Abstract
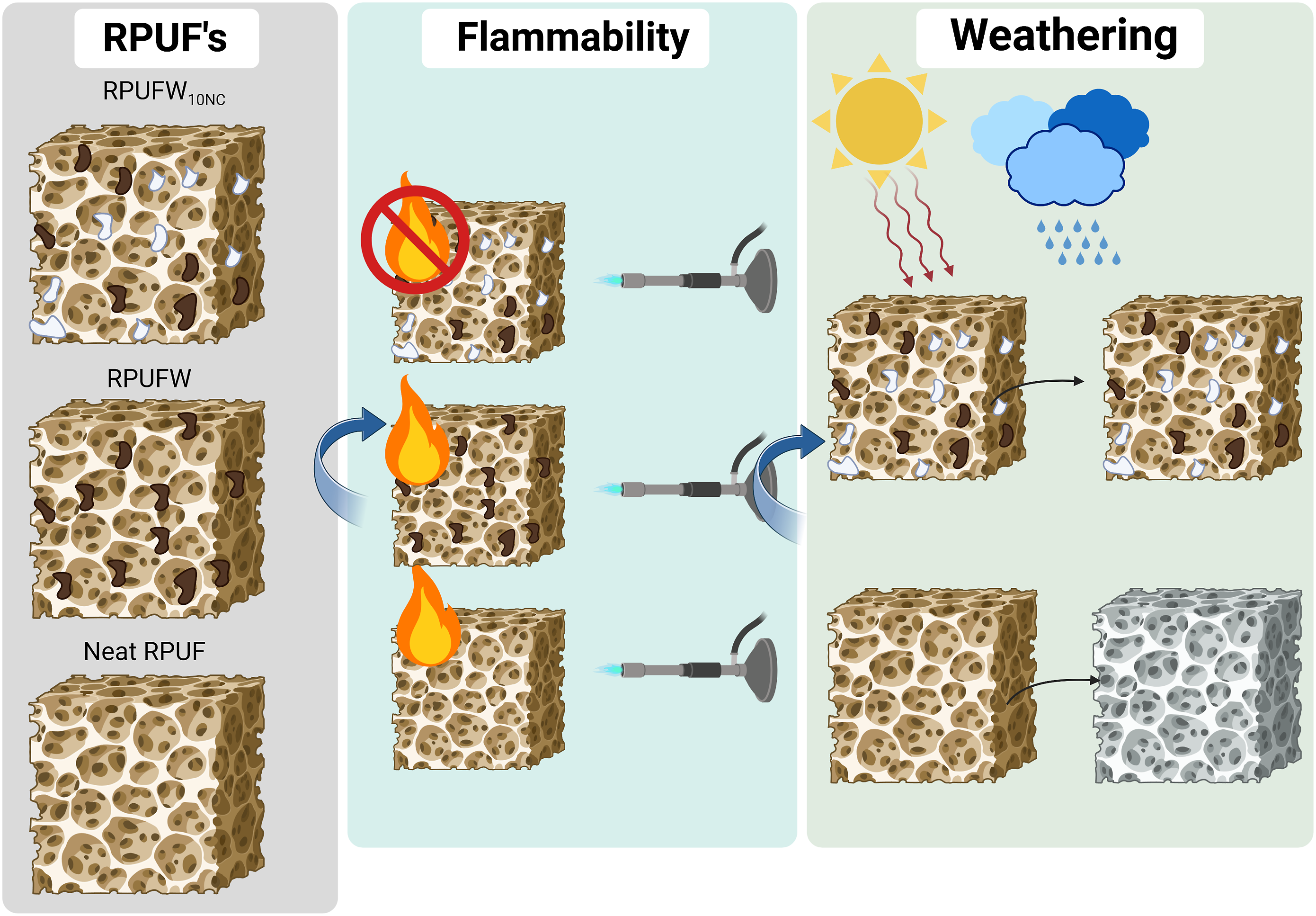
Rigid polyurethane foams (RPUF) filled with lignocellulosic fillers have gained considerable interest due to their mechanical performance and eco-friendly characteristics. However, their flammability and photodegradation resistance properties still need further improvement, which may be achieved by incorporating various particles. This study investigated the effect of adding 2.5% of wood flour and 5%–15% of an organophilic nanoclay (relative to the wood flour weight) on the RPUF morphology, density, compressive strength, thermal stability, flammability, and photodegradation resistance. The addition of wood flour and nanoclay made the RPUF cells more rounded and disrupted but did not affect density. The compressive properties were adversely impacted. Nevertheless, the nanoclay significantly improved both flammability and photodegradation resistance compared to the neat RPUF.
Introduction
Rigid polyurethane foams (RPUF) find application in diverse industrial sectors, including construction, transportation, thermal insulation, and packaging. Nevertheless, their production involves petroleum-based chemicals, with adverse environmental impact. Bio-based RPUF, which employ polyester polyols derived from vegetable oils, 1 chemical additives based on glycerin 2 and natural fibres or particles as fillers, 1 are more ecologically sound.
Due to their abundant availability, lignocellulosic materials and their derivatives are widely utilized as reinforcements or fillers. 3 These forestry products include wood flour, 1 individualized fibres,4,5 cellulose nanocrystals, 6 cellulosic pulps 7 and others. Reactive fillers may partially replace the polyol by chemically crosslinking to the cellular polymer.8,9 However, most of them are produced via expensive or tedious processes (i.e. micro- and nanoparticles from cellulose). Among the fillers, lignocellulosic materials and their derivatives are among the most used due to their large availability.
Rigid polyurethane foams are easily degraded by short wavelength ultraviolet (UV) rays from the sunlight, which mainly disrupts amines, leading to the formation of quinones. 10 This degradation adversely affects several RPUF properties, including a remarkable change from the natural yellow colour to the orange tone. 11 In this sense, several recent studies focused on mitigating this photodegradation. For instance, Delucis et al. 12 added wood flour, wood bark, kraft lignin and paper sludge to RPUF and achieved greater photodegradation resistance, especially for darker foams (those with wood bark and kraft lignin particles).
Another disadvantage of neat RPUF is their high flammability, resulting in short ignition times and rapid flame spread. 13 Much emphasis has been placed on safety regulations applicable to flame-retardant materials concerning toxicity and release of smoke products, such as carbon monoxide, carbon dioxide and hydrogen cyanide. 14 In this sense, several studies have focused on the insertion of fillers into RPUF to achieve improved thermal stability. Członka et al. 15 incorporated bleached Curauá fibres into bio-based RPUF and reported higher thermal conductivities and lower flammability. More recently, Bradai et al. 16 achieved a slight improvement in thermal stability, thermal conductivity and fire retardancy for RPUF reinforced with wood-based particles.
Furthermore, Babar et al. 17 reported improved thermal stability in RPUF reinforced with nanoclay. Indeed, various clays (e.g., montmorillonite and halloysite) have been used with flexible, semi-rigid and rigid foams. 18 These clays may create effective nucleation sites, and enhance mechanical and flammability properties. In this sense, Nik Pauzi et al. 19 introduced modified nanoclay into palm oil-based PU foam and achieved better thermal stability with higher nanoclay contents (6–10 %wt). Kim et al. 20 obtained slightly improved flame retardant properties for PUF with organically modified nanoclay (up to 3 php). As reported by Zheng et al., 21 in the combustion of RPUF reinforced by nanoclay, the filler may migrate to the surface and promote the formation of a charred layer that impair heat transfer and reduce mass loss. A similar mechanism was also reported for lignocellulosic fillers. 22
Although research with both wood and nanoclay fillers can be found in the literature, few articles investigated their combined use. Thus, this study focused on investigating RPUF filled with wood flour and nanoclay particles aiming at decreasing flammability and photodegradation resistance of this polymer matrix.
Methodology
Fillers
Forest-based wood flakes obtained from processing Eucalyptus spp. timber was collected in a sawmill situated in Southern Brazil. The flakes were subsequently oven-dried at 50°C until constant weight, milled using a Wiley mill, and sieved through a 100-mesh screen to obtain wood flour (WF). The WF was characterized via wet chemical analyses to obtain ethanol-toluene extractives (according to Tappi T264 Cm-97), 23 acid-insoluble lignin (Tappi T222 Om-02) 24 and holocellulose (remaining mass) contents of 3.50%, 21.30% and 79.30%, respectively, which is a usual chemical composition for a hardwood. Cloisite 30 B nanoclay (NC), a modified bentonite, was acquired from Sigma-Aldrich. This NC is frequently used as additive for polymers and, according to the supplier, has 2% moisture content, 1.98 g/cc specific gravity and 30% loss of ignition. 25
The WF and NC were subjected to Fourier-transform infrared spectroscopy (FTIR) using an IRSpirit device (Shimadzu® brand) equipped with an attenuated total reflection accessory (ATR). And thermogravimetric (TG) analysis was performed under nitrogen atmosphere using a TGA-1000 equipment (Navas brand), at a heating rate of 10°C.min−1, from room temperature (approximately 20°C) to 600°C.
Production of the RPUF
Rigid polyurethane foams were prepared using the free-rise pouring method, as illustrated in Figure 1. For that, fillers, polyol (a mixture of castor oil and crude glycerin oil in a 3:1 ratio), chain extender, and surfactant were mechanically stirred at 1000 rpm for 60 s. Subsequently, MDI and catalyst were incorporated into the reaction mixture (NCO/OH ratio: 1.2), which was manually mixed for 20 s and poured into an open wooden mould. Further details on each RPUF component can be found in previous studies of the research group.
7
Schematic representation of the production of the RPUF.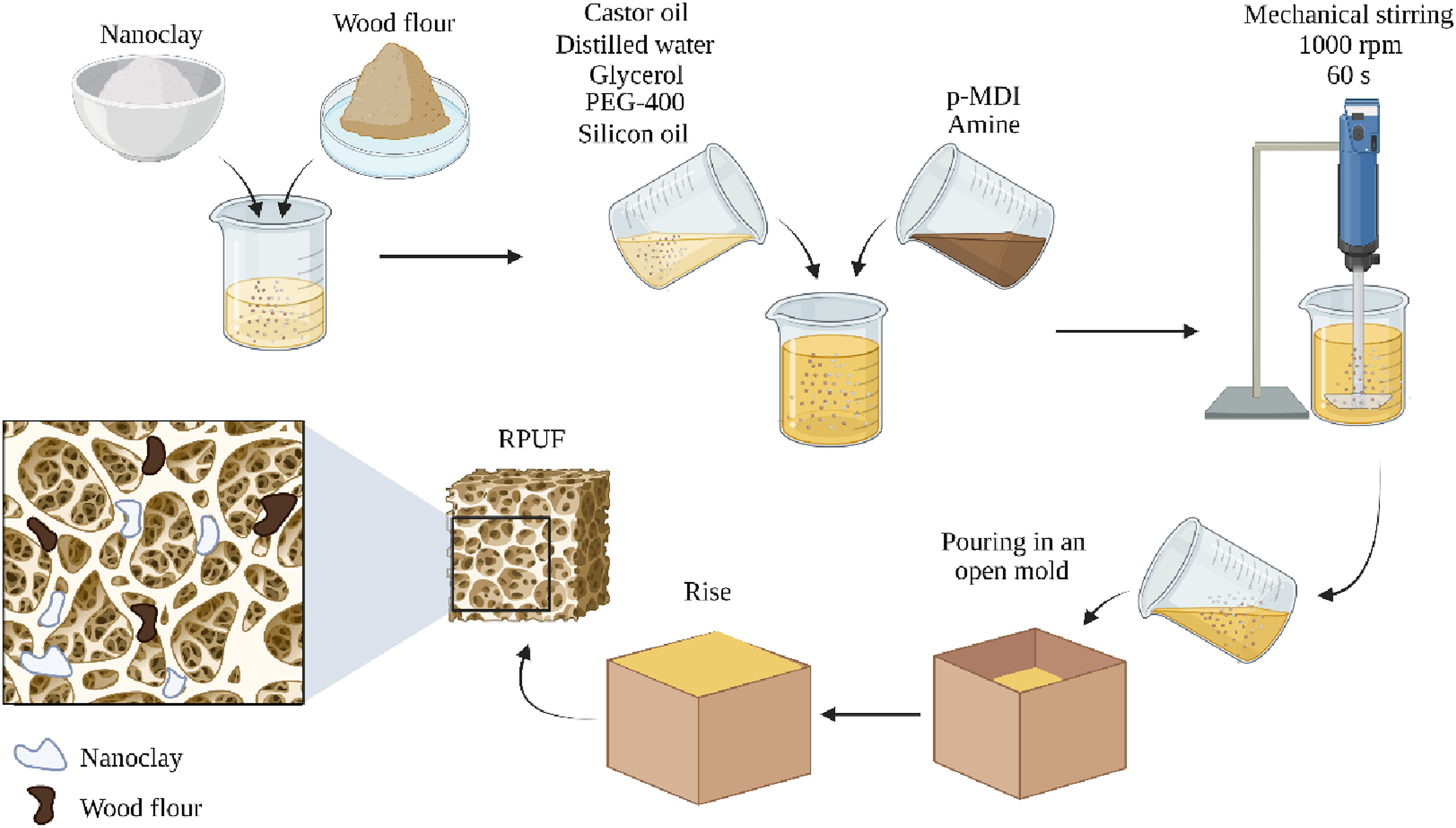
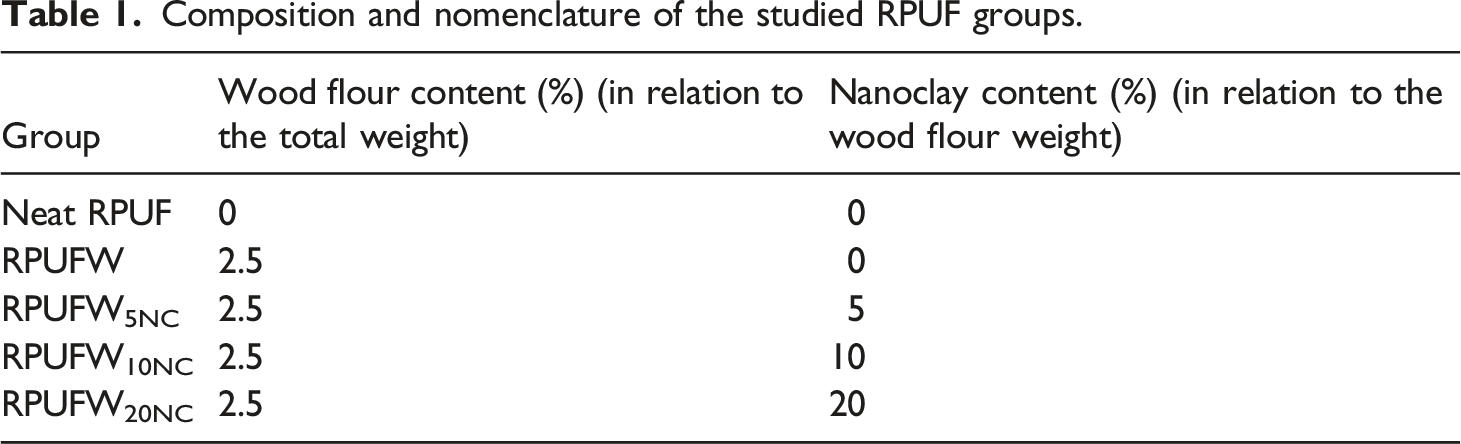
Composition and nomenclature of the studied RPUF groups.
Characterization of the RPUF
Both neat and filled RPUF were milled and subjected to FTIR and TG analysis using the previously described equipment and methodology. Morphological analysis was conducted perpendicular to the rise direction, using scanning electron microscopy (SEM) with an Evo MA10 equipment (Zeiss brand) to determine average cell size and anisotropy index (calculated using equation (1)) (Figure 2). Photographs and optical micrographs of the studied RPUF.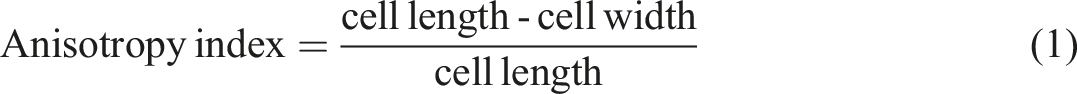

Five prismatic samples (5.0 cm × 5.0 cm × 2.5 cm; smaller dimension in the rise direction) per group were cut and weighed to determine apparent density according to. 26 The samples were also subjected to compression testing parallel to the rise direction, at a speed of 2.5 mm.min−1, using a EMIC 23-5D universal testing machine. The compressive strength was read at 3.30 mm displacement, according to. 27
Other prismatic samples (1.0 cm × 1.0 cm × 1.5 cm) were analysed for flammability based on the methodology described by Tondi and Pizzi. 28 For that, the RPUF sample was suspended on a rod at a vertical distance of 5 cm from the reducing flame of a Bunsen burner. Then, the burning was monitored with the aid of a digital camera.
Cubic foam specimens (2 cm sides) were placed inside a prismatic black wooden box with inner dimensions of 110 × 15 × 15 (length × width × depth, in cm). The top of face of these samples was exposed to UV radiation from a tubular lamp with a peak wavelength of 253.7 nm (UV–C) and germicidal action. The lamp had dimensions of 90 × 2.6 (length × diameter, in cm) and a power of 30 W. The surface colour was monitored for up to 16 days (384 h) of UV exposure. Colorimetric analysis was conducted in triplicate using a CR-400 colorimeter (Konica Minolta brand), configured to use a light source (illuminant) D65 and 10° viewing angle, based on the CIELab method.
Statistical analyses
Prior to ANOVA tests, all data, except the chemical and water uptake results, were tested for homogeneity of variances using Levene’s test and for data normality using the Shapiro-Wilk test. If the null hypothesis was rejected, Tukey tests were employed to compare the means. All statistical analyses were conducted at a significance level of 5%.
Results and discussion
Chemical characteristics of the fillers and RPUF
Fourier-transform infrared spectroscopy spectra of all studied fillers and RPUF are shown in Figure 3. Neat RPUF and the RPUF filled with WF presented similarly shaped spectra, which means that the WF did not induce a significant chemical change in the RPUF chemical structure, which was also reported in previous studies.
9
The prominent peaks at 2850 and 2930 cm−1 for the WF are attributed to low-molecular-weight compounds from its extractives, such as methyl and methylene groups.
29
The band around 1510 cm−1 in the WF spectra is related to the aromatic ring of lignin and the peaks at 1360 cm−1 and 1160 cm−1 are related to the presence of carbohydrates.
30
The broad peak at 3380 cm−1 refers to O-H stretching and can be related to adsorbed water molecules
9
This band was attenuated in RPUF filled with NC, indicating that WF has a more hydrophilic character than NC. Infrared spectra of the fillers and RPUF.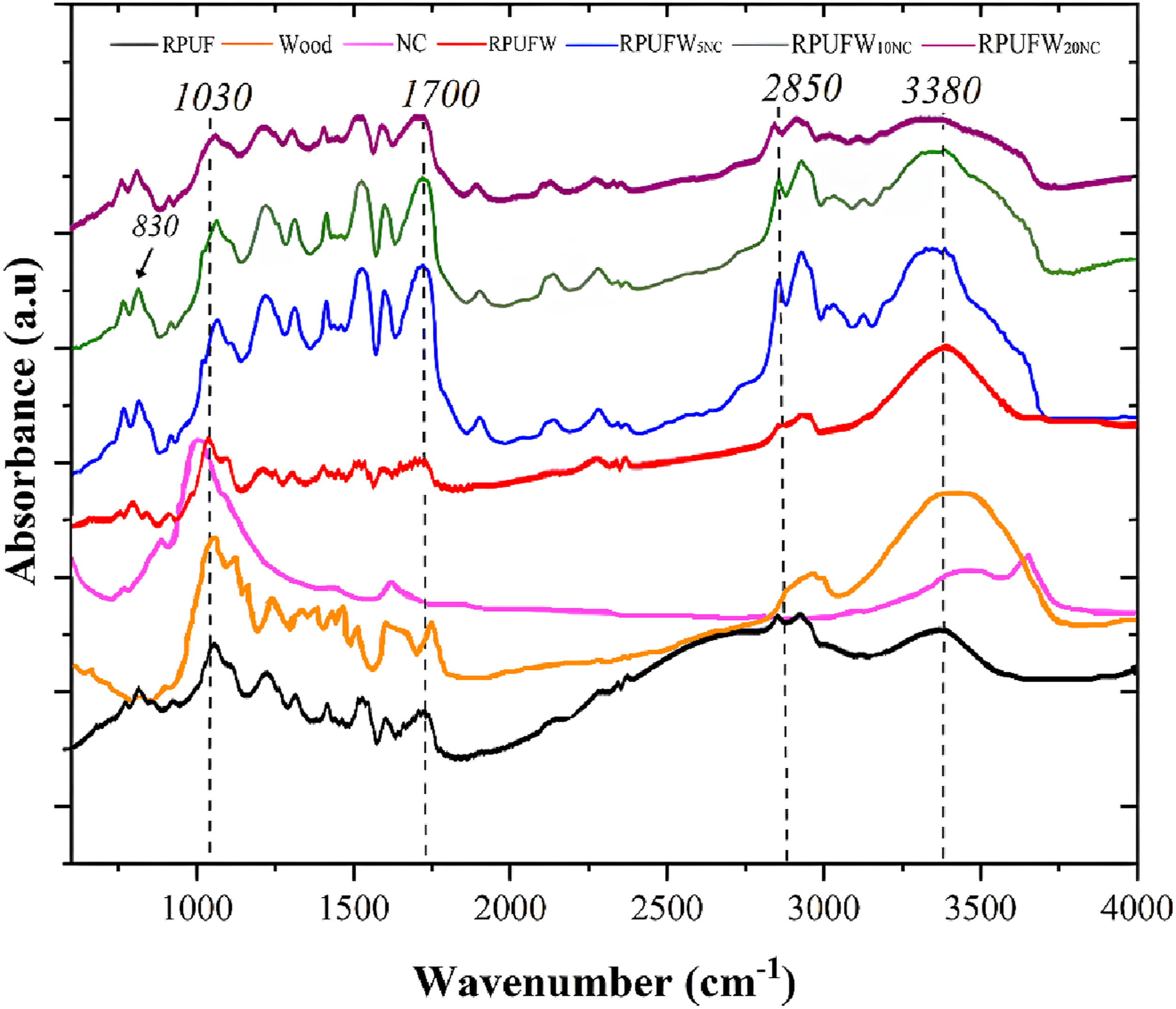
The increase in the 1700 cm−1 peak in the RPUF reinforced with WF is related to the presence of these bands in both materials, which are related to the presence of free carbonyl groups (C–O). 31 After NC insertion, the 830 cm−1 and 1030 cm−1 peaks also increased, which may be associated with symmetric Si-O-Si bonds. 32 Therefore, the increase in these bands for the NC-filled RPUF can be attributed to a chemical reaction between silicon (Si-O-R) groups from the NC and polyurethane, as reported in a previous study. 33 Finally, the prominent peak around 2280 cm−1 in both RPUFW5NC and RPUFW10NC are related to unreacted NCO groups, and the peaks around 1440 cm−1 indicate a carbonate group. 9
Morphological features of the RPUF
Figure 4 displays the SEM images with the morphological characteristics of the produced RPUF. The neat RPUF exhibited a typical cellular structure, mainly composed of closed cells with an elliptical shape oriented in the rise direction, indicating suitable foam formation. The insertion of WF did not affect the nucleation mechanism of the RPUF. This was also reported in previous studies on WF-filled RPUF9,29 and can be attributed to the chemical compatibility between WF and the polyurethane system. SEM images and morphological characteristics of the produced RPUF.
On the other hand, the presence of NC seems to have imparted cell disruptions. The NC probably attached itself to the cell wall, becoming a small nucleation site, weakening and breaking up some cells due to the weak interaction between RPUF and NC particles, impairing the rising processes of the filled foams and leading to the formation of more rounded shaped cells. 34 Even though NC insertion caused detrimental effects on the polymer cell structure, filler agglomerates were not observed.
Solid fillers, especially NC, are known to decrease cell size due to nucleation, which increases the number of cells per volume and, therefore, reduces the mean cell size. 35 The NC filler may also induce a steric hindrance effect that prevent the formation of linkages between OH groups from the polyol and NCO groups from the p-MDI, resulting in a more compact cell structure. The incorporation of NC further increased this effect, possibly due to its large surface area and ability to form hydrogen bonds with the polyol. This effect may also increase viscosity and compromise quality of the mixing of constituents. 36
Density and compressive properties of the RPUF
The decrease in cell size did not impact the apparent density of the RPUF (Figure 5), as reported in previous studies.1,36,37 Indeed, the added WF is a light filler, and was used in small amount, justifying the little impact on density. Density of the RPUF filled with NC was also little affected, ascribed to the low content of NC in relation to the RPUF weight, and the low rise of the filled foams in comparison with the neat foam.
9
Apparent density of the RPUF.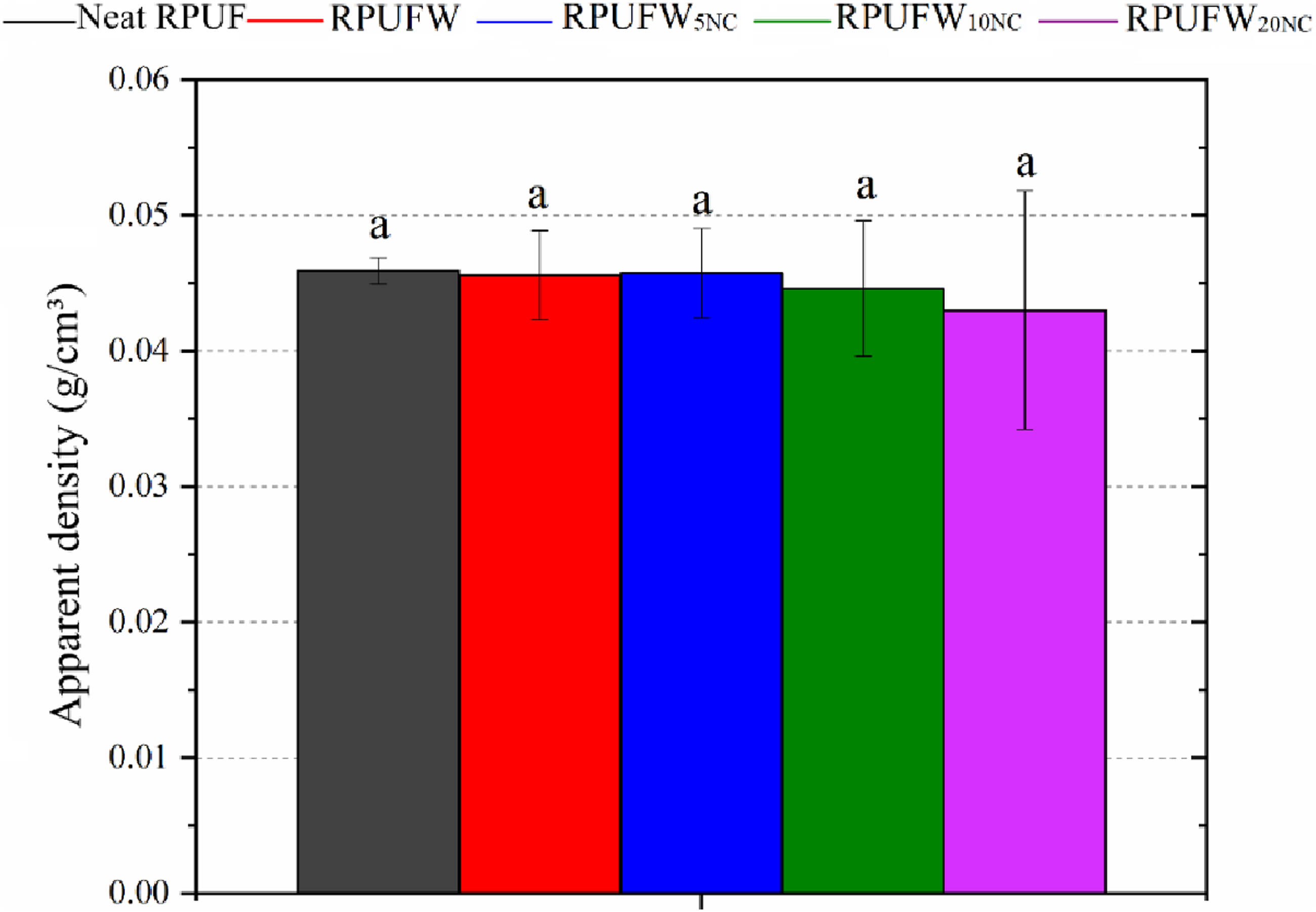
The incorporation of the NC decreased compressive strength of the foam (Figure 6), which can be ascribed to disruptions in cell edges shown in the previous SEM images. In earlier studies, Nayani et al.
38
and Kim el al.
39
also manufactured NC-filled RPUF and justified the decrease in mechanical properties based on the formation of open cells due to the presence of NC attached to the polymer cell wall. This weakens the cell structure, leading to ruptures in several points and resulting in overall lower compressive strength compared to neat RPUF and RPUFW. Nevertheless, all the produced RPUF achieved the minimum compressive strength of 65 kPa required for the use as core in structural panels according to ASTM E1730.
40
Representative stress versus strain curves (a) and compressive strength (b) and modulus (c) of the produced RPUF.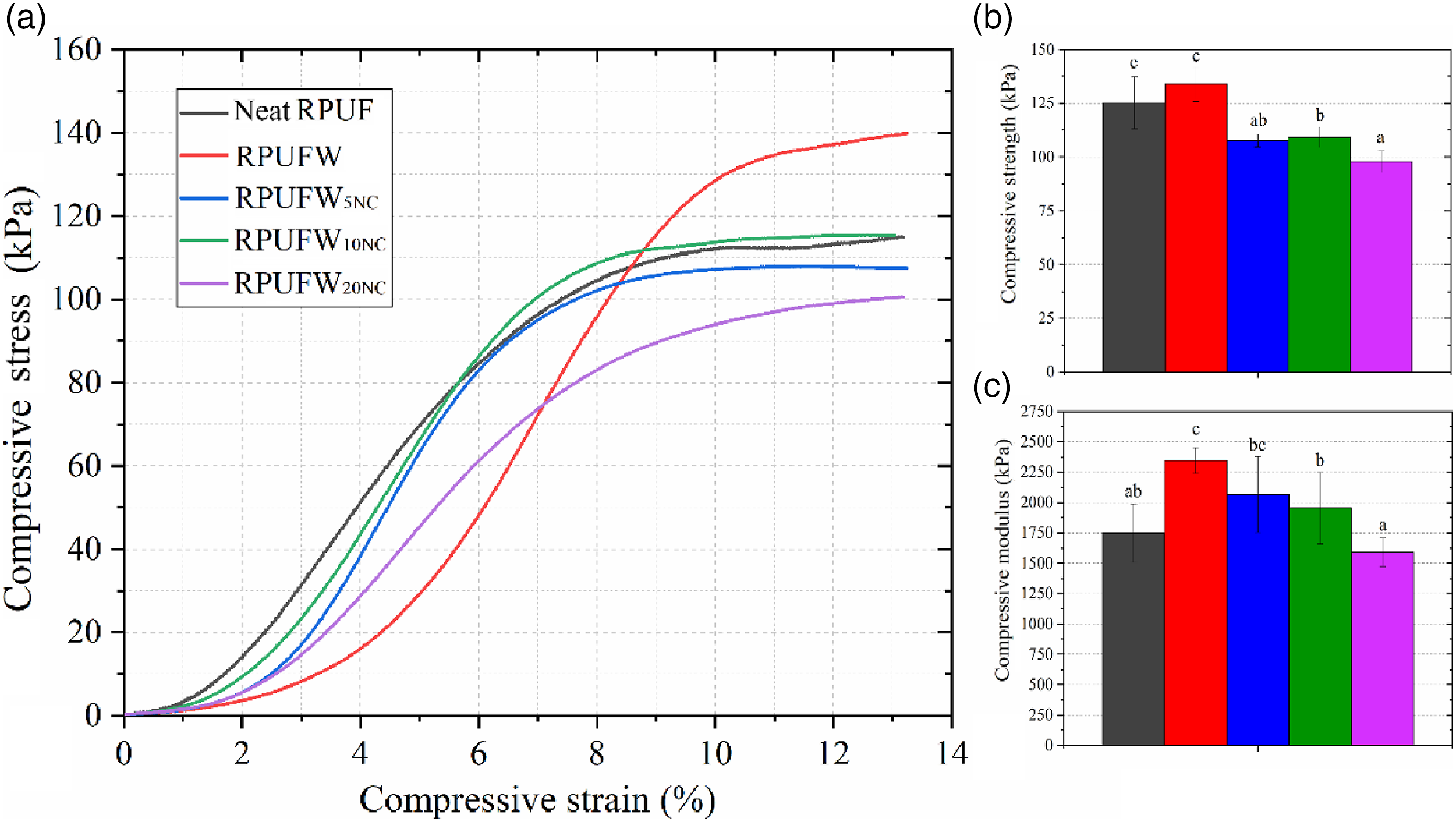
The compressive modulus of the foams increased with the inclusion of WF, and decreased with the inclusion of NC, returning to the original stiffness. According to Mangesh et al., 41 the mechanical response of RPUF loaded in the elastic region is a function of the urethane linkages formed between OH and NCO, which can be affected by the addition of p-MDI or by performing a rising under confinement. The possibility of a filler acting as an actual reinforcement depends on its reactivity in the PU system, which does not usually happen. 42
Thermal and combustion features of the RPUF
The results from the TG analysis are presented in Figure 7 and compiled in Table 2. They support the findings from the FTIR analysis, which indicated that the studied RPUF have similar chemical composition. No significant differences were observed when varying the NC content, perhaps due to the small sample size (in mg) used for this analysis. So, it is not possible to assign greater thermal stability to any of the filled RPUF compared to the neat RPUF. TG curves for the fillers and the produced RPUF. Main TG events for the fillers and the produced RPUF.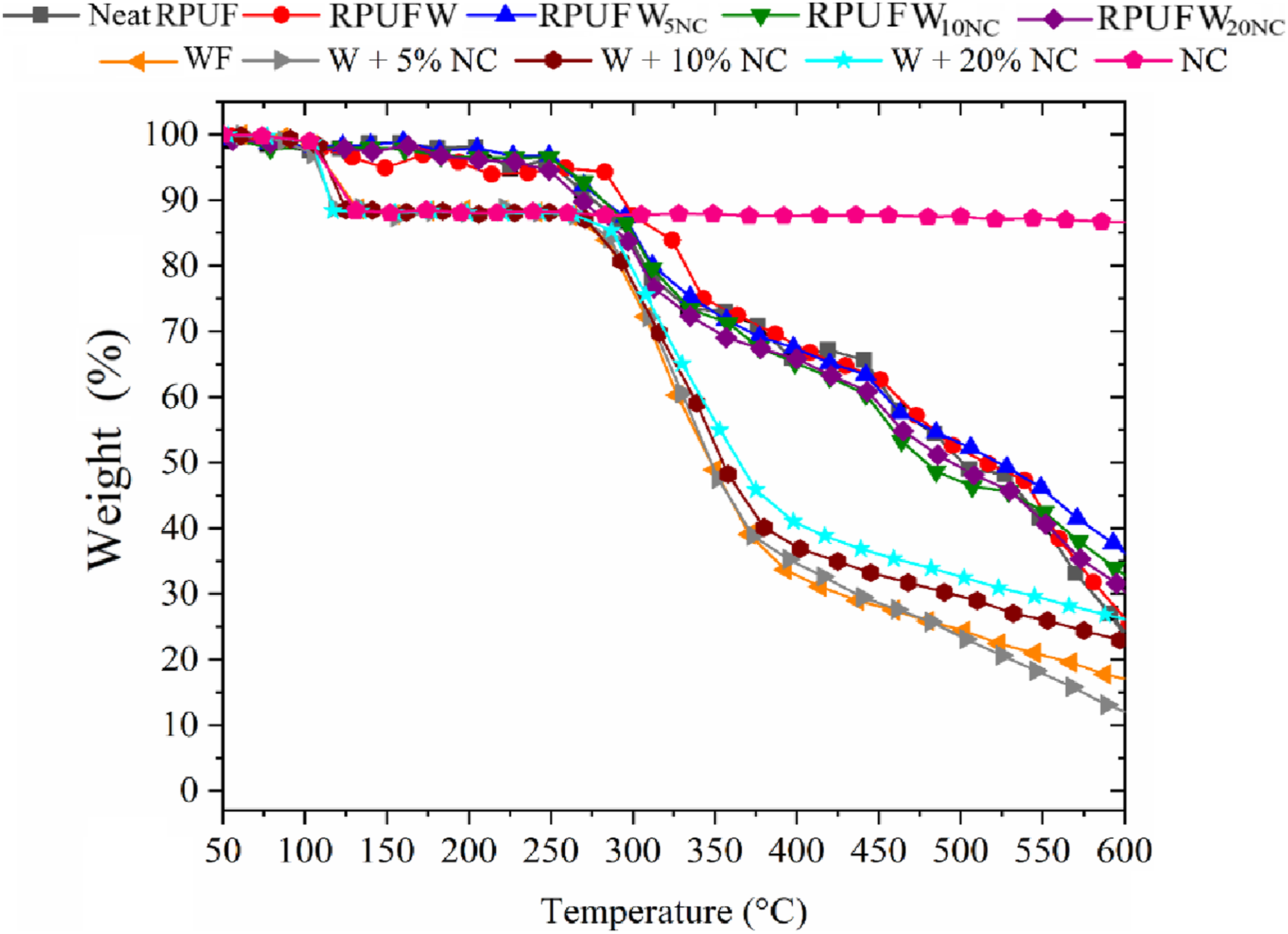
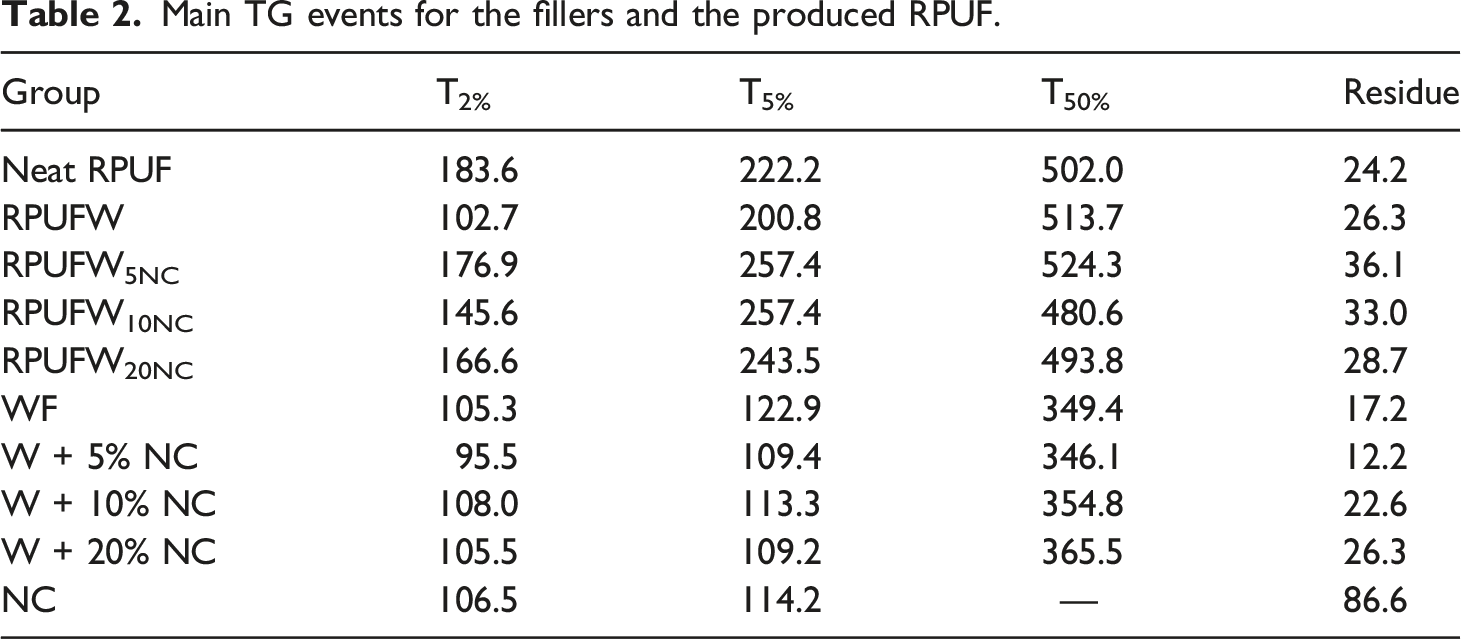
In Table 2, the main thermal events were defined as T2%, T5%, and T50% (the temperatures at which a weight loss of, respectively, 2%, 5% and 50% occurred). The T2% is associated with the release of moisture or low molecular weight substances, 43 and T5% and T50% are ascribed to the degradation of urethane bonds and urea groups, respectively. 44 The results attributed to the waste did not show a clear pattern, probably due to the low percentage of NC incorporated.
In general, the incorporation of NC increased the fire resistance of the RPUF, which can be explained by a formation of a barrier by the NC that hinders heat transfer and flame spreading, protecting the underlying layers from further burning, as discussed in previous studies.12,45 This phenomenon was confirmed by the flammability tests performed (Figure 8). The NC addition delayed the ignition time from 20 s to 35–40 s. It also reduced the consumption of RPUF, and larger residues were obtained for the RPUF filled with NC in comparison with the neat RPUF. Photographs following the flammability tests of the produced RPUF.
Colorimetric features and photodegradation resistance of the RPUF
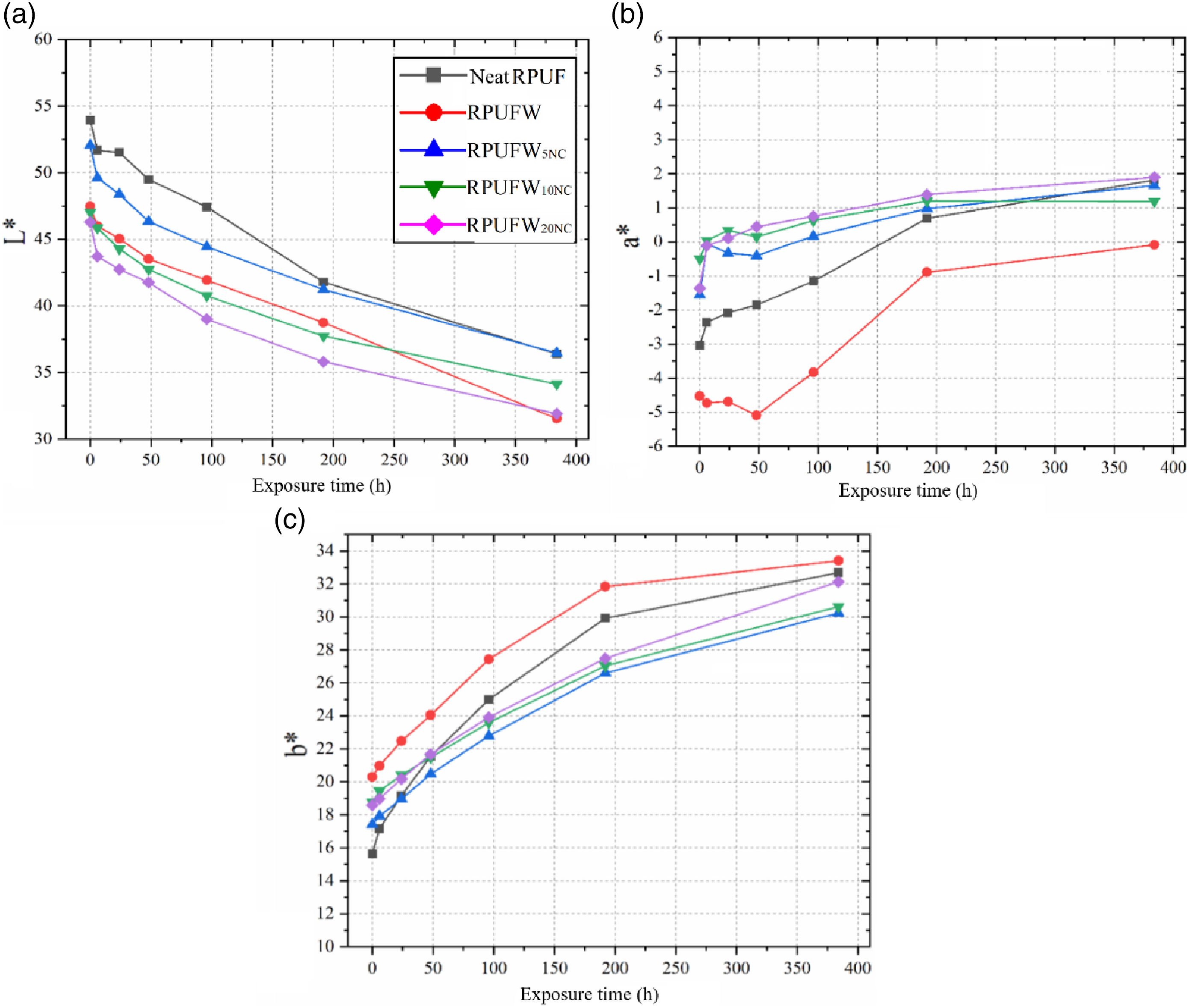
Figures 9 and 10 show the change in aesthetical features of the RPUF with UV exposure. The material degradation was marked by decrease in L* accompanied by the increase in a* and b*. The latter occurred until around 200 h, followed by the stabilization in these values. The changes in a* and b* justify the orange shades seen in the photographs. According to Rus and et al.,
46
this colour change can be explained by the formation of quinones due to the oxidation of aromatic groups of the polyurethane. Photographs of the produced RPUF exposed to continuous UV radiation. Evolution of the colorimetric parameters of the produced RPUF with continuous UV radiation. Where: L* is brightness; a* is green-red coordinate; b* is blue-yellow coordinate.
Conclusions
The results of this work indicate that the addition of WF and NC improved flammability and resistance to photodegradation characteristics of the polyurethane foams, without compromising other properties of the RPUF. The SEM images showed that the NC caused some disruption of the cellular structure, but the combination of fillers was able to yield similar density and compressive stiffness in relation to the neat polyurethane foam. The fillers brought a detrimental effect on compressive strength, although all RPUF reached the minimum demanded by ASTM E1730 for structural parts.
The introduction of WF and NC fillers did not alter the TG curves of the RPUF up to 500°C, but an increase in the TG residue was observed with the addition of NC. This is consistent with the improvement in flammability obtained in this study with the addition of NC fillers. In addition, the RPUF filled with NC exhibited more stable colorimetric properties compared to the unfilled foam, suggesting an improved resistance to photodegradation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Financing Code 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (301758/2019-0).
