Abstract
Periodontitis is a highly prevalent chronic inflammatory disease that progressively destroys the structures supporting teeth, leading to tooth loss. Periodontal tissue is innervated by abundant pain-sensing primary afferents expressing neuropeptides and transient receptor potential vanilloid 1 (TRPV1). However, the roles of nociceptive nerves in periodontitis and bone destruction are controversial. The placement of ligature around the maxillary second molar or the oral inoculation of pathogenic bacteria induced alveolar bone destruction in mice. Chemical ablation of nociceptive neurons in the trigeminal ganglia achieved by intraganglionic injection of resiniferatoxin decreased bone loss in mouse models of experimental periodontitis. Consistently, ablation of nociceptive neurons decreased the number of osteoclasts in alveolar bone under periodontitis. The roles of nociceptors were also determined by the functional inhibition of TRPV1-expressing trigeminal afferents using an inhibitory designer receptor exclusively activated by designer drugs (DREADD) receptor. Noninvasive chemogenetic functional silencing of TRPV1-expressing trigeminal afferents not only decreased induction but also reduced the progression of bone loss in periodontitis. The infiltration of leukocytes and neutrophils to the periodontium increased at the site of ligature, which was accompanied by increased amount of proinflammatory cytokines, such as receptor activator of nuclear factor κΒ ligand, tumor necrosis factor, and interleukin 1β. The extents of increase in immune cell infiltration and cytokines were significantly lower in mice with nociceptor ablation. In contrast, the ablation of nociceptors did not alter the periodontal microbiome under the conditions of control and periodontitis. Altogether, these results indicate that TRPV1-expressing afferents increase bone destruction in periodontitis by promoting hyperactive host responses in the periodontium. We suggest that specific targeting of neuroimmune and neuroskeletal regulation can offer promising therapeutic targets for periodontitis supplementing conventional treatments.
Introduction
Periodontitis is a chronic inflammatory disease affecting approximately 40% of the adult population in the United States (Eke et al. 2012). Periodontitis causes destruction of the periodontium, including alveolar bone and periodontal ligament, eventually resulting in tooth loss if left untreated. Current approaches to delaying the progression of the disease or regenerating lost periodontium are unsatisfactory, and novel treatment approaches are critically needed. The destruction of periodontal tissues during the progression of periodontitis is primarily due to the dysregulation of local host inflammatory or immune responses, in conjunction with microbial dysbiosis (de Vries et al. 2017; Lamont et al. 2018). Consequently, numerous factors regulating periodontal host responses can affect the progression of periodontitis.
The periodontium contains numerous sensory nerves that transduce noxious stimuli. These nociceptive afferents often contain neuropeptides and express high levels of the capsaicin receptor transient receptor potential vanilloid 1 (TRPV1), a Ca2+-permeable nonselective cationic channel (Chung, Jung, and Oh 2011). TRPV1+ peptidergic afferents modulate the function of barrier tissue in the skin, lung, and gut and also regulate host responses under bacterial infection and tissue injury (Baral et al. 2019). Nociceptive afferent-mediated modulation of host responses results in either protective or destructive outcomes in different contexts (Razavi et al. 2006; Talbot et al. 2015; Pinho-Ribeiro et al. 2018). However, their roles in modulating host responses in periodontitis are not well understood. Previous studies evaluating the neural control of bone remodeling and periodontal bone loss have produced contradictory results (Hill et al. 1991; Adam et al. 2000; Offley et al. 2005; Breivik et al. 2011; Takahashi et al. 2016), creating a need to clarify the role of TRPV1+ afferents in periodontitis.
In this study, we tested the hypothesis that TRPV1+ nociceptive afferents aggravate periodontitis by promoting hyperactive host immune responses.
Materials and Methods
Experimental Animals
All animal procedures were performed according to the NIH Guide for the Care and Use of Laboratory Animals (Publication 85–23, Revised 1996), a protocol approved by the University of Maryland Institutional Animal Care and Use Committee, and the ARRIVE guidelines. C57Bl/6,
Mouse Models of Periodontitis
Ligature-induced periodontitis was initiated by placing a 5–0 silk ligature around the maxillary left second molar (Abe and Hajishengallis 2013). The sutures were tied gently to prevent damage to the periodontal tissues. The ligatures remained in place in all mice throughout the indicated experimental period. In a separate experiment, bacteria-induced periodontitis was induced as described previously by oral inoculation of
Microinjection into the Trigeminal Ganglia
To selectively ablate TRPV1-expressing trigeminal nociceptors, resiniferatoxin (RTX) was directly injected into the trigeminal ganglia (TG) (Wang et al. 2019; Wang and Chung 2020). RTX is a highly efficacious agonist of TRPV1, and the activation of TRPV1 by RTX leads to ablation of nociceptors upon localized injection (Chung and Campbell 2016). In some experiments, adeno-associated virus (AAV) encoding inhibitory designer receptor exclusively activated by designer drugs (DREADD) receptor (AAV5-hSyn-DIO-hM4Di-mCherry) or green fluorescent protein (GFP; AAV5-hSyn-DIO-eGFP) was injected.
Osmotic Pump Implantation
Micro-osmotic pumps (Alzet; 100 µL) were filled with clozapine-N-oxide (CNO), a chemical actuator of DREADD, and inserted under back skin. This allows slow release of CNO for 14 d.
Micro–Focus Computed Tomography
After transcardial perfusion using 3.7% paraformaldehyde, the maxillae were hemisected, and micro–focus computed tomography (µCT) scanning was performed using Siemens Inveon Micro-PET/SPECT/CT (Siemens) with 9 µm spatial resolution. The linear distance from the cementoenamel junction (CEJ) to the alveolar bone crest (ABC) on the buccal side was measured (Abe and Hajishengallis 2013; Xiao et al. 2015).
Histological Assays
Immunohistochemical assays of TG and decalcified maxillae were performed as previously described (Chung, Lee, et al. 2011; Chung et al. 2012; Wang et al. 2017). Conventional immunohistochemical procedures were performed using rabbit anti-TRPV1 or chicken anti-GFP. For tartrate-resistant acid phosphatase (TRAP) staining, the maxillae were decalcified in 0.5 M EDTA, embedded in paraffin, and sectioned into 5-µm thickness. TRAP staining was performed using a commercial kit (Wako Pure Chemical).
Flow Cytometry
We prepared single-cell suspensions from gingival tissues according to a previously published method (Dutzan et al. 2016). The mice were transcardially perfused using >20 mL phosphate-buffered saline (PBS) to flush out the immune cells from the vasculature. Gingival tissues around 3 molars on both the buccal and palatal sides were dissected out, digested in type IV collagenase and DNase, and mashed through a cell strainer. Flow cytometry was performed using a Cytek Aurora flow cytometer (Cytek Biosciences). For detecting neutrophils, we used CD45-PE, CD11b-BV42, and Ly6G-PE-Cy7. 7-Aminoactinomycin D (7-AAD) was used to exclude dead cells from analysis. The results were analyzed using FCS Express 7 software.
Real-Time PCR
Maxillae were dissected out and total RNA was extracted using Trizol and purified using a Direct-zol RNA MicroPrep kit (Zymo Research). Real-time polymerase chain reaction (PCR) was performed and analyzed as described previously (Wang and Chung 2020) using the primer pairs described in Appendix methods. Relative quantification of messenger RNA (mRNA) was achieved using the 2−ΔΔCt method.
Luminex Multiplex Cytokine Assay and Enzyme-Linked Immunosorbent Assay
Maxillae were dissected out and ground in a Tris buffer containing a protease/phosphatase inhibitor cocktail, and the supernatant was used for the Luminex assays. Circulating norepinephrine was measured from blood collected retro-orbitally using a norepinephrine enzyme-linked immunosorbent assay (ELISA) kit.
16S Ribosomal RNA Sequencing
Bacterial 16S ribosomal RNA (rRNA) genes were PCR amplified with dual-barcoded primers targeting the V4 region. The amplicons were sequenced with an Illumina MiSeq, taxonomically classified, and clustered into operational taxonomic units (OTUs) using the Mothur software package. The α diversity was estimated using the Shannon index on raw OTU abundance tables. To estimate the β diversity across samples, we computed the Bray–Curtis indices, and the variation in community structure was evaluated using permutational multivariate analyses of variance (ANOVAs). The results were uploaded to NCBI SRA (PRJNA750467).
Statistical Analysis
All data are presented as mean ± SEM. The data were analyzed using Student’s
Results
TRPV1+ Afferents Drive Periodontitis-Induced Bone Loss
To determine the roles of TRPV1+ afferents in periodontitis, RTX was stereotaxically microinjected into the bilateral TG of adult mice (Fig. 1A). One week after RTX injection, the effects of nerve ablation were assessed using a ligature-induced periodontitis model. A ligature was tied around the maxillary left second molar, with the contralateral unligatured tooth serving as a control. The mice were euthanized 12 to 14 d after the placement of the ligature. The extent of bone loss was evaluated using µCT (Fig. 1B). Significant crestal bone loss around the second molars was noted in both vehicle (Veh) and RTX-treated groups (Fig. 1B, C). However, the extent of bone loss was approximately 45% lower in RTX-injected mice (
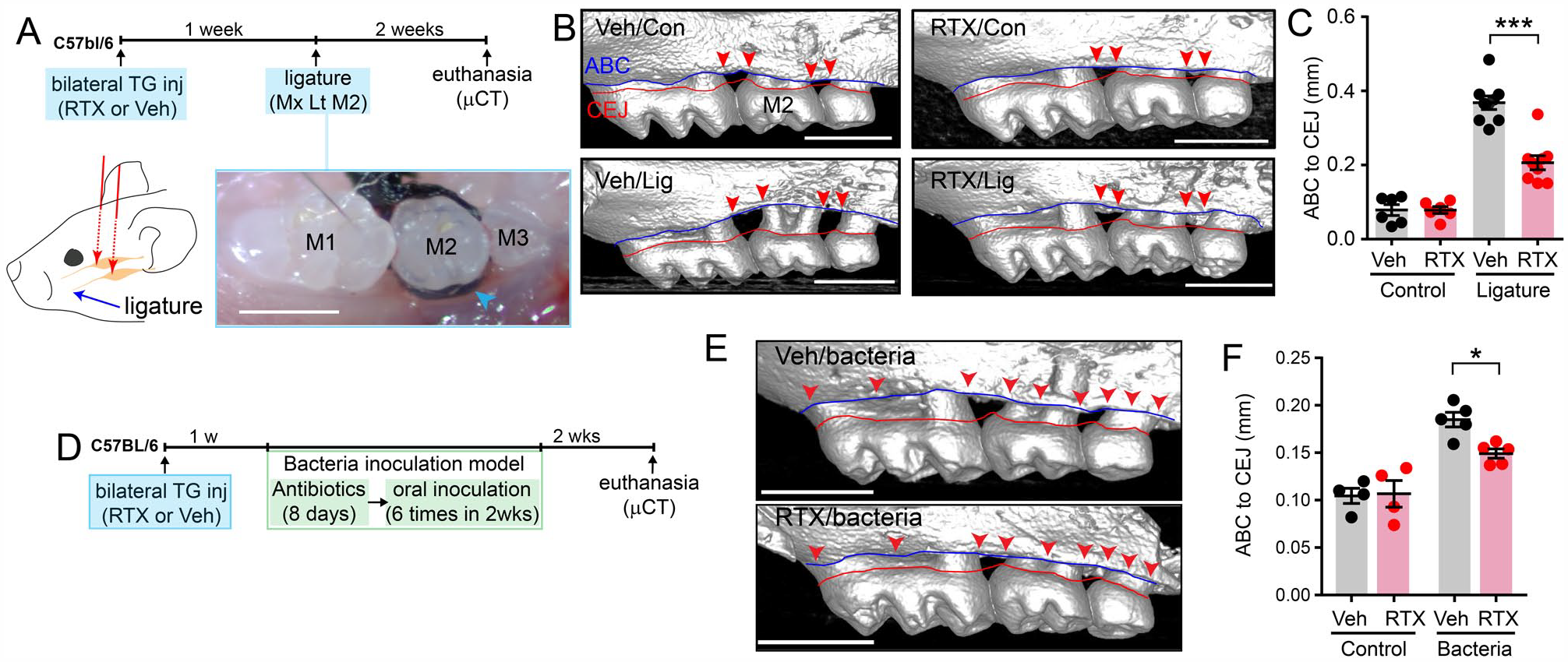
Ablation of transient receptor potential vanilloid 1+ (TRPV1+) trigeminal afferents attenuated periodontitis-induced bone loss in mice. (
We also determined the effects of ablation of TRPV1+ afferents on bone loss in an independent periodontitis model, by inoculation of oral bacteria (Fig. 1D–F). A week after intra-TG injections of RTX or Veh, we inoculated
Next, we determined the effects of nociceptor ablation on the number of osteoclasts using TRAP staining (Fig. 2A, B). Mice with ligature-induced periodontitis had more osteoclasts than unligatured controls. Significantly fewer osteoclasts were seen around the second molars in the RTX/ligature group than in the Veh/ligature group, while the numbers of osteoclasts in unligatured control teeth were not affected.
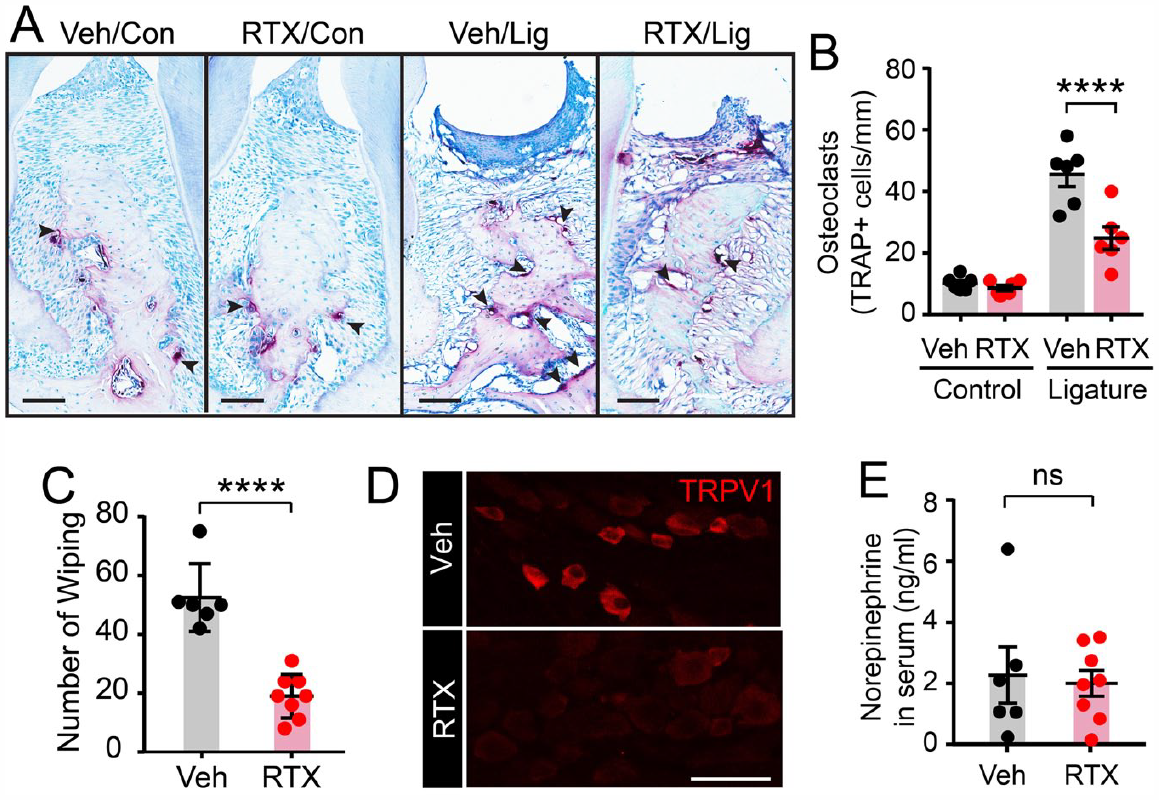
Ablation of transient receptor potential vanilloid 1+ (TRPV1+) trigeminal afferents attenuated osteoclasts in alveolar bone under periodontitis without altering circulating norepinephrine. (
We validated the effects of ablation by intra-TG injected RTX functionally and histologically. One week after intra-TG injection of RTX, eye-wiping behaviors in response to capsaicin application were significantly reduced (Fig. 2C). Postmortem histology indicated that the number of TRPV1+ neurons in TG was substantially reduced (Fig. 2D), as we have previously found (Wang et al. 2019). Three weeks after RTX injection, the levels of norepinephrine in circulating blood were the same in the RTX- and Veh-injected groups (Fig. 2E).
Chemogenetic Inhibition of TRPV1+ Afferents Reduces Bone Loss in Periodontitis
Crossing
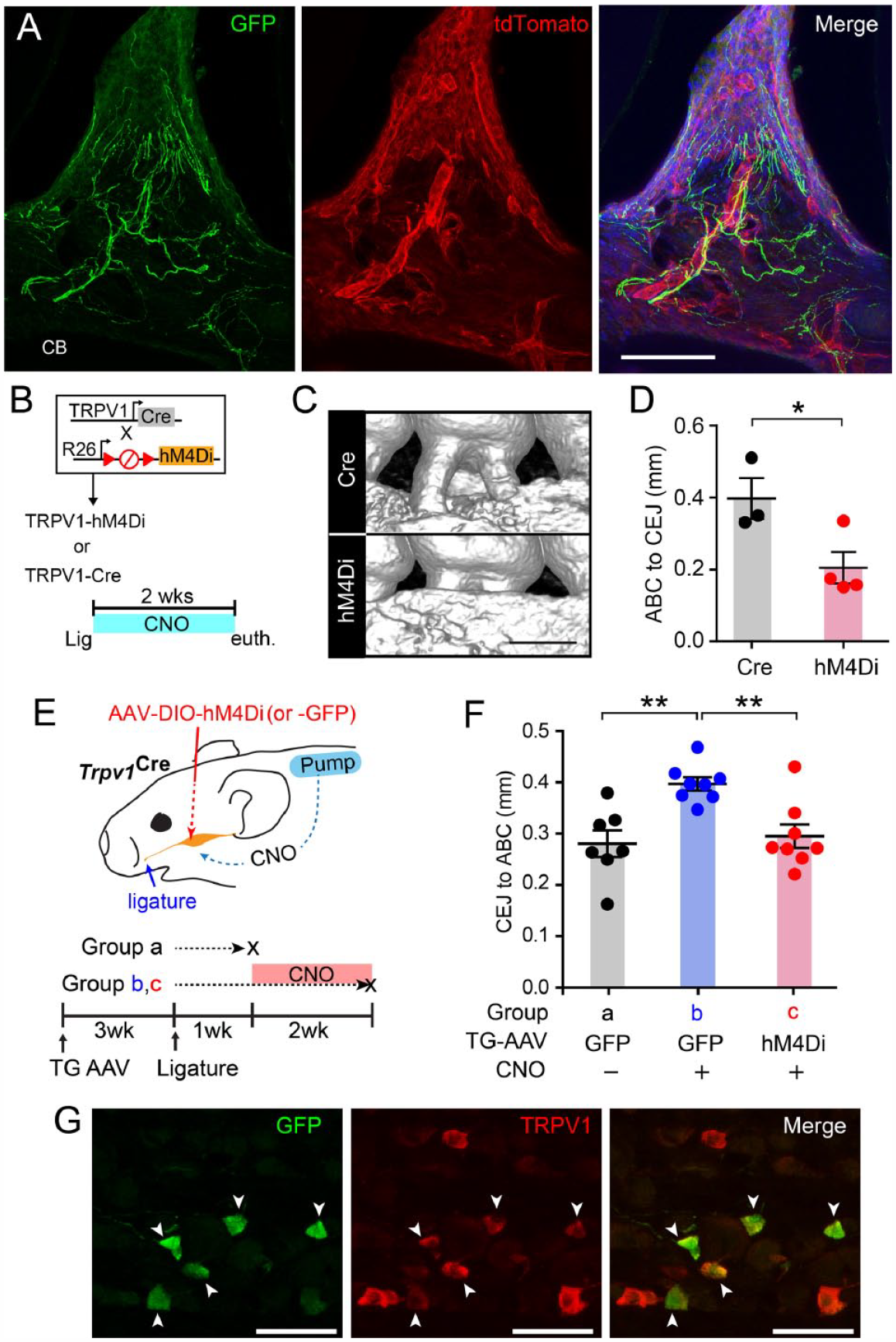
Noninvasive chemogenetic inhibition of transient receptor potential vanilloid 1+ (TRPV1+) afferents reduces ligature-induced bone loss. (
Ablation of TRPV1+ Afferents Decreases Local Host Responses in Periodontitis
Periodontal bone loss is mediated by dysregulated local immune responses in the periodontium (Garlet 2010; Graves et al. 2011). We therefore investigated whether TRPV1+ afferents contribute to the immune cell infiltration in the gingiva. Since the inhibitory effects of chemical ablation are more robust than those of chemogenetic silencing, we tested the effects of TRPV1+ afferent ablation on the profiles of gingival immune cells (Fig. 4A). One week after intra-TG injection of RTX or vehicle, the ligature was placed. Single-cell suspensions were prepared from gingival tissues around the maxillary molars, and flow cytometry was performed. In mice with Veh injection into the TG, the fraction of the CD45+ leukocytes were approximately 30% of live single cells in 2 wk after ligature placement, which is more than 5-fold greater than the unligatured control (Fig. 4A). The changes in the Ly6G+ CD11b+ population showed a similar trend, and the neutrophil percentage in the ligature group was significantly increased (Fig. 4B). In mice injected with RTX into the TG, ligature-induced increases in the number of CD45+ cells and neutrophils were significantly lower than those in vehicle-injected mice with ligature (Fig. 4A, B).
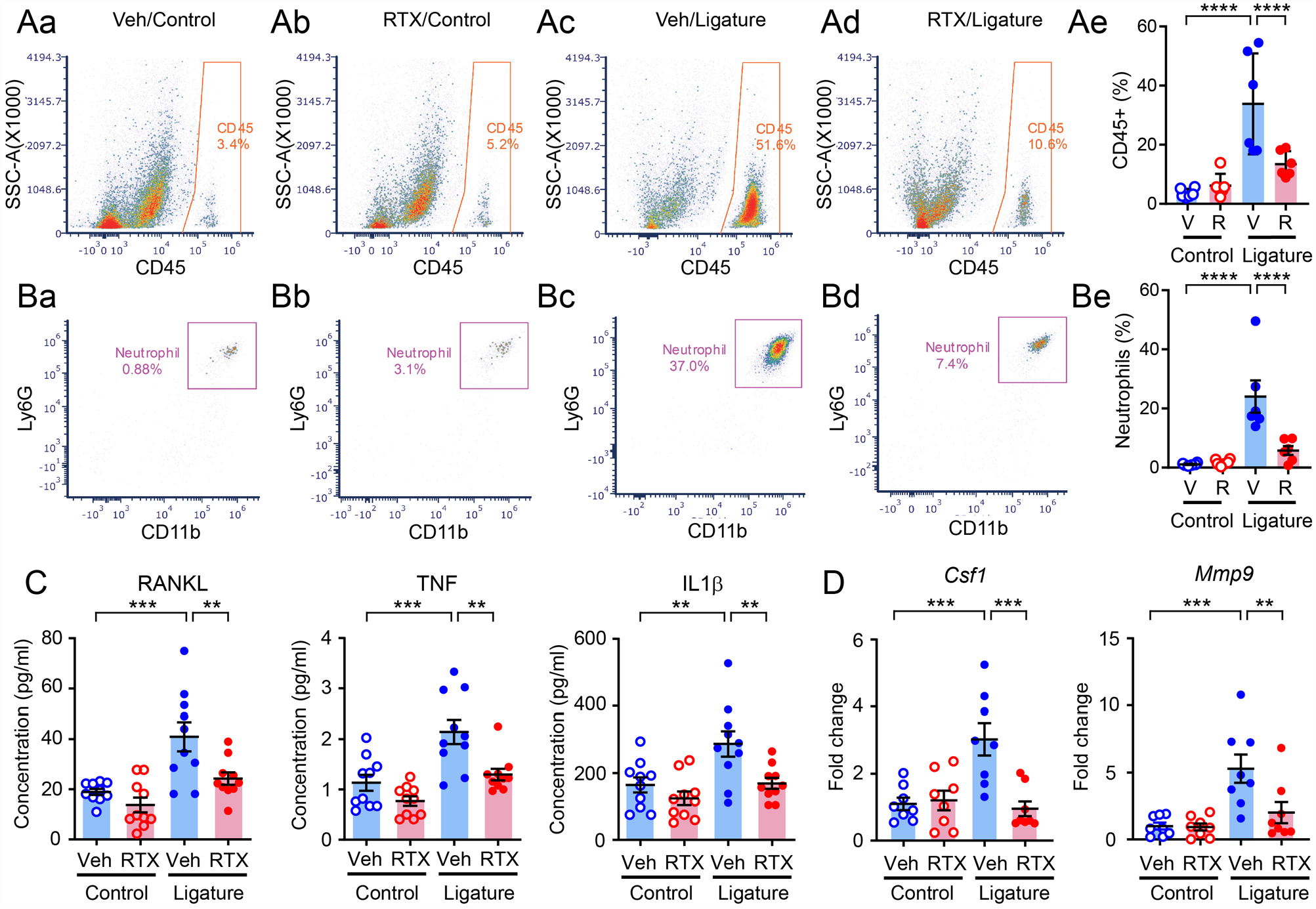
Ablation of transient receptor potential vanilloid 1+ (TRPV1+) afferents decreases host responses in the periodontium under periodontitis. (
We also analyzed the changes in proinflammatory cytokines, including tumor necrosis factor (TNF), interleukin (IL)–1β, and receptor activator of nuclear factor κΒ ligand (RANKL), which are associated with immune cells and periodontal bone destruction (Graves et al. 2011; de Vries et al. 2017). The expression levels of all of these cytokines were increased in the ligature group compared to the control, and the extent of increase was significantly lower in mice injected with RTX in the TG (Fig. 4C). We also investigated the changes in the mRNA levels of 2 additional genes implicated in periodontitis—
Ablation of TRPV1+ Afferents Does Not Alter the Microbiome in Periodontitis
We then investigated the effects of ablation of TRPV1+ afferents on the composition of periodontal microbiota, using 16S rRNA sequencing of bacterial samples recovered from the ligatures (Fig. 5A). The ligatures were recovered after 3 h (3h) from the right side (healthy control) or after 2 wk (2w) from the left side (periodontitis group) in mice in which RTX (R) or Veh (V) was injected into TG, and recovered bacteria were used for 16S rRNA sequencing. The 4 groups (i.e., V/3h, V/2w, R/3h, and R/2w) showed a variety of microbial compositions at the phylum level (Fig. 5B). The 4 groups showed no difference in α diversity (Fig. 5C), and β diversity was significantly different among groups (
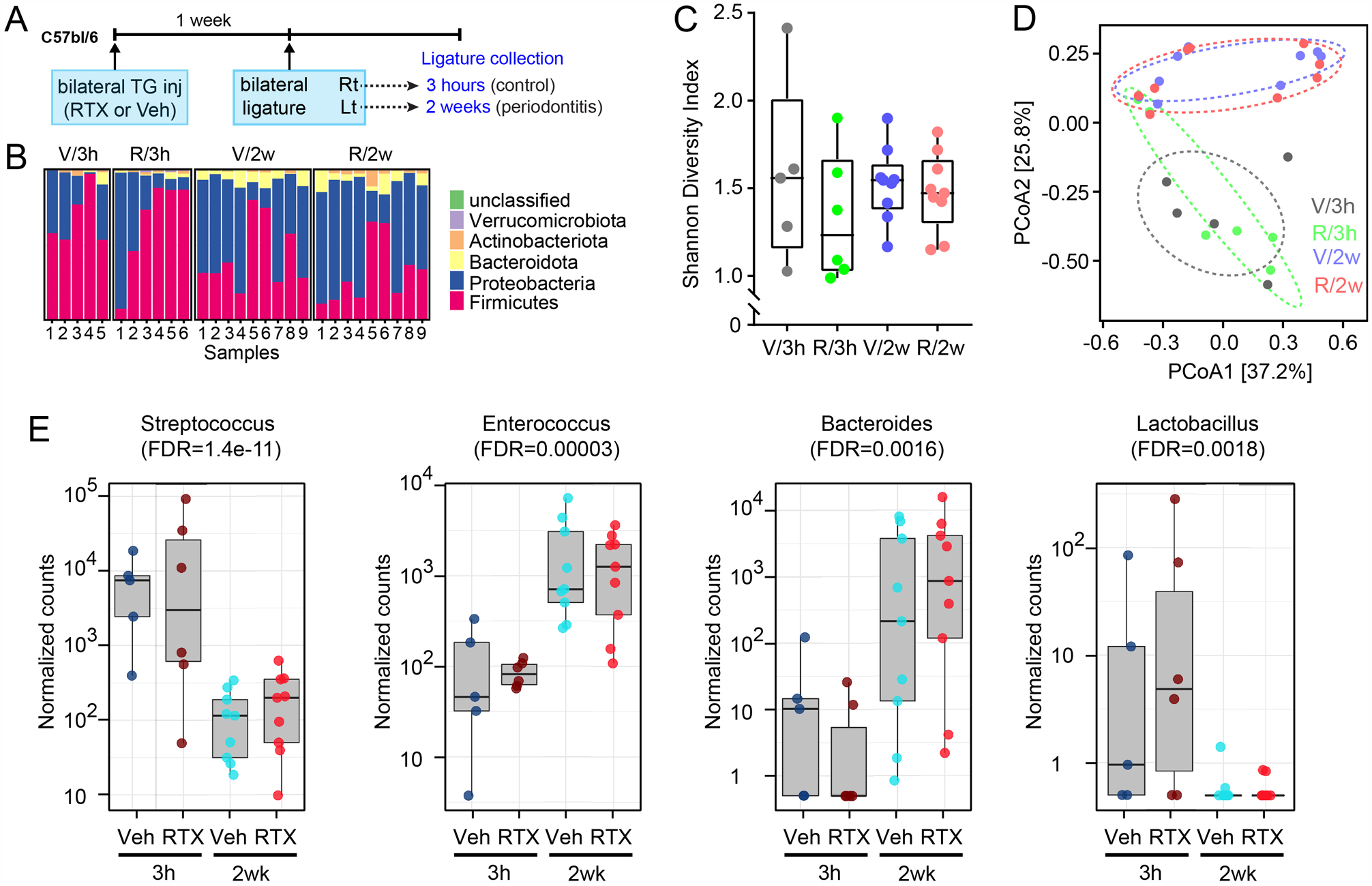
Ablation of transient receptor potential vanilloid 1+ (TRPV1+) afferents does not alter microbial dysbiosis under periodontitis. (
Discussion
In this study, we found that the ablation of TRPV1+ afferents confined to TG decreased bone destruction in 2 independent models of experimental periodontitis. The reduction was as efficacious as other pharmacological treatments, such as resolvin (Lee et al. 2016). The protective effects of nociceptor ablation on bone loss were accompanied by decreased osteoclast numbers in alveolar bone, reduced infiltration of leukocytes and neutrophils into the periodontium, and reduced levels of cytokines, such as RANKL, TNF, IL-1β, matrix metallopeptidase 9 (MMP9), and Colony stimulating factor 1 (CSF1), which are associated with innate immunity and osteoclastic differentiation. Not only chemical ablation but also chemogenetic functional silencing of nociceptive neurons displayed protective effects. These results unequivocally support that the TRPV1+ afferents signaling is critical for promoting dysregulated host responses and bone loss in periodontitis in vivo.
In contrast to our results, previous studies have found that the systemic injection of RTX or capsaicin accelerates alveolar bone destruction in periodontitis and reduces bone density (Offley et al. 2005; Takahashi et al. 2016). However, these results should be interpreted with care, because this treatment produces a deficiency of TRPV1+ neurons throughout the entire body, including the dorsal root ganglia and vagal ganglia, and probably involves strong compensatory processes in the nervous system. Many studies have shown that systemic capsaicin injection in neonates increases sympathetic activity in various organs (Luthman et al. 1989; Ralevic et al. 1995; Sann et al. 1995; Wang et al. 2001). Since sympathetic nerves are known to enhance bone resorption (Takeda et al. 2002; Khosla et al. 2018), it is difficult to attribute the bone changes following systemic treatment with vanilloids entirely to the selective manipulation of sensory neurons. For example, systemic genetic ablation of TrkA+ neurons, which mainly overlap with TRPV1+ neurons, in entire sensory neurons increases serum norepinephrine and bone resorption, which can be reversed by propranolol, suggesting the involvement of enhanced sympathetic activity (Chen et al. 2019). In our study, we used localized ablation of trigeminal afferents, limited to the ipsilateral ophthalmic/maxillary region of the TG (Wang et al. 2019), to minimize undesired exposure and compensatory effects. This manipulation did not produce changes in the level of circulating norepinephrine, which excludes the possibility that altered sympathetic activity mediated the neural regulation of bone loss.
Bone destruction by periodontal pathogens is mediated by innate and adaptive immune responses, and hyperactive host responses contribute to tissue destruction (Garlet 2010; Graves et al. 2011). Bacterial components trigger innate immune responses followed by a chain of host reactions, and neutrophils play important roles in the induction of bone loss through multiple pathways, including regulating other leukocytes, and secreting cytokines and tissue-destructing enzymes (Lee et al. 1995; Kantarci et al. 2003; Garlet 2010; Graves et al. 2011; Hajishengallis et al. 2016). Ablation of TRPV1+ afferents profoundly decreased the infiltration of leukocytes and neutrophils along with proinflammatory cytokines, which is consistent with the decreased bone loss in ligature-induced periodontitis. Therefore, TRPV1+ nerves contribute to exacerbating innate immunity in the periodontium under periodontitis. Ablation of TRPV1+ nerves apparently does not interfere with physiological bone regulation and local immunity without periodontitis. Furthermore, ablation of TRPV1+ nerves did not alter the microbiome in gingival sulcus in healthy conditions or in periodontitis. These results suggest that the inhibitory effects of nerve ablation on ligature-induced bone loss do not appear to be caused by the alteration, or normalization of the dysbiosis of the microbiome, but are more likely to be due to the decrease in dysregulated host responses. Therefore, selective interference of nociceptive nerve-induced aggravation of innate immunity can be a novel therapy to alleviate detrimental hyperactive host responses without interfering with homeostasis.
In this study, we focused on determining the effects of manipulating nociceptive nerves on bone loss, host responses, and microbial changes under periodontitis. Determining the mechanisms whereby TRPV1+ nerves aggravate innate immunity and bone loss is beyond the scope of this study, and we will further determine the mechanisms of neuroimmune and neuroskeletal regulation in periodontitis. Neural controls of other innate immune cells, such as monocytes and macrophages, or adaptive immunity need to be determined. Investigation of the upstream and downstream signaling associated with neuroimmune regulations is also critical. As previously suggested, neuropeptides released from TRPV1+ nerves regulate the infiltration and function of immune cells (Talbot et al. 2015; Baral et al. 2019), and the major neuropeptides contributing to bone loss in periodontitis need to be clarified. It is also important to understand the mechanisms underlying the “painless” contribution of nociceptive nerves to periodontitis. In our model, we presume that chronic periodontitis is accompanied by the persistent activity of nociceptive afferents. However, patients do not experience overt pain from the periodontium. Some unique pathobiology of the periodontium and periodontitis may contribute. Unlike lipopolysaccharides (LPS) from other sources, LPS from
In conclusion, we suggest that TRPV1+ afferents initiate neuroimmune signaling modulating bone loss in marginal periodontitis. Therefore, manipulating this neuroimmune axis (e.g., by the localized inhibition of TRPV1+ afferents or by the modulation of downstream signaling leading to neurogenic inflammation in affected gums) could provide novel therapeutic approaches for treating periodontitis, supplementing conventional therapies.
Author Contributions
S. Wang, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; X. Nie, X. Fan, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; Y. Siddiqui, contributed to conception and data interpretation, critically revised the manuscript; X. Wang, contributed to conception, design, data acquisition, analysis, and interpretation, critically revised the manuscript; V. Arora, contributed to design, data acquisition, and analysis, critically revised the manuscript; V. Thumbigere-Math, contributed to conception, design, data interpretation, critically revised the manuscript; M.K. Chung, contributed to conception, design, and drafted manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345211069956 – Supplemental material for Nociceptor Neurons Magnify Host Responses to Aggravate Periodontitis
Supplemental material, sj-docx-1-jdr-10.1177_00220345211069956 for Nociceptor Neurons Magnify Host Responses to Aggravate Periodontitis by S. Wang, X. Nie, Y. Siddiqui, X. Wang, V. Arora, X. Fan, V. Thumbigere-Math and M.K. Chung in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank Ms. Sinu Kumari for her technical assistance and Dr. Dana Graves at the University of Pennsylvania for helpful discussion.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health R01 DE027731 and R35 DE030045 to M.K. Chung, R00 DE028439, R03 DE029258, startup funds from the University of Maryland School of Dentistry to V. Thumbigere-Math, and P30 CA134274 and Maryland Department of Health’s Cigarette Restitution Fund Program to X. Fan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
