Abstract
Dental caries lesions are a clinical manifestation of disease, preceded by microbial dysbiosis, which is poorly characterized and thought to be associated with saccharolytic taxa. Here, we assessed the associations between the oral microbiome of children and various caries risk factors such as demographics and behavioral and clinical data across early childhood and characterized over time the salivary and dental plaque microbiome of children before clinical diagnosis of caries lesions. Children (
Introduction
The oral microbiome establishes, matures, and changes throughout life in response to various physiological and environmental factors (Kilian et al. 2016). To ensure that the oral ecosystem remains healthy, it needs to adapt to changes occurring in the oral environment (Zaura and ten Cate 2015; Marsh and Zaura 2017). When the natural equilibrium between the host and its oral microbiome shifts toward an imbalanced, also called dysbiotic, state, this can promote demineralization of a tooth surface and increase risk of dental caries (Kilian 2018). A dysbiotic shift in the oral microbiome toward a cariogenic state commences before the manifestation of a caries lesion and is, therefore, challenging to determine and characterize.
Here we used a longitudinal design to assess associations between children’s oral microbiome and various caries risk factors, such as demographics, oral health, diet, and the behaviors of the children and their caregivers. At different time points (study visits), we also compared the salivary and dental plaque microbiome of groups of children with different caries statuses: those who remained caries free at all time points (for the entire study period from 1–6.5 y of age); those who were diagnosed with dental caries at 6.5 y, 4 y, or 2.5 y of age but remained caries free before that time point (i.e., those with ecological dysbiosis toward caries before its clinical manifestation); and those who presented with early and advanced caries lesions at a specific time point (i.e., at 2.5 y and 4 y of age).
Materials and Methods
Current Study Population
This report conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. The study was part of a larger project, “Predicting Caries Risk in Underserved Toddlers in Primary Healthcare Settings” (Fontana et al. 2019). The current study is a longitudinal prospective cohort observational study. No intervention was performed during the study. The participants and study procedures have been described in detail elsewhere (Kahharova et al. 2020). Briefly, 266 children who completed 3 study visits (baseline or time point T1: ~1 y, T2: ~2.5 y, and T3: ~4 y of age) were included in the current study. At the age of 6.5 y (T4), 189 from 266 children underwent follow-up clinical oral examinations. Clinical oral examinations, the questionnaire administrations (Appendix Table 1), and the collection of the saliva and pooled dental plaque samples of children were performed at 3 time points (T1–T3). At baseline, saliva was collected from caregivers. Clinical oral examinations were performed using the International Caries Detection and Assessment System (ICDAS) criteria (Banting et al. 2012) by yearly calibrated examiners.
Sample Collection and Processing
Sample collection, storage, DNA isolation, polymerase chain reaction (PCR) amplification, sequencing, and data processing were performed as described previously (Kahharova et al. 2020). The zero-radius operational taxonomic units (zOTUs) were assigned taxonomy using HOMD v14.51 (Chen et al. 2010). The sequences are available in the NCBI BioProject database under accession number PRJNA803343.
Statistical Analyses
Detailed analyses are described in the Appendix. In this study, the data set was normalized in one of the following 2 ways: by random subsampling of the zOTU table or by trimmed mean of M-value (TMM) normalization of the zOTU data (Fig. 1). In addition, we analyzed the contributions of various caries risk factors (variables) to the salivary and dental plaque microbiome over time and associations of those factors with the children’s caries status (Fig. 1). Principal component analysis (PCA), group comparisons (β-diversity) using PERMANOVA, the Shannon Diversity Index, and species richness calculations were performed in PAST software version 3.21 (Hammer et al. 2001). The
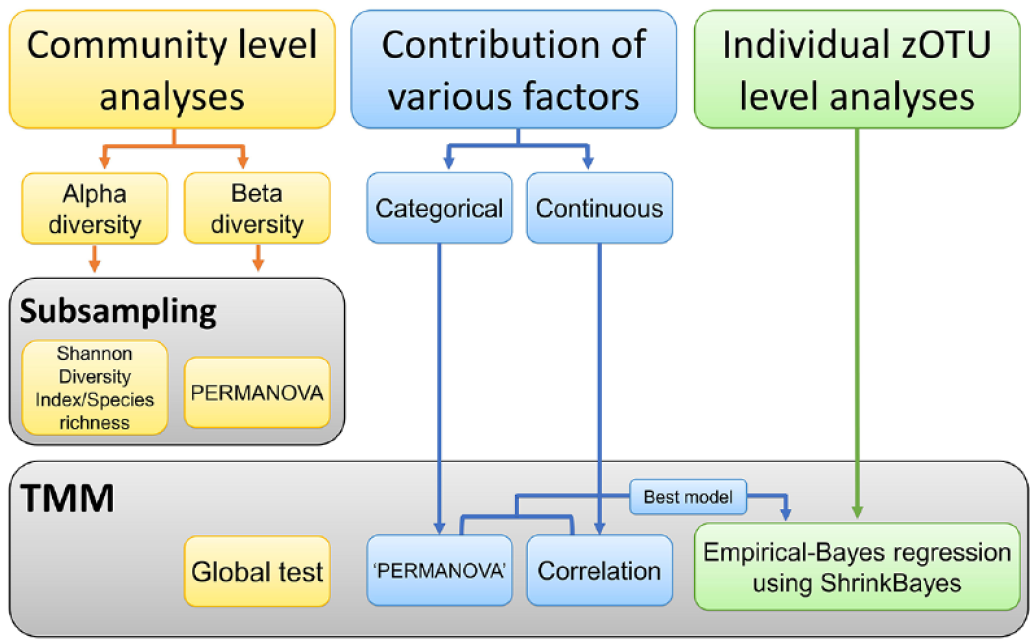
The flow diagram with the type of data set normalization per analysis type. Community-level analyses (yellow rectangles) were performed in both subsampled and trimmed mean of M-value (TMM) normalized data sets. Contribution of the various caries risk factors to the oral microbiome (blue rectangles) and individual zero-radius operational taxonomic unit–level analyses (green rectangles) were done on TMM normalized data sets.
To assess the contribution of the risk factors (variables) to the microbiome variance over time, the envfit function (vegan version 2.5_7 [Oksanen et al. 2020], R version 4.1.3 [R Core Team 2022]) was performed. The variables that yielded the best model by the bioenv function were included in the empirical Bayes regression analysis using ShrinkBayes (version 2.13.7 [van de Wiel et al. 2014], INLA version 22.5.03 [Rue et al. 2009]) as covariates. zOTUs with a Bayesian false discovery rate (BFDR) (Storey 2003) of at most 0.1 were deemed significant. The global test (Goeman et al. 2004) (R package version 5.42.0, R version 4.02) was used to assess the relationship between the microbiome and a response variable (caries status). For categorical variables, multinomial univariate logistic regression was conducted, while for continuous variables, the Kruskal–Wallis and the Mann–Whitney tests were performed (Bonferroni corrected), using IBM SPSS version 25 (SPSS, Inc.).
Results
Caries Status over Time
The children were assigned to different groups according to their caries status at each time point (Fig. 2A). Of the 266 children with microbial samples, 77 were lost to follow-up after 4 y of age (T3) (Fig. 2B). Across all time points (T1–T4 or 1–6.5 y of age), 50 of the 189 remaining children stayed clinically caries free Decayed, Missing due to caries, and Filled Teeth (dmft) = 0, d scored as ICDAS = 0 on all visible tooth surfaces) and were deemed the caries free at all time points (CFAT) group (Fig. 2A). While prevalence of dental caries (dmft ≥ 1; d scored as ICDAS ≥ 1) at the age of 1 y (T1) was 0.9% (
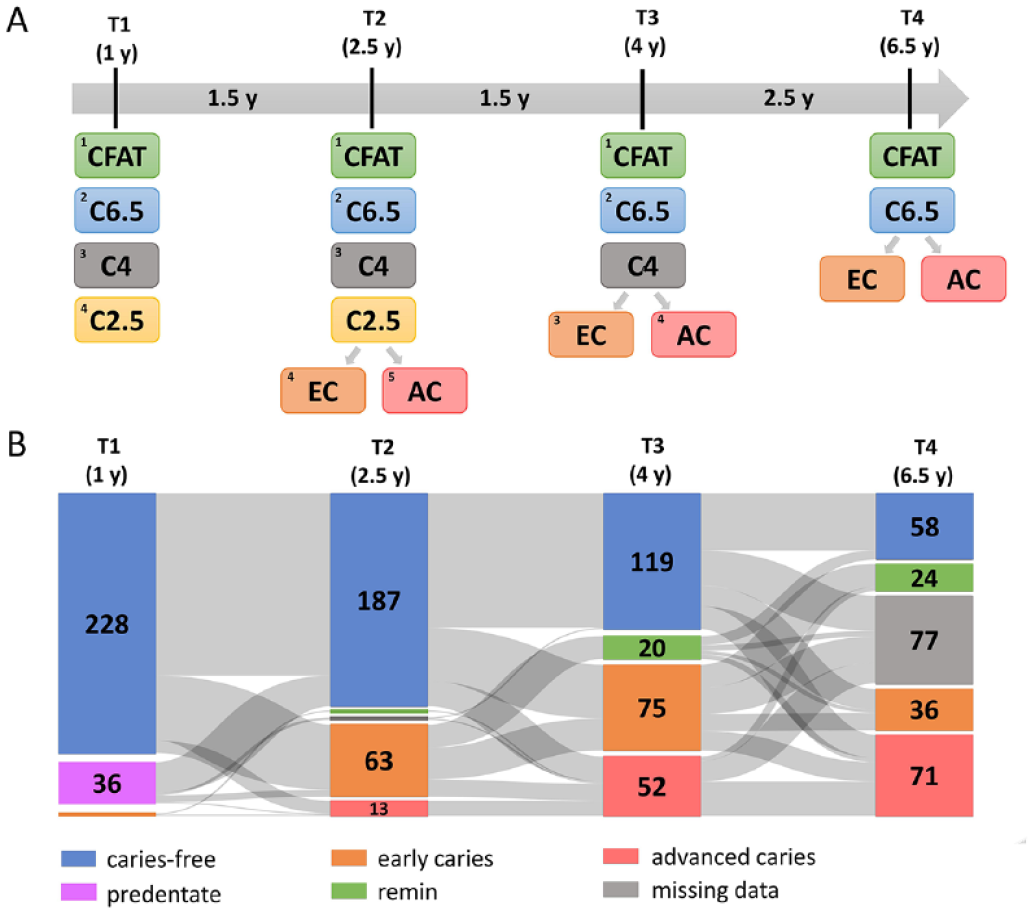
Distribution of the children based on the time of caries diagnosis and caries severity between 1 and 6.5 y of age. (
Oral Microbiome Analyses
Contributions of various caries risk factors to the oral microbiome composition
All analyzed variables are listed in Appendix Table 1, and a detailed description of the contributions of selected variables to the oral microbiome of the children over time is provided in the Appendix. In brief, at T1, exposure to antibiotics explained the largest variation in the salivary microbiome (Fig. 3A). At each time point, various factors, including demographics and the dietary habits of the children and their caregivers, were significantly related to the salivary microbiome community variation (Fig. 3A). Between the baseline (T1) and the later time points (T2 and T3), the proportion of the variation in salivary microbial composition explained by the included variables decreased (T1: 18.0%; T2: 11.4%; T3: 12.7%). For dental plaque, the largest variation in microbiome composition was explained by exposure to antibiotics, thick or tethered maxillary labial frenulum, frequency of sugary drinks and snacks, concentration of the bacterial DNA in the sample, race/ethnicity, and Medicaid status (Fig. 3B). At all time points, the frequency of sugary drink intake by both the children and their caregivers was significantly related to the dental plaque microbiome community variation of the children (Fig. 3B). In contrast with saliva, at T3, the proportion of the variation in plaque microbial composition explained by the variables was the highest (T1: 22.4%; T2: 16.6%; T3: 25.1%). At all time points, explained variation in plaque composition was higher than that of saliva (Fig. 3).
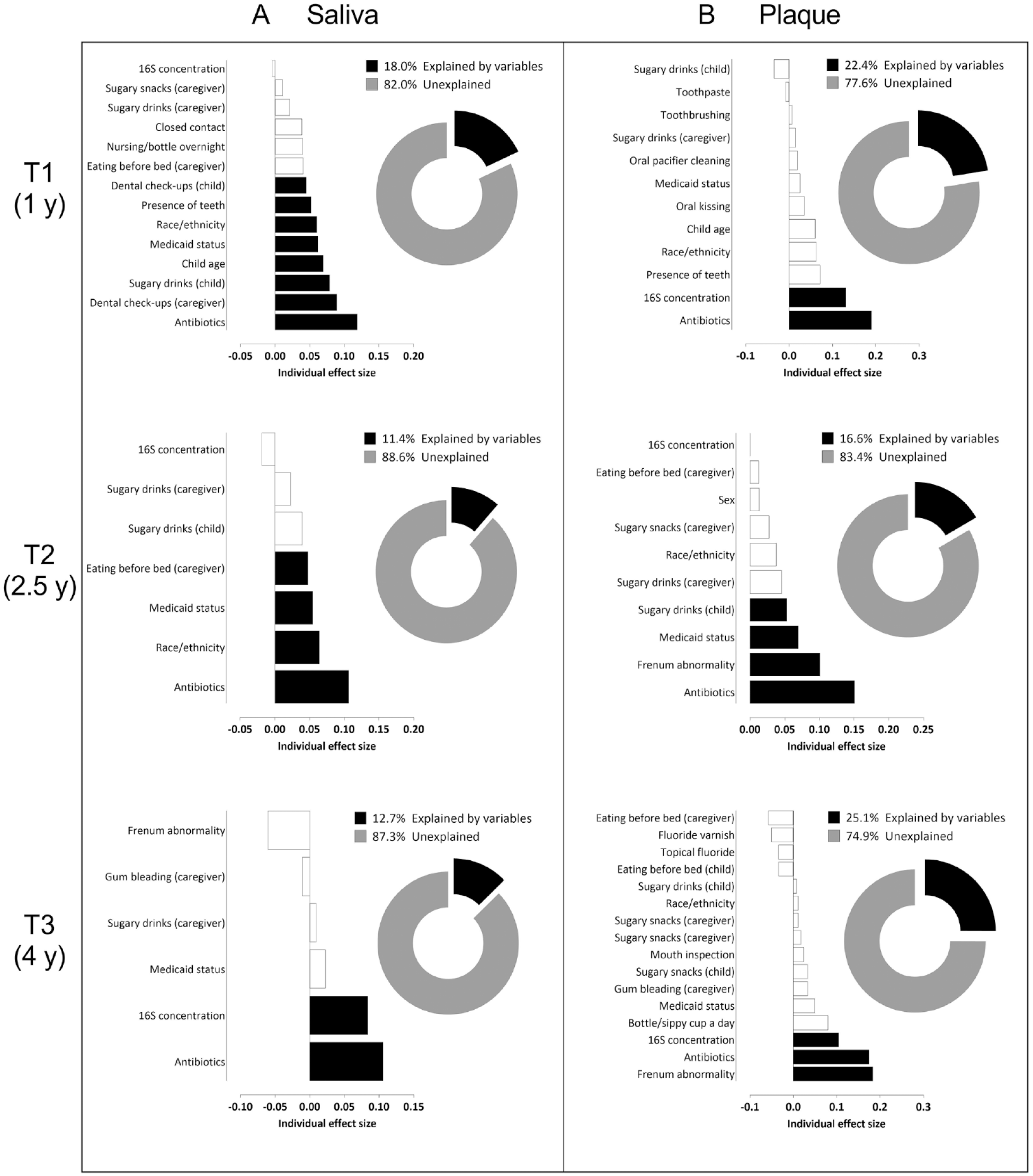
Variables associated with the salivary (
Microbial community-level differences
For the analyses below, detailed statistical outcomes are presented in the Appendix. In brief, at T1, the microbial profiles of saliva of the children, but not of plaque, differed significantly according to their caries status (Appendix Fig. 2A, B). The saliva of the 1-y-olds who would be diagnosed with caries by age 4 (C4) already differed from the saliva of those who remained clinically caries free throughout the study (CFAT) (Fig. 4A; Appendix Table 2A).
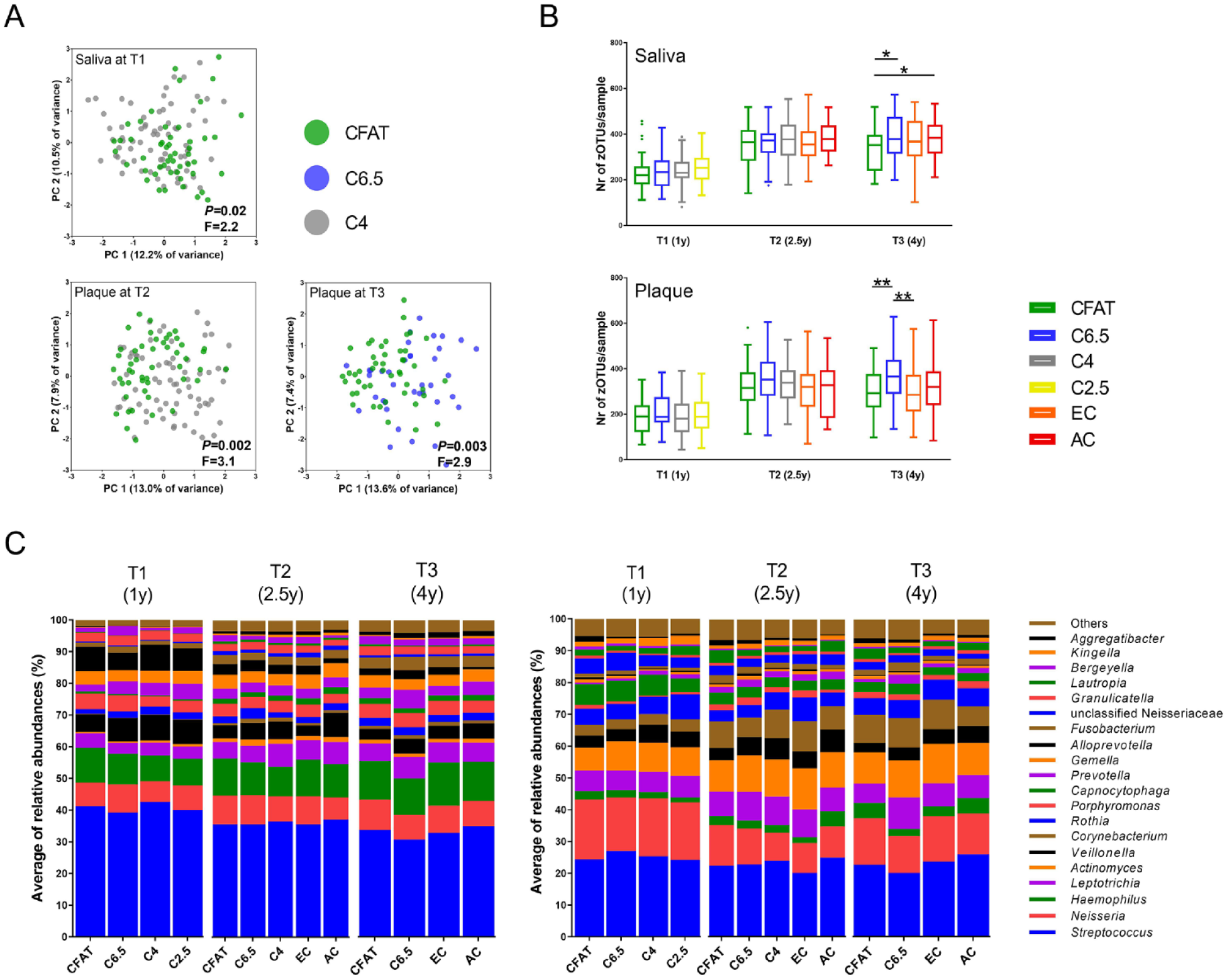
Microbial profile analyses of the salivary and dental plaque microbiome of the children over time (T1, T2, T3). (
At T2, the microbial profiles of dental plaque, but not of saliva, differed significantly according to the caries groups (Appendix Fig. 2A, B). The dental plaque microbiome of the C4 group differed significantly from those in the CFAT group (Fig. 4A; Appendix Table 2B). The plaque in the CFAT group was distinct from that of the children with early caries lesions at 2.5 y (EC) (Appendix Table 2C). In other words, at the age of 2.5, dental plaque of not only those children who presented with early caries at the age of 2.5 but also those who were diagnosed with caries at the next time point (at the age of 4) differed compositionally from dental plaque of the children who remained caries free throughout the study.
At T3, both the salivary and dental plaque microbiome profiles differed according to the caries groups (Appendix Fig. 2A, B). The saliva and plaque in the CFAT group were distinct from saliva and plaque in the C6.5 group (Fig. 5A, B; Appendix Table 3A, B) and in the EC (Appendix Table 3C) and AC groups (Appendix Fig. 3A, B; Appendix Table 3D, E). The dental plaque microbiome composition in the C6.5 group was distinct from that in the EC (Appendix Table 3F) and AC groups.
Differences in microbial composition between children with different caries status. The output of the global test comparing the composition of (
At T1 and T2, neither the salivary or plaque microbiome differed in α-diversity according to the caries groups, while at T3, the saliva samples in the CFAT group had significantly lower species richness than those in the C6.5 and AC groups (Fig. 4B). In plaque at T3, the microbial diversity was significantly higher in samples in the C6.5 group than in those in the CFAT group and in the EC group (Fig. 4B).
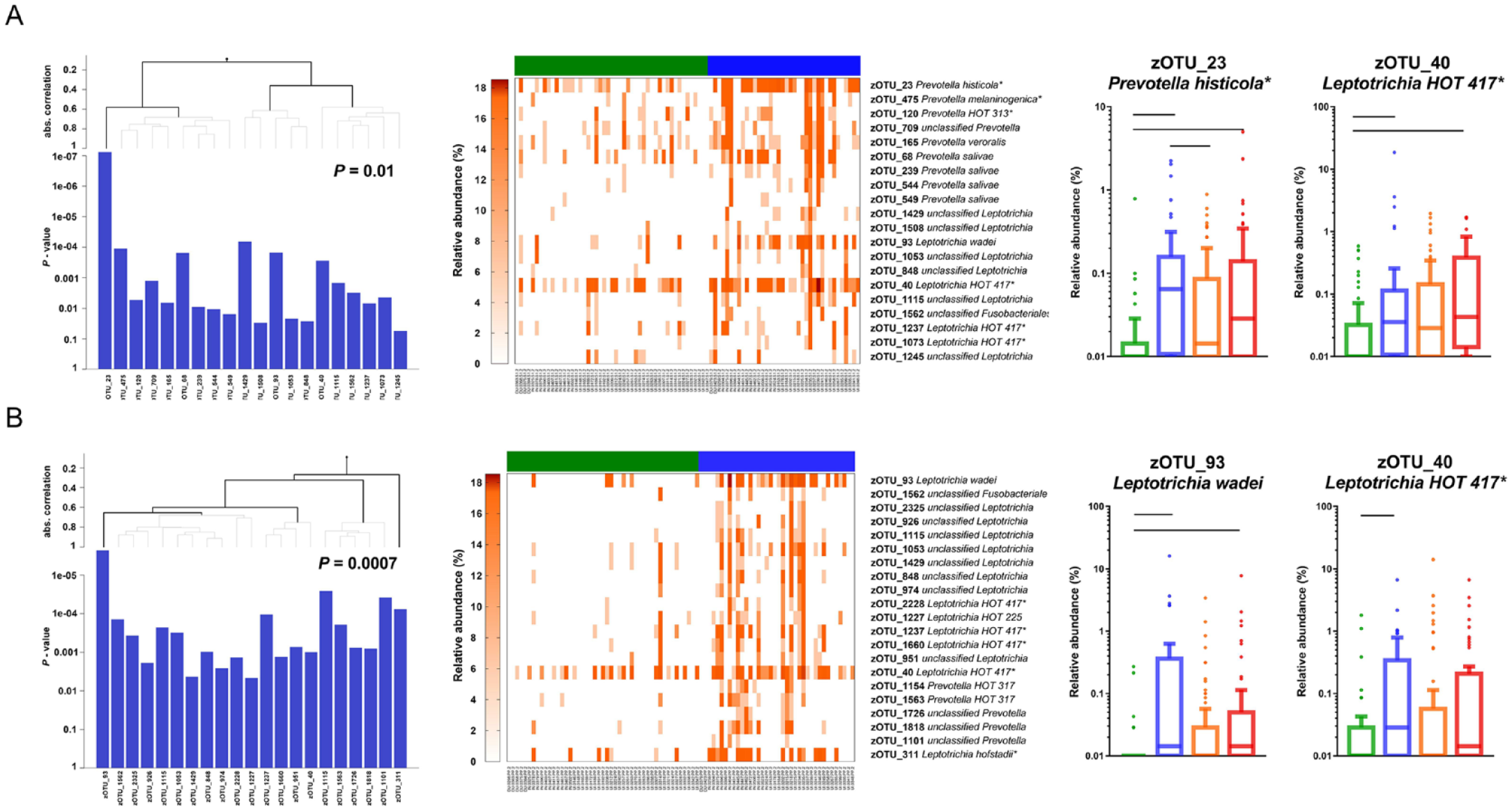
At the age of 4 (T3), in both saliva and plaque, groups of various species of the genera
Differences in individual taxa
First, we compared the top 20 most abundant genera in the salivary and dental plaque samples according to caries status (Fig. 4C). In saliva at T1, the relative abundance of genera
Next, we assessed the contribution of individual zOTUs to differences among the caries groups. For this, we used ShrinkBayes regression analysis, which included the most significant factors (best model) as covariates (Fig. 1). For saliva collected at T1, the following covariates were selected as the best model: child’s age, exposure to antibiotics, race/ethnicity, Medicaid status, number of teeth, the frequency of the child’s consumption of sugary drinks between meals, and the frequency of dental visits of the children and their caregivers. In total, 6 zOTUs differentiated between the C4 group and the CFAT group (Appendix Table 4A). The microbial profiles of dental plaque at T1 and those of saliva at T2 did not differ significantly according to the caries groups; therefore, the analyses on individual zOTUs were not performed. For T2 dental plaque, 4 covariates resulted in the best model: exposure to antibiotics, Medicaid status, tight or tethered maxillary labial frenulum, and frequency of the child’s consumption of sugary drinks between meals. Several zOTUs were significantly discriminatory between the dental plaque in the CFAT and samples from the C4 and EC groups (Appendix Table 4B, C).
For T3 saliva, exposure to antibiotics and the bacterial DNA concentration were included as covariates. Numerous zOTUs discriminated among the caries groups (Appendix Table 5A–C). For example, zOTUs such as zOTU_22 unclassified
Demographics, dietary and oral hygiene habits, and caries status
Child age, sex, delivery mode, and exposure to antibiotics did not differ across caries groups. However, Medicaid status and the recruitment site for all time points, income category for T2 and T3, and race/ethnicity for T3 did differ (results not shown) among the caries groups. The frequencies of consumption of sugary drinks by the children themselves and by their caregivers were significantly associated with the caries status at all time points (Appendix Fig. 7A, B). In addition, at T2, the frequency of the caregivers’ consumption of sugary snacks and caregivers’ consumption of anything other than plain water before going to bed was significantly associated with the caries status of their children. The frequency of caregivers’ inspection of the dentition of their child was significantly associated with caries status at T2 and T3.
Discussion
This study showed that the microbiome of children with clinically sound teeth, but imbalanced (dysbiotic) oral microbiome toward caries, differed from the microbiome of those who remained caries free at all time points and those who had clinically established early and advanced caries lesions. The oral microbiome of the children was affected by various factors such as antibiotic use, maxillary labial frenulum abnormality, bacterial and fungal DNA concentration in the samples, demographics, Medicaid status, and the eating and oral hygiene habits of the children and their caregivers.
We found that, 1 to 3 y prior to clinical caries detection, oral microbial communities were in a state of dysbiosis that was dominated by proteolytic taxa such as
One of the unexpected findings of our study was the resilience toward caries development (Rosier et al. 2017): even though the salivary or dental plaque microbiome presented signs of dysbiosis, some children were not diagnosed with caries during the second checkup at the age of 2.5 (1.5 y after the first assessment at the age of 1), but only at the third assessment at the age of 4. The interplay among microbial and host-related factors, such as the immune system (Zaura et al. 2014) and salivary properties (Pedersen et al. 2018), might play a role and should be included in future assessments.
Another unusual finding was the incongruity of the results in the 2.5-y-olds relative to those from the same children at a younger or an older age. At T2, when the children were approximately 2.5 y old, no significant differences in salivary microbiome across caries groups were observed, while saliva in both the 1- and 4.5-y-olds did distinguish children who remained caries free at all time points from the other groups. In addition, plaque samples collected from children with dysbiosis or caries at T2 had higher proportions of
In addition, currently assessed covariates could account only for a maximum of 18% of salivary or 25% of dental plaque microbiome composition variance, suggesting that a much broader view on the host genetic factors and immunological, environmental, and behavioral parameters might be needed to explain how the individual oral microbiome is shaped. The current study focused on questions that are associated with caries risk assessment only and can be self-administered to primary caregivers during medical or dental visits.
The current study sample exhibited high early and advanced caries prevalence, with high frequency of consumption of sweet drinks and snacks. Interestingly, in our study, not only the dietary habits of the children themselves but also those of their caregivers correlated positively with the caries outcome and affected the composition of the oral microbiome of the children, most likely due to higher availability of sugary snacks in certain households (Vepsäläinen et al. 2018).
Conclusion
Our findings demonstrate that the shift in the balance of the oral microbiome occurs before the clinical manifestation of dental caries. The dysbiosis state is observed 1 to 3 y before clinical detection of caries lesions and is associated with an increase of anaerobic and proteolytic taxa like
Author Contributions
D. Kahharova, V.Y. Pappalardo, M.J. Buijs, R.X. de Menezes, E. Zaura, B.W. Brandt, contributed to data acquisition, drafted and critically revised the manuscript; M. Peters, R. Jackson, A.T. Hara, G. Eckert, B. Katz, M.A. Keels, S.M. Levy, M. Fontana, contributed to conception and design, data acquisition, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345231160756 – Supplemental material for Microbial Indicators of Dental Health, Dysbiosis, and Early Childhood Caries
Supplemental material, sj-docx-1-jdr-10.1177_00220345231160756 for Microbial Indicators of Dental Health, Dysbiosis, and Early Childhood Caries by D. Kahharova, V.Y. Pappalardo, M.J. Buijs, R.X. de Menezes, M. Peters, R. Jackson, A.T. Hara, G. Eckert, B. Katz, M.A. Keels, S.M. Levy, E. Zaura, B.W. Brandt and M. Fontana in Journal of Dental Research
Footnotes
Acknowledgements
We thank the members of the Caries Risk Study Grant Group: Emily Yanca, Susan Flannagan, Barcey Levy, Jeanette Daly, John Warren, Justine Kolker, Alex Kemper, Dennis Clements, Jennifer Talbert, Fredrica Gallack, Brenda Pattison, Beth Patterson, Sue Kelly, Jen Tran, Sharon Gwinn, Lorena Galvez, and Lisa Robinson who contributed in data acquisition.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Fontana was a member of the National Scientific Advisory Committee of the Delta Dental Plans Association.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Institutes of Health (NIH) grant 5U01DE021412; NIH CTSA grants (UL1-TR000442, University of Iowa; 2UL1-TR000433, University of Michigan; and UL1-TR000006, Indiana University); Colgate-Palmolive, the University of Michigan, School of Dentistry; and a Consortium of Delta Dental Plans (Delta Dental of Iowa, Delta Dental of Wisconsin, the Renaissance Health Service Corporation for Delta Dental of Michigan). D. Kahharova was supported by Stichting Bevordering Tandheelkundige Kennis with NTvT Onderzoeksbeurs 2017 and by the ACTA Research Institute. V.Y. Pappalardo was funded by ACTA Research Institute, with ACTA 2019 Lustrum grant.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
