Abstract
Mitigation of irradiation injury to salivary glands was previously reported using a cell-free extract from mouse bone marrow. However, to bring this potential therapy a step closer to clinical application, a human bone marrow cell extract (BMCE) needs to be tested. Here, we report that irradiation-induced injury of salivary glands in immunocompetent mice treated with human BMCE secreted 50% more saliva than saline-injected mice, and BMCE did not cause additional acute inflammatory reaction. In addition, to identify the cell fraction in BMCE with the most therapeutic activity, we sorted human bone marrow into 3 cell fractions (mononuclear, granulocyte, and red blood cells) and tested their respective cell extracts. We identified that the mononuclear cell extract (MCE) provided the best therapeutic efficacy. It increased salivary flow 50% to 73% for 16 wk, preserved salivary parenchymal and stromal cells, and doubled cell proliferation rates while producing less inflammatory response. In contrast, the cell extract of granulocytes was of shorter efficacy and induced an acute inflammatory response, while that from red blood cells was not therapeutically effective for salivary function. Several proangiogenic (MMP-8, MMP-9, VEGF, uPA) and antiangiogenic factors (TSP-1, PF4, TIMP-1, PAI-1) were identified in MCE. Added advantages of BMCE and MCE for potential clinical use were that cell extracts from both male and female donors were comparably bioactive and that cell extracts could be stored and transported much more conveniently than cells. These findings suggest human BMCE, specifically the MCE fraction, is a promising therapy against irradiation-induced salivary hypofunction.
Introduction
Radiotherapy is a common treatment for millions of head and neck cancer patients worldwide. Unfortunately, a major sequela following irradiation (IR) is salivary gland (SG) hypofunction, which frequently remains permanent and adversely affects the quality of life of head and neck cancer survivors (Haddad and Limaye 2016). Less invasive techniques such as intensity-modulated radiation therapy are available to protect SGs from coirradiation damage (Braam et al. 2006; Trotti and Eisbruch 2011). However, in some cases, due to factors such as the location or severity of the tumor (Robar et al. 2007), SGs are still being injured. Recently, a cell-free biological therapy using an extract of bone marrow cells (BMCs) was proposed because it would be theoretically less thromboembolic and immunogenic than other experimental approaches, such as gene or stem cell–based therapy (Ferreira et al. 2018; Mohamed et al. 2022), being currently evaluated for restoring saliva secretion (Angeli et al. 2012). This cell-free extract consisted of soluble contents lysed from whole cells. We reported that the administration of the cell extract from mouse bone marrow (msBMCE) was therapeutically as effective as the transplantation of whole live BMCs in partially restoring saliva secretion to IR-injured SGs (Tran et al. 2013). In addition, we and other research groups had noted several clinically relevant advantages of using cell-free extracts. First, the administration of a cell extract would eliminate the problem of low cell survival during cell transplantation (Tongers et al. 2011). Second, cell extracts (CEs) could be stored frozen or be lyophilized and then be subsequently used when needed, as compared to requiring fresh cells (Fang, Su, et al. 2018). Third, lyophilized CEs could be an off-the-shelf product and easily transported at ambient temperature, as compared to cryopreserved cells requiring specialized facilities. Ultimately, these advantages would reduce treatment costs for patients and doctors (Su et al. 2018). However, in order to bring this proposed cell-free therapy for SG hypofunction to the clinic, a CE originating from human bone marrow (BM) needs to be tested.
Thus, the first objective of this study was to test the therapeutic effect of a cell extract from human bone marrow (BMCE) on IR-injured SGs in a mouse model. Second, because human whole BM is composed of heterogeneous cell populations, the second objective of this study was to determine whether there was any specificity among the major classes of BMCs. To do this, we separated human whole BM into its 3 major cell fractions: 1) red blood cells (RBCs), 2) granulocytes (GCs), and 3) mononuclear cells (MCs). The CE of each major cell fraction was then generated (red blood cell extract [RBCE], granulocyte cell extract [GCE], mononuclear cell extract [MCE]) and tested for their therapeutic efficiency in restoring salivary function. Finally, the third objective of this study was to compare the effect of CEs obtained from male versus female BM donors on the restoration of salivary secretion. In summary, our objectives were to test the effect of human BMCE and to investigate whether a specific major cell fraction in BM contributed more to the repair of IR-injured SGs. We also compared if BMCE therapeutic effect was affected by the sex of human BM donors.
Herein, we report that human BMCE provides a therapeutic effect in partially restoring function to IR-injured SGs. Furthermore, we identify multiple growth factors in human BMCE, RBCE, GCE, and MCE and unveil that the cell-free extract from the mononuclear cell fraction acts as the major effective constituent of human BMCE, while RBCE does not significantly repair IR-damaged SGs and that the GCE results in an acute inflammatory response. Finally, the sex of the BM donors does not affect CEs’ efficiency. These findings suggest human MCE as a promising therapy against IR-induced salivary hypofunction because MCE possesses these advantages: 1) better therapeutic efficacy than RBCE and GCE, 2) less inflammatory response, 3) more accessible and ample cell source, 4) no sex difference, and 5) convenient storage and transport.
Materials and Methods
Major Cell Fractions Isolated from Human Bone Marrow
Human bone marrow samples (n = 4; 2 males and 2 females; 20–34 y old) were purchased from Lonza. Ficoll-Paque PLUS density gradient media (GE Healthcare Life Sciences) was used to separate human BM samples into 3 fractions: red blood cells, granulocytes, and mononuclear cells (see Appendix).
Preparation of Cell Extracts
Cell extracts were prepared exactly as described in our previous studies (Tran et al. 2013; Su et al. 2020). In brief, cells were resuspended with 0.9% saline to a concentration of 107 cells/100 µL. Then, 3 cycles of freezing (−80°C) and thawing (37°C) were performed to lyse the cells. After centrifugation at 17,000 g for 30 min at 4°C, the supernatant (defined as the CE) was stored at −80°C until used for experiments.
Irradiation
The protocol to induce irradiation injury to SGs of mice was described previously (Fang, Shang, et al. 2018) (see Appendix).
Ninety-six mice were randomly divided into 7groups (n = 12–15 mice per group): 1) sham IR group (no irradiation, no injection), 2) saline group (IR + normal saline intravenous injection), 3) BMCE group (IR + human BMCE intravenous injection), 4) RBCE group (IR + human RBCE intravenous injection), 5) GCE group (IR + human GCE intravenous injection), 6) MCE group (IR + human MCE intravenous injection), and 7) msBMCE group (IR + mouse BMCE intravenous injection). According to their groups, mice were injected with either 100 μL normal saline or CE (2 mg/mL) through their tail vein at 5 to 7 d postirradiation, once per week for 2 consecutive weeks. Mice were sacrificed at 3 h after treatment, 24 h after treatment, 8 wk post-IR, or 16 wk post-IR. For the experiment comparing the effects of the sex of human CE donors, the scientist injecting the CE was blinded to the sex of the human donor into mice. There were no other criteria used for including or excluding animals.
Measurements
Salivary flow rate (SFR) and lag time measurements were performed at weeks 4, 8, 12, and 16 post-IR as described previously (Fang et al. 2015). Histological analyses, such as hematoxylin/eosin and proliferating cell nuclear antigen (PCNA), immunofluorescence staining using aquaporin 5 (AQP5, to detect acinar cells), α–smooth muscle actin(α-SMA, myoepithelial cells), cytokeratin 5 (CK5, basal ductal cells and a subpopulation of myoepithelial cells), GFRα2 (parasympathetic nerves), CD31 (endothelial cells), and quantitative real-time polymerase chain reaction (PCR) for EGF, VEGF, IGF-1r, HGF, FGF2, BMP7, NGF, MMP2, AQP5, and GAPDH, were done as previously described (Fang et al. 2015). Enzyme-linked immunosorbent assays were performed according to the manufacturer’s instructions to quantify interleukin (IL)–6, serum amyloid A (SAA), IL-10, IL-1Ra.
Statistics
SPSS version 19 software (SPSS, Inc.) was used to perform the statistical analyses. All data are presented as mean ± SD. One-way analysis of variance (ANOVA) with Tukey’s post hoc tests was used to determine statistical significance of differences (P < 0.05) between each group.
Results
Human BMCE Partially Restores Functionto IR-Injured SGs in an In Vivo Mouse Model
Because human BMCE has not yet been tested for treating SG hypofunction, this study used msBMCE as a positive control, since msBMCE was shown to be successful in previous studies to restore salivary function (Tran et al. 2013). By measuring SFRs, we found that both mouse and human BMCE partially restored salivary function, while IR mice injected with saline (negative vehicle control group) did not (Fig. 1A, P < 0.05). Human BMCE-treated mice had a 50% and 43% increase, respectively, in SFR at 8 and 16 wk post-IR when compared to saline-injected mice (Fig. 1A, P < 0.05). Another measure of salivary function is the lag time to salivation. This is evaluated by injecting pilocarpine and then measuring the delay of time (“lag time”) it takes to observe the first drop of saliva (Fig. 1B). Human BMCE- and msBMCE-treated mice had decreased lag time when compared to saline-injected IR mice, which indicates improved salivary function (Fig. 1B; P < 0.05). Lag time to salivation in human BMCE- and msBMCE-treated mice showed no statistical difference from the unirradiated sham controls (Fig. 1B; P = 0.17, denoted by symbol “#”).
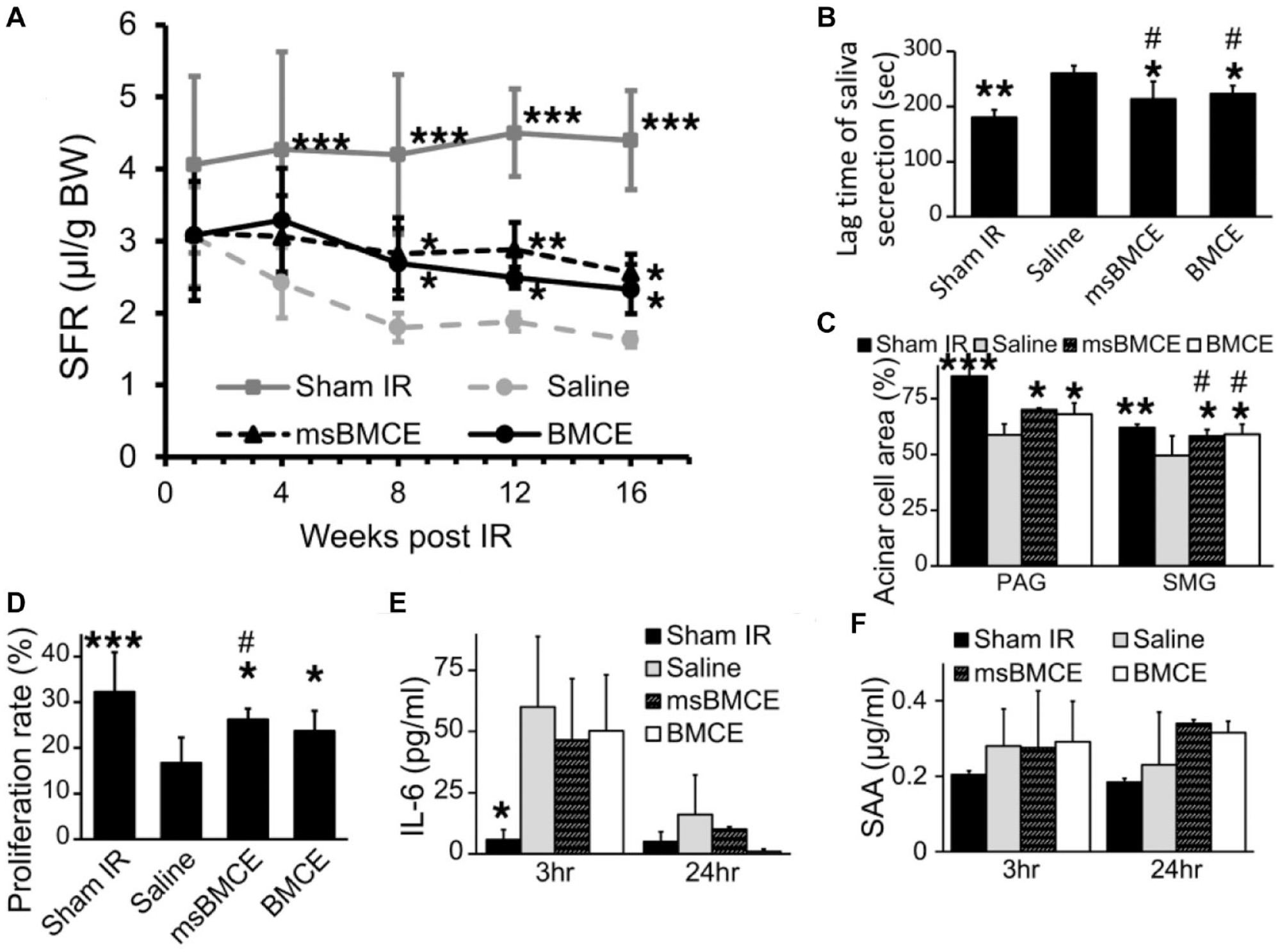
Cell extracts from both human bone marrow cell extract (BMCE) and mouse BMCE (msBMCE) can partially restore function to irradiation (IR)–injured salivary glands. (
Furthermore, we performed histological studies to correlate with salivary function measures. The percentage of acinar cell area was higher in the BMCE group (68.1% ± 5.1% in parotid gland [PAG] and 59.0 ± 4.5% in submandibular gland [SMG]) than in the saline group (59.4% ± 9.1% in PAG and 50.1% ± 5.7% in SMG) (Fig. 1C, P < 0.05). The percentage of acinar cell area in SMG was not statistically different from the unirradiated sham controls (Fig. 1C; P > 0.05). The cell proliferation rate (Fig. 1D), measured by staining for the PCNA, was significantly higher in the BMCE group (22.7% ± 4.4%) than that of the saline group (13.4% ± 4.9%, P < 0.05). To test whether an acute phase reaction occurs following administration of BMCE, we used enzyme-linked immunosorbent assay (ELISA) to measure plasma levels of acute phase proteins and found that injections of mouse and human BMCE did not increase IL-6 and SAA levels at both 3 h and 24 h postinjection when compared to saline injections (Fig. 1E, F). All these results suggest that human BMCE partially restores IR-damaged SGs with no additional acute inflammatory reaction.
Cell Extract of Mononuclear Cells Provides a Better and More Sustained Salivary Therapeutic Response Than Cell Extracts from the 2 Other Cell Fractions in Human Bone Marrow
To examine whether a specific extract of human BM cell fraction has a higher therapeutic effect in mitigating IR injury to SGs, we used Ficoll-Paque density gradient centrifugation to separate human BM into 3 cell fractions. The CE of each cell fraction was then generated (RBCE, GCE, and MCE) and injected into mice with IR-injured SGs. At 8 wk post-IR, MCE-treated mice had a 73% improvement in SFR when compared to the saline group (3.12 ± 0.72 µL/g vs. 1.8 ± 0.20 µL/g, P < 0.05, Fig. 2A), while the GCE-treated group had a 60% increase of SFR (2.88 ± 0.49 µL/g, P < 0.05). The RBCE-treated mice did not have a statistically significant increase in SFR compared to saline-injected mice (31% more SFR, 2.36 ± 0.55 µL/g, P > 0.05). SFR of the MCE-treated mice was not significantly different from the sham group at 8 wk (denoted by the symbol # in Fig. 2A). At 16 wk post-IR, MCE-treated mice still maintained a 50% increase in SFR (P < 0.05, Fig. 2A). This indicates that MCE treatment could sustain SFR at 8 wk but only partially restore SFR at 12 and 16 wk. Furthermore, the lag time of saliva secretion was significantly reduced in the MCE group when compared to the saline-injected group at 8 and 16 wk post-IR (Fig. 2B). Importantly, the lag time of saliva secretion, proliferation cell rate, and area of acinar cell area in MCE-treated mice showed no statistical difference from the unirradiated sham controls (Fig. 2B–D; P > 0.14).
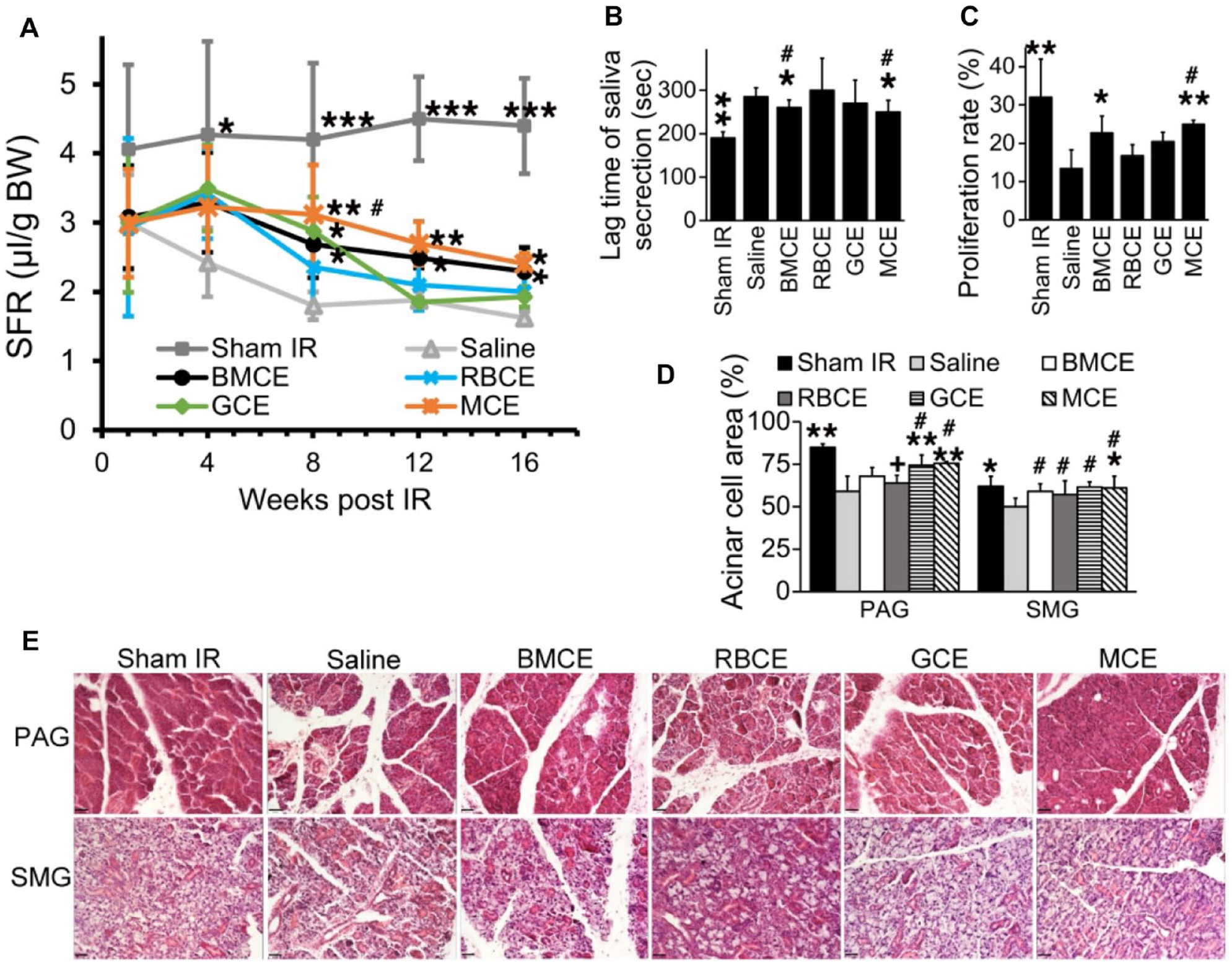
Cell extracts from mononuclear cells (MCE) and granulocytes (GCE) restored irradiation (IR)–injured salivary glands, with MCE having a prolonged and sustained therapeutic effect. (
MCE and GCE provided mitigation of IR injury for several types of cells in PAG and SMG. First, PCNA staining results showed that the percentage of proliferating cells in SMG was higher in the MCE group (25.2% ± 1.0%) versus that in the saline group (13.4% ± 4.9%, Fig. 2C, P < 0.01). Second, H&E photomicrographs revealed larger areas with acinar cells in both PAG and SMG in MCE and GCE groups when compared with that of the saline group (Fig. 2D, E, P < 0.05). Third, we measured the effect of human BMCE on additional types of salivary parenchymal (such as ductal) and stromal cells (endothelial, myoepithelial, nerves) using immunofluorescence staining for markers such as AQP5 to identify acinar cells, CK5 for salivary ductal basal cells, α-SMA for myoepithelial cells, CD31 for blood vessel endothelial cells, and GFRα2 for parasympathetic nerves. BMCE treatment resulted in a 2.5-fold increase in blood vessel relative fluorescence intensity (CD31) and a 2-fold increase in innervation (GFRα2) when compared to saline-treated mice (Fig. 3, P < 0.05) in PAG. Also, human BMCE-treated mice had a higher number of cells positive for AQP5, CK5, and α-SMA markers in PAG when compared to the saline-treated mice (Fig. 3B–F, P < 0.05). Cell extracts of BM cell fractions protected selected parenchymal (acinar, ductal) and stromal cell types (endothelial, myoepithelial, nerves) in IR-injured SGs (Fig. 3). For example, MCE protected parenchymal and stromal cells in PAG, while GCE protected acinar cells in SMG, ductal cells in PAG, and endothelial and nerve cells in PAG.
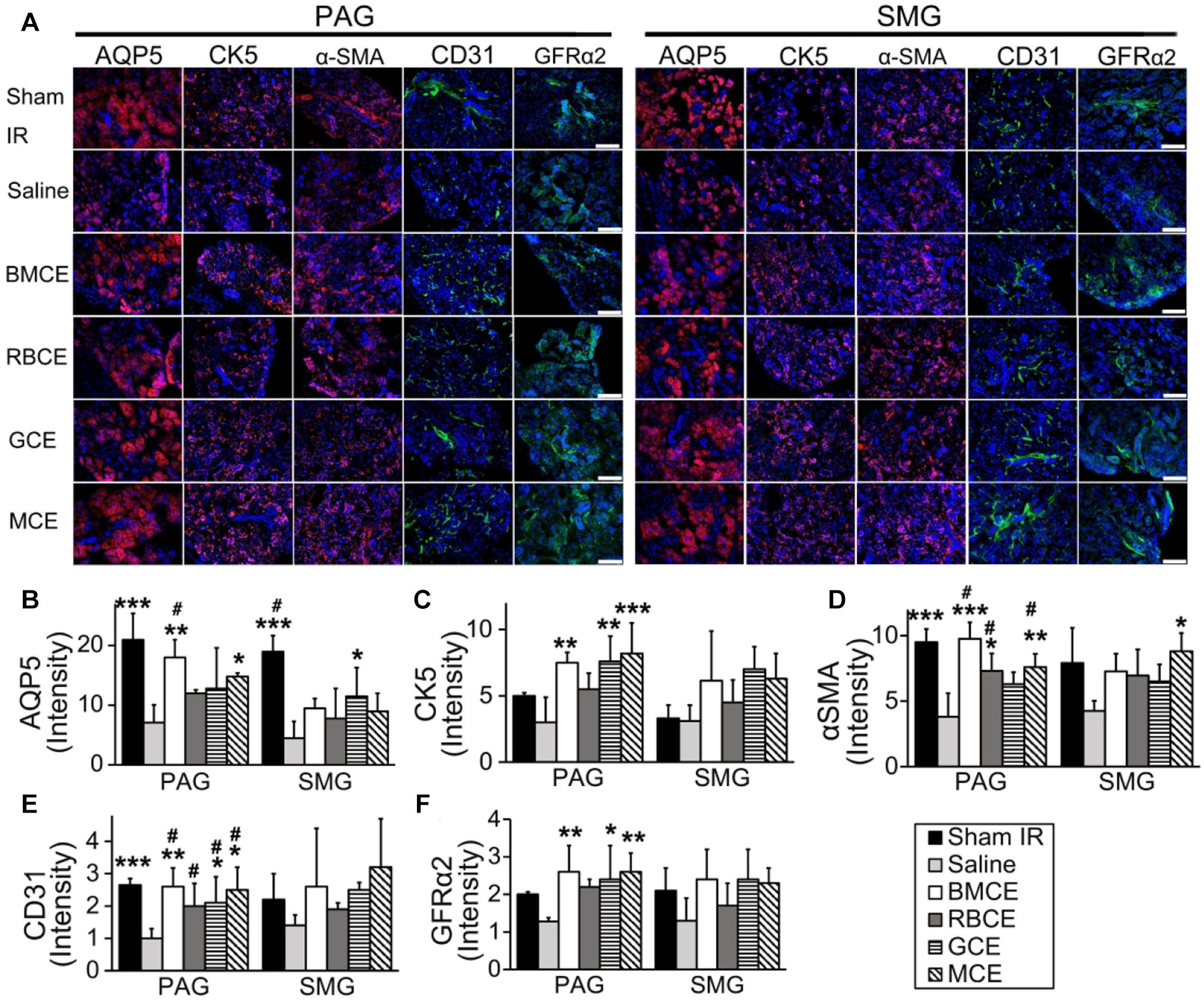
Cell extracts of mononuclear cells (MCE) and granulocytes (GCE) protected salivary parenchymal and stromal cells from irradiation injury. (
Fourth, we also quantified messenger RNA (mRNA) levels in SGs using quantitative reverse transcription PCR. In BMCE-treated mice, gene expression of growth factors involved in SG repair and development (such as EGF, IGF1r, BMP7), blood vessels, and nerve repair/regeneration (MMP2, NGF) was upregulated (Fig. 4A, P < 0.05). For CEs of BMC fractions, the gene expression levels of HGF, AQP5, and FGF2 were upregulated in the MCE group (Fig. 4A, P < 0.05). Furthermore, the expression of FGF2 and MMP2 in the MCE group was significantly higher when compared with that in BMCE and RBCE groups (Fig. 4A, P < 0.05).
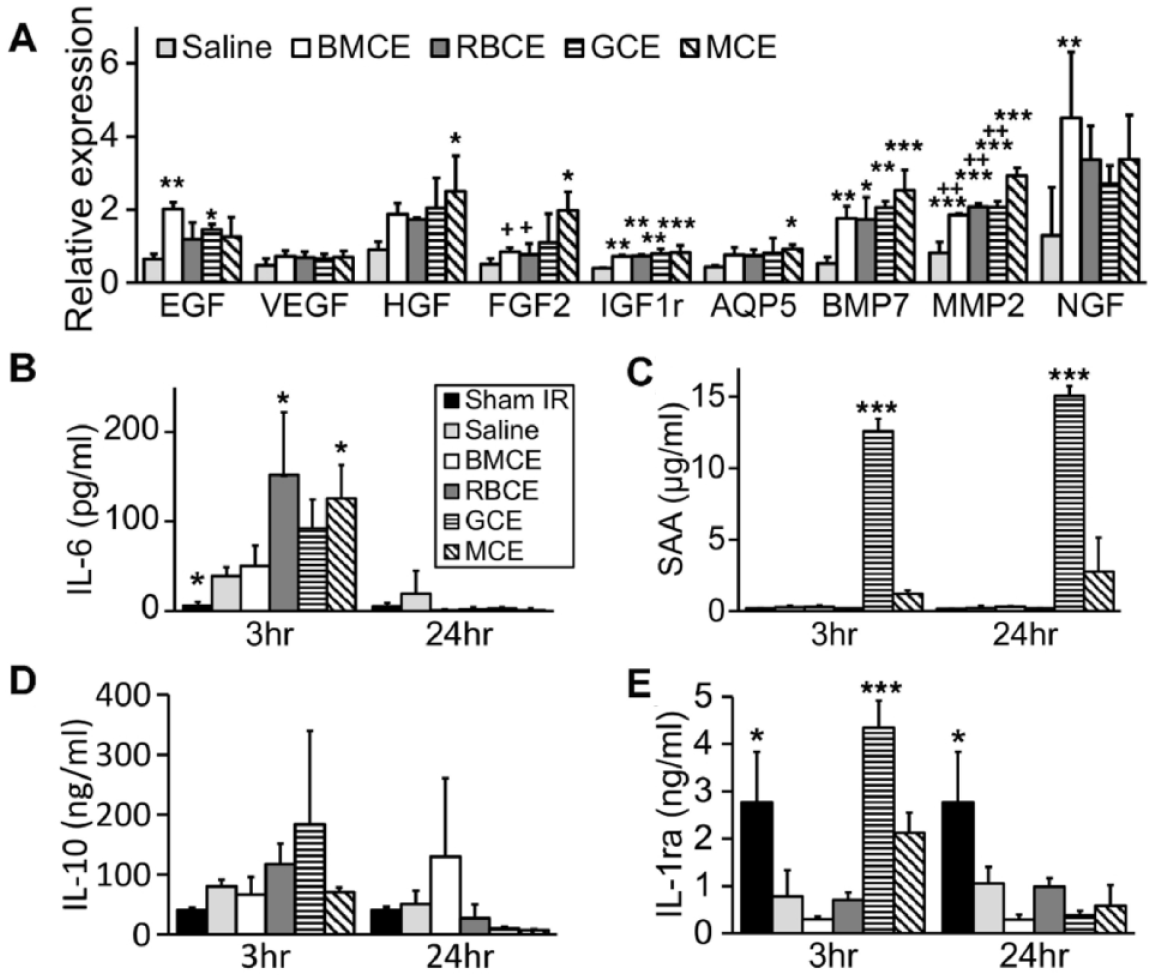
Immune responses elicited by bone marrow cell extract (BMCE) were concentrated in granulocyte cell extract (GCE), while mononuclear cell extract (MCE) induced a milder acute phase reaction. (
To test if an acute phase reaction occurs following administration of human CEs from BMC fractions, we used ELISA to measure levels of proinflammatory (IL-6, SAA) and anti-inflammatory (IL-10, IL-1Ra) cytokines in the plasma of mice. IL-6 levels were increased in CE-treated groups at 3 h postinjection when compared to the saline group (Fig. 4B). However, at 24 h postinjection, IL-6 levels were not different between CE- and saline-injected groups. SAA was significantly higher in GCE-treated mice at 3 h and 24 h postinjection (Fig. 4C, P < 0.001). GCE-treated mice also had higher levels of the anti-inflammatory cytokines IL-10 and IL-1Ra at 3 h postinjection (Fig. 4D, E). These results suggest that the immunological responses to BMCE are concentrated in GCE, whereas MCE induces a milder acute phase reaction. Taken together, these data indicate that MCE treatment partially restores the secretory function of SGs and maintains this effect at a clinically therapeutic level for 16 wk. GCE has a relatively short efficacy and results in an acute inflammatory response, while RBCE is not therapeutic in restoring the function of IR-injured SGs.
Characterization of Human BMCE and Cell Extracts Isolated from BM Cell Fractions
We profiled proteins in human BMCE using a protein array for quantifying angiogenesis-related growth factors based on the findings that BMCE-treated mice preserved ~2.5 times more endothelial cells (Fig. 3E, P < 0.05). As a control, we compared the growth factors of msBMCE (Fig. 5A). Several proteins in human BMCE were not detected in msBMCE, such as TSP-1, pentraxin3, PAI-1, ET-1, angiopoietin 2, persephin, and VEGF (Fig. 5A). When CEs of human BMC fractions were compared (Fig. 5B), MCE contained the most growth factors, including proangiogenic factors (MMP-8, MMP-9, VEGF, angiopoietin 2, uPA, IL-8, artemin, and ET-1), antiangiogenic factors (TSP-1, PF4, TIMP-1, PEDF, and PAI-1), and biphasic angiogenic factors (TGF-β1 and PTX3). Other factors such as uPA, TGF-β1, IL-8, TSP-2, and FGF-1 were not detected in RBCE, while VEGF, PEDF, CD26, and EG-VEGF were not detected in GCE. Although several identical growth factors were present in BMCE, RBCE, GCE, and MCE, their concentrations differed. For example, a higher level of TSP-1 was detected in MCE than in GCE. These data indicate that different mixtures of pro- and antiangiogenic factors exist in BMCE, RBCE, GCE, and MCE. Furthermore, we compared whether the sex of human BM donors had an effect on restoring the salivary function of IR-injured SGs. The CEs harvested from female versus male human donors were equally effective in repairing IR-injured SGs (Appendix Fig. 1).
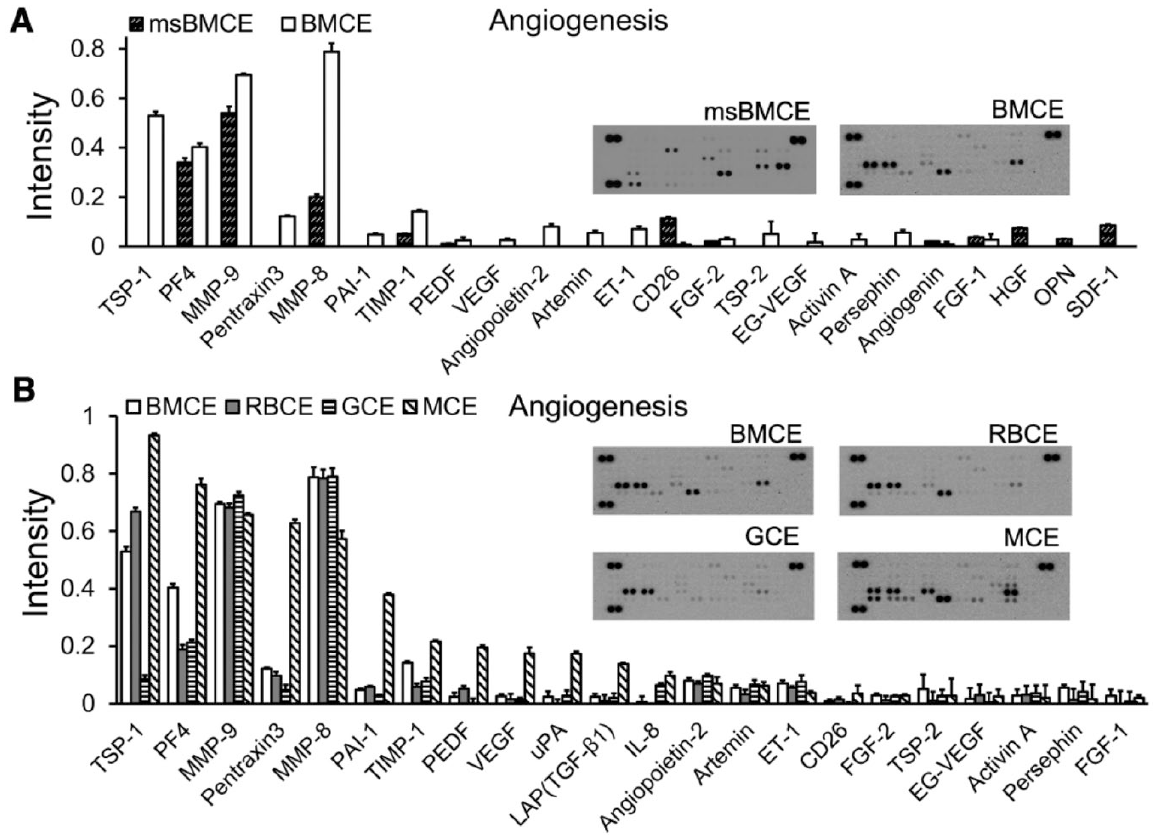
Different mixtures of pro- and antiangiogenic factors existed in bone marrow cell extract (BMCE), red blood cell extract (RBCE), granulocyte cell extract (GCE), and mononuclear cell extract (MCE). A protein array was used to identify angiogenesis-related growth factors present in (
Discussion
There are multiple other approaches under way to repair radiation injured SGs. Two promising approaches, gene- and cell-based therapies, are being tested in phase I/II clinical studies (Baumet al. 2012; Alevizos et al. 2017; Grønhøj et al. 2018; Sumita et al. 2020; Blitzer et al. 2022). Still, a longer period of observation is required to ensure safety concerns such as the potential risk of thromboembolism, tumorigenesis, or cell rejection. Our approach of using CEs is distinct from current approaches and offers advantages, such as avoiding the need to nurture stem cells to an appropriate differentiated stage or avoiding the need for gene therapy. Cumulatively, our data show that human BMCE partially restores salivary function of IR-damaged SGs to a clinically relevant therapeutic level. To be therapeutically successful, a 40% to 65% improvement in salivary function is considered clinically sufficient for alleviating the side effects of diminished salivary function in patients (Baum et al. 2012; Tran et al. 2017). We identify that the MCE provides the most effective and sustained therapeutic effect, which may be due in part to a mixture of pro- and antiangiogenic factors. The specific compositions of proteins varied both in presence and in concentrations between the 3 major cell fractions, with MCE having higher concentrations of angiogenic factors. In addition, several neurogenesis factors were identified, such as PTX3 (Rodriguez-Grande et al. 2015), persephin, and artemin. This may explain our recent report that BMCE decreased systemic inflammation and neuropathic pain in a mouse model of partial sciatic nerve injury (Sam Zhou et al. 2022). These findings suggest that interactions involving multiple growth factors in the CE, rather than 1 or 2 essential factors, are needed for the observed therapeutic effects.
MC, GC, and RBC are the 3 major cell fractions in BM that can be rapidly separated for clinical use. Other minor cell fractions (<0.1% in human BM samples) exist but are more time-consuming to separate, and their low yield would become a limitation for clinical applications. An additional reason for focusing on these specific major classes of BMC was the reported therapeutic potentials of MC or GC to treat conditions such as brain, cardiac, and SG injury (Kumar et al. 2009; Yeghiazarians et al. 2010; Cox et al. 2017; Dolati et al. 2019). MC is a heterogeneous fraction comprising diverse cell types, including differentiated cells (lymphocytes, monocytes) and progenitor cells (hematopoietic, endothelial progenitor, mesenchymal stromal, and small embryonic-like stem cells). The beneficial effects of MC treatment observed in clinical trials may be due to the combined effects of all MCs (Zhang et al. 2018; Wang et al. 2019). The advantage of MC therapy is that cells can be easily harvested and used with minimal processing when compared to cultured cells (Boyle et al. 2006; Sadek et al. 2009).
Human BMCE was reportedly successful as a xenograft when injected into an immune-competent mouse model of myocardial infarction, and it resulted in improved cardiac function without a severe immune response (Angeli et al. 2012). These results (and ours in SGs) suggest that CEs are relatively less immunogenic than the transplantation of intact cells, which opens possibilities for clinical allotransplantation of CEs. To test the immunogenicity of cell extracts, pro- and anti-inflammatory factors were assayed in this study. Mice in the GCE group had an increase in SAA at 24 h postinjection, while only a mild increase in SAA was detected in the MCE group, when compared to the saline group (Fig. 4C). IL-10 and IL-1Ra levels were higher at 3 h post-GCE injection (Fig. 4D, E). This indicated that GCE was involved in immunomodulation. Clinical studies have reported that mild acute reactions occurred and lasted for a week following infusions of autologous MCs to patients with brain or spinal cord injuries and that a low dose of MCs was safe (Kumar et al. 2009; Cox et al. 2017). Our results revealed that BMCE, which consisted of a lower concentration of GCE and MCE, induced comparable levels of inflammation to the saline group. In addition, this study injected human BMCE in immune-competent mice. The immunoreaction would be reduced if autologous or allogeneic human-derived CEs were injected in patients. Thus, theoretically, by further optimizing the concentration of CEs, this would be safe for patient testing.
Our findings that the MCE fraction is therapeutically active in mitigating radiation-injured SGs are in agreement with results from Sumita and colleagues, who reported an effective culture method to enhance the anti-inflammatory and vasculogenic phenotypes of peripheral blood mononuclear cells (PBMNCs) to treat radiation-injured SGs in mice (I et al. 2019). The same group is currently testing this approach of conditioning PBMNCs in a phase 1 clinical study for patients with severe radiation-induced xerostomia (Sumita et al. 2020). Our proposed MCE therapy can offer added benefits by being less immunogenic than injecting whole cells, and the CEs can be stored long term. Thus, MCE treatment can provide a promising alternative to cell-based therapies for SGs and other organs injured by radiotherapy.
Author Contributions
X. Su, S.D. Tran, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; Y. Liu, contributed to conception and design, data acquisition, analysis, and interpretation; critically revised the manuscript; O. ElKashty, J. Seuntjens, K.M. Yamada, contributed to conception and data design, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345221112332 – Supplemental material for Human Bone Marrow Cell Extracts Mitigate Radiation Injury to Salivary Gland
Supplemental material, sj-docx-1-jdr-10.1177_00220345221112332 for Human Bone Marrow Cell Extracts Mitigate Radiation Injury to Salivary Gland by X. Su, Y. Liu, O. ElKashty, J. Seuntjens, K.M. Yamada and S.D. Tran in Journal of Dental Research
Footnotes
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Canadian Institutes of Health Research (grant PJT-159577) and China Postdoctoral Science Foundation (2021M693622). The authors also acknowledge the financial support from the China Scholarship Council and thank the Center for Bone and Periodontal Research (McGill University). K.M. Yamada is supported by the Intramural Research Program of the National Institutes of Health, National Institute of Dental and Craniofacial Research. The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
