Abstract
Aging affects the number and function of gamma-delta (γδ) T cells in a tissue-specific manner, modifying the risk for inflammatory disease. These aging-related γδT-cell variations in gingival tissues that could increase the risk for inflammation and periodontal disease remain unknown. Here we sought to identify quantitative and qualitative variations in gingival γδT cells associated with aging that could have an impact in oral immunoinflammatory responses. For this, gingival tissues from young (4 mo) and aged (24 mo) male and female mice were collected and analyzed by flow cytometry. Cell suspensions were stimulated and stained with eFluor450 (cell viability), anti-CD45 (hematopoietic cells), anti-CD3 (lymphocytes), anti-TCRγδ (γδT cells), anti–IL-15rα (cell proliferation), and anti–Notch-3 (senescence marker). Detection of intracellular cytokines IL-17A and interferon γ (IFNγ) was performed. Gingival expression of specific γ- and δ-chains and cytokines was evaluated by quantitative reverse transcription polymerase chain reaction. A significantly higher number of IL-17A–producing γδT cells and IL-17A expression levels were observed in gingival tissues from aged females but not males. Similarly, the number of gingival Notch-3+ γδT cells increased with aging only in females. IL-15rα was not detected in gingival γδT cells. Chains γ1, 2, 4, 5, 6, and 7 as well as δ1, 2, 4, and 6 were detected. Detection levels of all γ chains except γ1 as well as δ1 and δ2 changed with aging in males, females, or both. Interestingly, number of IL-17A–producing conventional T cells similarly increased with aging only in females. Both sexes showed increased IFNγ+ conventional T-cell numbers with aging; however, it reached significance only in females. In conclusion, the number of gingival IL-17A–producing γδT cells and IL-17A expression increase naturally with aging specifically in females. This sexual dimorphism in gingival γδT and conventional Th17 cell numbers and phenotypes suggests distinct aging-related mechanisms of periodontitis in males and females.
Introduction
Although epidemiological data indicate that aging is a risk factor for periodontal disease (PD), the cellular and molecular basis for this observation remains to be elucidated (Ebersole et al. 2016). This is an important question because aside from the local effects/sequelae, PD is a risk factor for several systemic disorders such as diabetes, cardiovascular disease, and Alzheimer’s disease, which also increase with age (Lalla and Papapanou 2011; Lockhart et al. 2012; Dominy et al. 2019).
A common factor in aging-related diseases (including PD) is chronic inflammation, which involves quantitative and qualitative modifications of the immune system that lead to greater susceptibility to infections, neoplasia, autoimmune manifestations (immunosenescence), and a progressive increase in the proinflammatory state (inflammaging) (Fulop et al. 2017; Franceschi et al. 2018).
Specifically, gamma-delta (γδ) T cells are an essential component of the innate immunity at mucosal surfaces; reside in high abundance in epithelial surfaces, including the oral epithelium; maintain tissue homeostasis with the environment; and play a critical role for controlling intracellular bacterial infections due to their cytotoxic properties (Nielsen et al. 2017; Krishnan et al. 2018; Ribot et al. 2021). Different γδT-cell subsets have been described based on their gamma (γ) variable chain expression (Vγ1–7) in mice (Heilig and Tonegawa 1986), whereas human γδT-cell subsets are distinguished by Vδ chain usage (Nielsen et al. 2017). While Vγ6 cells develop only in the embryonic thymus and are maintained at local tissue sites by self-renewal, Vγ4 and Vγ1 cells develop in both the embryonic and adult thymus and can be reconstituted from the circulation throughout life. Moreover, Vγ6 cells appear to be dependent on the microbiota, while Vγ4 and Vγ1 cells are triggered by mechanical damage (Chen et al. 2022). Specific functions of immune surveillance and wound healing (Vγ4 and Vγ6 in skin and oral mucosa) and tissue repair (Vγ7 in the intestine) have been shown (Fischer et al. 2020; Ribot et al. 2021).
Recent evidence indicates that γδT cells maintain oral homeostasis through mutual interactions with the oral microbiota, with the majority of gingival γδT cells being tissue-resident Vγ6+ cells (persisting locally) and, to a lesser extent, Vγ1+ and Vγ4+ cells (Wilharm et al. 2019). Vγ6+ cells represent the major source of IL-17–producing cells in the gingiva; however, whether γδT cells play a protective or pathogenic role in PD remains controversial and seems to depend on the used models (ligature vs. Porphyromonas gingivalis infection) (Krishnan et al. 2018; Wilharm et al. 2019; Barel et al. 2022).
Aging affects the number and function of γδT cells in a tissue-specific manner. Thus, while the number of γδT cells is known to decrease by aging in the circulation (Argentati et al. 2002; Colonna-Romano et al. 2002), we have recently demonstrated that the number of resident γδT cells is increased (5-fold) in visceral adipose tissue of aged mice, which contributed to local and systemic chronic inflammation and metabolic dysfunction (Bruno et al. 2022). A recent report indicated that the number of gingival γδT cells is reduced in male mice immediately after birth until 40 wk of age (adult mice), and a prolonged ablation of γδT cells led to elevated gingival inflammation and subsequent alterations of the microbial diversity (Wilharm et al. 2019). Nevertheless, whether aging affects the number and phenotypes of gingival resident γδT cells remains unknown.
The factors and mechanisms contributing to tissue-specific differences in γδT-cell numbers and phenotypic changes associated with aging are not fully understood. Potential tissue-related environmental factors could render γδT cells either more resistant or susceptible to undergoing apoptosis or decreasing their proliferative capacity with aging. Tissue-specific cytokine signaling such as IL-15/IL-15rα or cellular senescence could contribute to these differences (Lodolce et al. 1998). It has also been shown that γδT cells directly recognize microbial antigens and respond, producing cytokines such as IL-17A and interferon γ (IFNγ), limiting transepithelial pathogen invasion and protecting mucosal tissues (Zeng et al. 2012; Edelblum et al. 2015). Therefore, aging-related variations in γδT-cell cytokine responses could also contribute to chronic infection and inflammation.
Herein, we sought to identify quantitative and qualitative variations in gingival γδT cells associated with aging in both males and females that could have an impact on oral immunoinflammatory responses.
Materials and Methods
Animals
All mouse procedures were performed in accordance with the approved protocol guidelines by the Institutional Animal Care & Use Committee (IACUC) of the University of Kentucky (Protocol # 2016-2385). Young (4 mo from Jackson Laboratory) and aged (24 mo from National Institute of Aging [NIA]) C57BL/6 mice were used (9 females and 9 males/age group). Gingival tissues from 3 quadrants/mouse were collected for flow cytometry analysis. A total of 6 pools (3 mice/pool) for each age group were independently analyzed. The remaining quadrants were used for RNA isolation and reverse transcription polymerase chain reaction (RT-PCR). These studies comply with the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines. See Appendix for further details on sample size estimation and methods for periodontal bone loss determination.
Flow Cytometry Analysis
Anesthetized mice with 5% isoflurane were euthanized by exsanguination and immediately perfused with 30 mL physiological saline via cardiac ventricles. Gingival tissues surrounding the upper and lower molars were dissected and collected in pools of 3. Tissues placed in RPMI (ThermoFisher) with 5% fetal bovine serum (FBS) (GE Health Care) were digested with Collagenase Type-II (Stem Cell) and 1 µL/mL DNAse (Life Technologies) in the presence of 100 U/mL penicillin/streptomycin (ThermoFisher) for 1 h at 37°C shaking. Then, 0.5 M EDTA (MilliporeSigma) was added during the last 5 min. After being filtered (70 µm Cell Strainer; VitaScientific) and resuspended in RPMI with 5% FBS, cells were counted and 3 × 106 cells stimulated with 50 ng/mL phorbol myristate acetate (Cayman), 2.5 μg/mL ionomycin (Cayman), and 1 µL/mL Cytoperm/Cytofix with Golgi Plug (BD) and incubated for 4 h in 37°C, 5% CO2. After being washed and resuspended in 1× phosphate-buffered saline (PBS) (Gibco), cells were incubated for 30 min at 4°C with eFluor450 for cell viability (Life Technologies) and Fc-block (Biolegend), followed by staining of extracellular receptors using anti-CD45 (APC-Cy7), anti-CD3 (FITC), anti-γδT (PerCP-CY5.5), IL-15rα (PE), and anti–Notch-3 (AF647) (Biolegend) for 30 min at 4°C (Appendix). Cells were then fixed with 4% paraformaldehyde (Biolegend) overnight, washed, and permeabilized with 1× BD Perm/Wash buffer (BD) for 15 min. Furthermore, cells were washed and resuspended in the same buffer containing IFNγ (AF700) and IL-17A (Violet 605) antibodies (Biolegend) for 30 min at 4°C. Finally, cells were washed and resuspended in 1× PBS with 5% FBS for flow cytometry analysis. For each antibody, corresponding isotype conjugates and UltraComp eBeads (LifeTechnologies) were used as control. For each sample, at least 10,000 events were read in a flow cytometer FACScalibur (BD Biosciences).
T-Cell Receptor–Variable-Segment γ and δ Profiling by RT-PCR
Total RNA was extracted from 3 pooled gingival tissues using the RNeasy Plus Mini-Kit (Qiagen). RNA (1 µg) was reverse transcribed using SuperScript III First-Strand Synthesis SuperMix for quantitative RT-PCR (qRT-PCR) (Invitrogen 11752050), and the resulting complementary DNA (cDNA) (50 ng) was amplified by use of Taq PCR Kit (New England Biolabs E5000S). The primers used to detect variable γ and δ chains were described previously and are listed in Appendix Table 2 (Heilig and Tonegawa 1986; Andrew et al. 2005). The following reaction conditions were used: denaturation at 94°C, annealing between 46° and 55°C (see Appendix Table 2 for specific temperatures), and extension at 72°C, for 38 cycles. PCR products were run on a 2% agarose gel and visualized using ethidium bromide on the ChemiDoc MP Imaging System (Bio-Rad).
Cytokine Expression by qRT-PCR
Expression levels for cytokines IL-1β, IL-6, and IL-17A were evaluated in gingival tissues from young and aged male and female mice. RNA was extracted using RNeasy Plus mini kit (Qiagen) and measured on a NanoDrop (ThermoFisher) for concentration and quality. RNA (1 µg) was then reverse transcribed using the qScript cDNA Synthesis Kit (Quanta). Amplification of mouse cytokine genes was performed with the QuantStudio 3 Real-Time PCR System (Applied Biosystems) using TaqMan Fast Advanced Master Mix (ThermoFisher) and TaqMan Assays for IL-1β (Mm00434228_m1), IL-6 (Mm00446190_m1), IL-17A (Mm00439618_m1), and endogenous control GAPDH (Mm99999915_g1) (Appendix Table 3). Gene expression was analyzed by calculating fold change values using the 2−ΔΔct method.
Statistical Analysis
Continuous variables were analyzed using Hotelling’s T-squared test, 2-sample t tests, and Mann–Whitney test. Regression analysis using simple linear regression was performed using gene counts as outcomes while using age, experimental group, and their interaction as covariates of interest. The level of significance used was α = 0.05. All analyses were performed in JMP Version 16 (SAS Institute) and R version 4.2.0 (R Core Team).
Results
The gating strategy for evaluating γδT cells in gingival cell suspensions from mice is shown in Figure 1A. The frequency and number of gingival γδT cells significantly increased about 2-fold (P = 0.05) and 10-fold (P = 0.014), respectively, in aged gingival tissues (Fig. 1B); however, when data were disaggregated by sex, significantly higher numbers of γδT cells (20-fold increase, P = 0.015) were seen in gingival tissues from females but not males (Fig. 1C). Interestingly, the total number of conventional αβT cells also increased significantly with aging in females (Appendix Fig. 1).
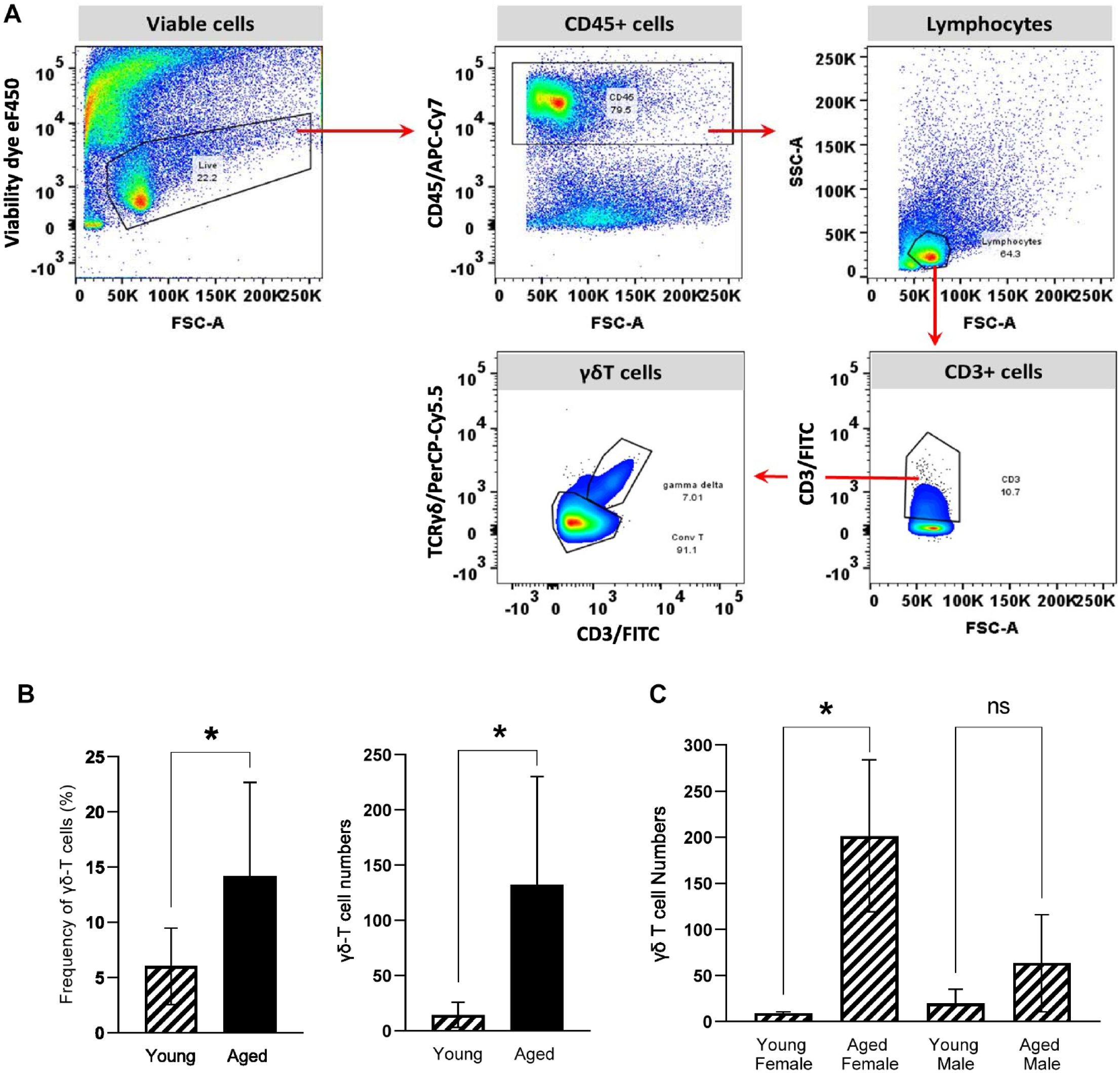
Effect of aging on numbers of gingival γδT cells. (
Representative plots to phenotype cells for intracellular cytokines are shown in Figure 2A. In general, γδT cells were positive only for IL-17A but not IFNγ; however, conventional αβT cells produced both cytokines, with IFNγ+ cells being the more abundant cell type. The number of gingival IL-17A+ γδT cells was significantly elevated with aging (10-fold, P = 0.0293); nevertheless, only females showed a significant increase in this subset of γδT cells (15-fold, P = 0.024). Variations in the number of IFNγ+ γδT cells were not seen with aging (Fig. 2B). Number of IL17A+ and IFNγ+ conventional αβT cells increased about 10-fold (P = 0.034) and 5-fold (P = 0.011), respectively, with aging; however, similarly as observed in γδT cells, data analysis by sex showed a significant increase only in gingival tissues from females for both cytokines (15-fold for IL-17A, P = 0.022; 12-fold for IFNγ, P = 0.0012) (Fig. 2C).
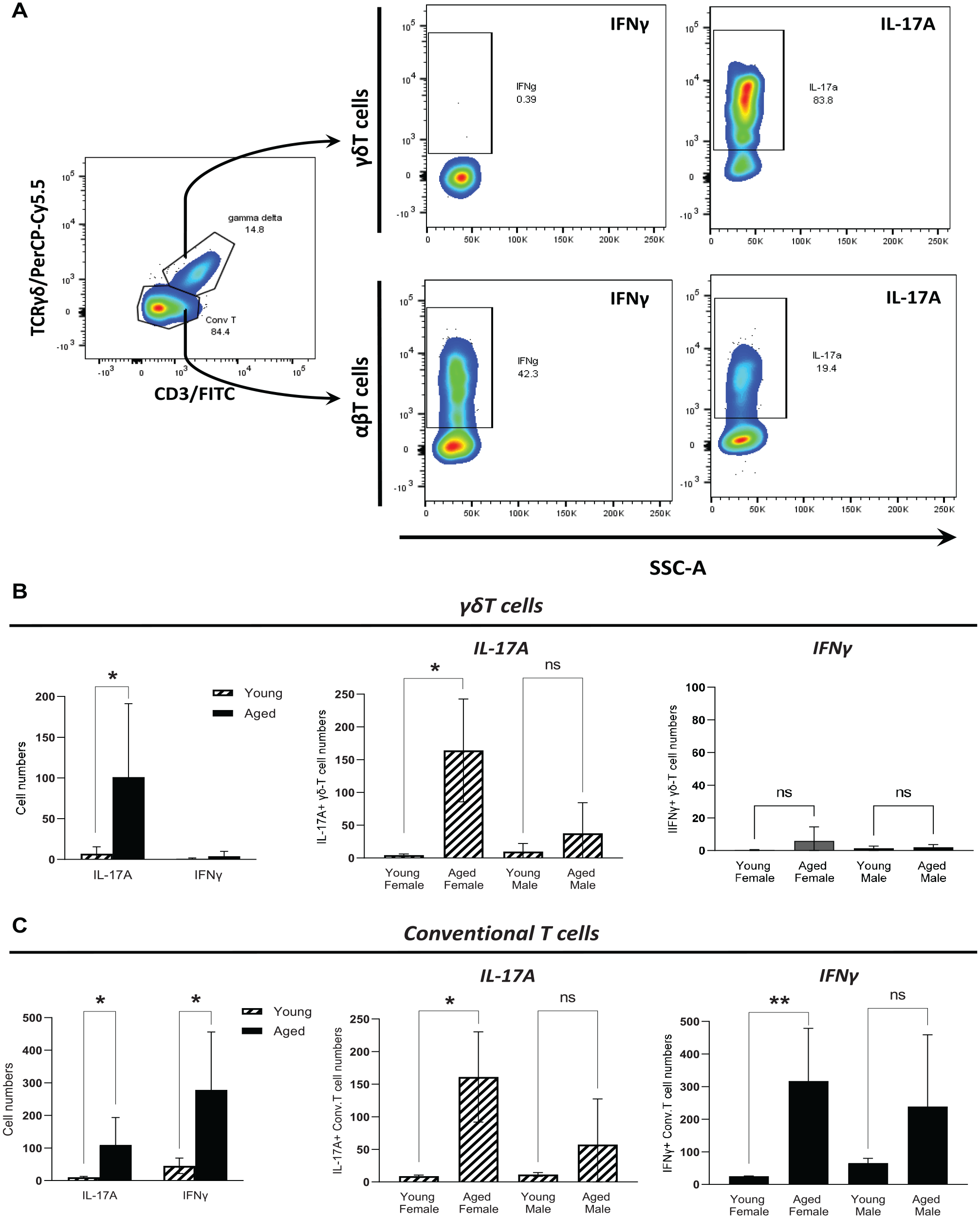
Effect of aging on cytokine responses of gingival γδ and conventional T cells. (
Representative figures for expression analysis of cell receptors associated with cell proliferation (IL-15rα) and senescence (Notch-3) by flow cytometry analysis are shown in Figure 3A and Appendix Figure 2. Expression of IL-15rα was not detectable in γδT cells; however, a small proportion of these cells were positive for Notch-3. Similarly, numbers of γδT Notch-3+ cells increased significantly about 3-fold (P = 0.0028) with aging only in female gingival tissues (Fig. 3B).
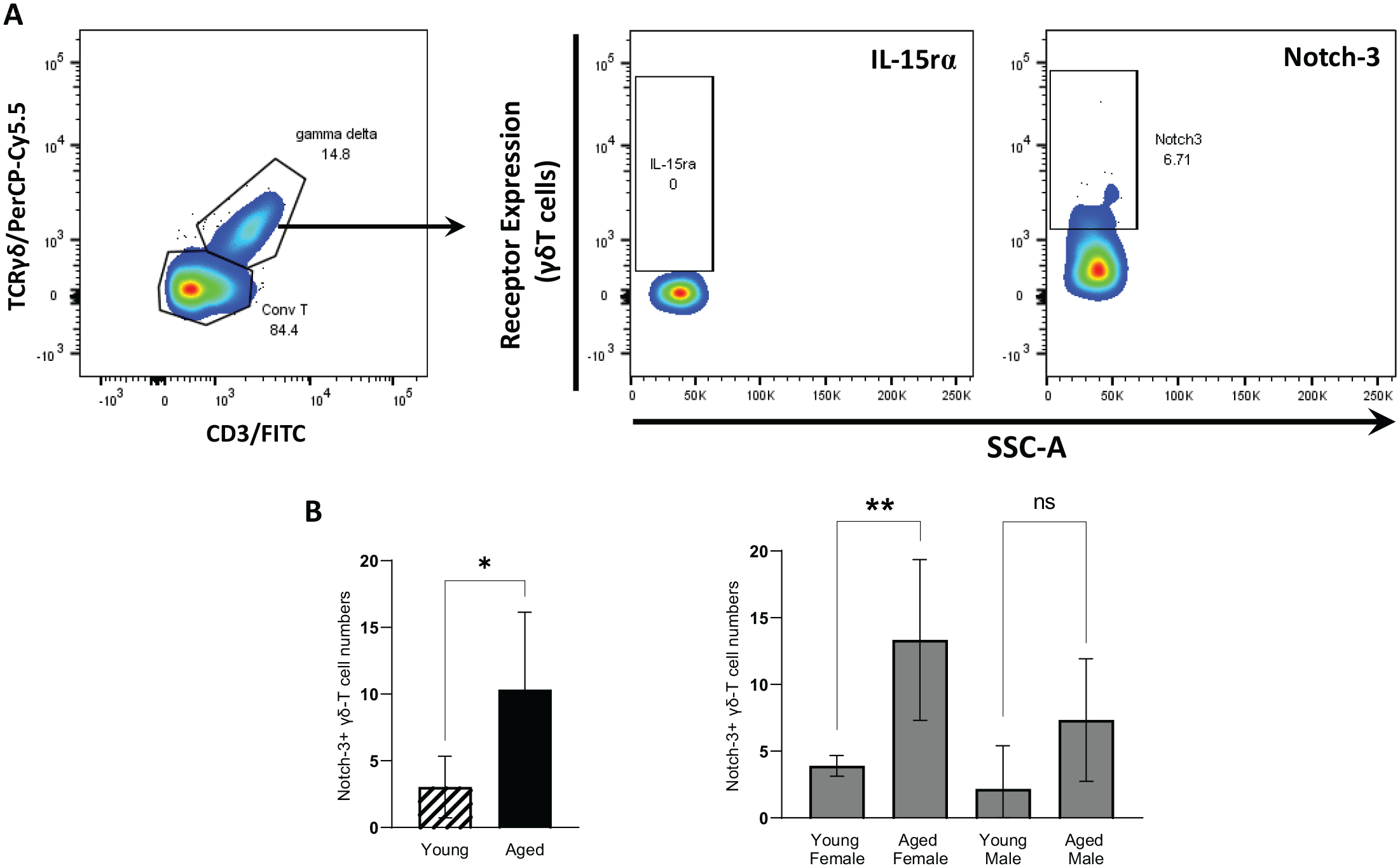
Effect of aging on Notch-3 receptor and IL-15rα expression of gingival γδT cells. (
Analysis of the effect of aging and sex in the gingival expression of specific gamma (γ) and delta (δ) T-cell V segments is shown in Figure 4. Only Vδ1, Vδ2, Vδ4, and Vδ6 chains were detected in gingival tissues, with Vδ1 exhibiting higher intensity. There appears to be mainly an age-associated variation in the detection of Vδ1 and Vδ2 subsets in both sexes, with the detection of the Vδ1 segment increasing with aging in both males and females, and Vδ2 increasing in females and decreasing in males. The expression of gingival Vγ segments was more diverse, with Vγ1, Vγ2, Vγ4, Vγ5, Vγ6, and Vγ7 detectable and Vγ1, Vγ6, Vγ2, and Vγ5 showing the greatest expression levels. Aging-related increased levels in Vγ5, Vγ6, and Vγ7 in both male and female and γ2 in male gingival tissues were observed. Levels of Vγ4 chain appear to be downregulated with aging only in males.
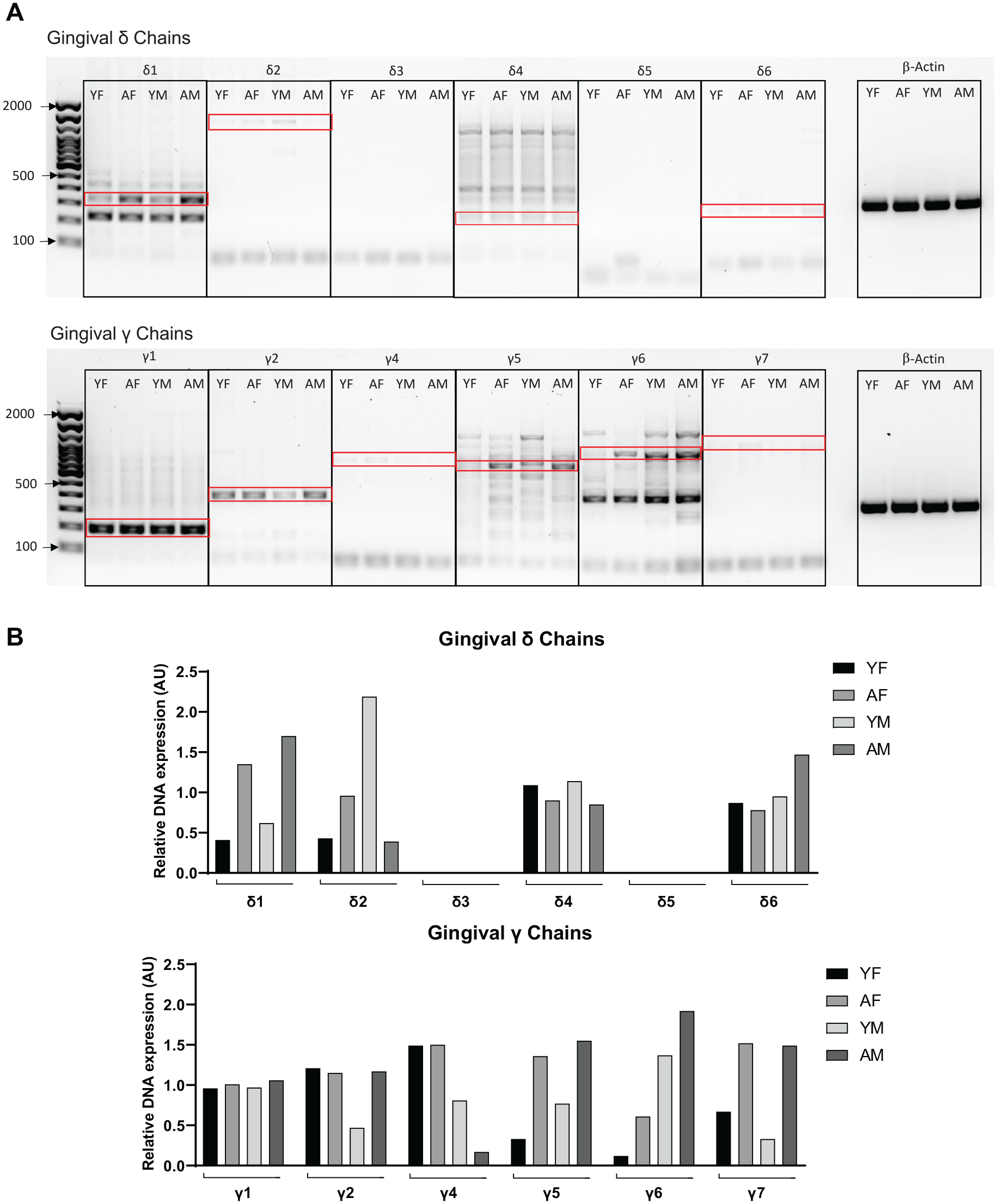
Effect of aging on gingival expression of Vδ and Vγ chain segments. (
Gene expression levels of IL-1β, IL-6, and IL17A in whole gingival tissues increased significantly with aging in females (P = 0.011; P = 0.043; P = 0.0021). About a 1- to 2-fold increase in IL-1β and IL-6 was observed in tissues from aged females and a 9-fold increase in IL-17A. In contrast, levels of IL-1β and IL-17A were comparable between young and aged males. The only significant increase in IL-6 levels was seen with aging in males (Fig. 5).
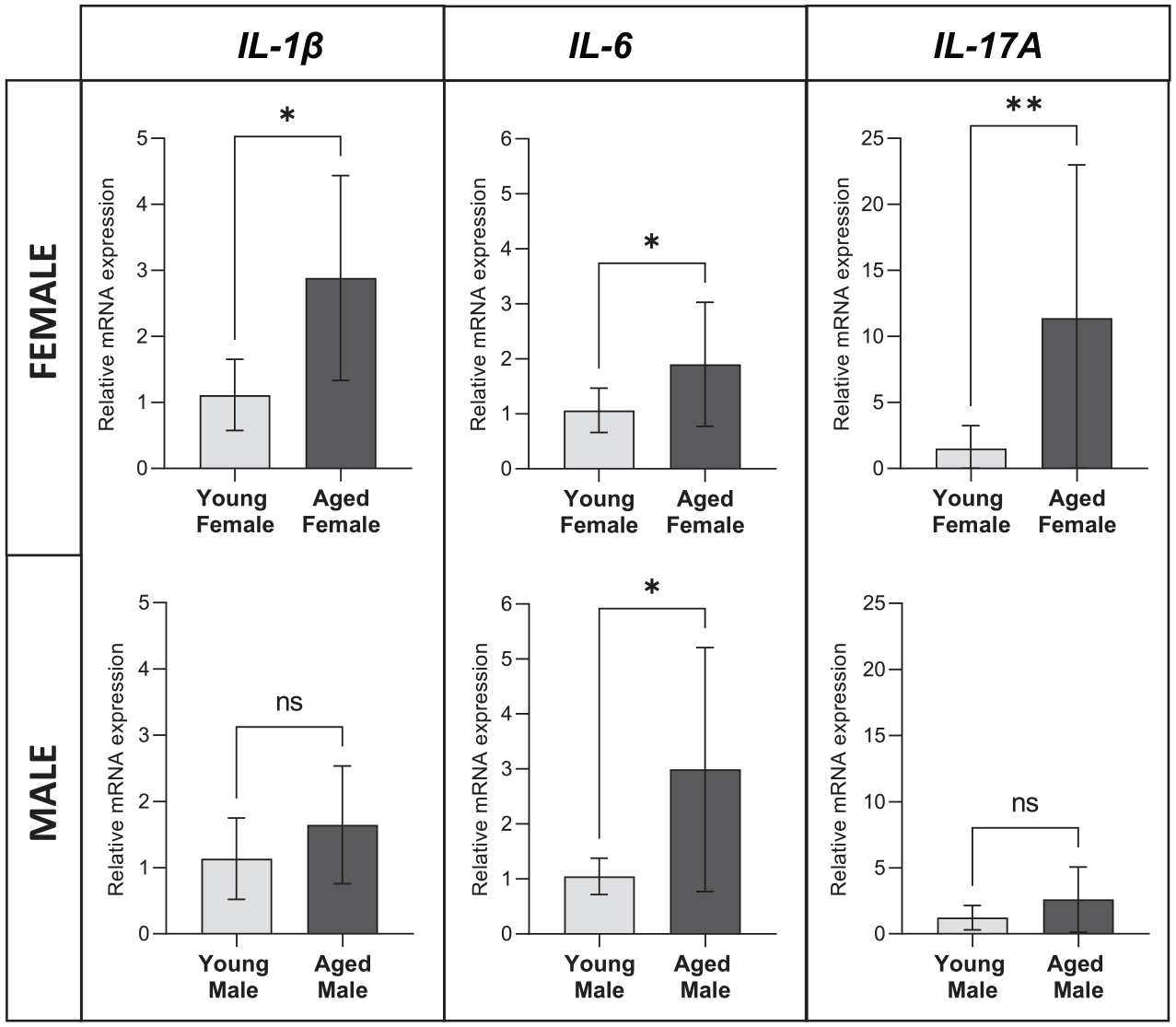
Effect of aging in the gingival expression of proinflammatory cytokines. Expression levels (messenger RNA) for IL-1β, IL-6, and IL-17A in gingival tissues from young and aged female and male mice are shown. Data from 4 mice per age group evaluated in duplicates are presented as mean ± SD. Statistical analysis involved Mann–Whitney test. *P ≤ 0.05. **P ≤ 0.001. ns, not significant.
Alveolar bone loss (ABL) increased with aging in both female and male mice; however, there were no statistically significant differences in ABL when aged female was compared with aged male (Appendix Fig. 3).
Discussion
Chronic inflammatory disease with aging (inflammaging) involves a perturbation of the innate immune mechanisms (Franceschi et al. 2018). Gamma-delta (γδ) T cells are an essential component of the innate immunity at mucosal surfaces, including the oral epithelium, that exhibit an activated phenotype with potent cytolytic and immunoregulatory capacities and modulate tissue repair, wound healing, and immune surveillance processes (Krishnan et al. 2018; Wilharm et al. 2019; Ribot et al. 2021). Given that aging affects the number and function of γδT cells in a tissue-specific manner, which modifies the risk for inflammatory disease, we sought to determine quantitative and qualitative changes of gingival resident γδT cells with aging.
It has been shown that gingival γδT cells develop prenatally in male mice, expand postnatally, and decline with age up to 40 wk of age (~9 mo old) (Wilharm et al. 2019), which corresponds to an adult mouse. Furthermore, these cells are reportedly negative for CD4 and CD8 cell surface markers and are predominately IL-17A as opposed to IFNγ producers (Krishnan et al. 2018). Nevertheless, whether number of gingival γδT cells is affected in aged mice (i.e., 18–24 mo) remains unknown. Here we found that frequency and numbers of γδT cells in gingival tissues were significantly increased with aging in females but not males. This suggests that biological processes affecting cell numbers within the tissues such as cell proliferation, apoptosis, or senescence of resident γδT cells could be particularly affected in females during aging. Evidence indicates that several immune cells, including γδT cells, express estrogen receptors, and estrogen is an important regulator of immune cells at mucosal surfaces (Andersson et al. 2015; Goodman et al. 2020). Particularly, estrogen has been shown to negatively regulate cellular senescence and enhance apoptosis (Ng and Hazrati 2022). In addition, estrogen attenuates T-cell proliferation (Cenci et al. 2003) and prevents γδT-cell migration from lymph nodes to peripheral tissues (Andersson et al. 2015). Consequently, an aging-related decrease in estrogen levels could be affecting these responses within the gingival milieu in females.
Evaluation of cell receptors associated with cell proliferation (IL-15rα) and cellular senescence (Notch-3) initially suggests that a fraction of gingival γδT cells in females could persist infiltrating the oral epithelium due to impaired cell death associated with senescence. These findings are consistent with evidence demonstrating sex differences in cellular senescence. Particularly, estrogen has a protective effect against senescence-inducing DNA damage and inhibits the expression of proteins that facilitate onset of cellular senescence (Ng and Hazrati 2022). Nevertheless, this and other potential mechanisms related to aging-associated changes in hormone levels, other cell receptors (e.g., IL2Rα, IL-7Rα), or the oral microbiome that could differentially activate and affect the number of gingival γδT cells in females need to be further elucidated and validated (Wilharm et al. 2019).
Consistent with previous studies, gingival γδT cells were mainly IL-17A producers but not IFNγ (Krishnan et al. 2018; Wilharm et al. 2019). The number of gingival IL-17A+ γδT cells was significantly elevated with aging only in females. Given that γδT cells are a major source of IL-17A in gingival tissues, with their number increasing in periodontal disease and facilitating pathogen-induced bone-destructive immune responses (Gemmell and Seymour 1995; Dutzan et al. 2017; Barel et al. 2022), higher numbers of these cells in gingival tissues with aging may increase the risk for pathologic inflammation and periodontal disease in aged females. It is likely that other environmental changes (e.g., oral dysbiosis) could be enhancing oral inflammatory responses mediated by these innate cells. Interestingly, a significant increase of IL-17A+ conventional αβT-cell numbers was also seen with aging only in females, which supports the hypothesis that there could be a sex-related effect of aging on IL-17A gingival T-cell responses involving both γδ and conventional αβ T cells. Previous studies reported that the transcription factor retinoic acid–related orphan receptor-γt (RORγt) gene, expressed by Th17 and IL-17+ γδT cells, harbors several estrogen response element sites in its promoter region (Chen et al. 2015). Therefore, a decline in estrogen levels with aging would support an alteration of IL-17A responses in females that make them more prone to IL-17A–associated inflammatory disease.
Elevated gingival expression levels of IL-1β and IL-17A with aging were observed in females but not males, while IL-6 expression was increased in both sexes. IL-1β, but not IL-6, is required for the optimal expansion of IL-17A+ γδT cells (Ness-Schwickerath and Morita 2011), and thus this sex-specific observation reinforces the possibility of a sex-related effect of aging on gingival IL-17A T-cell responses. Nevertheless, these steady state immunoinflammatory differences were not associated with greater or lower alveolar bone loss, which could suggest a destructive or protective effect, respectively, in females with aging. Although this observation is not coincident with greater periodontal disease frequency and severity in men that has been described in epidemiological studies (Eke et al. 2012), future research using experimentally induced periodontitis mouse models would be needed to determine the effect of increased IL-17A gingival responses with aging in periodontal disease under inflammatory/dysbiotic conditions in females. These sex-related oral immunoinflammatory differences, rather than differentially modifying the severity of disease, could instead provide a unique potential mechanism involved in aging-associated periodontitis in females.
Qualitative gene expression analysis of gingival resident γ and δ chains was consistent with previous findings indicating that Vγ1 and Vγ6 are the most prevalent in gingival tissues, followed by Vγ5, Vγ2, Vγ4, and Vγ7 (Krishnan et al. 2018; Wilharm et al. 2019). Interestingly, aging appeared to affect some subsets of gingival resident γδT cells regardless of sex (e.g., Vγ5, Vγ6, Vγ7, Vδ1), where there seems to be an increased presence of these subsets with aging, with few exceptions specifically in males showing aging-related reduced levels (e.g., γ4 and δ2). Consequently, aging-related variations that may not be reflected in the global number of resident gingival γδT cells could be occurring within specific cell subsets, which needs to be further confirmed by other approaches such as single-cell RNA sequencing.
It has been shown that the oral microbiota regulates gingival γδT-cell homeostasis (Wilharm et al. 2019) and the oral microbiome is affected by aging (Kirakodu et al. 2019; Ebersole et al. 2021). Studies exploring the role of specific features of the oral microbiome associated with aging and sex that may affect gingival γδT-cell subsets need to be developed.
In conclusion, the number of resident gingival IL-17A–producing γδT cells and IL-17A gingival expression levels increases naturally with aging, specifically in females. These innate immune cells and perhaps other immune cell populations (e.g., Th cells, macrophages, and neutrophils), whose proinflammatory activity has also been shown to be regulated by estrogen (Chakraborty et al. 2023), could be contributing to increase the risk for aging-associated periodontitis in postmenopausal women (Haas et al. 2009; Penoni et al. 2017; Romandini et al. 2020). This is a relevant question given that although the frequency of periodontitis increases with aging in both men and women (Eke et al. 2012), the role of aging as an important variable modifying the risk for oral inflammation distinctly in males and females remains undetermined (Ioannidou 2017).
Author Contributions
V. Tubero Euzebio Alves, M.E.C. Bruno, L. Wang, R.J. Danaher, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; S. Mukherjee, contributed to data acquisition and interpretation, critically revised the manuscript; L. Su, contributed to data analysis and interpretation, critically revised the manuscript; M.E. Starr, contributed to conception, design, data analysis and interpretation, critically revised the manuscript; O.A. Gonzalez, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231205210 – Supplemental material for Sex-Related Effect of Aging in Gingival Gamma-Delta T Cells
Supplemental material, sj-docx-1-jdr-10.1177_00220345231205210 for Sex-Related Effect of Aging in Gingival Gamma-Delta T Cells by V. Tubero Euzebio Alves, M.E.C. Bruno, S. Mukherjee, L. Wang, R.J. Danaher, L. Su, M.E. Starr and O.A. Gonzalez in Journal of Dental Research
Footnotes
Acknowledgements
We thank the Flow Cytometry and Immune Monitoring Core from University of Kentucky for their technical support.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health/National Institute of General Medical Sciences (grants P30GM110788-05 and R01GM129532) and National Institute of Aging (R56 AG061508).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
