised the manuscript; W. Ji, contributed to conception, design, data interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231205273 – Supplemental material for Endochondral Repair of Jawbone Defects Using Periosteal Cell Spheroids
Supplemental material, sj-docx-1-jdr-10.1177_00220345231205273 for Endochondral Repair of Jawbone Defects Using Periosteal Cell Spheroids by J. Zhu, S. Zhang, S. Jin, C. Huang, B. Shi, Z. Chen and W. Ji in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank John F. Edelbrock from Northwestern University for his great assistance in PDMS microarray master mold fabrication and illustration, as well as Prof. Lijia Ma from Westlake University and Prof. Yiwei Li from Huazhong University of Science and Technology for their insightful discussions and suggestions.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A patent pertaining to this work has been filed and is pending: “Preparation of cartilaginous microtissue for rapid repair of jawbone defects (in Chinese)” (inventors: W. Ji, J. Zhu, S. Zhang, and S. Jin).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 82170931), the Fundamental Research Funds for the Central Universities (No. 2042022kf1172), the Wuhan University Specific Fund for Major School-level Internationalization Initiatives (WHU-GJZDZX-RC07), and the ITI research grant (1721-2022).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
