Abstract
The use of carbamate to foam epoxy depends significantly on the precured modulus to stabilize the cellular structure. The optimum precured modulus is developed from the reaction of epoxy resin and the neat amine. The selection of the neat amine relies on its reaction temperature with epoxy, which is required to be below the decomposition temperature of carbamate. This study investigates the effect of three different neat amines on the rheological behavior of foaming epoxy-carbamate-amine. They are bisphenol-A diglycidyl ether epoxy (DGEBA), isophorone diamine carbamate (IDPA.CO2), N-aminoethylpiperazine (AEP), 2,4-Diamino-1-methyl-cyclohexan (DMC) and isophorone diamine (IDPA). The mixtures of DGEBA-amine-carbamate are filled in 25% and 75% of the volume of a closed mold. Precuring is carried out at 60°C for 2 h. The foaming and complete curing are conducted at 180°C for 1 h. Having H-active at piperazine, AEP reacts with DGEBA faster and develops a higher precured modulus compared to DMC and IDPA. It is important to note that DGEBA-AEP-IDPA.CO2 exhibits viscoelastic behavior beyond 138°C, seen by its rheological storage modulus lower than loss modulus and its tan delta larger than 1. The reaction between DGEBA and the H-active piperazine of AEP leads only to linear linkage and is unable to further crosslink compared to the primary amine (-NH2). This results in a lower glass transition temperature Tg of DGEBA-AEP-IPDA.CO2. The effect of amine on foaming is more obviously at 25% filling level. DGEBA-AEP-IPDA.CO2 has more spherical and homogeneous cellular structure and the density of 285 kg/m3. Having quite similar chemical structure, both DGEBA-DMC-IPDA.CO2 and DGEBA-IPDA-IPDA.CO2 produce the epoxy foams having cell-interconnection and coalescence; their densities are also similar 301 kg/m3 and 305 kg/m3, respectively. All the foams are closed-cell at 75% of filling level. The cell morphologies are well reflecting the foaming modulus and tan delta behavior.
Introduction
Epoxy foam can be produced from a mixture of resin and chemical blowing agents [1-3] and by solid-state foaming with physical blowing agents [4-5], with chemical blowing agents [6] or even without blowing agent [7]. Epoxy foam is also fabricated by applying an aqueous emulsion-template [7]. Besides, epoxy syntactic foams are produced by the addition of microballoons [8-10]. Compared to foaming techniques, epoxy-carbamate foaming offers a better way to control the density and the cellular structure of the final epoxy foam [11-13]. The carbamate has a dual function as a foaming agent and curing agent for the epoxy resin system. The applied IPDA-carbamate in this study decomposes when heated above its decomposition temperature of 70°C [14]. The IPDA-carbamate revives the original amine and CO2 as shown in Scheme 1. The released CO2 foams the epoxy matrix, while the revived amine reacts with the epoxy resin to stabilize the cellular structure and achieve the fully three-dimensional (3D) crosslinking networks. This dual function accounts for the advantage of carbamate in foaming epoxy resin compared to other chemical blowing agents.
The effectiveness of foaming is highly dependent on the viscosity or modulus of the epoxy matrix when the gas phase is generated. Too low a viscosity will cause the growing cells to coalesce. Conversely, too high a modulus restricts cells growth and eventually leads to cracking. Therefore, an optimum viscosity or modulus is important to develop and support foaming. The best practice for developing a suitable foaming modulus is to pre-react the epoxy resin with a free neat amine. This process generates a precured network having large molecular oligomer chains/networks compared to the monomer structure of the epoxy resin. The precured network is therefore able to hold/trap the gas phase and grow with it to build the cellular structure in the epoxy matrix. In this advanced technique, carbamate proves to be the best candidate for blending with other neat amines. However, it is important to note that the precured reaction must be conducted below 70°C to ensure that IPDA-carbamate does not decompose. For this concern, the low molecular weight amines are the appropriate candidates.
There are two neat amines were selected to compare the foaming behavior with isophorone diamine (IDPA) for this study. They are N-aminoethylpiperazine (AEP), and 2,4-Diamino-1-methyl-cyclohexan (DMC). Their chemical structures are shown in Scheme 2. The active hydrogen (H-active) in the amine groups of the three amines plays the key role in the reaction with bisphenol-A diglycidyl ether epoxide (DGEBA). AEP has two H-active at the primary amine and one H-active at the piperazine. DMC has four H-actives from the two primary amines directly linked to cyclohexane. IPDA also has four H-actives from the two primary amines, but only one primary amine is directly linked to cyclohexane. Based on the chemical structure, the three amines react with DGEBA at different rates [11]. In this manuscript, the influence of the amines on the rheological behavior and properties of the final epoxy foams is investigated.
Experimental
Materials
Epoxy resin DGEBA (DER331) was purchased from Huntsman, Texas, United States. The resin DER 331 has an epoxide equivalent weight (EEW) of 182 g/eq, and a viscosity at 25°C of 11,000–14,000 mPas. The synthesized carbamate (IPDA.CO2) has a Mw of 214.3 g/mol, an amino hydrogen equivalent (AHEW) of 53.6 g/mol and a density of 1.26 g/cm3. The decomposition temperature is about 70°C and the CO2 content was 21 wt% of carbamate [11]. AEP (Sigma Aldrich, St. Louis, Missouri, United States) has a Mw of 129 g/mol and an AHEW of 43.1 g/mol. DMC (Aradur XB50021 B, Huntsman, Texas, United States) has a Mw of 128,22 g/mol and an AHEW of 32,1 g/mol. IPDA (Aradur 22962, Huntsman, Texas, United States) has a Mw of 170.3 g/mol, an AHEW of 42.6 g/mol. They were used as received.
Methods
Formulation and the filling weight of 25% and 75% of mold volume.
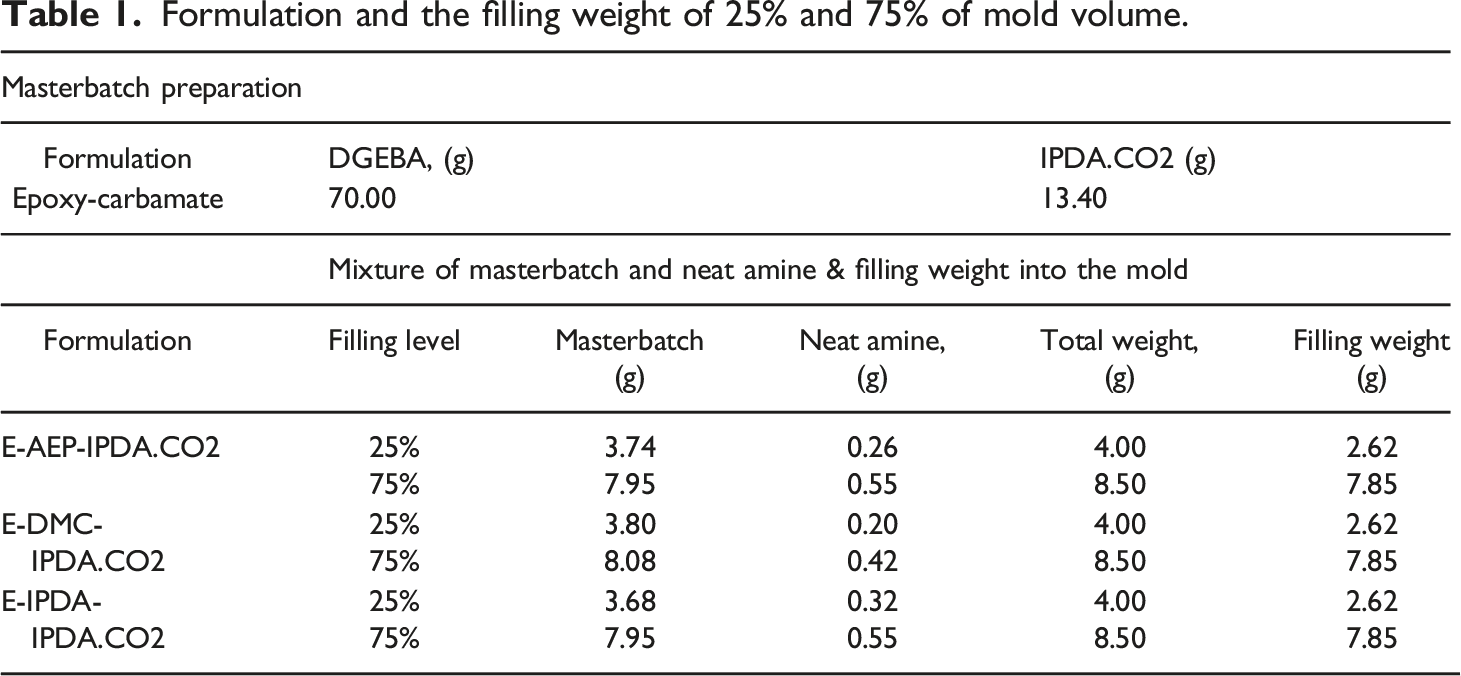
The mixture of masterbatch and neat amine of each formula was prepared in 4 g for 25% filling level and 8.5 g for 75% filling level. The weights were prepared in excess to eliminate the mixture adhering in the PP cups. The masterbatch in the PP cup was taken out from the refrigerator and kept at room temperature of 25°C for 2 h. The required neat amine was weighed into the masterbatch PP cup. They were mixed using a DAC 150.1 FVZ bicentrifugal mixer (Hauschild Engineering, Hamm, Germany) at 3000 rpm for one minute.
An aluminum mold manufactured in-house at the mechanical workshop with the inner rectangular cuboid of dimensions 3 cm × 3 cm x 1 cm was used. The mold volume is 9 cm3. The mold was placed on the scale and the actual weight was added into the mold according to Table 1. Assumed that the three compounds have a similar density. The filling weight is aimed to achieve the density 290 kg/m3 (25% filling level) and 870 kg/m3 (75% filling level). The density of the mixture is estimated based on the density of DGEBA, carbamate and neat amines.
The mold with the resin mixture was placed in a preheated PW 20 hot press (P/O/Weber, Remshalden, Germany). The holding force of the hot press was 10 kN. The resin mixture was precured at 60°C for 2 h. The hot press was heated to 180°C with an effective heating ramp of 7°C/min to foam and further cure the samples for 60 min. The final hot press force was kept at 15–20 kN. Finally, the mold was placed in the cooling press at 10 kN for 10 min before the foam samples were released. Two epoxy foams were prepared for each formula.
Characterization and Testing
3. Results and Discussion
3.1 Propose the reaction rate of epoxide groups with the original H-actives and the newly formed H-actives
Theoretically, the primary amine will convert to secondary amine after reacting with the epoxide group. Since only 35% of H-active of the neat amines are available, the epoxide groups will react with the newly formed dimer. As shown in Scheme 3, the reaction of epoxide group and AEP forms a dimer with an H-active at the secondary amine and an H-active at the piperazine. The epoxide group could react more easily with the H-active at the piperazine [11]. It is important to note that for the same amount of 35% AHEW, the number of AEP molecules (having 3 H-active) are more than that of DMC and IPDA (having 4 H-active). In the case of DMC and IDPA, the H-active at primary amine of the neat amine (rate E&D1, rate E&IP1) reacts more readily with the H-active at the active at the primary amine of the newly formed dimer (rate E&D2, rate E&IP2). It is possible that the H-active at the primary amine of IPDA react faster than H-active at the primary amine at the cyclohexane ring of DMC. However, it is noted that the dimer E&D has less steric hindrance than the dimer E&IP. Hence, rate E&D2 is faster than rate rate E&IP2. This proposed hypothesis is evident from the rheological characteristic.
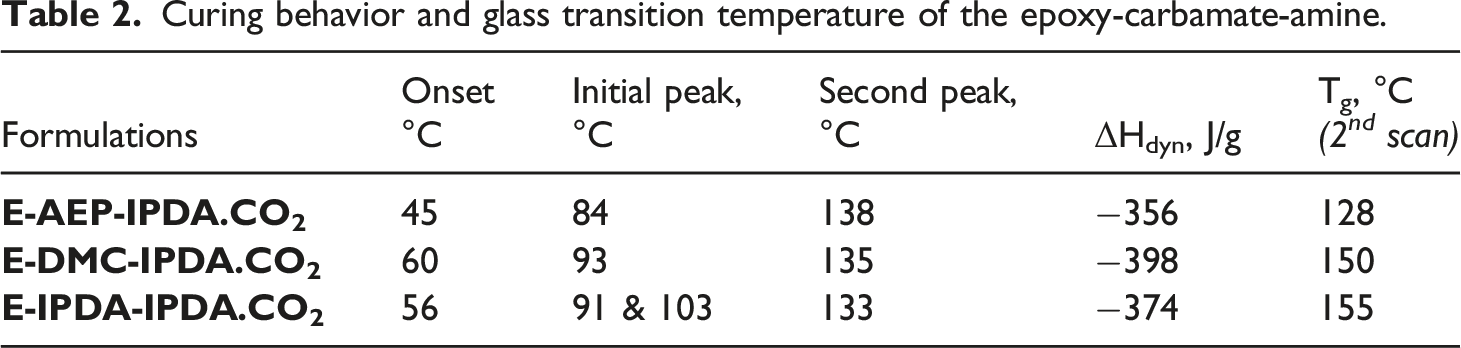
3.2 Curing enthalpy and glass transition temperature from DSC measurements
The precuring and curing behaviors of the epoxy-carbamate-amine measured by DSC are shown in Fig. 1a. The glass transition behavior of the epoxy foams is present in Fig. 1b. The peak temperature and the curing enthalpy were determined and listed in Table 2. The DSC curves show that the epoxy groups start to react with AEP at 45°C, with DMC at 60°C, and with IPDA at 56°C (Fig. 1a and Table 2). The initial temperature peaks are originated to the reaction of epoxy groups and amines. These results are consistent with the rheological data and the discussion on reaction scheme 3. The reaction of DGEBA – AEP showed the fastest first temperature peak at 84°C. The reaction of DGEBA – DMC exhibited the initial temperature peak at 93°C. The reaction of DGEBA – IPDA showed two temperature peaks. The small peak at 91°C could represent the reaction of epoxy group and IPDA to form dimers, while the large peak at 103°C could exhibit the reaction of dimers and higher oligomers. The board and deep peaks around 133°C – 138°C correlated to the reaction of oligomers and the revived IPDA. As expected, the total curing enthalpy was lowest for the formulation of E-AEP-IPDA.CO2, followed by the second value of E-IPDA-IPDA.CO2 and the highest value of E-DMC-IPDA.CO2. The steric hindrance of the amine directly bound to the cyclohexane ring of DMC required the highest energy to react. The Tg of the epoxy foams was determined from the second DSC scan and reported in Table 2. Comparison with the Tg determined by DMA is discussed in Section 3.6. (a) the first DSC scan shows the precuring and curing behaviors the epoxy-carbamate-amine and (b) the glass transition behavior from the 2nd DSC scan. Curing behavior and glass transition temperature of the epoxy-carbamate-amine.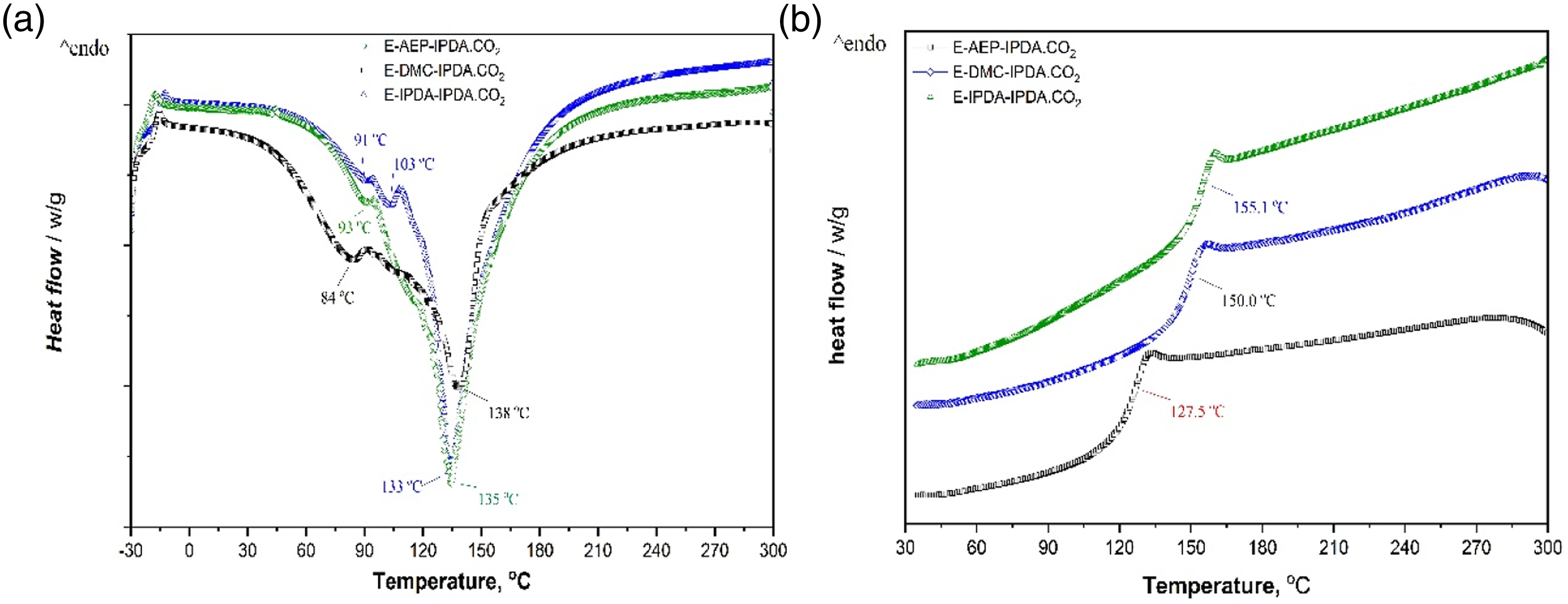
3.3 The effect of amine on the rheological behavior of epoxy-carbamate-amine
The viscosities of epoxy mixtures precured at 60°C for 2 h and heated to 180°C and maintained for 1 h are shown in Figure 2. Initially, the viscosities decrease to their lowest value due to the heat and then increase due to the reaction of DGEBA and amines, producing higher molecular weight oligomers. In the case of E-AEP-IPDA.CO2, the viscosity increases at a temperature of 45°C after 1.6 min; the viscosity increases significantly and reaches the plateau. This confirms the fact that the AEP reacts faster than DMC and IPDA, because AEP has more molecules as discussed above. For DMC and IDPA, the increase in viscosity started after 16.8 min and 10.8 min, respectively. Although, the reaction started later in DMC than in IDPA, DMA developed higher precured viscosity compared to IDPA. It is because dimer, trimer and oligomer of DGEBA-DMC have lower steric hindrance and react faster as discussed at scheme 3. After pre-curing and heating to 180°C, all the compounds start foaming and show a peak in the reduction of their viscosities. In fact, the viscosity results do not provide information about the gelation and foaming, which can be verified from the storage and loss modulus and the tan delta of the rheological behavior. The amine affects the viscosity of the epoxy-carbamate-amine with precuring and foaming and curing time, process 1: heating from 30°C to 60°C, process 2: holding at 60°C for 2h, process 3: heating from 60°C to 180°C, process 4: holding at 180°C for 1h.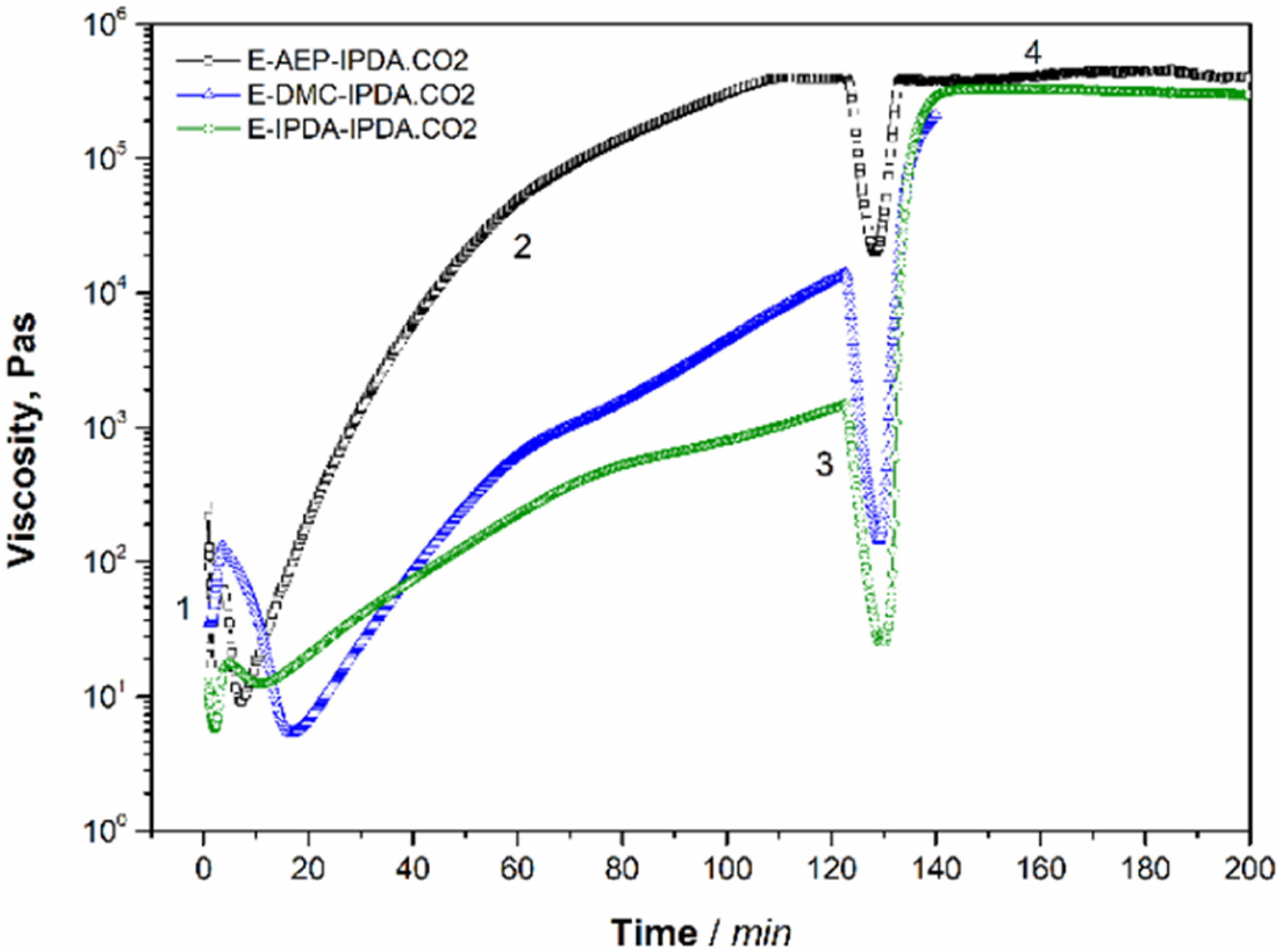
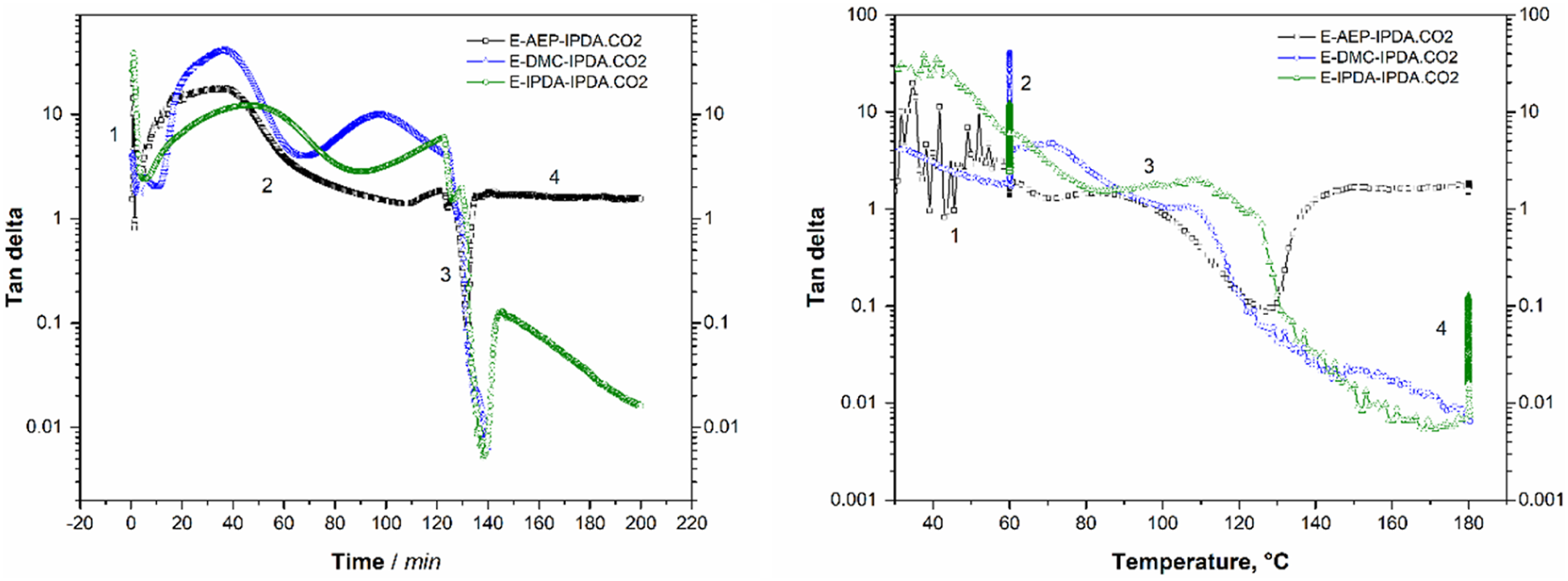
Figure 3 shows the effect of the amine on the behavior of the storage and loss modulus as a function of time (Fig. 3a) and temperature (Fig. 3b). This is totally different from the rheological characteristic of DGEBA + 100% IPDA.CO2 as shown in Fig. 10 at appendix. The gel point is determined as the intercept between the storage modulus and the loss modulus [5, 16]. All three compounds do not gel after 2 h of pre-curing at 60°C (Fig. 3a). Gelation occurs during heating from 60°C to 180°C. Specifically, gelation occurs at 99°C for E-AEP-IPDA.CO2, at 109°C for E-DMC-IPDA.CO2 and at 121°C for E-IPDA-IPDA.CO2 (Fig. 3b). The most important distinguishing features is E-AEP-IPDA.CO2, whose storage modulus after foaming is lower than its loss modulus. This means that E-AEP-IPDA.CO2 has a tan delta value higher than 1 and behaves viscoelastic (Fig. 4). The viscoelastic behavior could allow a different foaming of E-AEP-IPDA.CO2 compared to the elastic behavior of E-DMC-IPDA.CO2 and E-IPDA-IPDA.CO2. This assumption will be discussed in section 3.5 on cellular morphology. The amine affects the gel point as the interception of storage modulus (solid line) and loss modulus (dash line) of the epoxy-carbamate-amine, a) with time, b) with temperature; process 1: heating from 30°C to 60°C, process 2: holding at 60°C for 2h, process 3: heating from 60°C to 180°C, process 4: holding at 180°C for 1h. Tan delta of E-AEP-IPDA.CO2 is greater than 1, showing the viscoelastic behavior during foaming and curing with rev-IDPA, while tan delta of E-DMC-IPDA.CO2 and E-IPDA-IPDA.CO2 is less than 1, process 1: heating from 30°C to 60°C, process 2: holding at 60°C for 2h, process 3: heating from 60°C to 180°C, process 4: holding at 180°C for 1h.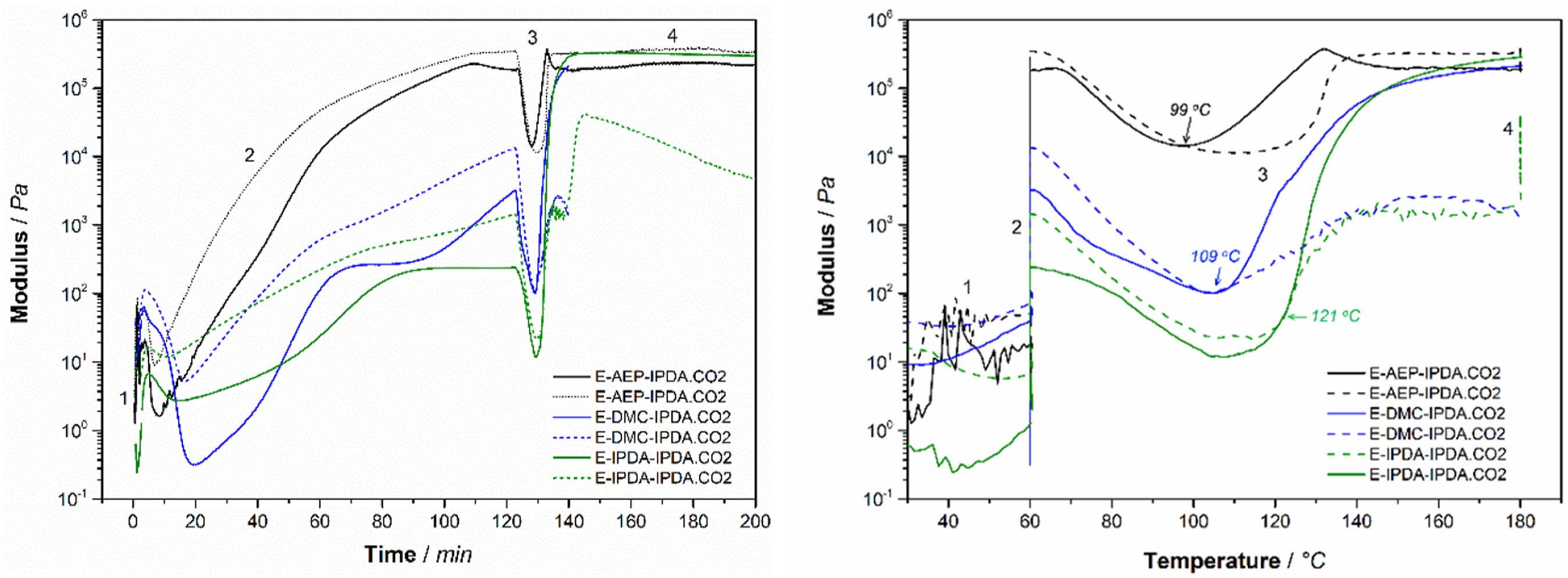
3.4 Surface and cross-section
The top surface and cross-section of epoxy foams are shown in Fig. 5. As expected, only the E-AEP-IPDA.CO2 foam has a fully expanding surface at 25% filling level, and its cellular structure is more homogeneous. Both E-DMC-IPDA.CO2 and E-IPDA-IPDA.CO2 do not have fully expanding surfaces (this is more evident in E-IPDA-IPDA.CO2). In addition, their cellular structures are less homogeneous and have many large cells. This reflects well the rheological results and clearly present the influence of the chemical structure of the amine hardener on the epoxy-carbamate foam. The top surface and cross-section of epoxy foams at 25% and 75% filling level.
At 75% filling level, the color of E-AEP-IPDA.CO2 becomes more yellowish from the core to the outer foam. This shows the inhomogeneous expansion caused by the 75% as an excess fill level. The color of E-DMC-IPDA.CO2 and E-IPDA-IPDA.CO2 is whiter and more uniform for the whole cross-section.
3.5 Cellular morphology of epoxy foams
Density and cell size of epoxy foam at 25% and 75% filling level.
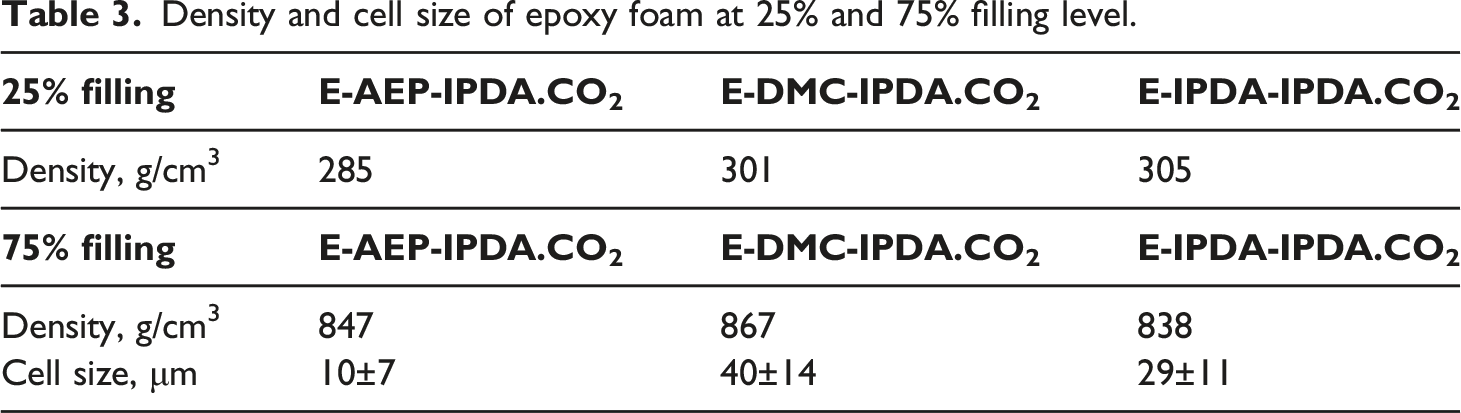

All epoxy foams at 25% filling level a) E-AEP-IPDA.CO2 with spherical cell and less interconnection, b) E-DMC-IPDA.CO2 and c) E-IPDA-IPDA.CO2 with inhomogeneous cell and more cell-interconnection.
These features illustrate the foaming mechanism and cell stabilization. The cell stabilization depends on the modulus developed from the reaction of precured-DGEBA-amine networks and rev-IDPA (which is released from carbamate). As shown in Fig. 7, the storage and loss moduli of E-AEP-IPDA.CO2 are so high that fewer cell-interconnection occurs. E-IPDA-IPDA.CO2 behaves more elastically after foaming, with its storage modulus really higher than its loss modulus. This is a response from the crosslinking by primary amine directly attached to cyclohexane of DMC (Scheme 3). In the case of E-IPDA-IPDA.CO2, the storage and loss moduli equalize for a period of time. This reflects well the chemical structure of IDPA with three methyl groups and one methyl-primary amine attached to cyclohexane (Scheme 3.) Therefore, more and larger cell-interconnections are observed in E-IPDA-IPDA.CO2 as shown in Fig. 6c. The foaming mechanism is observable on the behavior of the storage, G’ (solid line) and loss modulus G” (dash line). G’ < G″ in E-AEP-IPDA.CO2 while G’ > G″ in E-DMC-IPDA.CO2 and G’ ≥ G’ in E-AEP-IPDA.CO2.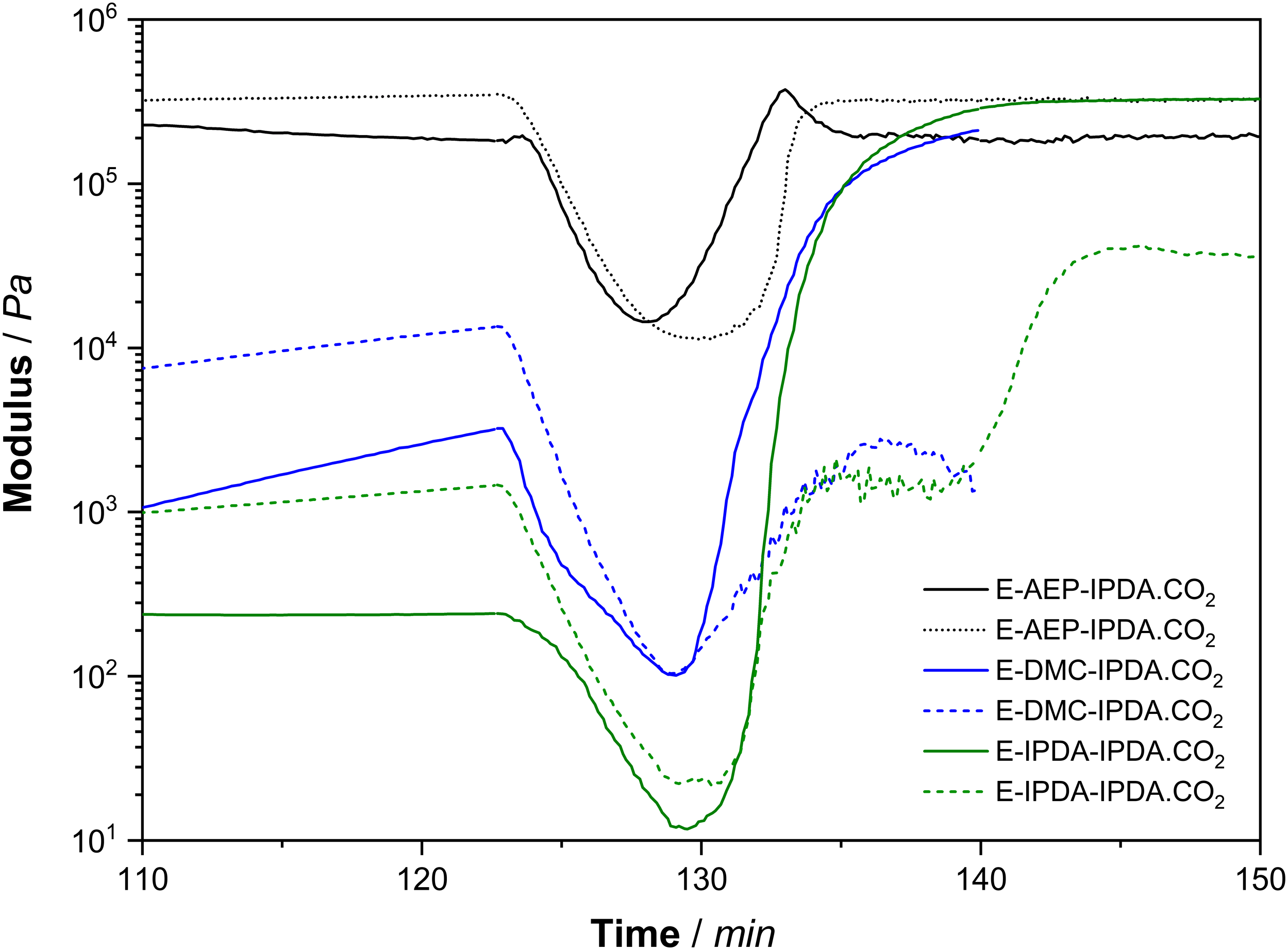
At 75% filling level, all epoxy foams are closed-cell. This is because the expanding volume is limited (only 25% of the volume is free in the mold). Since E-AEP-IPDA.CO2 behaves viscoelastic during foaming (tan delta > 1), it supports the nucleation of CO2 gas, resulting in numerous micronscopic cells as shown in Fig. 8a and 8d. Larger cell size was observed in E-DMC-IPDA.CO2 shows larger cell size (Fig. 8b) and cell-flocculation at 400X magnification (Fig. 8e). E-IPDA-IPDA.CO2 (Fig. 8c) exhibited slightly smaller cell size compared to E-DMC-IPDA.CO2 (Fig. 8b). Cell-flocculation is also observed in E-IPDA-IPDA.CO2 and a group of micron-cell in coalescence as shown in Fig. 8f. These features proved the lowest storage and loss modulus of 30IPDA-70IP.CO2 during foaming as displayed in Fig. 7. All epoxy foams at 75% filling level: 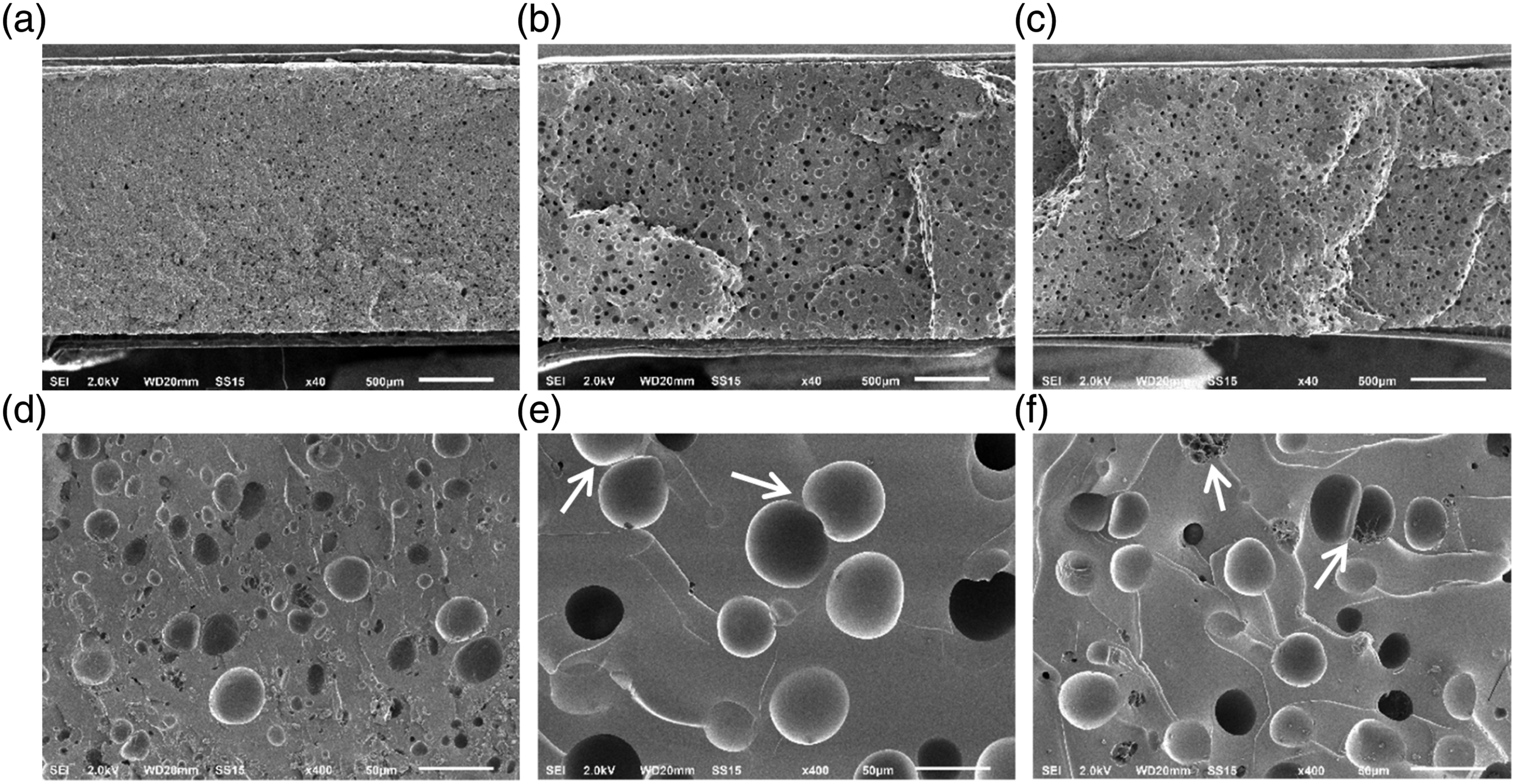
3.6 Dynamic mechanical properties of epoxy foams
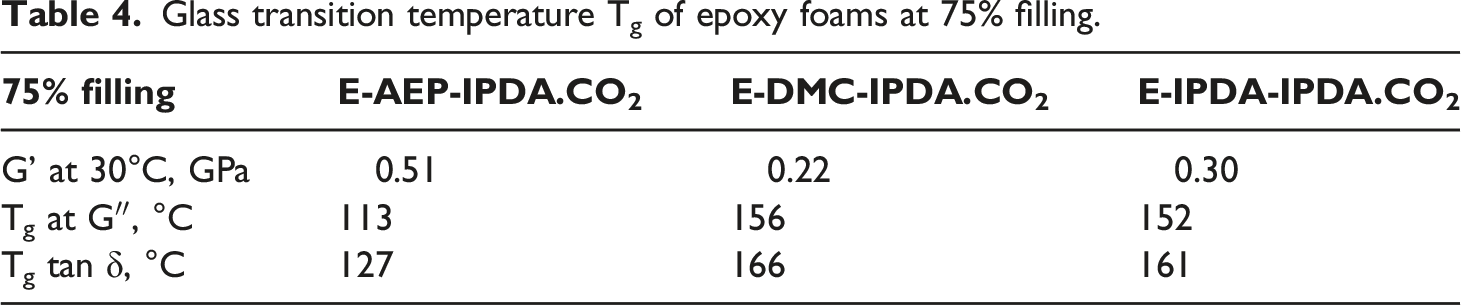
Glass transition temperature Tg of epoxy foams at 75% filling.
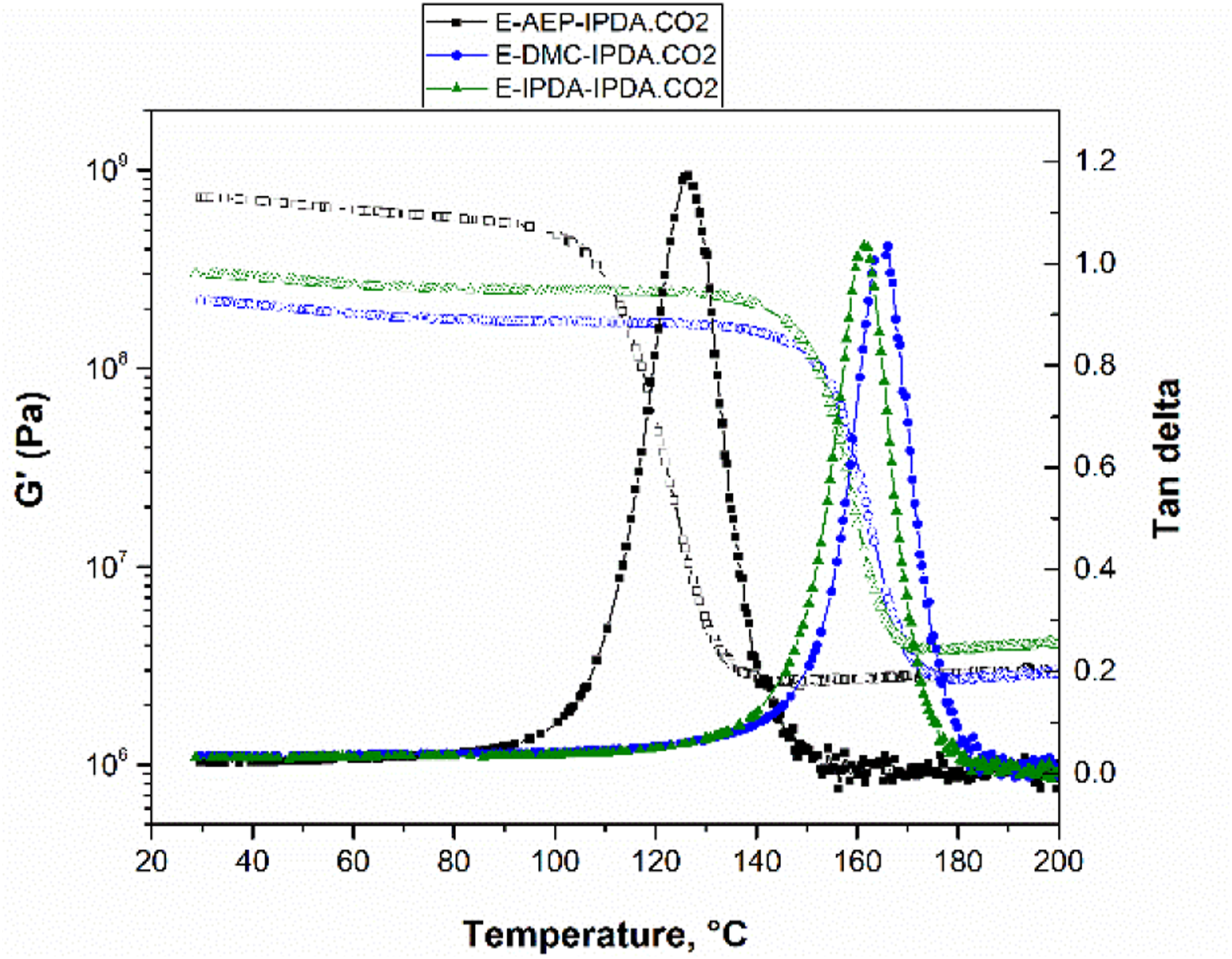
The effect of neat amine on the storage modulus and tan delta of epoxy foams.
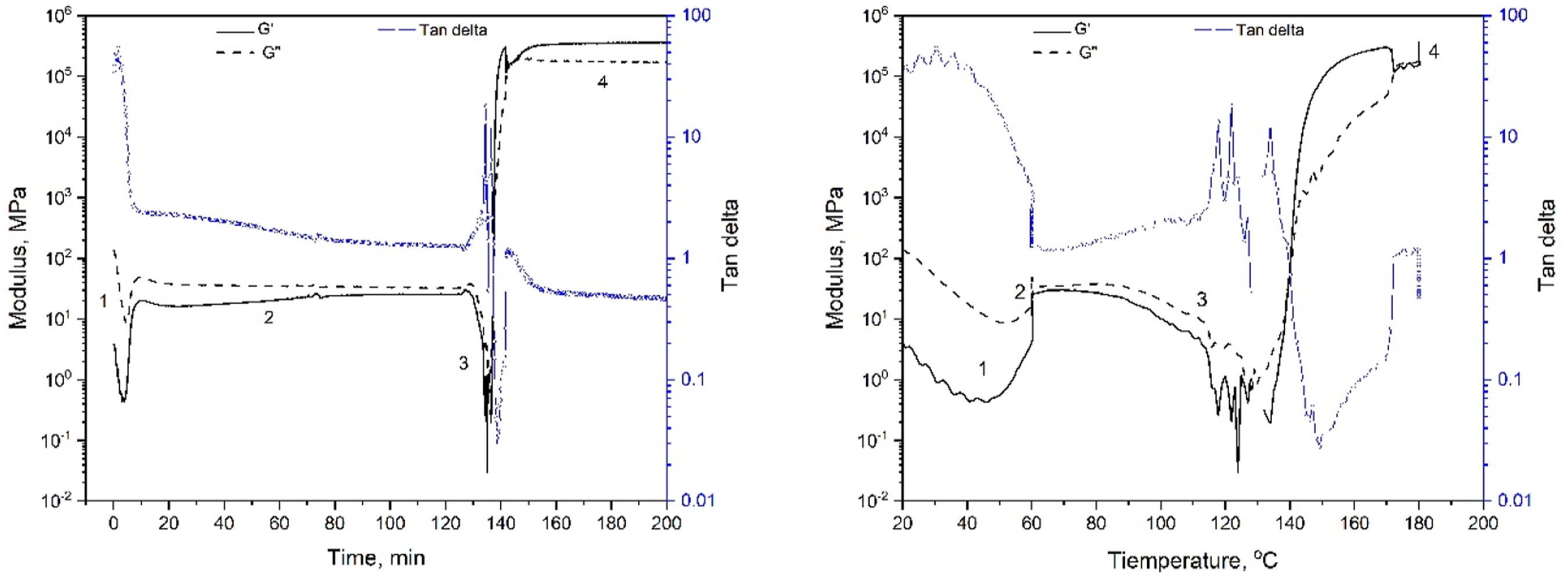
The rheology characteristic of DGEBA + 100% IPDA.CO2 (0% neat amine), a) time, b) temperature exhibits no reaction during precuring at 60°C for 2 h, process 1: heating from 30°C to 60°C, process 2: holding at 60°C for 2h, process 3: heating from 60°C to 180°C, process 4: holding at 180°C for 1h.
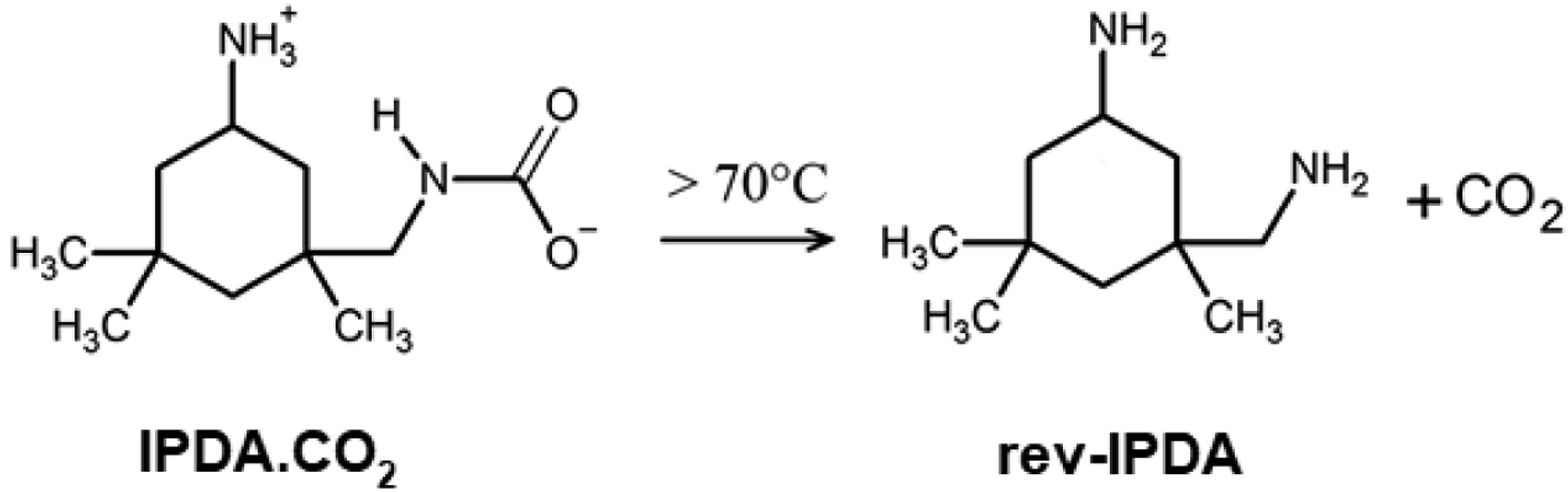
Decomposition reaction of IDPA-carbamate (IDPA.CO2) to form a revived IPDA (rev-IPDA) and CO2 gas [142].
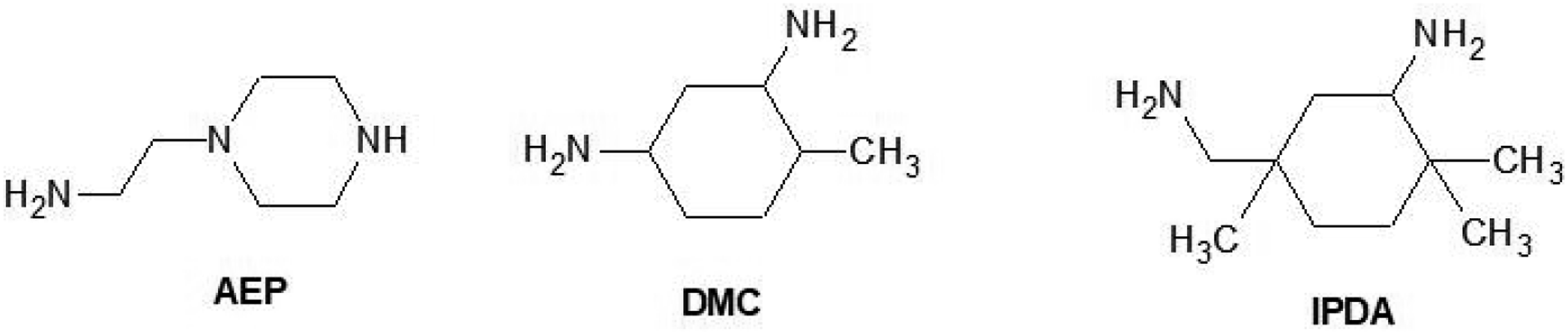
Chemical structure of N-aminoethylpiperazine (AEP), 2,4-Diamino-1-methyl-cyclohexane (DMC) and isophorone diamine (IDPA).
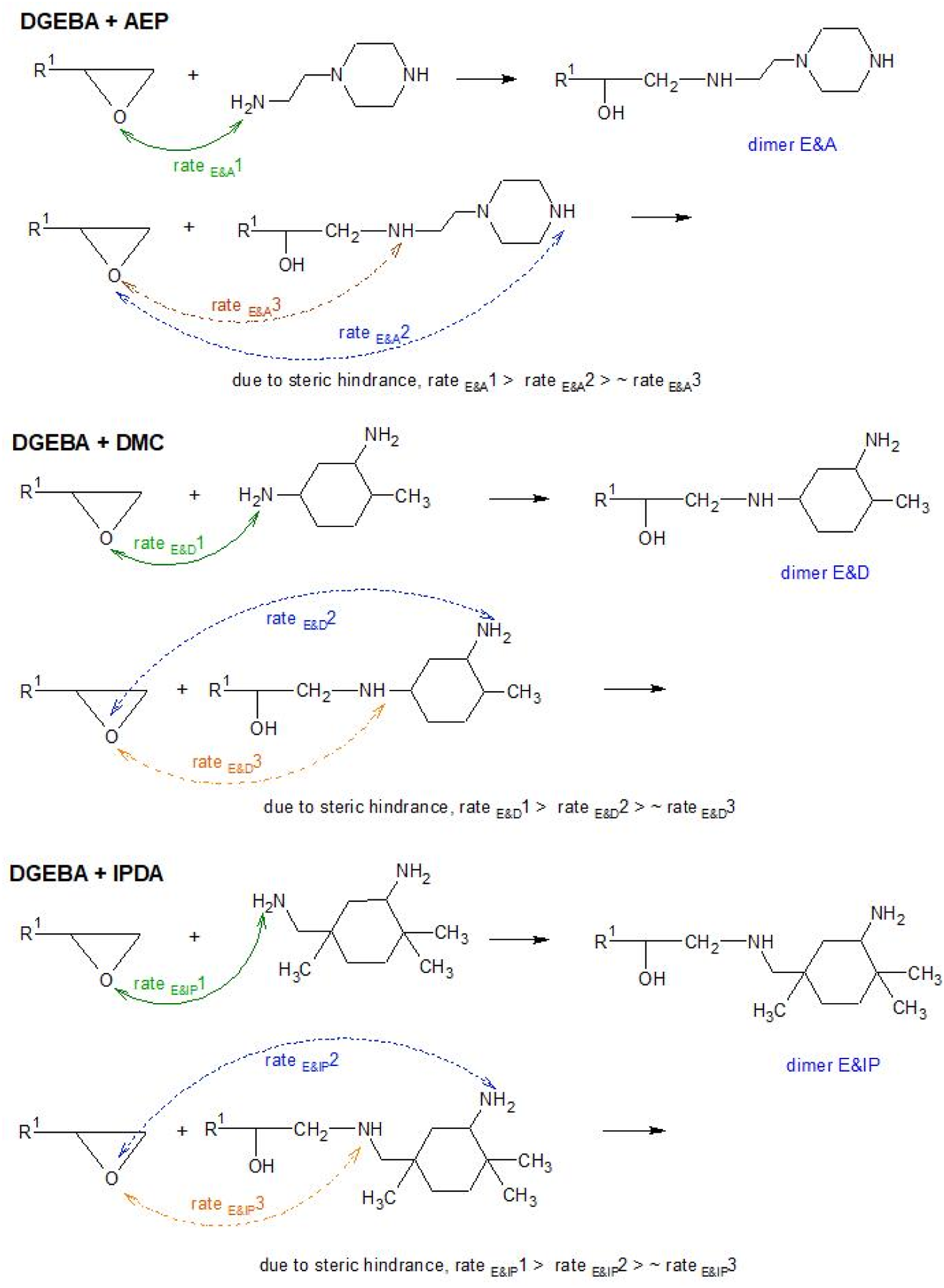
The suggestion of reaction rate of epoxide groups and H-active of the neat amines and the H-active in a newly formed dimer.
Conclusion
The foaming of epoxy with carbamate provides a controllable processing window for the production cell-interconnection and closed-cell epoxy foam. The cell-morphology can be designed by selecting the appropriate amine chemical structure. AEP exhibits the faster reaction with DGEBA and therefore develops a high precured-modulus. Moreover, the H-active piperazine in AEP leads to the viscoelastic behavior of E-AEP-IPDA.CO2. The cell-morphology is spherical and the foam is well obtained at 25% mold filling level. DMC and IPDA react more slowly with DGEBA, and provide lower precured-moduli. Both E-DMC-IPDA.CO2 and E-IPDA-IPDA.CO2 foams are closed-cell and well obtained at 75% mold filling level. The storage modulus at 30°C of E-AEP-IPDA.CO2 is the highest (0.51 GPa), while the glass transition temperature of E-DMC-IPDA.CO2 is the highest (166°C).
Footnotes
Acknowledgements
The authors express the appreciation to the GERMAN RESEARCH FOUNDATION (DFG) for financial support (project number 452210919: AL 474/49-1) as well as the Eurostars-2 joint program with co-funding from the European Union Horizon 2020 research and innovation program with the grant number 01QE1816 B. In addition, the authors would like to thank the Bavarian Polymer Institute (BPI) for the opportunity to work in their laboratories and use their equipment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the GERMAN RESEARCH FOUNDATION (DFG) for financial support (project number 452210919: AL 474/49-1) as well as the Eurostars-2 joint program with co-funding from the European Union Horizon 2020 research and innovation program with the grant number 01QE1816 B.