Abstract
Propranolol is a nonselective β-adrenergic receptor antagonist that is efficacious in reducing facial pain. There is evidence that its analgesic efficacy might be modified by variants of the catechol-O-methyltransferase (
Keywords
Introduction
Symptoms of painful temporomandibular disorder (TMD) are reported by 5% of U.S. adults, one-fifth of whom use prescription medication to manage the pain (Slade and Durham 2020). However, analgesics for TMD lack efficacy (Mujakperuo et al. 2010), and “action is urgently needed to improve care for individuals with a TMD” (National Academies of Sciences 2020). Yet, formidable barriers restrict the development of new analgesics, including the many years required to discover new analgesics and the high probability that they fail to show efficacy in phase III trials (Chaplan et al. 2010).
Effective analgesics might instead be identified using strategies from precision medicine (Niculescu et al. 2019). One strategy is to repurpose drugs developed for other conditions, using them to target biological pathways that modulate pain. Another strategy is to tailor drug therapy based on patients’ genotypes (Wilentz and Cowley 2017). Both strategies are applicable to propranolol, a nonselective β-adrenergic receptor antagonist, which was developed in the 1960s and is effective for migraine prevention (He et al. 2017). Recent findings from preclinical studies have shown that propranolol inhibits pain processes that amplify trigeminal nociception (Khasar et al. 1999; Shields and Goadsby 2005; Rodrigues et al. 2006; Ciszek et al. 2016; Boyer et al. 2017). Other studies suggest that propranolol’s analgesic efficacy is modified by genetic variation in the catechol-O-methyltransferase (
These studies underpinned our premise that propranolol’s analgesic efficacy is enhanced in people whose
This study aimed to determine if
Methods
The study was approved by Institutional Review Boards at the 3 enrollment sites: University of North Carolina at Chapel Hill, University of Florida, and University at Buffalo. All study participants provided signed informed consent. Full details of the study methods are reported elsewhere (Tchivileva et al. 2020) and summarized below using headings from the CONSORT statement (Moher et al. 2010).
The trial design was a double-blind, placebo-controlled, parallel-group, phase 2b RCT. Study participants were 200 adults aged 18 to 65 y with TMD myalgia (with or without arthralgia), as determined using the diagnostic criteria for TMD (Schiffman et al. 2014). There were 16 exclusion criteria based on propranolol’s contraindications and health conditions that could have affected pain ratings. Study participants were randomized using 1:1 allocation stratified by study site using fixed permutated blocks of 4. Randomization was initiated by study coordinators via an electronic web response system, thereby masking study personnel and all study participants. Study participants were allocated to one of the investigational products: capsules of 60-mg extended release propranolol or a matching placebo, both administered once daily for 1 wk and then twice daily for 8 wk.
As specified a priori in the study protocol, the primary outcome was a facial pain index, calculated as the product of daily facial pain intensity (reported on a 0 to 100 numeric rating scale) and pain duration (% of day with facial pain) divided by 100. Each was recorded in a daily pain diary, and their product (i.e., daily facial pain index) was averaged across the 7 d prior to study visits conducted 1, 5, and 9 wk after the randomization visit. The secondary outcome was the patient’s global impression of change (PGIC; Hurst and Bolton 2004) evaluated at weeks 5 and 9.
Genotyping
DNA was extracted from venous blood samples collected at the baseline visit. Genotyping was performed by LGC Genomics (Beverly, MA, US) using proprietary KASP™ assays based on competitive allele-specific polymerase chain reaction.
Statistical Methods
To reduce population stratification bias (Cardon and Palmer 2003), this analysis was restricted to non-Hispanic Whites. Regression models tested for an interaction of genotype and allocated treatment group to evaluate the hypothesis that the A allele of rs4680 is associated with greater analgesic efficacy of propranolol. For the primary endpoint of facial pain index, efficacy was quantified as change in the index, computed by subtracting the baseline value from the value at each follow-up visit (i.e., weeks 1, 5, and 9). The continuous measure of change was analyzed as the dependent variable using a linear mixed model for repeated measures (MMRM), while logistic MMRMs evaluated reductions of ≥30% and ≥50% relative to baseline, both of which are clinically meaningful thresholds of improvement in pain (Dworkin et al. 2009).
As specified a priori in the study protocol, the number of A alleles was modeled as a continuous fixed-effect variable (i.e., assuming an additive genetic model). Other fixed-effect variables were treatment group (2 groups), study visit (3 postbaseline categories), and all 2-way and 3-way interactions of genotype, treatment group, and visit. Fixed-effect covariates were baseline pain index (modeled as a continuous variable), study site (3 categories), and sex (2 categories). Study participants were included as a random effect. MMRMs used data from all available follow-up visits and programming statements (Davis 2014) tested for a 2-way interaction of treatment group with genotype at week 9. Other programming statements calculated odds ratios or mean differences between treatment groups at week 9 for each genotype. Model-adjusted mean values or proportions were calculated and plotted for descriptive purposes. A comparable logistic MMRM was used to analyze global impression of change, dichotomized from the 7-point response scale to classify patients who reported feeling “somewhat better” through “a great deal better” (Hurst and Bolton 2004).
To determine the efficacy across the full range of clinically meaningful reduction in pain, logistic MMRMs for reductions ranging from ≥20% to ≥70% were used to construct cumulative response curves (Farrar 2010). The percentage area under curve (%AUC) was calculated for each treatment group, with the %AUC difference representing the efficacy estimate. AUC values were calculated using the trapezoid rule, and bootstrap estimates of the standard error were obtained from 1,000 samples of the observed data. Genotype-group differences in %AUC were evaluated using Wald’s test.
The threshold for statistical significance of effect modification was
Sample Size Justification
SOPPRANO was planned with a target sample of 200 randomized study participants, providing 90% statistical power for the primary aim of overall analgesic efficacy of propranolol. As initially envisaged, the sample was to be limited to non-Hispanic Whites, providing 61% power to detect an interaction between
Results
Baseline Characteristics of Study Groups
Between August 2015 and January 2018, 200 study participants were randomized to either propranolol (
Baseline Characteristics of Participants in the 2 Treatment Groups.
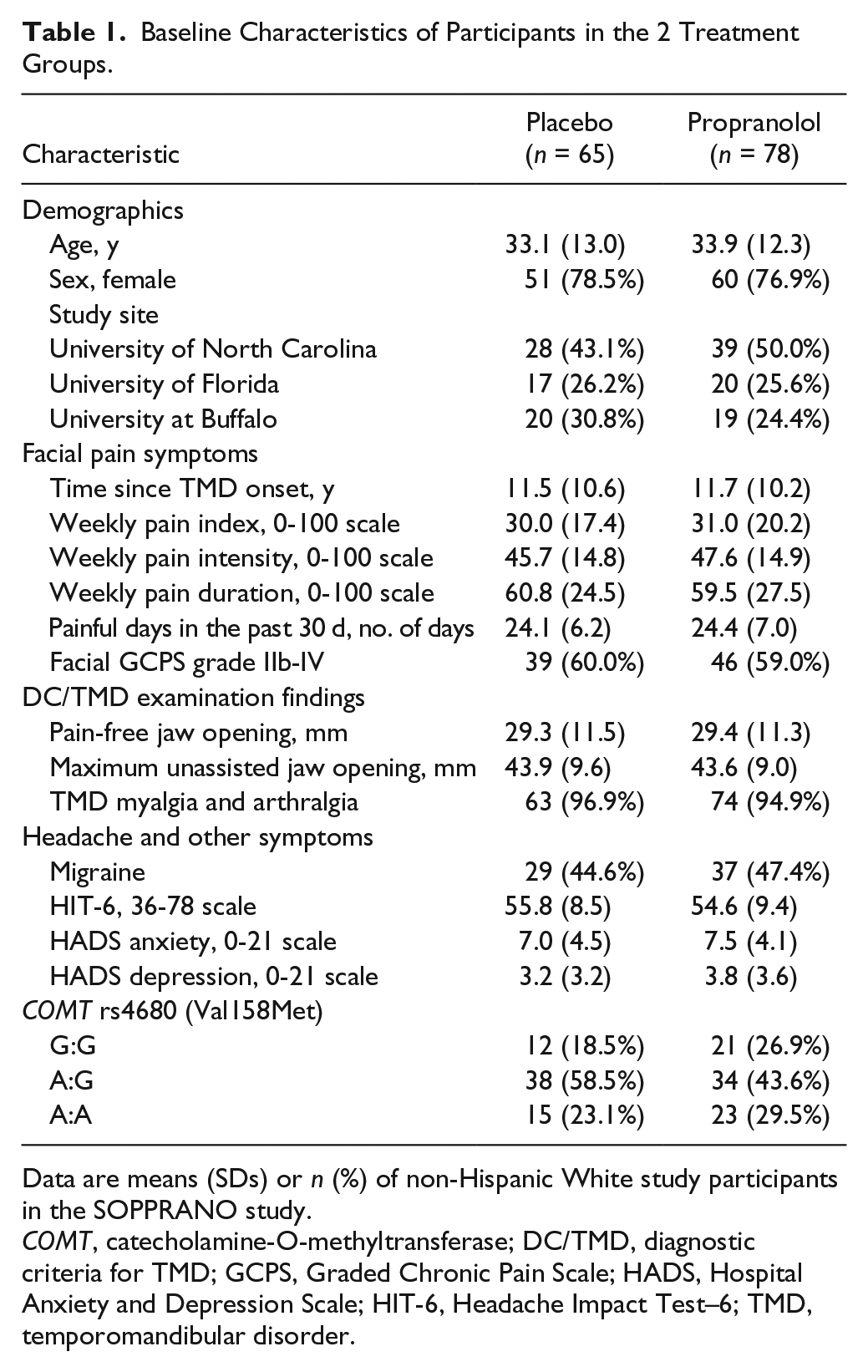
Data are means (SDs) or
Baseline Characteristics of Participants Classified by
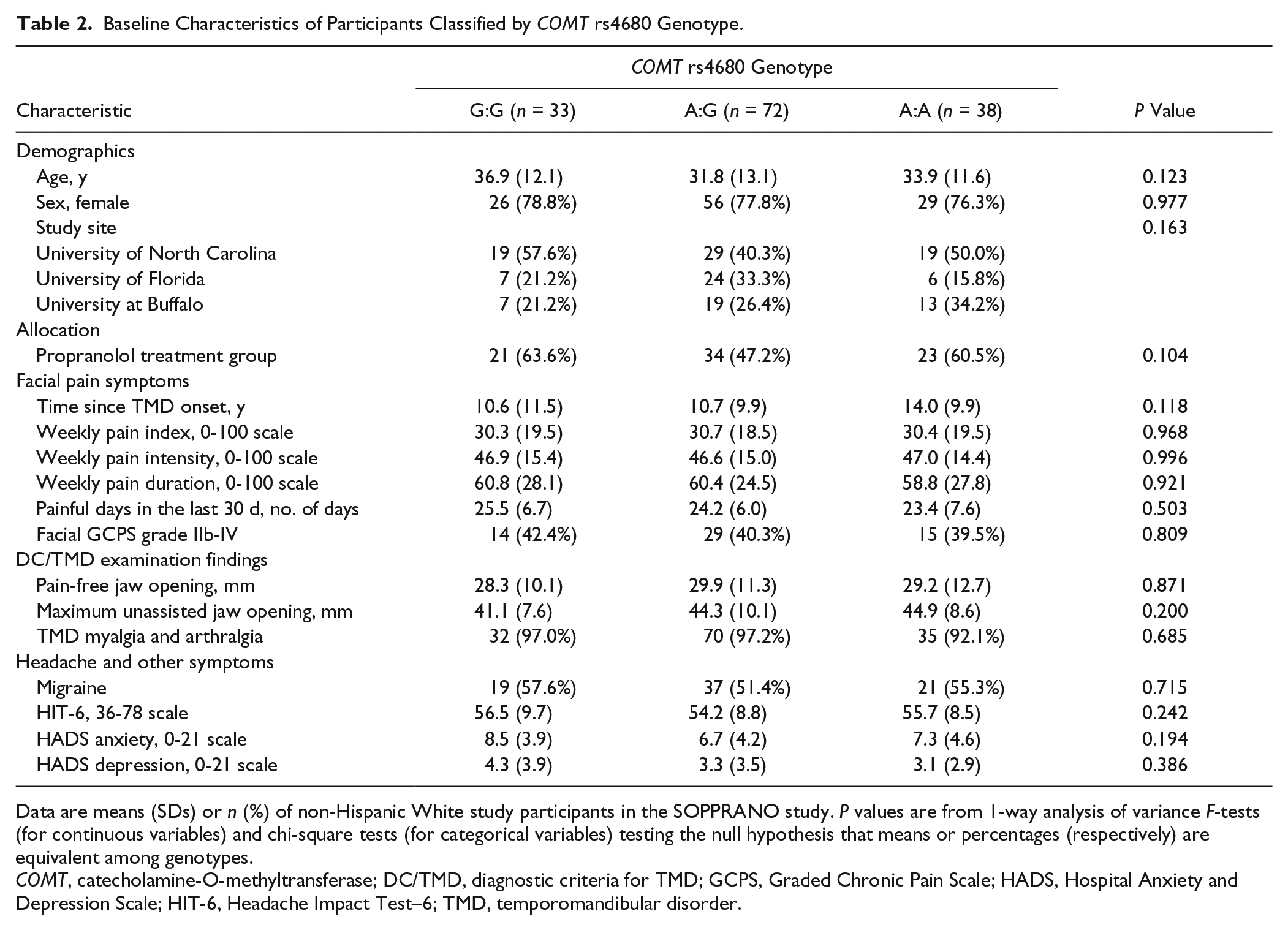
Data are means (SDs) or
Effect Modification of the Primary Endpoint by the COMT Genotype
Results for the linear MMRM analysis of the continuous measures of pain index revealed greater efficacy for G:G homozygotes than for A:A homozygotes, although the interaction was not statistically significant (Table 3; Suppl. Fig. S1). Among the
Summary of Efficacy Estimates and Tests for Gene × Treatment Group Interactions.
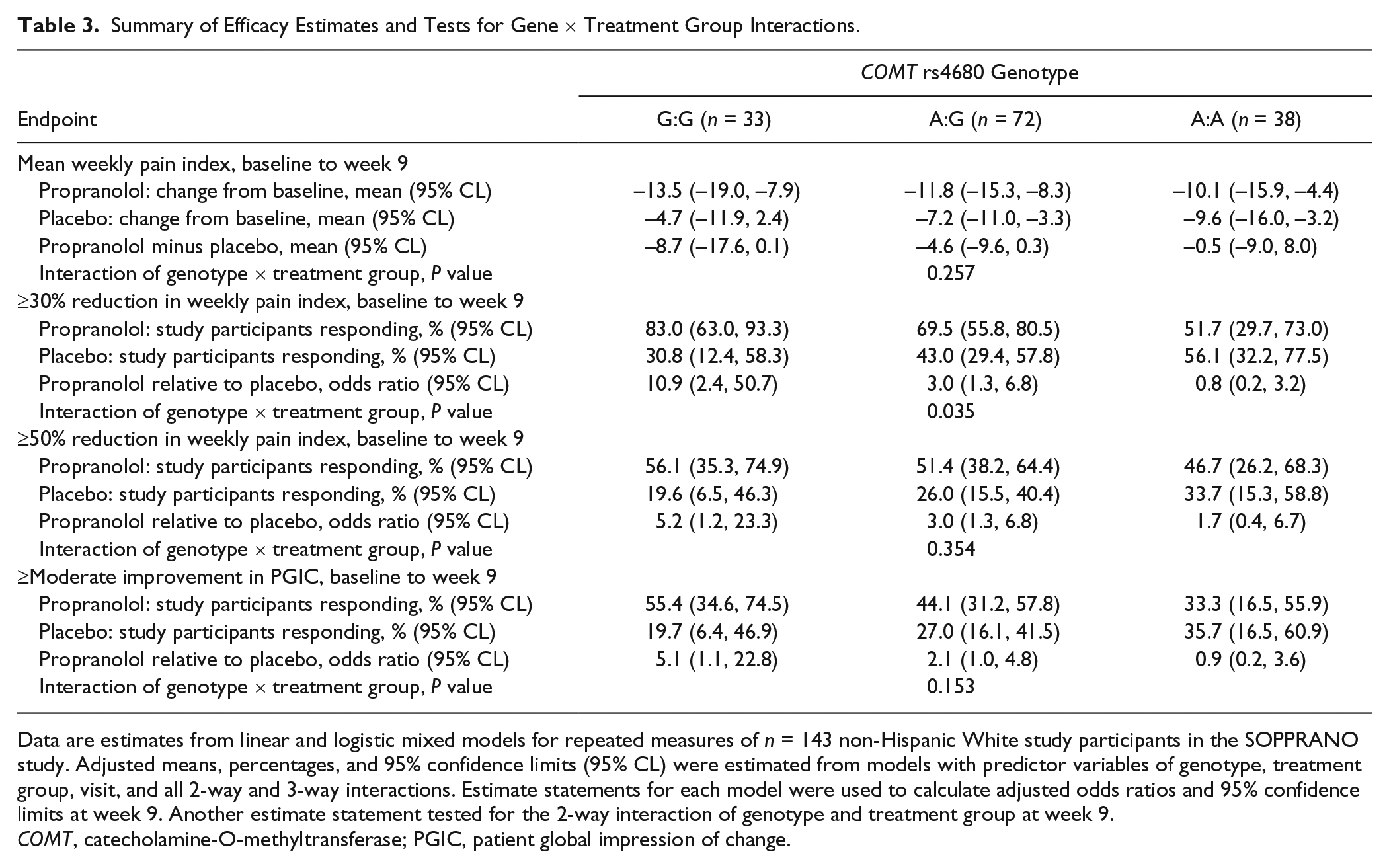
Data are estimates from linear and logistic mixed models for repeated measures of
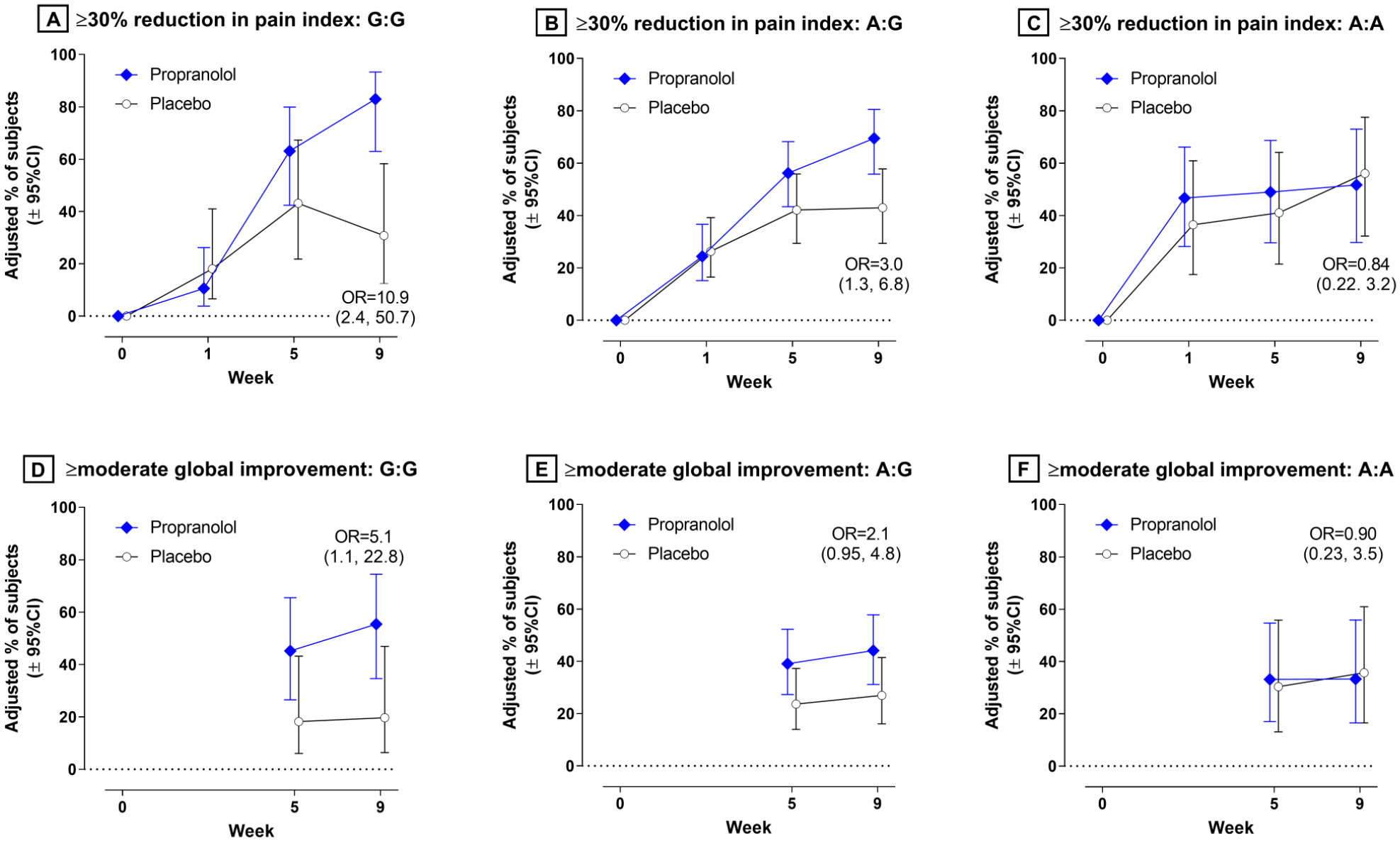
Change in 2 endpoints at 3 follow-up visits for study participants in 2 treatment groups stratified according to genotype of the rs4680 single nucleotide polymorphism of
When results were integrated for thresholds of 20% to 70% reduction in pain index, the propranolol-versus-placebo difference in cumulative response curves was greater for G:G homozygotes (43.7 percentage point difference in AUC, 95% CL = 15.4, 72.1) than for A:A homozygotes (6.5 percentage point difference in AUC, 95% CL = −30.2, 43.2). This genotype difference in treatment effect estimates was statistically significant (
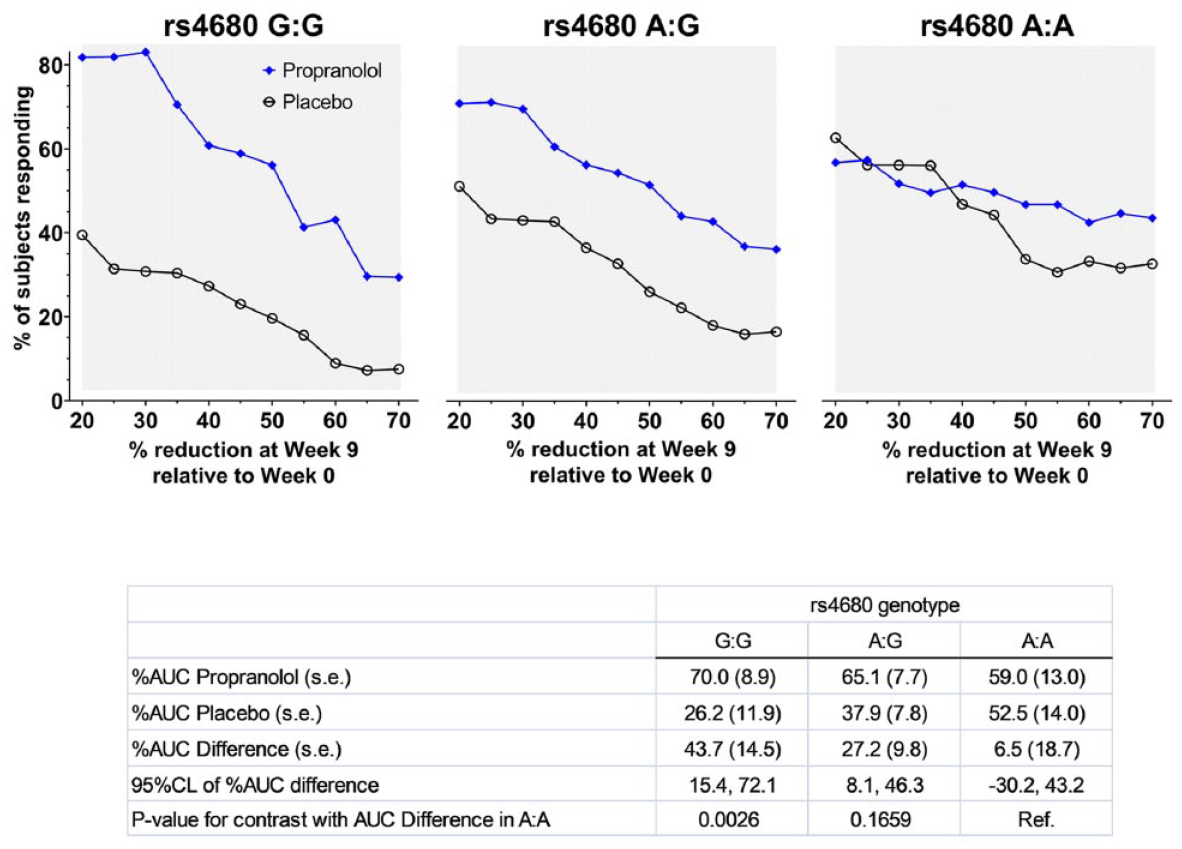
Cumulative response curves for facial pain index among study participants in 2 treatment groups stratified according to genotype of the rs4680 single nucleotide polymorphism of
Effect Modification Analysis Using the PGIC Secondary Endpoint
Model-adjusted percentages of study participants reporting at least moderate improvement in their overall condition ranged from 19.7% (placebo) to 55.4% (propranolol) in G:G homozygotes (Fig. 1D), with a corresponding odds ratio of 5.1 (95% CL = 1.1, 22.8; Table 3). Odds ratios were 2.1 (95% CL = 1.0, 4.8) for A:G heterozygotes (Fig 1E:) and 0.9 (95% CL = 0.2, 3.6) for A:A homozygotes (Fig. 1F), although the interaction of genotype and treatment group at week 9 was not statistically significant (
Qualitative Evaluation of Credibility of the Overall Findings
The analysis fulfilled all 5 study design criteria used to evaluate the credibility of effect modification in RCTs (Sun et al. 2010), namely, 1)
Sensitivity Analysis
When the
Discussion
This RCT of study participants with painful TMD found some evidence that propranolol’s analgesic efficacy was modified by
Before considering the implications of this unexpected finding, the study’s limitations warrant consideration. For this secondary genetic objective, the statistical analysis plan specified that the primary endpoint be analyzed both as a continuous measure and after dichotomizing it at thresholds of ≥30% and ≥50% pain reduction. The 3 endpoints were consistent in showing antagonistic directions of effect for the A allele, although the gene × treatment group interaction was statistically significant only for the ≥30% threshold and in secondary analysis of cumulative response curves. This is counter to the conventional expectation that continuous variables yield greater statistical power (Snapinn and Jiang 2007). However, as noted for the primary paper from this study (Tchivileva et al. 2020), many study participants in the placebo group experienced reductions of <30% in the facial pain index, thereby reducing the efficacy signal from the continuous endpoint. Instead, there was greater power to detect a ≥30% reduction in pain, a measure that is favored as clinically meaningful for patients (Dworkin et al. 2009). In addition, calculated statistical power for effect modification was only 47% using the continuous endpoint. Nonetheless, the binary endpoint revealed statistically significant gene × treatment group interactions that were consistent with clinically meaningful differences in efficacy, although 95% confidence intervals were wide, making the estimates imprecise. However, we caution against overinterpreting the interaction
The current study has several strengths, including its randomized, placebo-controlled design with allocation concealment and masking. Consistent with the best practices for clinical trials of pain (Edwards et al. 2016), the primary endpoint was measured using daily pain diaries. The 9-wk follow-up period exceeded the minimum of 4 wk recommended for phase 2b trials (Gewandter et al. 2014). Pain-related characteristics measured at baseline did not differ appreciably between the treatment groups or COMT genotypes, making it unlikely that the gene × treatment group interaction was confounded by those characteristics. The sensitivity analysis revealed very similar results for different genetic models, and we therefore favor the additive model because it parsimoniously depicts the biological gradient.
The opposing directions of effect modification in the current and pilot study require scrutiny. Although both studies used the same endpoint, there were notable differences: the pilot study used a crossover design, enrollment was restricted to 40 women, the dose of propranolol was lower, and efficacy was analyzed after 1 wk. In general, parallel-group designs are favored for clinical trials because they require fewer assumptions when testing efficacy (Dubey 1986). The pilot study (Tchivileva et al. 2010) used a haplotype of
A more salient difference between studies was the placebo group’s response: in the pilot study, there was effectively no reduction in facial pain in the placebo phase, whereas in the current study, there were substantial reductions in facial pain in the placebo group. In this study, the absence of efficacy for the A:A homozygotes was due as much to a heightened response (relative to G:G homozygotes) in the placebo group as it was to a diminished response in the propranolol group. Sizable responses in placebo groups are the norm in clinical trials of pain, suggesting that the antagonistic effect of the A allele seen here is more credible than the amplifying effect suggested by the pilot study. This effect of
Seemingly inconsistent effects of
Compared with the pilot study, these results provide stronger evidence that
Author Contributions
G.D. Slade, contributed to conception, design, data analysis, and interpretation, drafted and critically revised the manuscript; R.B. Fillingim, R. Ohrbach, contributed to conception, design, data acquisition, and interpretation, critically revised the manuscript; H. Hadgraft, contributed to data acquisition, critically revised the manuscript; J. Willis, contributed to data analysis, critically revised the manuscript; S.J. Arbes Jr., contributed to conception, design, and data interpretation, critically revised the manuscript; I.E. Tchivileva, contributed to conception, design, data acquisition, and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
DS_10.1177_0022034520962733 – Supplemental material for COMT Genotype and Efficacy of Propranolol for TMD Pain: A Randomized Trial
Supplemental material, DS_10.1177_0022034520962733 for COMT Genotype and Efficacy of Propranolol for TMD Pain: A Randomized Trial by G.D. Slade, R.B. Fillingim, R. Ohrbach, H. Hadgraft, J. Willis, S.J. Arbes and I.E. Tchivileva in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank the SOPPRANO research staff and patients who participated in the trial. The authors also thank cardiologists Drs. Alan Hinderliter (UNC at Chapel Hill), David Sheps (University of Florida), and Thomas Cimato (University at Buffalo) for monitoring participants’ safety.
A supplemental appendix to this article is available online.
The trial is registered at ![]() , NCT02437383. Full details of the trial protocol are available at ClinicalTrials.gov.
, NCT02437383. Full details of the trial protocol are available at ClinicalTrials.gov.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the National Institutes of Health (NIH)/National Institute of Dental and Craniofacial Research (NIDCR) R34-DE022088 and U01-DE 024169 grants.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
