Abstract
Rodent animal models for vital pulp therapy are commonly used in dental research because their tooth anatomy and cellular processes are similar to the anatomy and processes in humans. However, most studies have been conducted using uninfected sound teeth, which makes it difficult to adequately assess the inflammatory shift after vital pulp therapy. In the present study, we aimed to establish a caries-induced pulpitis model based on the conventional rat caries model and then evaluate inflammatory changes during the wound-healing process after pulp capping in a model of reversible pulpitis induced by carious infection. To establish the caries-induced pulpitis model, the pulpal inflammatory status was investigated at different stages of caries progression by immunostaining targeted to specific inflammatory biomarkers. Immunohistochemical staining revealed that both Toll-like receptor 2 and proliferating cell nuclear antigen were expressed in moderate and severe caries-stimulated pulp, indicating that an immune reaction occurred at both stages of caries progression. M2 macrophages were predominant in moderate caries-stimulated pulp, whereas M1 macrophages were predominant in the severe caries-stimulated pulp. Pulp capping in teeth with moderate caries (i.e., teeth with reversible pulpitis) led to complete tertiary dentin formation within 28 d after treatment. Impaired wound healing was observed in teeth with severe caries (i.e., teeth with irreversible pulpitis). During the wound-healing process in reversible pulpitis after pulp capping, M2 macrophages were predominant at all time points; their proliferative capacity was upregulated in the early stage of wound healing compared with healthy pulp. In conclusion, we successfully established a caries-induced pulpitis model for studies of vital pulp therapy. M2 macrophages have an important role in the early stages of the wound-healing process in reversible pulpitis.
Keywords
Introduction
Dental pulp consists of connective tissue, nerve fibers, blood vessels, and multiple cell types with specialized functions that maintain the biological and physiological vitality of the tooth (Yu and Abbott 2007). With advances in understanding the important role of dental pulp for long-term tooth survival, there is increasing interest in developing vital pulp therapy (VPT) strategies to avoid pulp removal (Schmalz et al. 2020). Despite the implementation of conservative strategies in dentistry, caries remains the most common infectious disease and the main cause of endodontic treatment (Frencken et al. 2017; Lin et al. 2020). VPT for teeth with carious pulp exposure is to remove only the inflamed portion to maintain and preserve the healing potential of the pulp, which is called pulpotomy, or perform direct pulp capping (DPC) in the exposed site. The success rate of VPT depends on an accurate diagnosis of the pulpal status (reversible or irreversible pulpitis), which influences the repair process (Ricucci et al. 2019). However, because of the fluctuating inflammatory condition of the remaining pulp, the outcomes have been difficult to predict, and little is known about the healing capacity of pulp under inflammatory conditions. Various clinical diagnoses (e.g., cold/heat response and electric pulp testing) have been used to assess the status of sound or diseased dental pulp (Abbott and Yu 2007; Hilton 2009), but many histological studies have shown a weak relationship between clinical symptoms and the severity of pulpal inflammation (Mejàre et al. 2012). In 1 clinical study, almost 40% of clinically asymptomatic teeth had histologically irreversible pulpitis (Michaelson and Holland 2002). Such inconsistency may explain the lower success rate of VPT. To improve clinical outcomes, further investigations are needed concerning accurate differential diagnosis between reversible and irreversible pulpitis; there is also a need to fully investigate the correlation between caries-induced inflammation and the wound-healing process in dental pulp.
Multiple immune cells have diverse roles in protecting dental pulp from foreign bodies after pulp exposure because of structural damage to the tooth. The innate immune effector cells in dental pulp are neutrophils, dendritic cells, macrophages, and lymphocytes (Hahn and Liewehr 2007; Galler et al. 2021). Among these, macrophages and dendritic cells are resident since initial tooth development; macrophages have key roles in both inflammatory and healing processes (Italiani and Boraschi 2014). Previous studies have shown that macrophages can polarize into 2 subtypes during wound healing depending on different tissue microenvironments. M1 macrophages are predominant during the inflammatory phase; they mediate host defense against pathogens by releasing proinflammatory cytokines such as interleukin (IL)–6 and tumor necrosis factor α. In contrast, anti-inflammatory M2 macrophages contribute to tissue repair by expressing high levels of proteins such as mannose receptor, IL-10, and transforming growth factor β (Italiani and Boraschi 2014; Krzyszczyk et al. 2018). Therefore, we presumed that evaluations of macrophage polarization in inflamed pulp could help to identify the threshold separating reversible and irreversible pulpitis.
When developing new materials for VPT, animal models are necessitated for validating biological effects or safety before clinical trials (Greek et al. 2012). Until now, most animal studies initiating pulp capping materials have been conducted on healthy teeth to investigate effects on hard tissue formation and inflammation (Dammaschke 2010; He et al. 2017). Other studies have used the pulpitis model, obtained by drilling into healthy teeth and opening the pulp or adding the bacterial component, for subsequent pulp-capping experiments (Aubeux et al. 2021; Chen et al. 2021). However, such models do not accurately represent the clinical pulpal pathology during caries progression, in which caries-induced inflammation already occurs before pulp exposure (Ricucci et al. 2018; Richert et al. 2022). Therefore, new animal models that involve caries-induced inflammation to mimic clinical conditions are necessary for investigating outcome of VPT.
In this study, we attempted to build a pulpitis model based on the conventional caries model. We hypothesized that the inflammatory status of dental pulp would differ according to the stage of caries progression; moreover, the wound-healing process after DPC would differ between sound teeth and teeth with caries-induced pulpitis. We first assessed the inflammatory status of teeth at different stages of caries progression, as determined by micro–computed tomography (CT). Then, we performed DPC to investigate hard tissue formation. Finally, we examined inflammatory changes in sound teeth and teeth with caries-induced pulpitis after DPC.
Materials and Methods
Animals
All animal studies were conducted with the approval of the Institutional Animal Care and Use Committee of Osaka University Graduate School of Dentistry (No. R-01-017-0); they also followed the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines. Forty 14-d-old Sprague–Dawley rats (weighing 23–30 g; CLEA Japan, Inc.) were obtained and housed in plastic cages with corncob bedding under a controlled light cycle. The animals were given powdered food containing tetracycline (4 g/kg) (FUJIFILM Wako Pure Chemical Corporation) and water containing penicillin G potassium (4,000 U/mL) (Meiji Seika Pharma Co., Ltd.) for 3 d to eliminate nonspecific pathogens before the caries induction procedure.
Caries Induction and Characterization by Micro-CT
The caries induction timeline and procedures are shown in the Appendix text and Appendix Figure 1. The rats were fed a cariogenic “Diet 2000” powdered diet (sucrose 56%, wheat flour 7%; CLEA Japan, Inc.) ad libitum during the caries induction period. Stages of caries progression were classified as shallow, moderate, or severe according to the depth of the demineralized layer of dentin, as determined by micro-CT with the following conditions: tube voltage of 90 kV, tube current of 160 μA (R-mCT2; Rigaku). Shallow caries was defined as demineralization that remained within the outer third of dentin, moderate caries was defined as demineralization that reached the middle third of dentin, and severe caries was defined as demineralization that reached the inner third of dentin.
Direct Pulp-Capping Procedure
As described in the caries induction timeline (Appendix Fig. 1), when rats are reared to 7 to 9 wk old, sodium pentobarbital (Somnopentyl; Kyoritsu Pharmaceutical) (30 mg/kg) was administered via intraperitoneal injection; subsequently, micro-CT was used to confirm whether the caries progression had reached a moderate or severe level, then subjected to DPC. During the procedure, infected dentin was visualized using a caries detector dye (Kuraray Dental); pulp exposure was intentionally induced with a 0.5-mm round bur (Dentsply Sirona) after complete caries removal in the moderate caries group. In the severe caries group, pulp was exposed during caries removal; after the completion of caries removal, the exposed pulp in both groups was irrigated with saline for 15 s, then gently dried with a cotton pellet for hemostasis. Subsequently, the cavity was capped with ProRoot MTA (Dentsply Sirona) and sealed with glass ionomer cement (Fuji IX, GC). Inflammatory changes after DPC were investigated on days 1, 3, 7, and 14. Tertiary dentin formation was analyzed by micro-CT and quantified 4 wk after treatment using TRI 3D-BON (Ratok) software, as previously described (Okamoto et al. 2018). DPC in 8-wk-old male Sprague–Dawley rats with sound teeth (n = 20, weighing 180–220 g; CLEA Japan, Inc.) was used as a control condition.
Histopathological Evaluation
Teeth specimens at different stages of caries progression were subjected to hematoxylin and eosin (H&E) staining to evaluate general inflammatory conditions. Brown and Brenn (B&B) staining was performed to observe bacterial invasion into the dentinal tubules and pulp. Immunohistochemical staining for Toll-like receptor 2 (TLR2), proliferating cell nuclear antigen (PCNA), and double immunofluorescent staining of CD68 and CD206 (to determine macrophage polarization) was conducted to assess the pulpal inflammatory status before and after capping. The detailed methodology is shown in the Appendix.
Statistical Analysis
Sample size was determined according to the resource equation approach (Festing 2006); 4 rats with bilateral teeth were subjected to DPC in each condition. The analysis excluded specimens with biased sample measurements due to unrelated elements like cavity size, sealing material loss, and so on. The statistical significance among 4 groups was measured using One-way analysis of variance and Tukey’s honestly significant difference. Differences between 2 groups were determined by Student’s t-test. All analyses were performed using IBM SPSS Statistics 22 (IBM Corp.). The statistical significance threshold was set at P < 0.05.
Results
Investigation of Inflammatory Responses According to the Stage of Caries Progression
Carious teeth were evaluated by micro-CT and histopathological analysis to determine the overall pulpitis status at each stage of caries progression. Micro-CT images revealed teeth reached shallow, moderate, and severe carious progression (Fig. 1A). In the shallow caries group, H&E staining showed no significant inflammatory cell infiltration (Fig. 1B(a), Appendix Fig. 3(a–c)); B&B staining showed minimal bacterial invasion into dentinal tubules (Fig. 1C(a)), and limited TLR2-positive and PCNA-positive cells were observed beneath carious lesions (Fig. 1D(a), E(a)). Therefore, the shallow caries group was excluded from subsequent experiment. In the moderate caries group, reactionary dentin formation was observed beneath carious lesions, while the odontoblastic layer remained stable, and inflammatory cell infiltration was observed next to the caries lesion but not infiltrated to the coronal pulp (Fig. 1B(b), Appendix Fig. 3(d–f)); B&B staining revealed bacterial invasion into dentinal tubules, but pulp was unaffected (Fig. 1C(b)). TLR2-positive cells and PCNA-positive cells were distributed in the peripheral zone of the pulp near carious lesions (Fig. 1D(b), E(b)). In the severe caries group, H&E staining showed a disordered odontoblast layer with extensive inflammatory cell infiltration under carious lesions even infiltrated to the coronal pulp with blood vessel dilation (Fig. 1B(c), Appendix Fig. 3(g–i)). In addition, B&B staining confirmed the invasion of Gram-positive bacteria deep into dentinal tubules (Fig. 1C(c)). Finally, large numbers of TLR2-positive and PCNA-positive cells were present near carious lesions (Fig. 1D(c), E(c)).
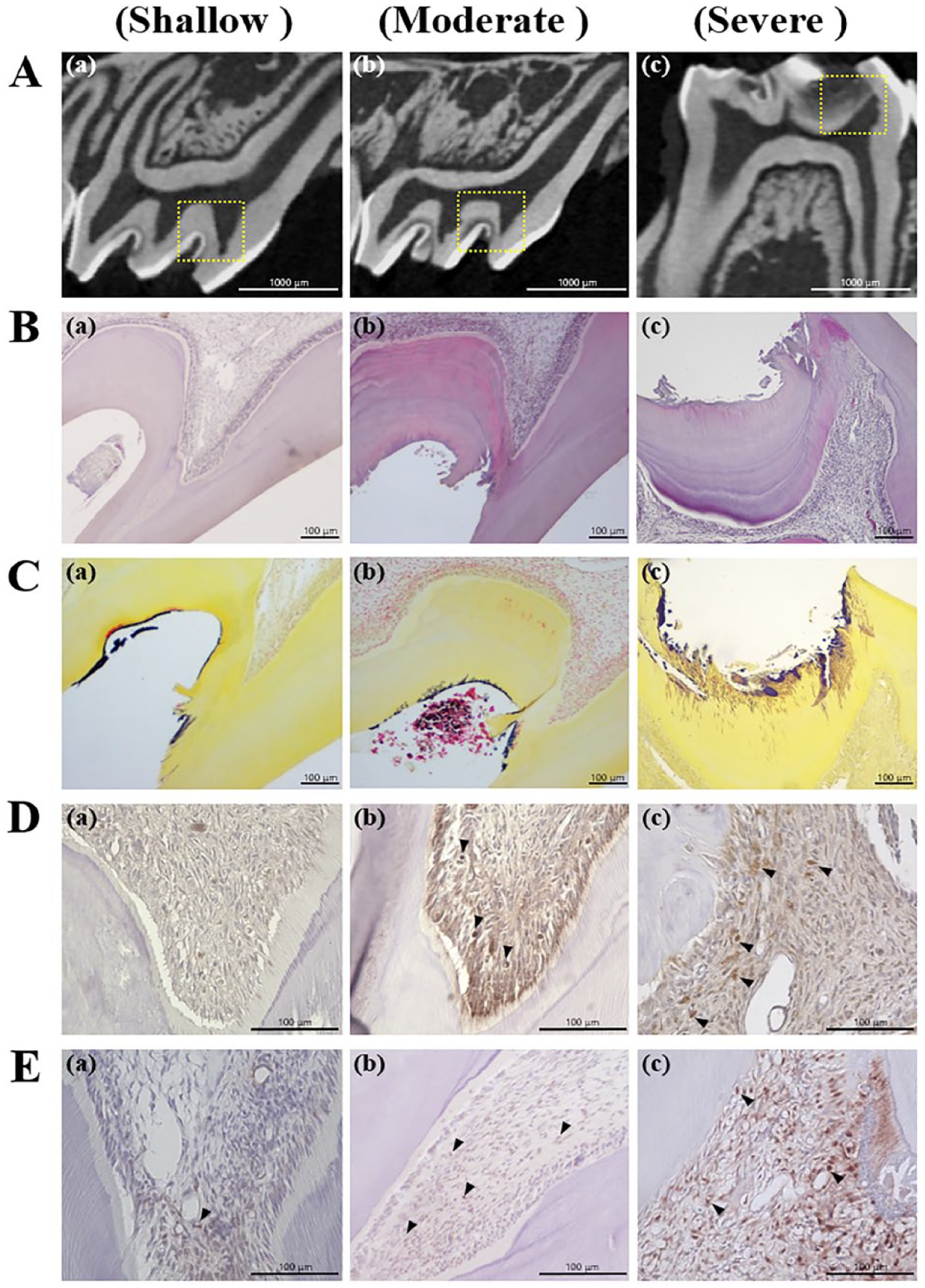
Characterization of pulpal inflammatory status at different stages of caries progression. (
Next, we examined the populations of M1 and M2 macrophages in the moderate and severe caries groups. CD68-positive/CD206-negative cells were regarded as M1 macrophages, whereas CD68-positive/CD206-positive cells were regarded as M2 macrophages, based on the definitions in a previous report (Gu et al. 2019). In the moderate caries group, few M1 macrophages were observed near the odontoblast layer close to carious lesions; M2 macrophages showed a generally wider distribution and larger population in the inflamed pulp (Fig. 2C(a)). In the severe caries group, macrophages exhibited an M1-dominant phenotype, while a smaller number of M2 macrophages were distributed around the M1 macrophages (Fig. 2C(b)). In addition, we investigated the macrophage population in the carious teeth with pulp exposure. The results showed that M1 macrophages were predominant beneath the exposure area, but no M2 macrophages were observed (Fig. 2C(c)). M2 macrophages were observed only in areas distant from sites of exposure.
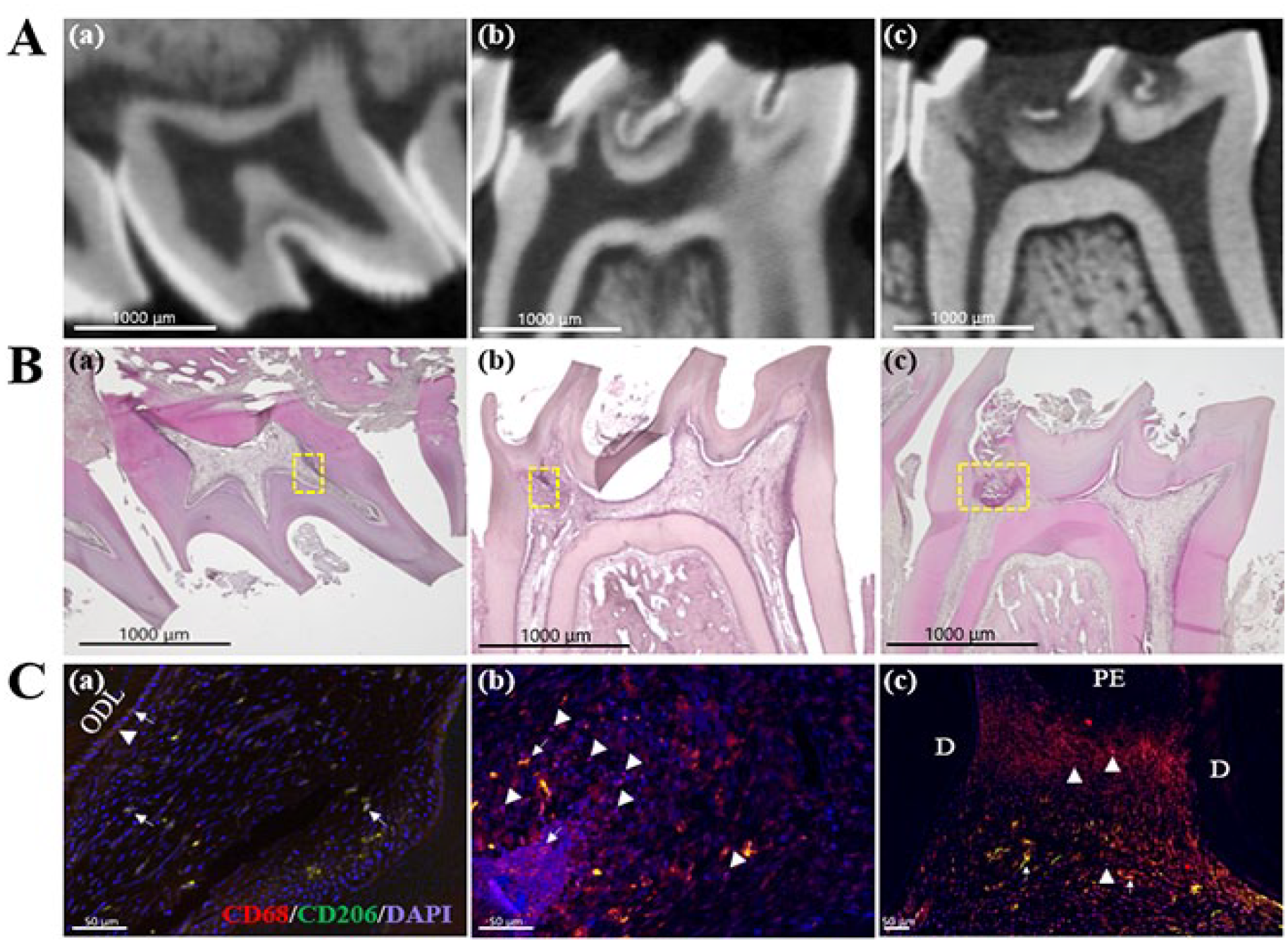
Macrophage polarization at different stages of caries progression. (
Tertiary Dentin Formation under Inflammatory Conditions
To differentiate caries-induced reversible and irreversible pulpitis, we performed DPC and investigated tertiary dentin formation in teeth with moderate caries and severe caries, as well as sound teeth. Micro-CT and H&E staining showed that complete tertiary dentin was formed in the sound and moderate caries groups (Fig. 3A(a–f)) but not in the severe caries group (Fig. 3A(g–i)). Moreover, there was significantly less newly formed tertiary dentin in the moderate caries group than in the sound group (Fig. 3B, C). In the sound group, newly formed dentin bridges possessed arranged dentinal tubules (Appendix Fig. 4); the odontoblast-like layer was arranged below the dentin bridge (Fig. 3A(c)). In the moderate caries group, dentinal tubules were oriented in an irregular direction with a less-organized odontoblast-like layer (Fig. 3A(f)). The severe caries group showed incomplete hard tissue formed at the area detached from the capping site; some cells were entrapped into calcified tissue, and strong eosin-stained connective tissue with high cell density was observed beneath the calcified tissue (Fig. 3A(i)).
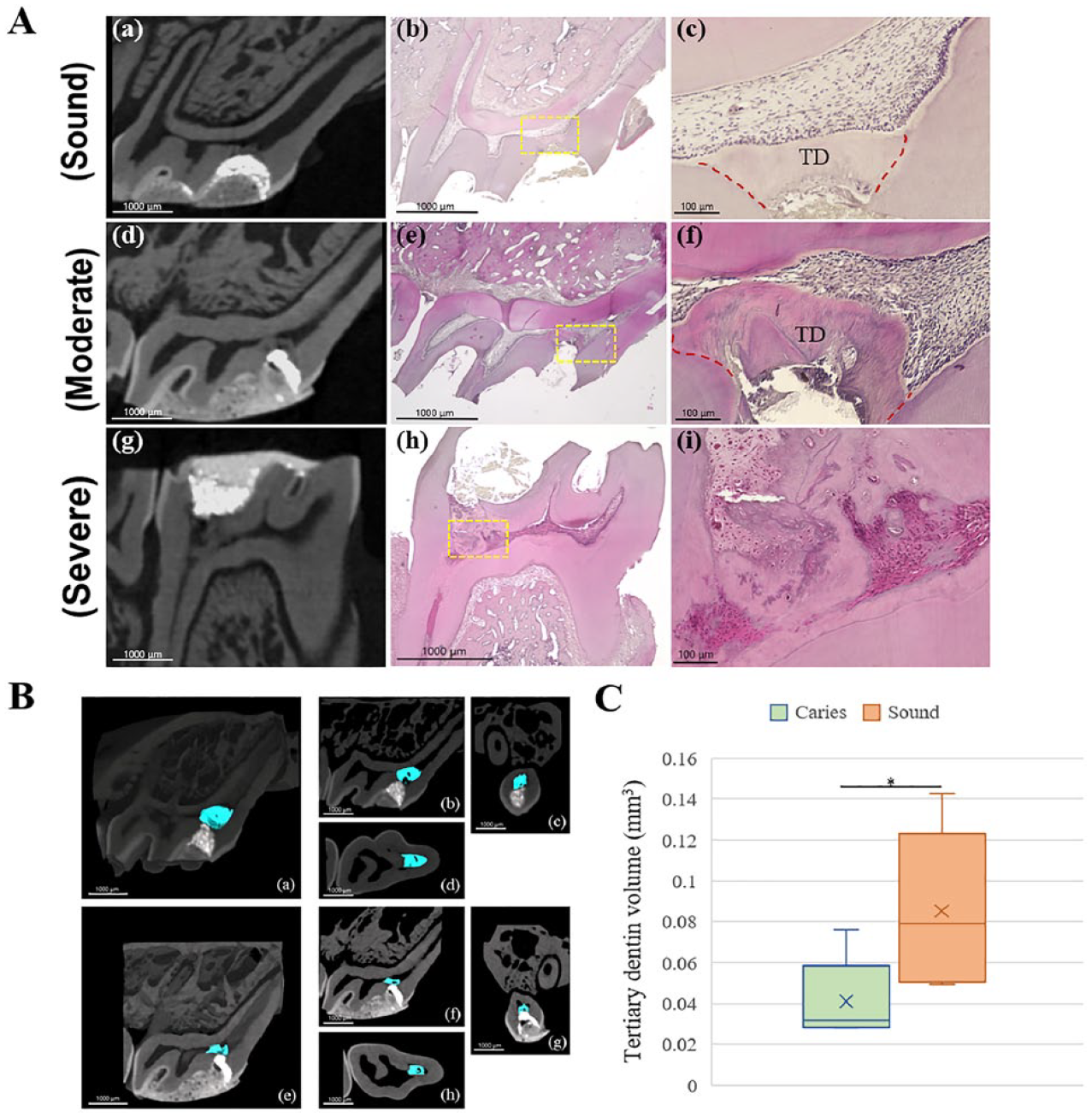
Tertiary dentin formation after pulp capping in teeth at different stages of caries progression. Direct pulp capping in sound teeth was used as a control. (
Comparison of Inflammatory Changes during the Wound-Healing Process between Sound Teeth and Teeth with Reversible Pulpitis
To investigate whether caries-induced inflammation affects the wound-healing process, inflammatory changes were investigated by H&E staining and spatiotemporal localization of macrophages after DPC in sound and moderate carious teeth. H&E staining showed high cell density throughout the pulp chamber in the moderate caries group on days 1 and 3 (Fig. 4A(a, b)). In contrast, inflammatory cell infiltration was limited to the pulp horn beneath the cavity in the sound group (Fig. 4A(e–h)); residual pulp distant from the cavity still remained stable. Immunofluorescence staining showed a limited number of M1 macrophages in the moderate caries group, whereas M2 macrophages were predominant at each time point after capping (Fig. 4B(a–d)). In addition, there was a distinct shift in the number of M2 macrophages in the moderate caries group during the wound-healing process—the largest population of M2 macrophages was observed on day 3, and a noticeable decrease began after day 7 (Fig. 4C(a)). In the sound group, there was no significant shift in the number of M2 macrophages (Fig. 4C(b)). Moreover, the numbers of M2 macrophages were higher in the moderate caries group than in sound group on days 1 and 3 (Fig. 4D).
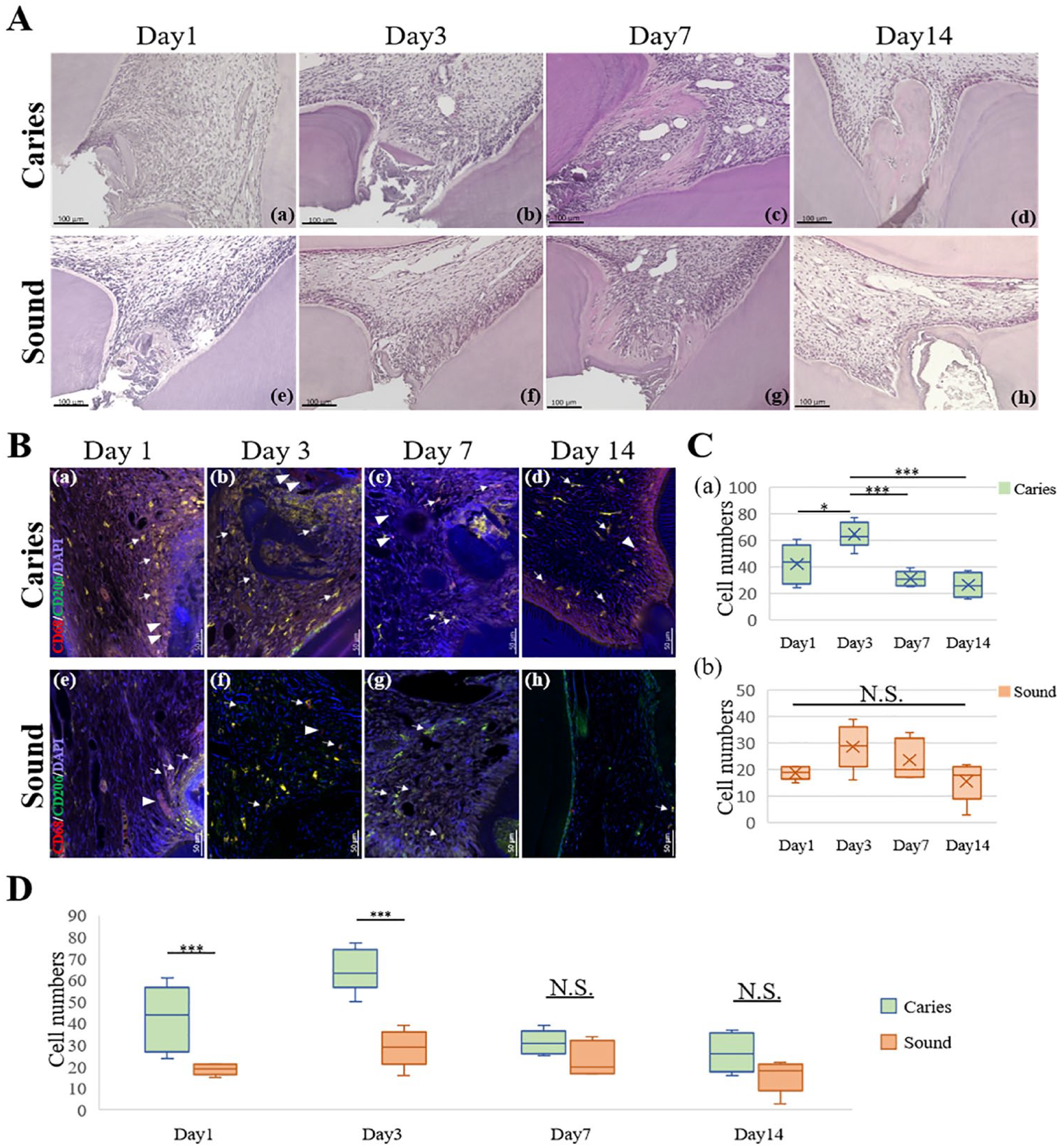
Wound-healing process in sound teeth and teeth with moderate caries. (
Finally, we investigated the proliferative capacity of dental pulp cells during the wound-healing process. The moderate caries group exhibited a wide distribution of PCNA-positive cells on days 1, 3, and 7, whereas the sound group exhibited PCNA-positive cells in a limited peripheral area close to the capping site (Fig. 5A). The largest number of PCNA-positive cells was observed on day 3, and the number began to decrease on day 7 in both groups (Fig. 5B). In addition, PCNA expression was significantly higher in the moderate caries group than in the sound group on days 1 and 3 (Fig. 5C).
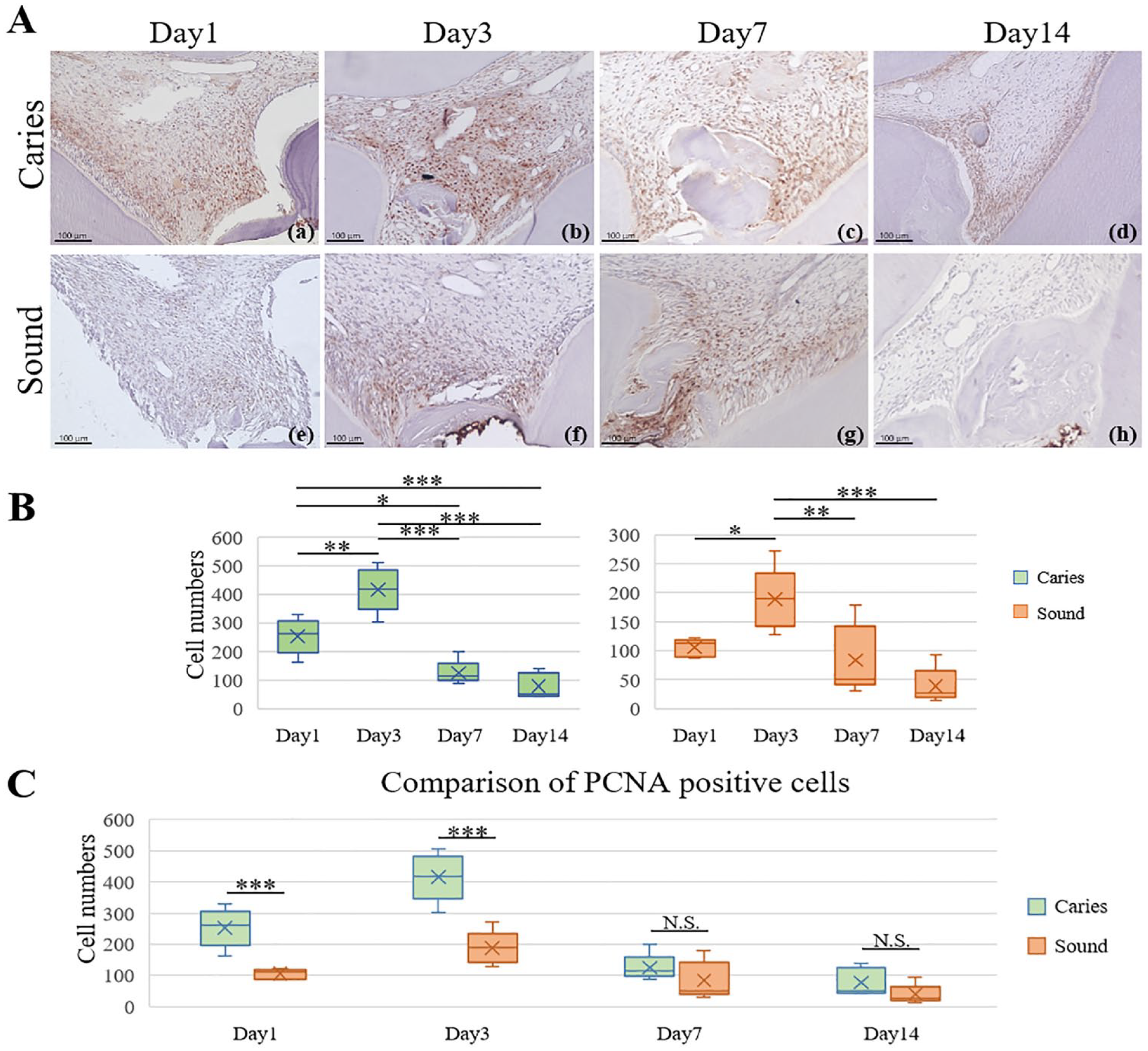
Proliferating cell nuclear antigen (PCNA) expression during the wound-healing process. (
Discussion
Rodent caries models have long been used to investigate human oral diseases, producing much of our current understanding of caries pathology, treatment, and prevention (Bowen 2013). However, few studies have used caries models to assess the outcome of VPT due to the lack of research on correlating pulpitis and caries lesions. In this study, we examined the pulpitis status at different stages of caries progression in a rat caries model. The odontoblastic layer, located in the outermost layer of dental pulp, controls innate immunity in the dentin–pulp complex. It can sense pathogens by activating pattern recognition receptors (PRRs) during caries progression, thus triggering innate and/or adaptive immune responses, as well as secreting reactionary dentin to rescue the tooth from various external stimuli (Farges et al. 2015; Yumoto et al. 2018; Galler et al. 2021). Therefore, we investigated the expression of TLR2, a PRR that recognizes Gram-positive bacteria and B&B staining. Bacterial invasion into dentinal tubules, positive expression of TLR2, and significant inflammatory cell infiltrations by histological observation suggest successful activation of the immune response in the moderate and severe carious teeth. In our investigation of macrophage polarization in inflamed pulp according to the stage of caries, the moderate caries group primarily exhibited M2 macrophages, whereas the severe caries group exhibited M1 macrophages. This result was consistent with a previous clinical study that M2 macrophages were predominant in human caries-stimulated pulp, whereas M1 macrophages were predominant in exposed pulp associated with caries (Yoshiba et al. 2020). The present findings suggest that the inflammatory response in rat caries-induced pulpitis model is comparable with human carious teeth. In addition, when DPC was performed on rat carious teeth, the moderate carious teeth induced complete hard tissue formation, while the severe carious teeth formed impaired hard tissue and displayed continuous, severe inflammation. These results suggest that inflammation can be rescued when caries proceed to a moderate stage that M2 macrophages predominated in the pulp tissue, indicating reversible pulpitis. Conversely, when the severe stage of caries progression is reached and characterized by M1 macrophage dominance, it can readily compromise the wound-healing process through uncontrolled inflammation. And with current pulp capping material having a limited effect on turning macrophages to M2 phenotype (Yuan et al. 2018), pulpitis can hardly be alleviated, indicating irreversible pulpitis. Therefore, we suggest that identifying the predominance of macrophage phenotype in the remained pulp will help differentiate reversible/irreversible pulpitis and predict the treatment outcome.
The general wound-healing process is a highly programmed process that is involved in various cellular and molecular events. It includes hemostasis, inflammation, proliferation, and remodeling phases (El karim et al. 2021). In the present study, PCNA expression in moderate carious teeth indicated that the inflammatory and proliferative phases have overlapped during caries progression, and when DPC was performed, acute inflammation might have been triggered again due to pulp exposure (Guo and DiPietro 2010; El karim et al. 2021). As a result, the wound-healing process in carious teeth underwent a more complex process that is mainly affected by the surrounding environment (e.g., infection, immune regulation). Although the proliferative capacity was upregulated in moderate carious teeth during the initial stage of wound healing, the speed of dentin bridge formation was not significantly enhanced compared with teeth in the sound group—the connective tissue started to form on day 3, and partial tertiary dentin was formed on day 7 and became complete on day 14. Notably, the shift in the population of PCNA-positive cells was similar to the macrophages. Macrophages tend to proliferate locally, rather than recruit new monocytes to injury sites (Jenkins et al. 2011). We suspected that some of the PCNA-positive cells were proliferating macrophages. Therefore, we investigated PCNA and CD68 double expression after DPC. As shown in Appendix Figure 5, we found that some PCNA-positive cells also expressed CD68, which supported our hypothesis.
Furthermore, we compared hard tissue formation between sound and moderate caries groups after DPC with ProRoot MTA. After 4 wk of treatment, the moderate caries group had a significantly lower volume of tertiary dentin and the structure was more disorganized than the sound group, indicating caries-induced inflammation hinders the natural repair process. These results are consistent with clinical findings that the lowest quality of reparative dentin is usually associated with pulpal inflammation (Trowbridge 1981; Ricucci et al. 2014). We supposed that the absence of a regular dentinal tubular structure may interfere with the ability of newly differentiated odontoblastic-like cells to receive required signals to produce reactionary dentin, thereby resulting in reduced hard tissue formation in carious teeth.
The role of macrophages during the wound-healing process has been studied intensively over the past decades. Numerous studies have shown the positive effect of M2 macrophages on promoting wound healing by upregulation of stem cell activity, angiogenesis, collagen depositions, and matrix remodeling (Novak and Koh 2013; Neves et al. 2020). Although the population of M2 macrophages was higher in the moderate carious teeth than in the sound teeth, it did not accelerate hard tissue formation. We assume that the presence of M2 macrophages in the inflammatory environment caused by dental caries may serve to further encourage angiogenesis under hypoxia conditions (Kieu et al. 2022). More studies are needed to fully elucidate the specific role of macrophages under different inflammatory conditions.
In conclusion, we successfully established a rat caries-induced pulpitis model for VPT research through an evaluation of its pulpal inflammatory status: the demineralized layer reached the middle third of dentin in moderate carious teeth, indicating reversible pulpitis with M2 macrophage predominance, whereas the demineralized layer reached the inner third of dentin in severe carious teeth, indicating irreversible pulpitis with M1 macrophage predominance. The limitation of this study is that only qualitative analysis of inflammatory biomarkers was performed to determine the different caries progression–induced pulpitis. However, our findings suggest M2 macrophage predominance might play pivotal role in the successful wound healing from reversible pulpitis. Further quantitative analysis will be needed to clarify the factors affecting the failure of wound healing in irreversible pulpitis.
Author Contributions
H. Huang, contributed to conception and design, data, acquisition, analysis, and interpretation, drafted and critically revised the manuscript; M. Okamoto, contributed to conception and design, data interpretation, critically revised the manuscript; M. Watanabe, S. Matsumoto, K. Moriyama, S. Komichi, M. Ali, S. Matayoshi, contributed to acquisition and data analysis, critically revised the manuscript; R. Nomura, K. Nakano, contributed to data conception, critically revised the manuscript; Y. Takahashi, M. Hayashi, contributed to conception and design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345221150383 – Supplemental material for Development of Rat Caries-Induced Pulpitis Model for Vital Pulp Therapy
Supplemental material, sj-docx-1-jdr-10.1177_00220345221150383 for Development of Rat Caries-Induced Pulpitis Model for Vital Pulp Therapy by H. Huang, M. Okamoto, M. Watanabe, S. Matsumoto, K. Moriyama, S. Komichi, M. Ali, S. Matayoshi, R. Nomura, K. Nakano, Y. Takahashi and M. Hayashi in Journal of Dental Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grants-in-Aid for Scientific Research (22H03268, 21K09915, 21K16967) from the Japan Society for Promotion of Science.
Data Sharing Statement
Data that support the findings in this study are available from the corresponding author upon reasonable request.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
