Abstract
Improving the ablative resistance of carbon/phenolic (C/Ph) composites is an urgent need in the field of thermal protection of hypersonic vehicles. In this paper, novel C/Ph composites are fabricated by introducing different contents of ZrB2 ceramic particles, and the anti-ablation properties of the composites are tested in plasma wind tunnel. Under mainstream conditions with enthalpy of 21.84 MJ/kg, heat flux of 1.6 MW/m2, and pressure of 1.6 kPa, the effects of ZrB2 ceramic content on the thermal stability, linear ablation rate and surface temperature of C/Ph composites are further analyzed. The maximum ablative surface temperature is positively correlated with the content of ZrB2. As the ZrB2 content increases, the back face temperature of the materials decreases gradually, while the linear ablation rate increases slightly after a significant decrease. When the ablation time is 50 s, The linear ablation rate of the sample with 7% ZrB2 content is the lowest (0.0184 mm/s). These dates of the plasma wind tunnel test could provide a reference for applications of C/Ph composites. The oxidation of ZrB2 in the materials contributes significantly to improving its anti-ablation properties in severe environments. Accordingly, optimizing the ZrB2 content enables the development of high-performance C/Ph composites with enhanced thermal properties, and reduced ablation rates, making them well-suited for thermal protection applications.
Keywords
Introduction
Thermal Protection System (TPS) is critical in hypersonic vehicles subjected to severe aerodynamic heating.1,2 Low-density carbon/phenolic (C/Ph) composites have been extensively applied in the heatshield of hypersonic vehicles because of their excellent ablation resistance. 3 The thermal protection mechanism is that C/Ph composites absorb heat mainly through phenolic pyrolysis and the thermal blocking effect generated by pyrolysis gas to prevent heat transfer to the back face.4,5 However, the surface of the char is consumed through oxidation by the boundary-layer gases, causing the surface of traditional C/Ph composites to recede and affect the aerodynamic shape. It is still a rather complex and challenging issue to improve thermal and anti-ablation properties.
The modification of C/Ph composites by ceramic particles is one of the current research focuses. Many researchers have been committed to enhancing the performance of C/Ph composites in recent years.6–10 Ceramic particles containing zirconium are particularly suitable for anti-ablation modification of materials due to the ultra-high melting point of the oxide.11–13 Zhaoqi Niu et al. 14 studied Zirconium chelated hybrid phenolic resin thermal insulation composites, and the results showed that the formation of ZrC and ZrO2 thermal barrier ceramics is the main reason for enhanced thermal and anti-ablation properties. Masoud Salavati-Niasari et al.15–21 studied nanoparticles in the catalysis application. In our previous study, the effects of ZrC content on C/Ph composites were investigated. 22 The results showed that the thermal stability and anti-ablation resistance are improved with increasing ZrC content. ZrB2 is a kind of ultra-high temperature ceramic, its melting point is very high in the absence of oxygen, and the volatilization of boron oxide (B2O3) produced at high temperatures may enhance the thermal blocking effect.23,24 Amirsardari et al. 25 studied the thermal stability of GO/C/Phenolic Nanocomposite by introducing zirconium boride (ZrB2) under oxyacetylene tests. They found that the ZrO2 layer plays an essential role in improving anti-ablation properties, but did not conduct detailed research on the effect of ZrB2 content. It is necessary to study further the effect of ZrB2 content on the mechanical, thermal stability, and anti-ablation properties of C/Ph composites.
It is of great significance to investigate the thermal barrier effect under a severe environment with high heat flux, high enthalpy, high pressure, and even high-velocity erosion characteristics.26–28 The widely used methods of ablation tests include oxyacetylene flame, 29 laser ablation, 30 plasma wind tunnel, 31 arc-jet wind tunnel, 32 and so on.4,13 The first two methods can only simulate the high-temperature oxidizing atmosphere instead of the severe environment of high enthalpy, high velocity, and plasma non-equilibrium state, 33 and combustion products produced by oxyacetylene fuel will affect the ablative result of the materials. It is important to consider the interaction between the plasma environment and the materials. The dissociation and ionization of gas molecules in the shock layer of the hypersonic vehicle shouldn’t be ignored. The parameters of the two wind tunnel tests are more comprehensive. The plasma wind tunnel is superior to arc-jet in screening materials as it can provide pure environments rather than the copper contamination in arc-jet. 31 The pure flow field formed by the plasma wind tunnel can simulate the service environment more realistically and consider the effects of gas composition, pressure, and thermochemical ablation comprehensively. The difference of gas medium has a great influence on material ablation. Compared with other methods of testing carbon phenolic materials, the plasma wind tunnel is more scientific and reasonable. It is very meaningful to study the effect of ZrB2 content on the anti-ablation performance of C/Ph composites under the plasma wind tunnel condition, but there is limited research nowadays.
Based on this, we innovatively tested ZrB2 modified C/Ph composites with plasma wind tunnel. Anti-ablation performance and microstructure of C/Ph composites with different ZrB2 content were studied and the thermal protection mechanism was explored. We studied the effect of ZrB2 contents on material microscopic properties, thermal stability, and the linear ablation rate. The best-modified particle content was obtained.
Experiment
Materials
Materials and reagents include absolute ethanol, boron modified phenolic resin, needle-punched carbon fiber felt, acetone, nitric acid, ZrB2 particles, and hyperdispersant 7423. Phenolic resin THC-400 (Shanxi Taihang Impedefire Polymer Co., Ltd, China) is insoluble in water but soluble in ethanol. Needle-punched carbon felt as the preform (0.145 g/cm3, Tianniao High and New Technology Co., Ltd, Jiangsu, China) has 2.5 D-stitched fibrous architecture consisting of carbon fiber felt (T700SC-12,000-50C, Toray, Japan) in the thickness direction and needled fiber tows (3 K) in the through-thickness direction. The macrograph and microstructure of carbon fiber needle felt are shown in Figure 1. Particle size distributions of ZrB2 (6.1 g/cm3, Ningbo Jinlei Nanomaterial Technology Co., Ltd) are 1∼2 μm. The chemical reagents used in the experiments are given in Table 1. Macrograph and microstructure of carbon fiber needle felt. Chemical reagents used in the experiments.
Fabrication
The schematic illustrations of the preparation process of ZrB2 modified C/Ph (ZrB2-C/Ph) composites are presented in Figure 2. These composites were fabricated in turn by surface treatment, vacuum impregnation, drying, and curing. Firstly, surface treatment of the felt was conducted: it was treated with acetone for 24 h to desize carbon fibers; and then, the felt was dried in the oven after washing with distilled water; afterward, the felt was processed with concentrated nitric acid for 2 h, and dried in the oven. Secondly, the phenolic resin was dissolved in absolute ethanol to prepare the 25% phenolic ethanol solution; after that, ZrB2 particles and hyperdispersant 7423 were added into the prepared solution. The addition amount of ZrB2 was 3%, 5%, 7%, 9%, and 11%, respectively, and the hyperdispersant 7423 which was 7% of ZrB2 in the phenolic ethanol solution, and used dispersion homogenizer (JFS 750S) to mechanically stir at 1500 r/min for 40 min until completely dissolved. The surface-treated fiber felt was impregnated in the mixed solution at room temperature in the vacuum oven. Thirdly, the impregnated carbon fiber felt was placed in the drying oven to dry thoroughly. Finally, ZrB2-C/Ph composites were prepared after ZrB2 curing by the double-stage method 35: the first was performed at 120°C for 1 h and the second was performed at 170°C for 2 h. The names of materials with their basic parameters are summarized in Table 2. Preparation process diagram of ZrB2-C/Ph composites. Parameters of the processed composites.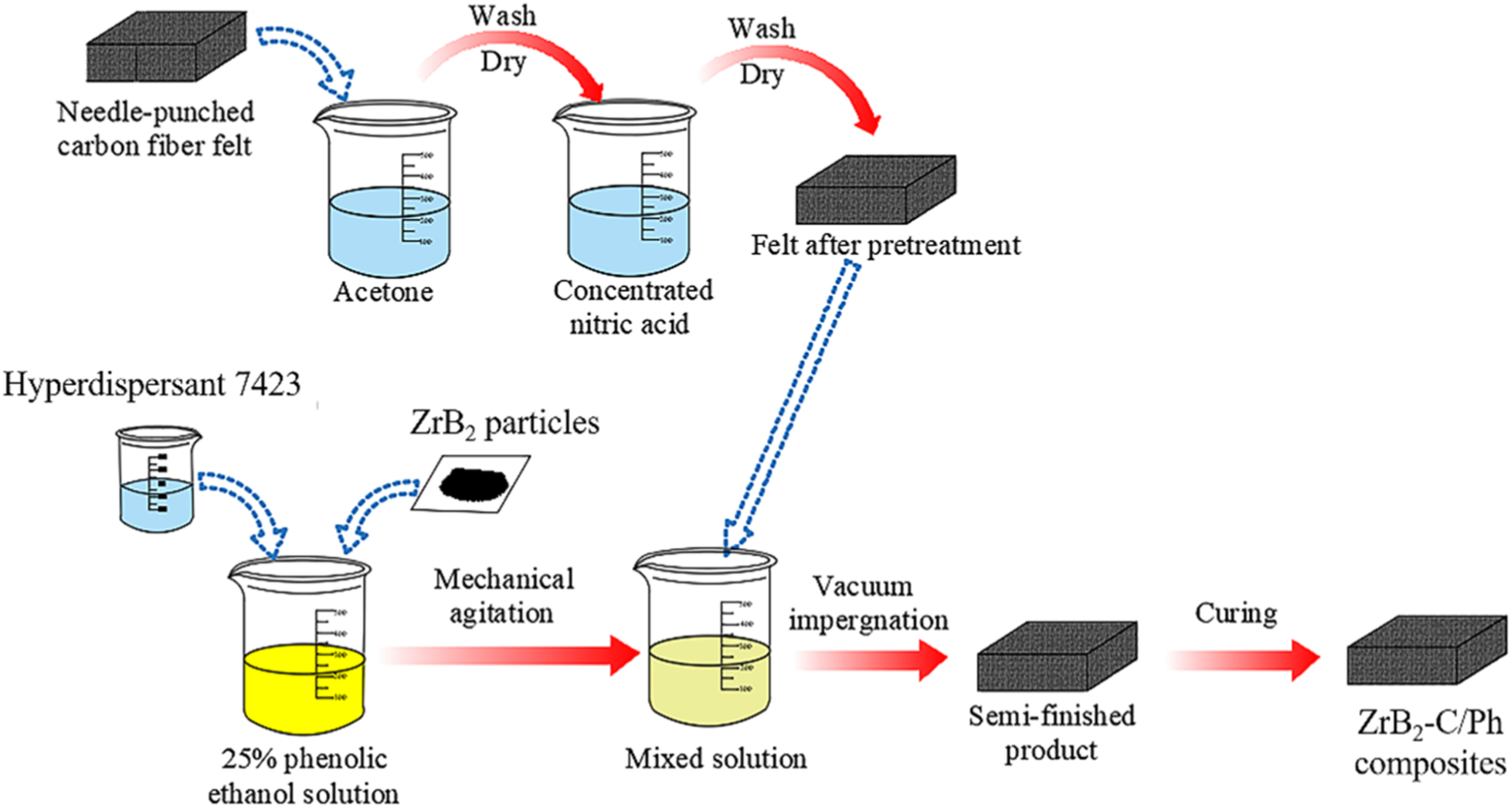
Characterizations
In order to study the dispersion effect of hyperdispersant 7423 and determine the optimal addition amount, the stability of ZrB2 particles in phenolic ethanol solution containing different amounts of dispersants is studied using the sedimentation test. Hyperdispersant 7423 was added to the solution containing 25% phenolic resin and 5% ZrB2 particles, The dispersion state of ZrB2 particles was observed, and the settling process of ZrB2 was recorded within 180 min.
Compression strength and thermal conductivity of the samples were measured, respectively. Thermogravimetric (TG) analysis was conducted to estimate thermal stability. Anti-ablation performance was evaluated by the plasma wind tunnel. The test conditions were a surface pressure of 1.6 kPa, enthalpy of 21.84 MJ/kg, heat flux of 1.6 MW/m2, and test time of 50 s. Before testing, the specimens were machined with a size of Φ30 mm × 25 mm, and held in a graphite mold on the water-cooled manipulator arm. The ablated surface was 50 mm from the nozzle outlet, perpendicular to the direction of the flame. The infrared pyrometer measured the temperature of the ablated surface, while a K-type thermocouple measured the temperature of the back face. Simultaneously, the shape change of samples during the test was recorded by the camera. The thickness of the ablated sample is measured after stripping the white ZrO2 layer, and the linear ablation rate is calculated. The linear ablation rates of samples can be calculated by equation (1):
Results and discussions
Properties of carbon/phenolic composites with different ZrB2 contents
The sedimentation process of ZrB2 with different dispersant contents is shown in Figure 3. The addition amount of hyperdispersant 7423 from left to right are 0%, 3%, 5%, 7%, 9%, and 11% of the content of ZrB2 in order. After hyperdispersant 7423 was dispersed in phenolic ethanol solution for 180 min, ZrB2 particles with 7% dispersant content had the best uniform dispersion. Sedimentation experiment of ZrB2 with different dispersants.
Figure 4 shows the microstructure and element mapping of C/Ph composites with 7% ZrB2 content. It can be seen that the fibers are connected by phenolic resin, and the carbon fibers on the surface are disordered, and there is no obvious fiber orientation. The O, Zr, and B elements are well-distributed in the C/Ph composite. At the same time, there is no obvious agglomeration of ZrB2 particles, which also proves that the dispersion of ZrB2 particles in phenolic resin is uniform. A uniform ceramic protective layer is formed during ablation, which is beneficial to the ablative properties of the material. Element mapping of C/Ph composite with 7% ZrB2 contents.
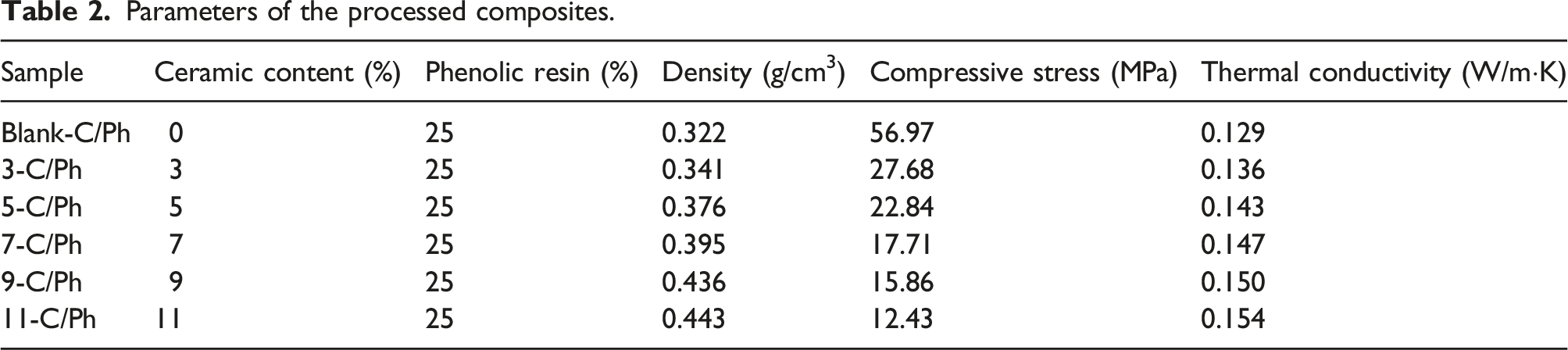
It can be seen from Table 2 that the density and thermal conductivity of the materials gradually increase as the ZrB2 content increases. When the ceramic content is 0%, the density of C/Ph composites is the lowest (0.322 g/cm3), and when the ZrB2 content is 11%, the C/Ph composites density is the highest (0.443 g/cm3). The introduction of ZrB2 can form a local high thermal conductivity area inside the C/Ph material, and with the increase of ZrB2 content, the high thermal conductivity area increases, resulting in an approximately linear increase in the thermal conductivity of C/Ph. The addition of ZrB2 particles prevents the resin matrix from forming a continuous mesh structure, resulting in a decrease in the compressive stress of C/Ph composite.
TG curves of composite materials containing different contents of ZrB2 tested in a nitrogen environment are shown in Figure 5. Below 700°C, the variation trend of composites is similar, and it can be observed that the main weight changes occur at this stage between 300°C and 700°C. But the temperature of the maximum degradation rate for the ZrB2-modified materials is significantly higher than that of the blank-C/Ph composite (571°C). The weight loss of the blank-C/Ph composite (21.56%) at this stage is almost three times that of the 7-C/Ph composite (7.76%) and four times that of the 11-C/Ph composite (5.49%), indicating that the introduction of ZrB2 particles significantly improves the thermal stability of phenolic. Because ZrB2 can oxidize with oxidizing gases (such as CO, CO2, and H2O) generated from phenolic pyrolysis to increase the mass. Therefore, when the temperature exceeds 700°C, the mass loss of composites without ceramic particles increases. In contrast, the mass loss of several groups of materials modified by ZrB2 particles is almost unchanged. TG curves of C/Ph composites with different contents of ZrB2.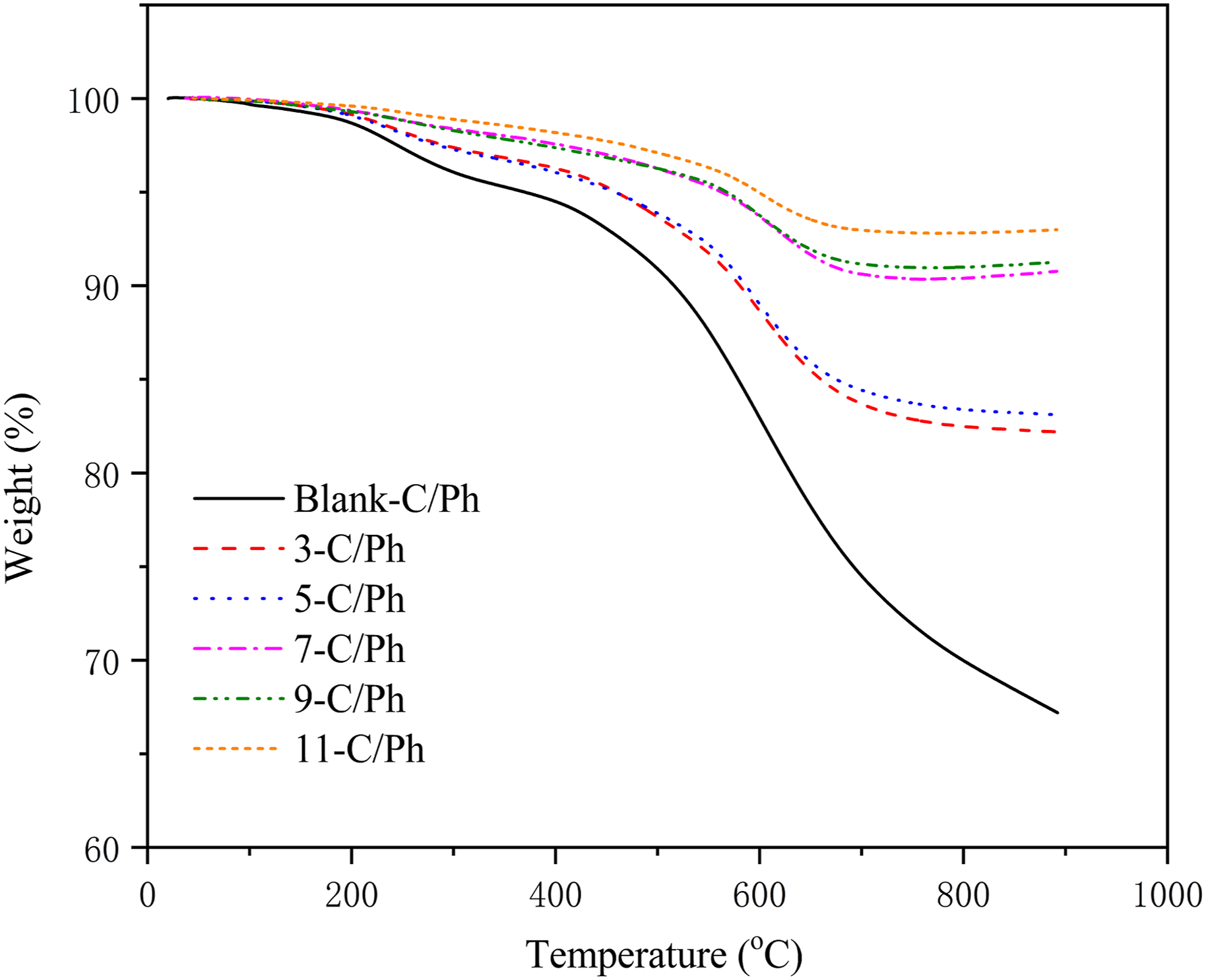
Morphological characterization
The states of blank-C/Ph and 7-C/Ph composites before, during, and after the ablation are shown in Figure 6. The two pre-test samples are displayed in Figure 6(a) and (d); there is no observable difference between the two pre-test samples. Figure 6(b) and (e) provide macrographs of samples during the plasma wind tunnel test. Figure 6(c) and (f) presents the post-test samples of the composites, respectively. It is clear to see that the white-colored layer on the ablated surface in Figure 6(f), significantly differs from blank-C/Ph composites. Similar phenomena and related mechanisms have been discussed in our previous work.
22
Macrographs of the composites before, during, and after the ablation test, respectively: (a-c) C/Ph; (d-f) 7-C/Ph.
Linear ablation rates and microstructure characterization
Figure 7 shows the results of linear ablation rates of the materials after the test. The calculation of the ablation rate does not include the white layer. The addition of ZrB2 decreases the linear ablation rate. The lowest linear ablation rate is obtained when the ZrB2 content in the materials is 7%, which is 0.0184 mm/s. When the content of ZrB2 exceeds 7%, the linear ablation rate increases slightly, but it is still lower than that of blank-C/Ph composites. The linear ablation rate of 11-C/Ph composites is 0.0236 mm/s, which is 0.0052 mm/s higher than that of 7-C/Ph composites, but 69% lower than that of blank-C/Ph composites. ZrB2 particles strongly influence the reduction of the linear ablation rate. Compared with the oxy-acetylene ablation test results of Amirsardari et al,
25
line ablation rates of C/Ph composites are higher in plasma wind tunnel, the linear ablation rates of 7-C/Ph compared with blank-C/Ph decreased by 75% in plasma wind tunnel, but decreased by 90% under oxy-acetylene ablation. Because the thermal environment provided by the plasma wind tunnel is more realistic and severe. This could be considered that the mainstream in the plasma wind tunnel has higher recovery enthalpy and mechanical denudation than oxy-acetylene flame, which needs further comparative study in future work. Linear ablation rates of ZrB2-C/Ph composites with different contents of ZrB2.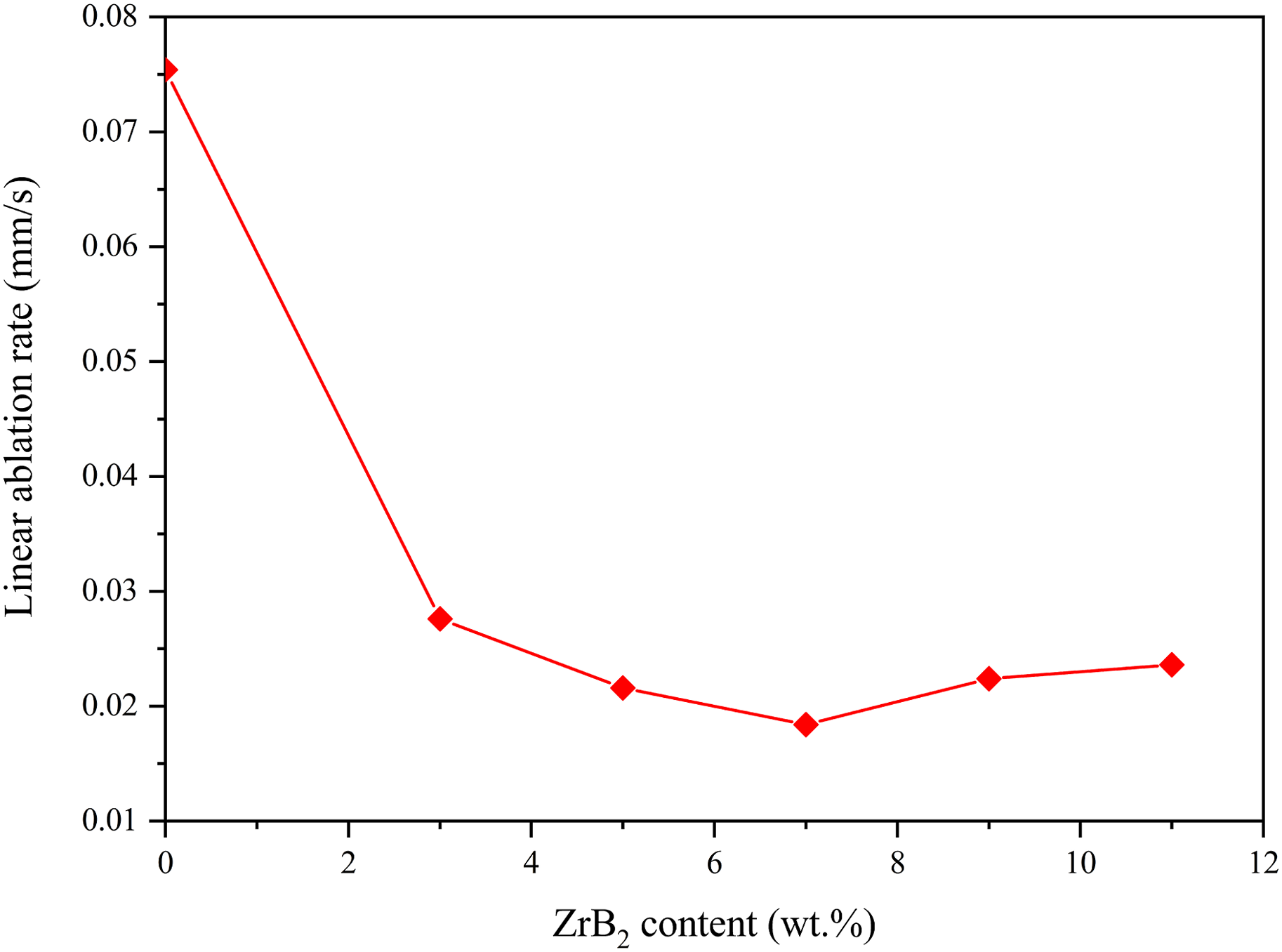
After ablation, the surface ceramic coatings are removed, and the microstructure of the samples is observed in Figure 8. When the ZrB2 content is less than 7%, the matrix on the ablated surface gradually increases as the content increases. However, When the ZrB2 content in the material is greater than 7% (Figure 8 (e) and (f)), the matrix does not increase correspondingly, but the pore size increases slightly with the increase of ZrB2 content. The matrix coated on the surface of carbon fiber can reduce the oxidation rate. There may be pyrolytic carbon, ZrO2, ZrC, and unoxidized ZrB2 in the matrix. In the wind tunnel test process, phenolic resins are pyrolyzed to generate pyrolytic carbon, and ZrB2 is oxidized to generate ZrO2 and B2O3, the production of B2O3 is less than the evaporation at the surface ablation temperature.
34
SEM micrographs of the ablated surface of ZrB2-C/Ph composites with different contents of ZrB2: (a) blank; (b) 3%; (c) 5%; (d) 7%; (e) 9%; (f) 11%; (g)-(h) SEM micrographs of white colored layer on the ablated surface.
The presence of ceramic particles and their reaction products can improve the strength of the carbonization layer and reduce the effect of mechanical denudation, thus reducing the ablation rate of the composites. 35 When the content of ZrB2 is low (less than 7%), the thermochemical ablation occupies the dominant position and the linear ablation rate decreases as the ZrB2 content increases. When the ZrB2 content in the material is excessive, the increase of the content of ZrB2 would bring about the increase of the density of the matrix; the ZrO2 coating would be so dense that the gases generated in the ablation process accumulate in the material, resulting in the peeling of the ZrO2 layer. Therefore, when the content of ZrB2 increases from 7% to 11%, the linear ablation rate is slightly increased by the combined action of mechanical denudation and thermochemical ablation. Figure 8(g)-(h) shows the micrographs of the white-colored layer on the ablated surface. Almost all of the carbon fiber is oxidized, and the pores formed by the fiber can be clearly seen.
The ablated surface temperatures
The temperature-time curve of the ablated surface of the sample measured by the infrared temperature measurement system during the plasma wind tunnel test is shown in Figure 9. The maximum ablated surface temperature is below 2200°C, and all curves show the same trend of rapid heating and constant surface temperature with the increase of ablation time. When the content of ZrB2 varies from 0% to 7%, the content of ZrB2 is positively correlated with the maximum ablated surface temperature; when the content is more than 7%, the maximum ablated surface temperature is almost constant. Such a result would confirm a limit to the effect of the ZrB2 content on the ablated surface temperature. With the increase of the content of ZrB2, the main factors affecting the maximum ablated surface temperature also change. Temperature of the ablated surface of ZrB2-C/Ph composites with different content of ZrB2.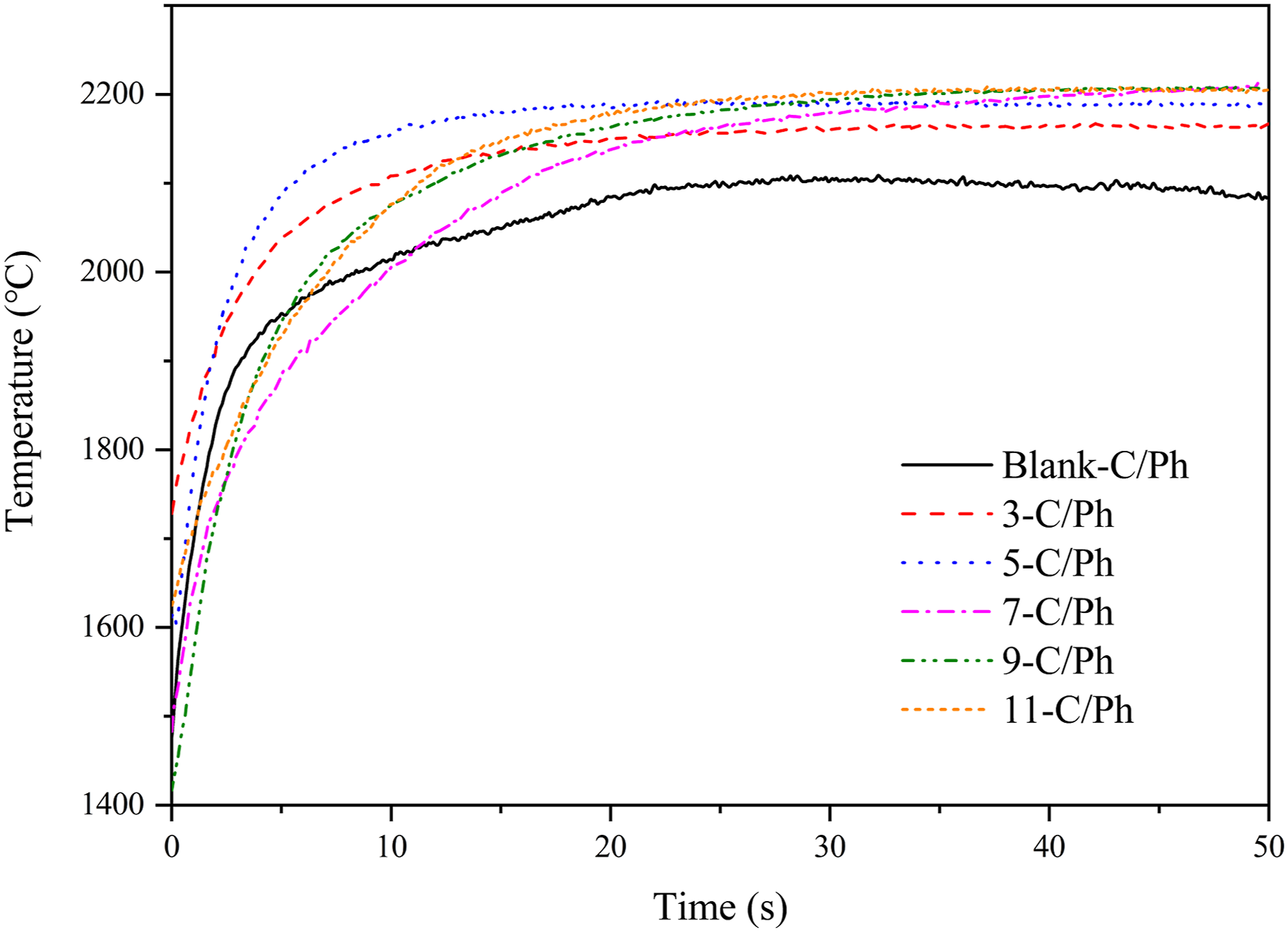
For all the test samples, the external heating conditions are the same, and the presence and content of ZrB2 mainly cause the difference in ablated surface temperatures. Due to the high ablated surface temperature, the radiative heat dissipation plays a dominant role more than the exothermic oxidation of ZrB2, the heat absorption of B2O3 gasification and the heat dissipation from pyrolysis gas escape. ZrO2 has a lower radiation coefficient compared with pyrolytic carbon and carbon fiber, which leads to the increase of ablated surface temperature by adding ZrB2. As the content of ZrB2 increases, the ZrO2 produced on the ablated surface tends to densify. Hence, the ablated surface temperature remains unchanged when the ZrB2 content is more than 7%.
The back face temperatures
Figure 10 shows the temperature-time curve of the back face measured by a thermocouple. The back face temperature decreases as ZrB2 content increases. The 11-C/Ph composite has the lowest back face temperature, and is lower than Blank-C/Ph about 50% at 50 s. The back face temperature is controlled by multiple factors such as ablated surface temperature, thermal conductivity, thickness, and chemical reactions of ceramic particles. When the ZrB2 content increases from 0 to 7%, the linear ablation rate of the composites decreases gradually, and the thickness increases, the ZrO2 layer effectively prevents heat transfer from the material surface to the back, which is conducive to preventing the back face temperature from rising rapidly. When the content increases from 7% to 11%, the thermal conductivity of the material increases and thickness decreases, but the ablated surface temperature remains unchanged; the increase of ZrB2 content leads to the increase of B2O3, volatilization and thermal blocking effect of B2O3 becomes the main reason to suppressed the heat transfer to the back face. Temperature of the back face of ZrB2-C/Ph composites with different contents of ZrB2.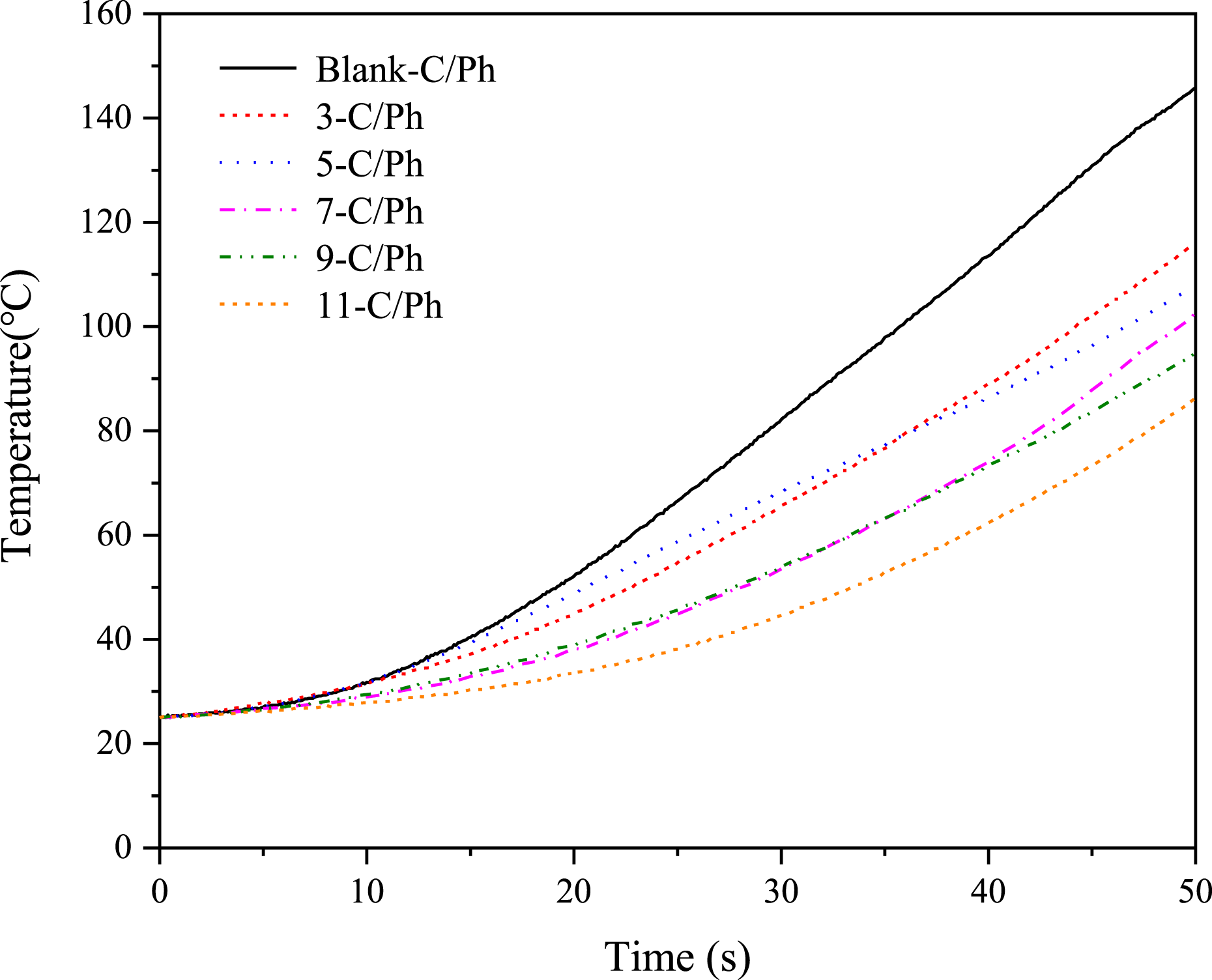
Conclusions
In short, we successfully prepared ZrB2-C/Ph with different ZrB2 contents and investigated their ablative properties at a heat flux of 1.6 MW by plasma wind tunnel test. It is found that materials have excellent comprehensive performance when the content of ZrB2 is 7%. Due to the oxidation of ZrB2 to ZrO2 ceramic protective layer, and the evaporation of B2O3 enhances the thermal blocking effect, the ablative resistance of ZrB2-C/Ph is significantly improved. As the ZrB2 content increases, the back face temperature of the materials decreases gradually, and the line ablation rate decreases first and then increases. The linear ablation rates of 7-C/Ph compared with blank-C/Ph decreased by 75%, this result is higher than that obtained under the condition of oxyacetylene ablation. The back face temperature of the sample with 11% ZrB2 content rose the slowest, 50% lower than that of the composite without ZrB2. ZrB2 ceramic particles could be promising materials to improve the anti-ablation performance of carbon/phenolic composites. These results measured in the plasma wind tunnel can provide a reference for engineering applications. The effect of different proportions of multicomponent ceramic particles added to C/Ph composites on ablative properties could be investigated in the future.
Footnotes
Acknowledgments
The authors sincerely acknowledge the support from the National Natural Science Foundation of China (grant numbers 92271106).
Author contributions
Andi Lin contributed to the conceptualization, methodology, formal analysis, investigation, and writing the original draft. Haiming Huang and Shuang Wang contributed to supervision, experimental design, review, and editing. Ye Tian and Jie Huang contributed to experimental investigation and data analysis. Xinmeng Wang contributed to review and editing. All authors discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China; (92271106).
