Abstract
Fissure sealant (FS) and fluoride varnish (FV) are effective in preventing dental caries when compared with a no-treatment control. However, the relative clinical effectiveness of these interventions is uncertain. The objective of the study was to compare the clinical effectiveness of FS and FV in preventing dental caries in first permanent molars (FPMs) in 6- to 7-y-olds. The study design was a randomized clinical trial, with 2 parallel arms. The setting was a targeted-population program that used mobile dental clinics in schools located within areas of high social and economic deprivation in South Wales. A total of 1,016 children were randomized 1:1 to receive either FS or FV. Resin-based FS was applied to caries-free FPMs and maintained at 6-mo intervals. FV was applied at baseline and at 6-mo intervals for 3 y. The main outcome measures were the proportion of children developing caries into dentine (D4-6MFT) on any 1 of up to 4 treated FPMs after 36 mo. At 36 mo, 835 (82%) children remained: 417 in the FS arm and 418 in the FV arm. A smaller proportion of children who received FV (n = 73, 17.5%) versus FS (n = 82, 19.6%) developed caries into dentine on at least 1 FPM (odds ratio [OR] = 0.84; 95% CI, 0.59 to 1.21; P = 0.35), a nonstatistically significant difference between FS and FV treatments. The results were similar when the number of newly decayed teeth (OR = 0.86; 95% CI, 0.60 to 1.22) and tooth surfaces (OR = 0.85; 95% CI, 0.59 to 1.21) were examined. In a community oral health program, semiannual application of FV resulted in caries prevention that was not significantly different from that obtained by applying and maintaining FS after 36 mo (EudraCT: 2010-023476-23; ISRCTN: ISRCTN17029222).
Introduction
Children vary in their susceptibility to dental caries, with disease prevalence being closely linked to social and economic disadvantage (Locker 2000; Watt et al. 2016). Furthermore, teeth differ in their susceptibility to dental caries. The occlusal surface of first permanent molars (FPMs) is particularly prone to dental caries, often within a short period after eruption into the mouth. Decay on this surface accounts for the majority of affected tooth surfaces in adolescents and adults (Chestnutt et al. 1996; Carvalho et al. 2001; Marthaler 2004; Hopcraft and Morgan 2006). Management of occlusal caries has proven to be a great challenge to the dental profession (Carvalho 2014), and preventing dental caries on the occlusal surfaces of FPMs in high-risk children is a key objective in preventive dental care.
Two preventive dental technologies have the potential to be targeted at the occlusal surfaces of FPMs: pit and fissure sealant (FS) and fluoride varnish (FV). A 2013 Cochrane systematic review of sealants for preventing dental decay in permanent teeth concluded that in 12 trials where resin-based sealants were compared with no sealant controls, the sealed teeth were significantly less likely to be carious at 2-y follow-up (odds ratio = 0.12; 95% CI, 0.07 to 0.19; Ahovuo-Saloranta et al. 2013). The clinical effectiveness of FV was also the subject of a Cochrane review (Marinho et al. 2013). This identified 13 studies that compared FV with a placebo or no treatment, concluding that the pooled D(M)FS prevented fraction was 43% (95% CI, 30 to 57%; P < 0.0001).
Thus, while it is generally accepted that FS and FV are effective in the prevention of dental caries, which technology is clinically superior is unknown. Ahovuo-Saloranta et al. (2016) published a Cochrane systematic review on the relative effectiveness of FS versus FV. This updated a version of the review published in 2010 (Hiiri et al. 2010). The more recent review identified 4 trials that compared resin-based FS with FV. Two of these studies, involving 358 children, suggested that FS prevented more caries in FPMs at 2-y follow-up. The pooled odds ratio was 0.65 (95% CI, 0.50 to 0.94; P = 0.02). The authors stated that the body of evidence was assessed as being low quality. Ahovuo-Saloranta et al. concluded,
Scarce and clinically diverse data are available on the comparison of sealants and fluoride varnish application, therefore it is not possible to draw clear conclusions about possible differences in effectiveness for preventing or controlling dental caries on occlusal surfaces of permanent molars. The conclusion of this updated review remains the same as the last update in 2010.
The objective of the trial reported here was to compare the clinical effectiveness of FS and FV in preventing dental caries in FPMs among 6- to 7-y-olds. The cost-effectiveness of the interventions and the acceptability of these treatments were also examined and will be reported elsewhere.
Methods
The full trial protocol was published at the commencement of the trial (Chestnutt et al. 2012) and is reported here in summary, in line with the CONSORT guidelines (Schulz et al. 2010).
Trial Design and Setting
The trial design is illustrated in Figure 1. The 2-arm, parallel-group study was a phase IV randomized trial. Participants were randomized to receive resin FS or FV. Clinical examinations and treatments were undertaken in schools via mobile dental clinics (MDCs) as part of Designed to Smile, a national oral health improvement program in Wales (Welsh Government 2016). Participants were recruited in 2 cohorts between October and January in the 2011–2012 and 2012–2013 school years. The fluoride level in the local water supply was <0.1 ppm.
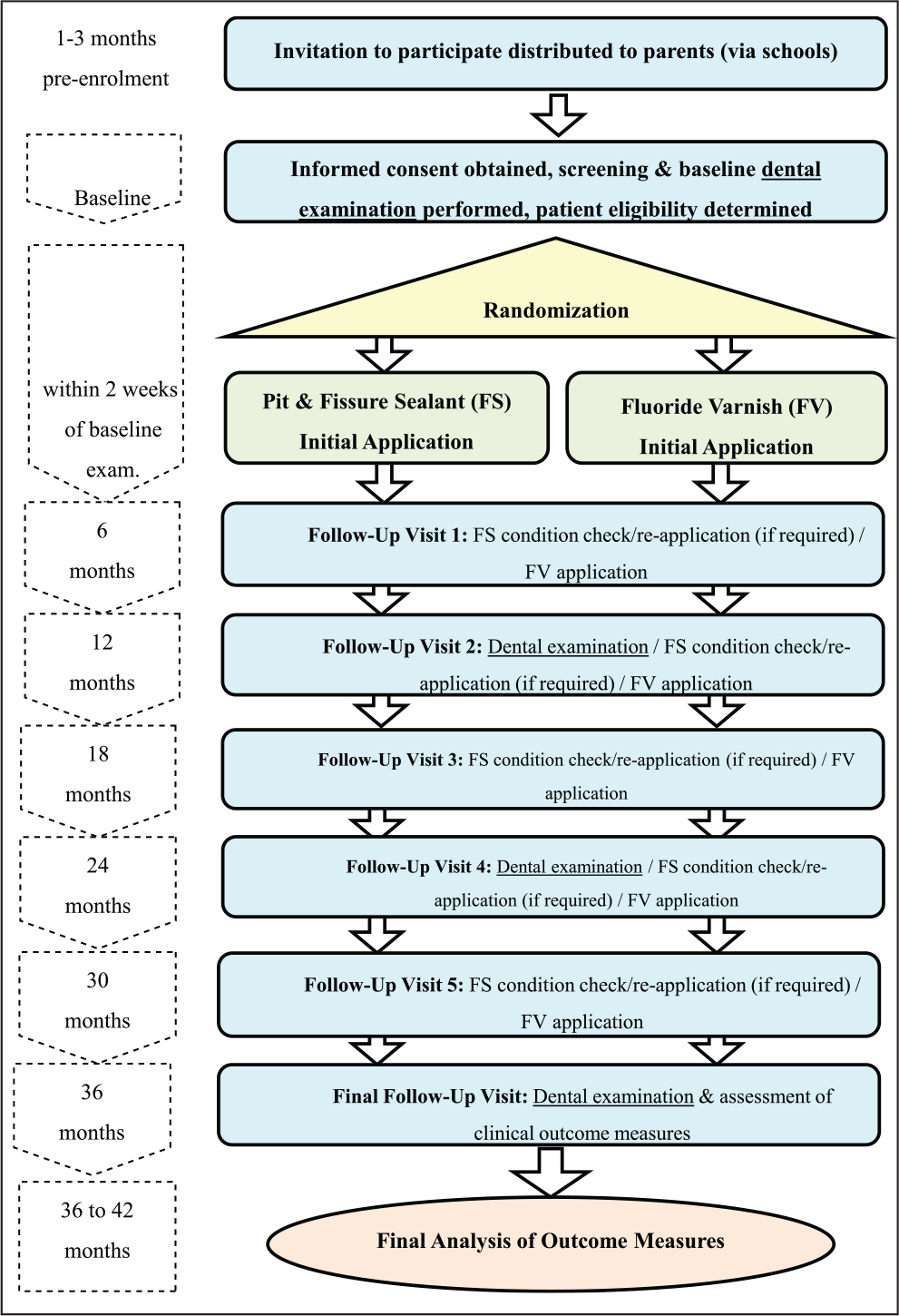
Trial design.
Participants
The target population comprised children aged 6 to 7 y attending 66 primary schools in Communities First areas—these localities have been identified as areas of social and economic deprivation by the Welsh Government. All children in such schools are deemed at high caries risk and qualify for FS/FV application (Public Health England 2014; Scottish Intercollegiate Guidelines Network 2014). To be included in the study, written consent from the child’s parent or guardian was required, and at least 1 fully erupted FPM needed to be present at the baseline examination. Children who had a known sensitivity to colophony (an ingredient in the FV), who had a history of severe allergies or had been hospitalized due to asthma, or who were participating in another Clinical Trial of an Investigational Medicinal Product were excluded from participation.
Interventions
Clinical Examinations
Study participants were examined supine in the MDC, under a standard overhead dental clinical light, with a plane dental mirror and ball-ended probe. The probe was used only to remove debris and to determine surface texture. It was not used to probe for cavitation. Teeth were not dried prior to clinical dental examination. Gross debris was removed with a toothbrush.
Caries status was assessed at baseline and 12, 24, and 36 mo by trained and calibrated dentists at the d1/D1 to d6/D6 level per ICDAS criteria (International Caries Detection and Assessment System; https://www.icdas.org/). In this report, caries presence is reported at the ICDAS codes (4 to 6) into dentine level. The clinical dental examinations were undertaken by experienced community dental officers. A total of 6 were used across the study, with 1 examiner involved in all years of the project. A training and calibration exercise was undertaken in advance of each round of clinical examinations. As part of the annual caries assessment, approximately 5% of study participants were reexamined to determine intraexaminer reproducibility.
Technologies Evaluated
The FS used was the Delton Light Curing Opaque Pit & Fissure Sealant (CE0086; Dentsply Ltd). The standard clinical protocol, as described by the product manufacturer, was used to apply FS to the occlusal surfaces of the included FPMs. Initial application of FS occurred within 2 wk of the baseline dental examination, performed by a qualified and trained dental hygienist. In the case of partially erupted molars, where sufficient tooth surface was available, sealant was applied. This situation arose particularly in the case of upper molars. The same 2 dental hygienists provided treatments throughout the trial using 2 MDCs. The condition of the FS was reexamined at 6, 12, 18, 24, and 30 mo. FS was reapplied if the existing sealant had become detached or if occlusal coverage was considered insufficient—either due to further eruption of the tooth or due to part of the sealant becoming lost.
The FV used was Duraphat 50-mg/mL dental suspension (PL 00049/0042; Colgate-Palmolive Ltd), equivalent to 22,600-ppm fluoride. Dosage per single application did not exceed 0.4 mL. The standard clinical protocol was used to apply the FV to all surfaces of the FPM. FS was applied by a dental hygienist in the MDC within 2 wk of the baseline clinical examination and at 6-mo intervals for 30 mo. The study protocol dictated that reapplication should occur within a 4-wk interval on either side of the 6-mo anniversary of the previous application.
Caries Risk–Related Habits and Dental Care during the Trial
An annual parental questionnaire asked about toothbrushing frequency, whether the child brushed on his or her own or with parental assistance, the type of toothpaste used, and the quantity of toothpaste dispensed to the toothbrush. Enquiry was also made of the age at which toothbrushing started. The use of mouthwash, fluoride drops, and fluoride tablets was determined, as was previous application of FV by the child’s own dentist. Attendance at a dentist outside the Designed to Smile program was ascertained, as well as the frequency of dental attendance. Parents were asked about lifetime residency in South Wales. The annual questionnaire also collected data on dietary habits, with an emphasis on the frequency of the consumption of sugar-rich food and drinks. The questionnaire was sense checked and designed with patient and public representatives from a school not involved in the study, prior to trial commencement.
Children attended their usual sources of dental care during the trial, but their dentists were asked to refrain from providing FS or FV treatments. Children and their parents continued with their usual oral hygiene regimes, the details of which were gathered via the annual questionnaire.
The occurrence of any serious adverse events or serious adverse reactions were ascertained and recorded with the study serious adverse event form.
Outcome Measures
The prespecified clinical outcome measures were as follows. The primary outcome was the proportion of children developing new caries (D4-6MFT) on any surface of up to 4 treated FPMs. Secondary caries models at the child, tooth, and surface levels were as follows:
The number of FPMs remaining free of caries into dentine per child for those FPMs included in the trial
The caries status of treated or untreated caries on each surface of each FPM
The binary outcome of caries occurrence on occlusal versus nonocclusal surfaces of each FPM
Sample Size
Data from a previous cohort study of local primary schoolchildren were used to derive the caries incidence in children (mean age, 6.5 y) with at least 1 erupted FPM (Treasure et al. 2005). By the age of 10 y, 40% had caries in ≥1 of their FPMs. Based on recent Cochrane reviews, it was estimated that FV would reduce the 3-y incidence from 40% to 30% in this population (Marinho et al. 2013), whereas FS would reduce it further to 20% (Ahovuo-Saloranta et al. 2013). For an individually randomized trial at a power of 80% with a significance level of 5%, at least 313 children per group were required for a comparison of caries incidence of 20% versus 30% at 36-mo follow-up.
Randomization
Randomization of participants was stratified by school and balanced for sex and primary dentition baseline caries levels with minimization in a 1:1 ratio for treatments. A random component was added to the minimization algorithm (Altman and Bland 2005) such that it was not completely deterministic (Brown et al. 2005).
Sequence Generation
Randomization was carried out by the South East Wales Trials Unit, independent of the recruiting and examining personnel in the MDC, using lists of pupil sex and caries data charts collected at baseline.
Allocation Concealment Mechanism and Implementation
Allocation lists were produced and provided to the MDC staff within a 2-wk window before they returned to the school for the baseline treatments.
Blinding
The physical nature of the technologies under test limited the scope for blinding. Both the participant and the dental hygienist were aware of the treatment provided. The dentist undertaking the clinical dental examinations at baseline and 12, 24, and 36 mo was not informed of the arm to which the participant had been randomized. However, the presence or absence of FS at assessment would obviously indicate the likely treatment received.
Statistical Methods
All comparative analyses were carried out on an intention-to-treat basis (without imputation). The primary outcome was the proportion of children experiencing caries into dentine at ICDAS levels 4 to 6 on any 1 of up to 4 FPMs in the trial at 36 mo. The D4-6MFT variable was calculated (and converted to a binary outcome) from the full caries charts of those children attending the 36-mo examination and included only those FPMs in the trial. FPMs that were already sealed, carious into dentine, filled, or affected by posteruptive breakdown at baseline were excluded from the trial.
The primary outcome was analyzed per a logistic regression model. The results are presented as unadjusted and adjusted odds ratios for the FV arm versus the FS arm. The intervention was carried out within schools, so a 2-level logistic model was used to account for clustering by school. If clustering was found to be negligible, the primary analysis was taken to be a single-level model. Two- and 3-level logistic regression models were used for the analysis of tooth- and surface-level caries outcomes. Ordinal regression was used to analyze the number of FPMs with caries. All models were adjusted for the randomization-balancing variables: sex and baseline caries in the primary dentition. Baseline caries (d4-6mft) was categorized as none, 1 or 2 primary teeth, or ≥3 primary teeth. The number of FPMs per child in the trial was added to the models as a covariate but removed if nonsignificant. Other covariates added to the primary analysis model were those from the dental health questionnaire.
Statistical analysis was conducted with SPSS 20 (IBM Corp) and STATA (StataCorp).
Approvals
Research ethics approval was granted by the Research Ethics Committee for Wales 3 (11/MRE09/6). The trial was regulated by the UK Medicines and Healthcare Regulatory Authority as a Clinical Trial of an Investigational Medicinal Product.
There were no significant changes to the trial methodology after trial commencement.
Results
Participant flow through the trial is illustrated in Figure 2. In total, 1,303 children for whom parental consent had been obtained were screened for participation in the trial. Of these, 1,016 were deemed eligible for inclusion, but 1 participant subsequently withdrew consent to participation and to use of any of his or her data. At screening, 287 children were excluded mainly due to lack of FPM eruption. Children were randomized to receive FS (n = 514) or FV (n = 501).
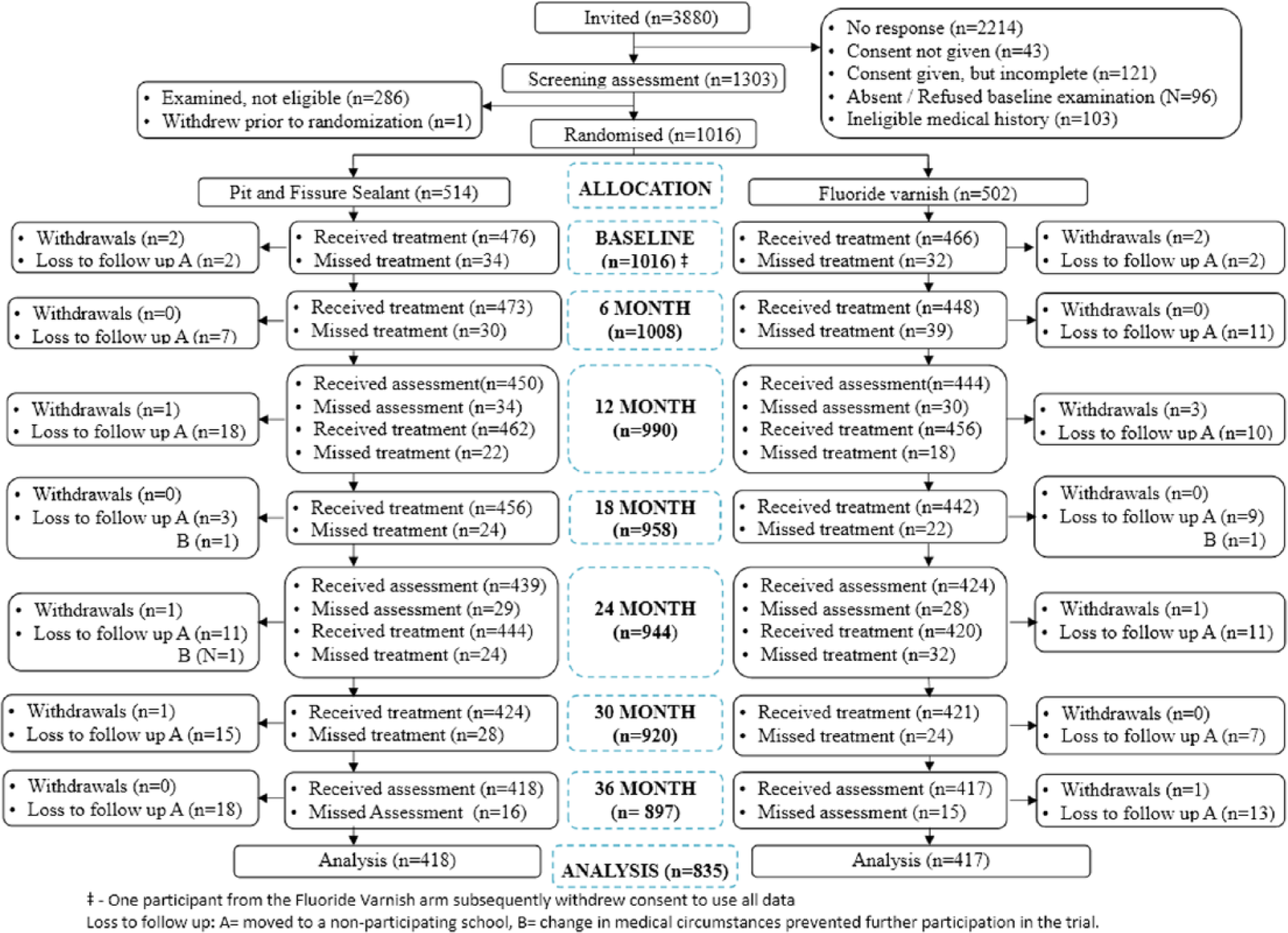
Participant flow through the trial.
The baseline characteristics of the study participants are shown in Table 1. In line with the targeted-population approach of Designed to Smile, 78.7% of the study population were residents in the second-most and most deprived quintiles of deprivation. There were no apparent differences between trial arms in sex or the proportion of children with caries experience in their primary dentition. Within deprivation quintiles, the distribution of children across trial arms was similar, and 78.6% of children lived in the bottom 2 quintiles of deprivation. There were no marked differences in baseline dental caries experience at baseline of the 835 children who completed the trial or the 180 who were lost to follow-up or withdrew.
Characteristics at Baseline of the 1,016 Children Randomized to Participate in the Trial.
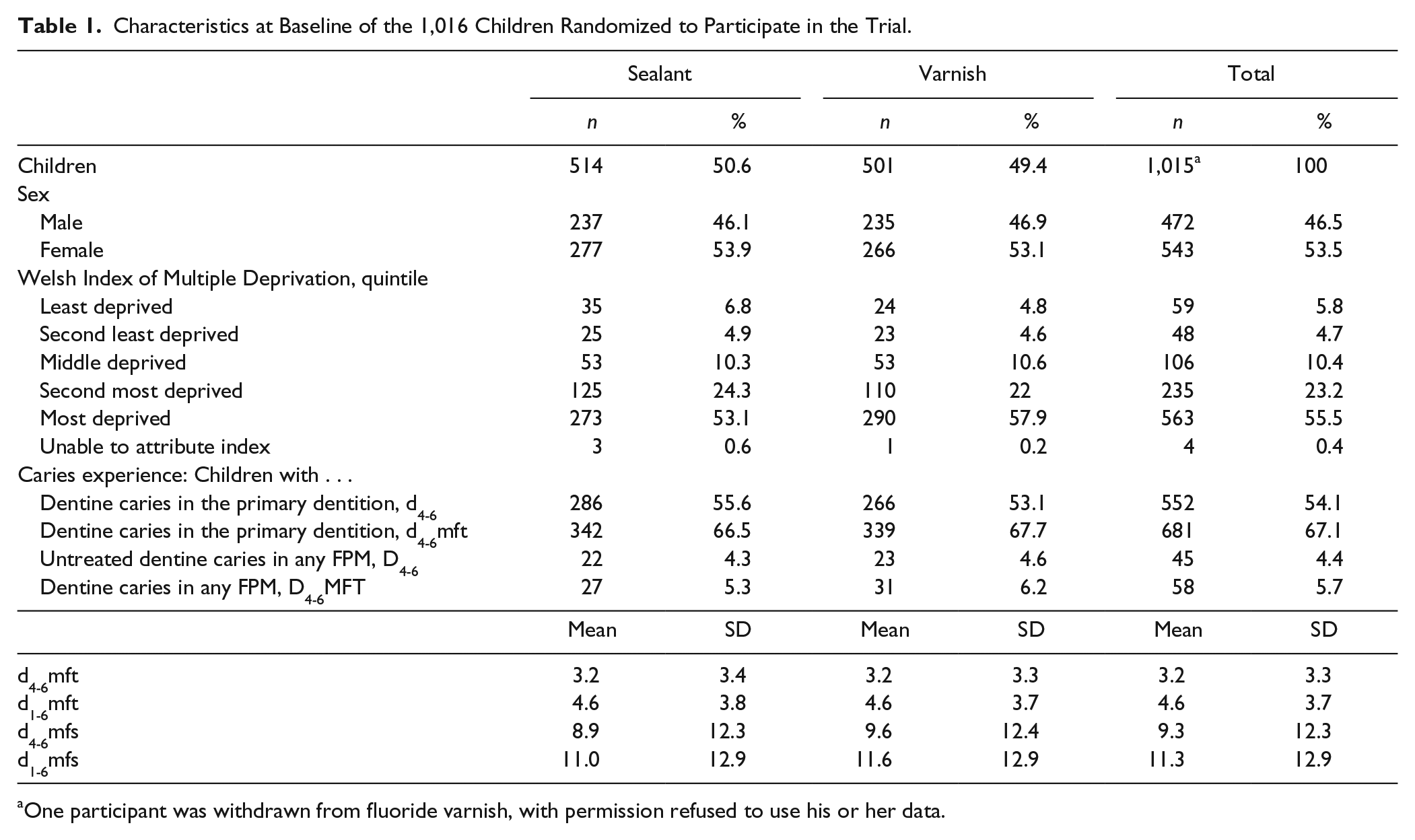
One participant was withdrawn from fluoride varnish, with permission refused to use his or her data.
At 36 mo, 835 (82.2%) children underwent a final clinical examination: 418 in the FS arm and 417 in the FV. The most common reason for not completing the trial was moving away from the area or to a school that was not participating in the trial, reported as lost to follow-up (Fig. 2). Five children withdrew from the FS arm and 7 from the FV arm. Dropout bias was assessed with baseline data and was not identified; missing data were therefore assumed to be missing completely at random. We also ascertained that dropout was low, mainly due to moving, and was not associated with treatment arm. This all pointed to a conclusion that missing data were missing completely at random.
Trial fidelity was high: 95% of children received at least 5 of the 6 scheduled treatments, and 97.6% had their treatment on time or outside the 4-wk window only once. Sealant retention was high. At 36 mo, in the maxillary FPMs, 74.5% were intact, 23.3% were partially intact, and 0.5% were lost. Corresponding figures for the mandibular FPMs were 91.4%, 5.1%, and 0.8%. No adverse effects were reported during or in the 48 h after treatment in either group. Inter- and intraexaminer reproducibility were both high, with mean kappa scores of 0.82 and 0.89, respectively.
The proportion of children who developed dentine caries (D4-6MFT) on at least 1 FPM at 36 mo was broadly similar in both the FS arm (19.6%) and the FV arm (17.5%; Table 2). Since sex and baseline caries prevalence were used to balance the randomization, an adjusted model was also performed and taken as the primary analysis. The odds ratio for developing caries in the FV arm was 0.84 (95% CI, 0.59 to 1.21) in the adjusted model. The final model (Table 2) shows no significant difference in the proportion of children with dentine caries (D4-6MFT) on any FPM in the trial at 36 mo whether the children received either FS or FV. Children who had >3 carious primary teeth at baseline were significantly more likely to develop caries into dentine on a FPM at 36 mo. There was no difference between the proportion of boys and girls developing caries into dentine on at least 1 FPM. None of the covariates altered the main effect for arm.
Proportion of Children with Dentine Caries (D4-6MFT) on Any FPM in the Trial at 36-mo Follow-up, by Trial Arm and Influence of Covariates.
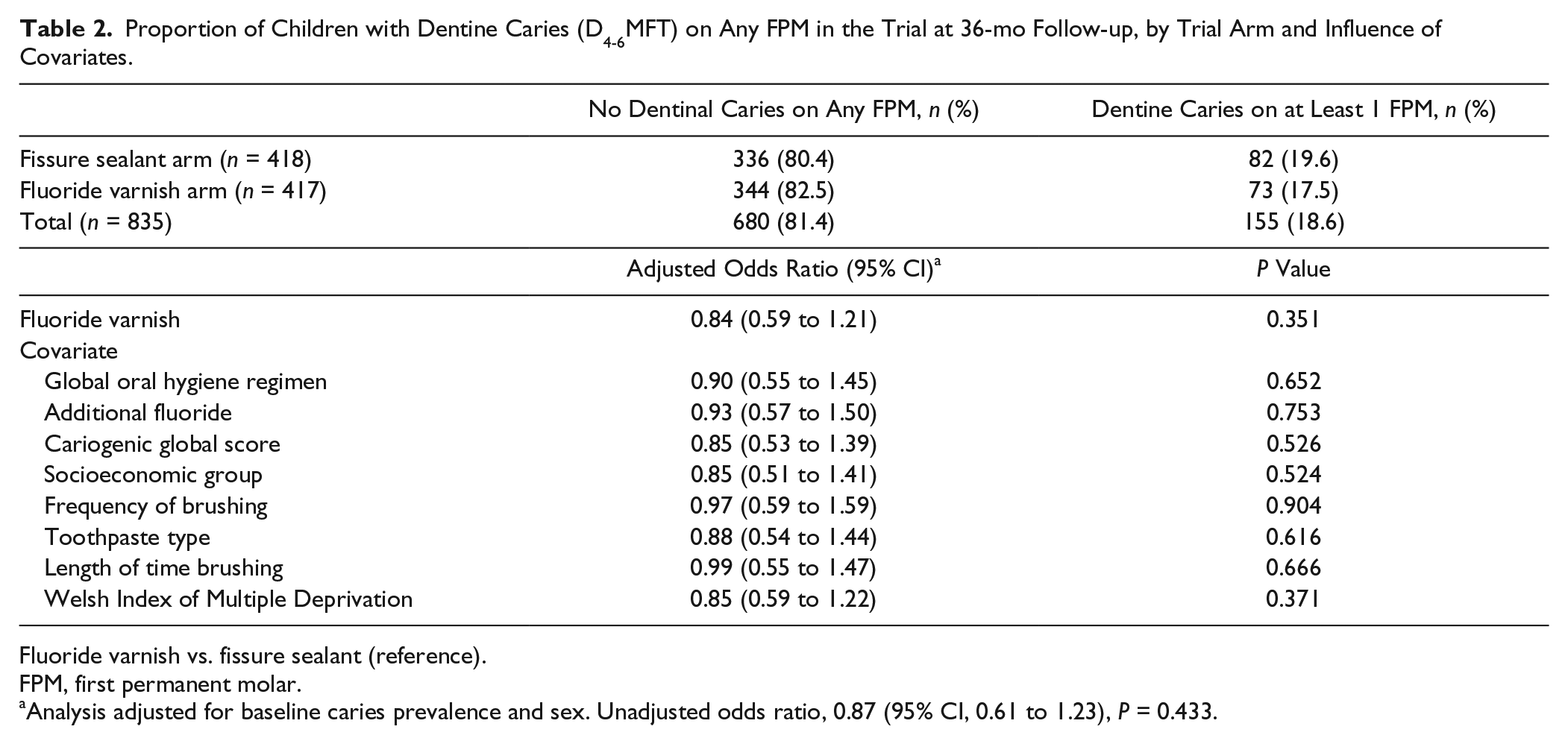
Fluoride varnish vs. fissure sealant (reference).
FPM, first permanent molar.
Analysis adjusted for baseline caries prevalence and sex. Unadjusted odds ratio, 0.87 (95% CI, 0.61 to 1.23), P = 0.433.
The findings for caries outcome models at the level of the tooth, tooth surface, and occlusal versus nonocclusal surface are shown in Table 3. No significant differences between the interventions tested were observed, with the number of teeth developing caries in both trial arms being very similar.
Development of Dentine Caries (D4-6MFT) at Tooth, Tooth Surface, and Occlusal versus Nonocclusal Surface on FPMs by Intervention Arm at 36 mo.
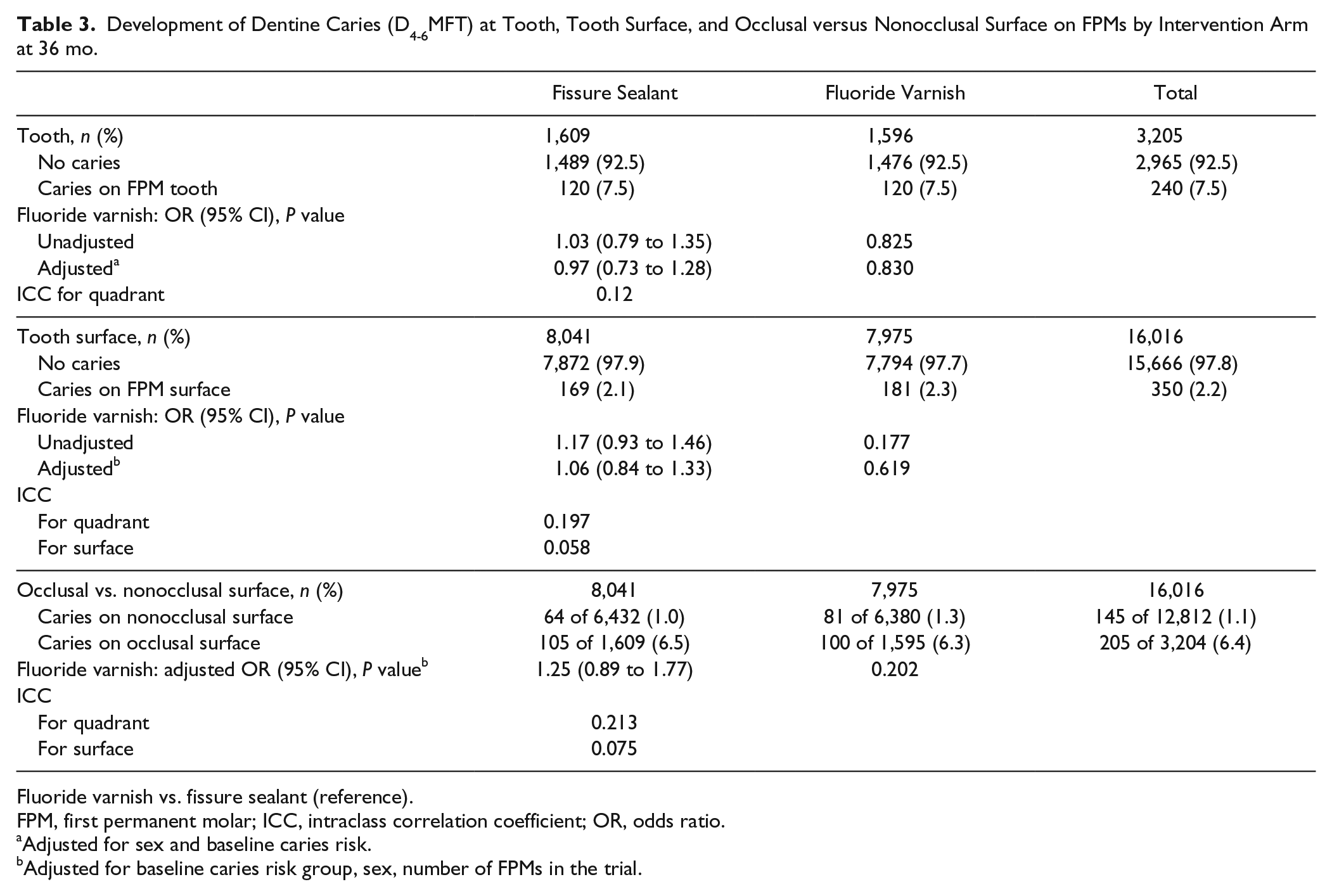
Fluoride varnish vs. fissure sealant (reference).
FPM, first permanent molar; ICC, intraclass correlation coefficient; OR, odds ratio.
Adjusted for sex and baseline caries risk.
Adjusted for baseline caries risk group, sex, number of FPMs in the trial.
Discussion
This study is the largest ever to address the question of the clinical and cost-effectiveness of FS and FV. The very high standard to which this work was conducted and the excellent fidelity obtained mean that the findings here are robust.
Given that the recent systematic review of the effectiveness of the technologies tested suggested that FS may be clinically superior (Ahovuo-Saloranta et al. 2016), the results obtained in this trial are perhaps contrary to what many pediatric and public health dentists would have expected. However, the evidence available to inform that review was very weak. There have only been 4 studies that directly compared FS and FV (Raadal et al. 1984; Bravo et al. 2005; Liu et al. 2012; Salem et al. 2014). Three of these contributed to the Cochrane review. Two studies of 2-y duration—a split-mouth study at high risk of bias conducted in 1984 (Raadal et al. 1984) and a parallel-group study (Liu et al. 2012) at unclear risk of bias—together provided a total of 358 children to the analysis. Bravo et al. (2005) reported on a comparison of FS and FV at 4 and 9 y, but the final outcome was based on just 75 children and was deemed by the Cochrane group to be at high risk of bias (Ahovuo-Saloranta et al. 2016).
The current trial clearly showed that while <1 in 5 children had developed decay into dentine in their FPMs at 36 mo, there was no clinically important difference in the proportion of children developing decay (D4-6MFT) on any FPM whether treated with FS or FV. When tested against no-treatment controls, the effectiveness of FS is generally accepted and has been reinforced by the findings of 2 recent systematic reviews (Ahovuo-Saloranta et al. 2013; Wright, Tampi, et al. 2016). Both the Department of Health in England (Public Health England 2014) and the American Dental Association (Wright, Crall, et al. 2016) endorse the use of sealants as effective caries-preventive agents. However, the current study suggests that 6-mo application of FV results in a caries-preventive effect that is not significantly different from that obtained by the use of sealants. This may contradict the recent recommendations of the American Dental Association (Wright, Crall, et al. 2016) of a preference for FS over FV in preventing occlusal caries.
Conclusion
The findings of this trial demonstrate that in community oral health programs targeted at children at high caries risk, the application of FV as a caries-preventive measure will result in caries prevention that is not significantly different from that obtained by applying and maintaining FS after 36 mo.
Author Contributions
I.G. Chestnutt, B.L. Chadwick, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; R. Playle, contributed to design, drafted the manuscript; S. Hutchings, J. Nuttall, contributed to design, critically revised the manuscript; S. Morgan-Trimmer, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; D. Fitzsimmons, contributed to conception, design, data acquisition, analysis, and interpretation, critically revised the manuscript; N. Aawar, L. Angel, S. Derrick, C. Hoddell, N. Kirby, C. Scoble, J. Townson, B. Withers, contributed to data acquisition, critically revised the manuscript; C. Drew, contributed to data acquisition, analysis, and interpretation, critically revised the manuscript; K. Hood, I. Humphreys, T.M.M. Lau, C. Lisles, M.Z. Morgan, K. Onishchenko, contributed to data analysis and interpretation, critically revised the manuscript; S. Murphy, contributed to conception, design, data analysis, and interpretation, critically revised the manuscript; C. Phillips, contributed to conception and design, critically revised the manuscript; T. Pickles, contributed to conception and design, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Footnotes
This report presents independent research funded by the National Institute for Health Research (grant 08/104/04). The views expressed are those of the authors and not necessarily those of the National Health Service, the National Institute for Health Research, or the Department of Health.
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
