Abstract
This work is intended for life science researchers and novice users who are currently employing or planning to employ high-density electromyography (HD-sEMG) techniques. An HD-sEMG system measures the electrical activity of muscles by generating detailed activation maps using multiple, densely arranged electrodes placed over a limited area of the skin. Electromyography systems are composed of several components, which can be systematically identified through a structured literature review. This review focuses exclusively on 2D HD-sEMG systems and adopts a descriptive, narrative-based approach aligned with established guidelines for systematic literature reviews. It compares similarities and differences across studies to identify the key elements that should be reported when describing a 2D HD-sEMG system. Data were extracted from 48 articles, which were analyzed and categorized into three main domains: technology, instrumentation, and implementation. Based on this analysis, a set of minimum reporting guidelines for HD-sEMG systems was developed, accompanied by a checklist designed to ensure comprehensive and standardized documentation.
Keywords
Introduction
Electromyographic systems, as described in,1,2 are designed to estimate muscle strength, provide feedback on muscle activity levels, identify muscle activation patterns, and enable the control of prosthetic devices, robotic systems, or rehabilitation technologies. Surface electromyography (sEMG) is a non-invasive technique employed to evaluate muscle function by recording the electrical potentials generated during muscle activation. These signals represent the summation of motor unit action potentials detected at the recording site, modeled as a discrete stochastic process.
This technique involves placing electrodes on the skin surface overlying the target muscles, allowing for the evaluation of neuromuscular activity without penetrating the skin. A key feature of these electrodes is the number of signal channels they use. Single-channel electrodes have one channel that generates the EMG signal, while multichannel configurations include several channels that correspond to the muscle being analyzed.
In, 3 the configurations of multichannel electromyography are described, including linear arrays and two-dimensional (2D) arrays. Linear arrays consist of electrodes arranged in a single line, commonly used for tracking muscle activation along a specific axis or in the direction of a muscle fiber. In contrast, 2D arrays distribute electrodes across a surface in a grid-like pattern, allowing for spatial mapping of muscle activity over a broader area.
Among the variations of bidimensional electrode arrays, two main types can be distinguished: high spatial resolution surface electrodes (HSR-sEMG) and high-density surface electrodes (HD-sEMG). HSR-sEMG arrays are designed to capture fine-grained spatial details of muscle activation, offering enhanced resolution for detecting subtle neuromuscular patterns and improving the selectivity in identifying motor unit parameters. HD-sEMG arrays, on the other hand, feature many closely spaced electrodes, providing comprehensive coverage and detailed spatial information, making them ideal for advanced signal processing and clinical diagnostics.
HD-sEMG is an advanced, non-invasive technique for recording muscle electrical activity, enabling detailed spatial and temporal analysis of electromyographic signals. 4 By employing a dense array of electrodes placed on the skin surface, HD-sEMG provides high-resolution mapping of muscle activation patterns, facilitating a more comprehensive assessment of neuromuscular function. 2D electromyography refers to a matrix array of monopolar electrodes arranged in rows and columns, which allows for the establishment of a spatial relationship. Linear arrays, on the other hand, are monopolar configurations with only multiple rows and only one column. HSR arrays use double differential or Laplacian filters to improve signal clarity. These electrode grids cover a large portion of the muscle surface, providing additional spatial information that complements traditional temporal data. 5
Despite the growing interest in HD-sEMG systems, no systematic review has been identified that specifically addresses the technological, instrumentation, and implementation aspects of multichannel two-dimensional configurations. Existing reviews primarily focus on conventional sEMG or isolated clinical studies, without delving into the technical components that comprise these advanced systems. In, 3 a review is presented, but it is limited solely to electrodes and does not consider the other elements that comprise an HD-sEMG system. Reference 6 presents a systematic review that explores the clinical applications of HD-sEMG.
Therefore, this systematic review aims to identify and analyze the key components involved in the design and application of two-dimensional HD-sEMG systems, including technological components, instrumentation setups, and signal processing methods. The research question guiding this review is: Which technological, instrumentation, and implementation components are most relevant in two-dimensional HD-sEMG systems developed for research prototypes?
Elements of a HD-sEMG system
The review presented in 3 is an analysis of electrodes required for an HD-sEMG; however, the key components were not critically examined to assess their roles in data acquisition, processing, and overall system performance.
Electrode design and configuration
Multiple electrodes in HD-sEMG allow for the extraction of anatomical and physiological information at the muscle or motor unit level. 7 Four main advantages of using HD-sEMG arrays can be identified: flexibility in array size to cover larger areas, cost-effectiveness based on sensor materials, device reusability, ease of application, and low maintenance requirements.
The characteristics of the electrodes to be analyzed involve several factors: the number of electrodes, which varies depending on the muscle being examined; the configuration, which may be monopolar or bipolar; and the shape and size, which can be rectangular or circular, with typical electrode sizes ranging from 0.3 to 5 mm. 6
The inter-electrode distance (IED) 6 is recommended to range between 2.5 and 10 mm, and this spacing should remain consistent in all directions across the electrode array. Signals obtained with different electrode sizes or distances should not be compared, as this can affect their amplitude and spectral characteristics. 8 Although large electrodes may reduce signal variability, they also lose important information due to low-pass spatial filtering, which limits the detail of the analysis. 8
Additionally, the grid size, defined by the row-to-column ratio, varies according to the number of electrodes and the specific muscle under investigation. The arrangement of rows and columns is flexible, depending on the number of electrodes, with a minimum of two electrodes and no defined maximum, as determined by the manufacturer’s specifications.
In the design of commercial electrode grids for surface electromyography (sEMG), it is common practice to exclude one or more corner positions to adjust the total number of electrodes to a power of 2. This approach is driven by technical considerations related to the efficiency of electronic scanning and digital signal processing. For instance, a grid with 13 × 5 positions contains 65 electrodes; by removing one corner, the total becomes 64 electrodes, which corresponds to 2. 7 Such configurations facilitate integration with electronic systems that operate optimally with binary structures, such as multiplexers and analog-to-digital converters. The use of conductive gel is optional, and signal filtering can be performed either through hardware or software. Signal amplification is carried out solely by circuitry.
Proper positioning of electrodes relative to the innervation zone is essential and should be determined according to the specific objectives of the investigation. Accurate placement ensures reliable signal acquisition and minimizes cross-talk, thereby enhancing the validity of the data collected for specific experimental purposes. Conversely, improper positioning may result in distorted signal interpretation, particularly in HD-sEMG, where spatial resolution is a critical factor. As discussed in reference, 9 the location of the electrodes with respect to the innervation zone significantly affects both the amplitude and morphology of the recorded signals and must therefore be carefully considered during experimental design.
Data acquisition
In the analog-digital (A/D) conversion process, sampled sEMG values are transformed into binary numbers that can assume any of 2n possible values (ranging from 0 to 2n−1), where n represents the number of bits used by de A/D converter. HD-sEMG signals are inherently analog. According to the reference, 10 sEMG can be understood as a two-dimensional distribution of electric potential across the skin surface that evolves over time. This continuous signal is converted into a sequence of digital samples through electrode systems and electronic converters, enabling the generation of spatial and temporal maps of muscle activity. To store and process these signals using digital devices, it is necessary to convert them into a digital format through two fundamental processes: sampling and A/D conversion.
Sampling is the process by which a continuous analog signal, such as sEMG, is converted into a series of discrete values taken at regular time intervals. To avoid distortion due to aliasing, 10 the sampling frequency must exceed twice the highest frequency present in the signal.
A/D conversion is the process that transforms each sample of an analog signal into a binary number, allowing the signal to be stored and processed by digital devices. This conversion depends on the number of bits in the A/D converter, which determines the resolution and precision with which each sample is represented. A minimum resolution of 12 bits is recommended; however, 16 bits is preferred, as it provides acceptable quantization errors even for low-amplitude signals amplified with fixed gain. A resolution of ≤0.5 μV between adjacent levels is advised to ensure signal fidelity. 10
In addition to these technical considerations, information regarding the electromyography equipment used for signal acquisition will be included, focusing on details such as manufacturer, model, number of channels, and other relevant specifications.
Signal processing
Several techniques are employed to process HD-SEMG signals. This review aims to identify the information extracted from the electromyographic signals, the tools used for their processing, and the specific methods and software applied. According to, 11 EMG signals must be processed to eliminate noise, and appropriate algorithms are required to enable feature extraction and classification.
In 12 proposes various techniques for EMG signal processing, including time-domain analysis, frequency-domain analysis, signal normalization, fast Fourier transform (FFT), and discrete wavelet transform (DWT). Similarly, 13 suggests approaches such as wavelet analysis, time-frequency approximations, autoregressive models, artificial intelligence, and higher order statistics (HOS) models. Depending on the specific processing strategy, these techniques can be further subdivided. Among the most commonly applied methods are Wavelet Transform (WT), Fast Fourier Transform (FFT), and Wavelet Packet Transform (WPT), each offering distinct advantages for EMG signal analysis. 12
Testing of the subject muscles being investigated
Paper 3 considers several critical aspects, including the specific muscle under investigation, the experimental setup, and the placement of electrodes on the skin surface. Multiple references, including 14 and, 15 provide detailed guidelines for the appropriate electrode placement for each muscle in the human body.
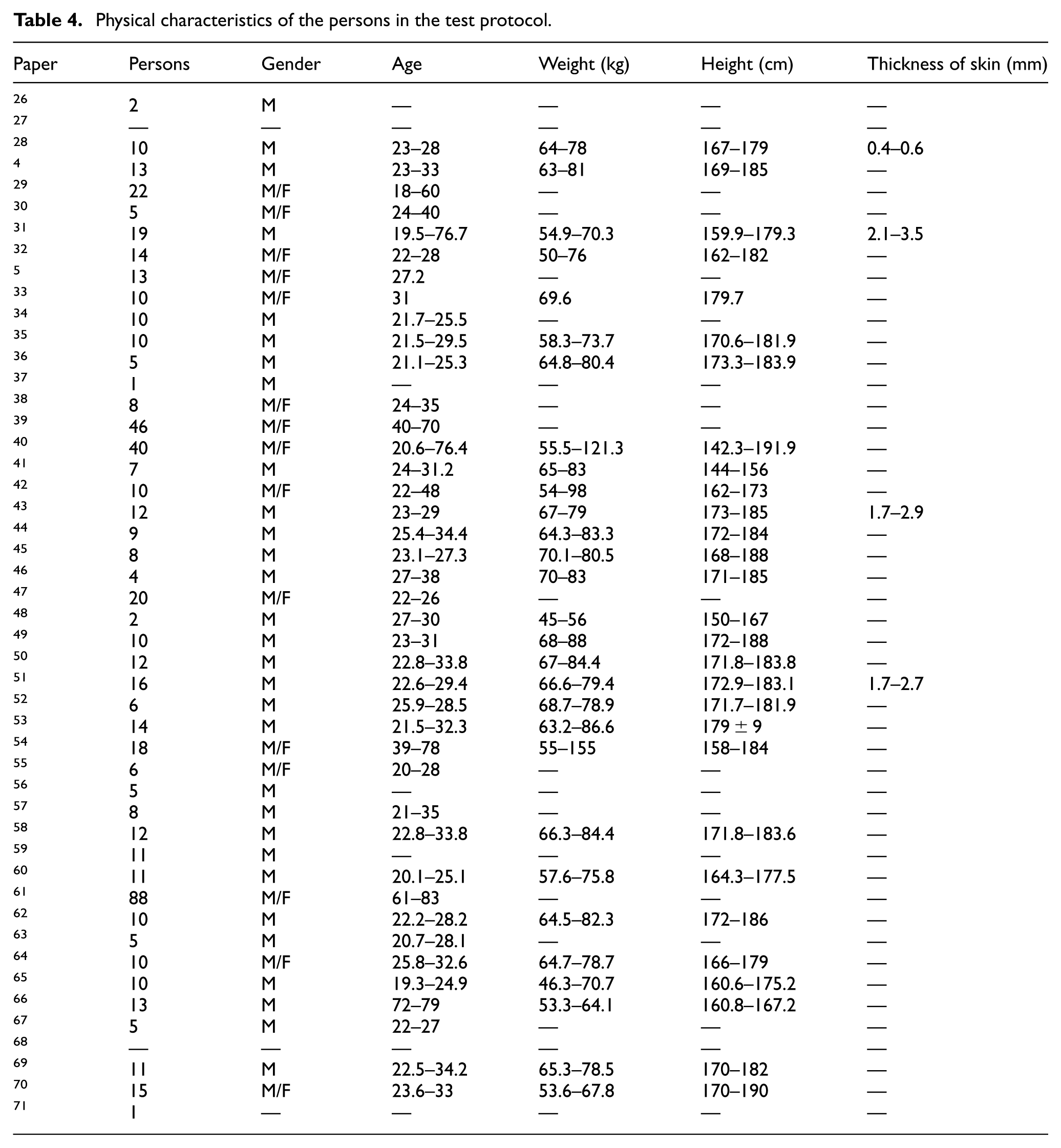
Additionally, data concerning the physiological characteristics of the participants—such as sample size, sex, age, weight, height, and skin thickness—were incorporated to support a comprehensive and contextually relevant analysis.
The experimental design addresses key procedural aspects, including skin area cleansing, adherence to the Declaration of Helsinki, obtaining informed consent from participants, and approval by the appropriate ethics committee.
Methodology for systematic review
This review is descriptive in nature and follows the narrative review format. It is based on established guidelines for conducting narrative synthesis, wherein the integration of data typically involves a narrative juxtaposition of evidence, 16 highlighting similarities and differences across studies. A descriptive review aims to examine the current state of the literature related to a specific research question, topic, or concept. Within this category, narrative reviews focus on compiling information that provides contextual background and general arguments from the authors‘ perspective.
Data were extracted from each selected article and analyzed to determine how the characterization of two-dimensional high-density surface electromyography (2D HD-sEMG) was addressed. In references 16 and, 17 the review process is structured into three main stages: planning, conducting, and reporting. Reference 16 further subdivides these stages into eight specific steps, which are outlined below.
Step 1. Formulate the problem
This systematic review aims to identify the technology, instrumentation, and implementation in components involved in prototypes utilizing 2D HD-sEMG, and analyze the elements integrated into such systems. No prior reviews specifically addressing these aspects in 2D or high-density configurations were found. Existing literature primarily focuses on general surface electromyography or multichannel studies oriented toward clinical analysis or electrode-specific evaluations.
Step 2. Develop and validate the review protocol
The research questions and objectives of the study were defined during the development and validation of the review protocol. The purpose of this review is to systematically examine the various components that constitute a 2D HD-sEMG system, with particular emphasis on technology, instrumentation, and implementation aspects.
The research questions establish a clear connection between the focus of the investigation and the methodology approach. This review addresses the following five research questions:
RQ1 How has multichannel surface electromyography developed?
RQ2 What elements should be considered when configuring a 2-D electromyography system?
RQ3 What types of sensors have been employed in the construction of 2-D sEMG electromyography systems?
RQ4 What features must the acquisition system include to assemble the 2-D sEMG electromyography system?
RQ5 What type of post-processing techniques are applied in 2-D sEMG electromyography systems?
Step 3. Search the literature
This step marks the beginning of the review phase, initiated through a systematic search of electronic databases. Specifically, five databases were consulted: Science Direct, Web of Science, Springer, IEEE, and Scopus.
These sources were selected due to their technological rather than clinical focus. The keywords used in the search, aligned with the research questions, included surface electromyography, multichannel, and 2-D. Synonyms and abbreviations such as sEMG, multichannel, 2D, and two-dimensional were also incorporated.
The search was conducted in two directions to ensure comprehensive coverage of all possible keyword combinations and relevant documents. The results obtained from each direction were compared to verify consistency and completeness in the final set of selected articles.
The initial search yielded a total of 853 papers: 244 from Science Direct, 26 from Web of Science, 552 from Springer, 1 from IEEE, and 30 from Scopus. Titles were subsequently screened to eliminate 29 duplicate entries.
As part of the literature search, two relevant books were identified. Electromyography (EMG) Techniques for the Assessment and Rehabilitation of Motor Function 18 and sEMG: Barriers Limiting Widespread Use of sEMG in Clinical Assessment and Neurorehabilitation. 19 The first volume comprises 19 chapters, of which only 1 specifically mentions HD-sEMG in the title. 20 This chapter was excluded from further analysis because it relied on data obtained from a pre-existing database, rather than original experimental recordings.
The second volume consists of 18 chapters, none of which explicitly reference HD-sEMG in their titles. The concept of high-density EMG is only briefly defined or mentioned in five chapters authored by,21,22,23,24 and. 25 However, these references are limited to general definitions and do not offer substantial methodological or analytical contributions relevant to the scope of this study.
Step 4. Screen for inclusion
Once the two-way review was completed, selection criteria were established to define the scope of the study. The inclusion criteria were as follows:
IC1: Studies incorporating multichannel electromyography systems in two-dimensional (2-D) configurations.
IC2: Studies that identify and define the components of the surface electromyography system, including architectural descriptions using block diagrams or system design schematics.
IC3: Studies that implement electromyography systems with both pre-processing and post-processing of signals.
The following exclusion criteria were also defined:
EC1: Studies involving single-channel or three-dimensional (3-D) electromyography systems.
EC2 Studies that do not utilize surface electromyography, such as multi-channel intramuscular approaches using needle or wire.
EC3: Articles written in a language other than English.
Additionally, the following criteria were excluded from the review:
DC1: Articles that analyze signals obtained from pre-existing databases.
DC2: Articles focused solely on sensor usage without system-level integration.
DC3: Studies employing ultrasound technology.
Following the establishment of inclusion and exclusion criteria, abstracts were reviewed, resulting in the elimination of 30 articles that did not meet the specified requirements, as illustrated in Figure 1.
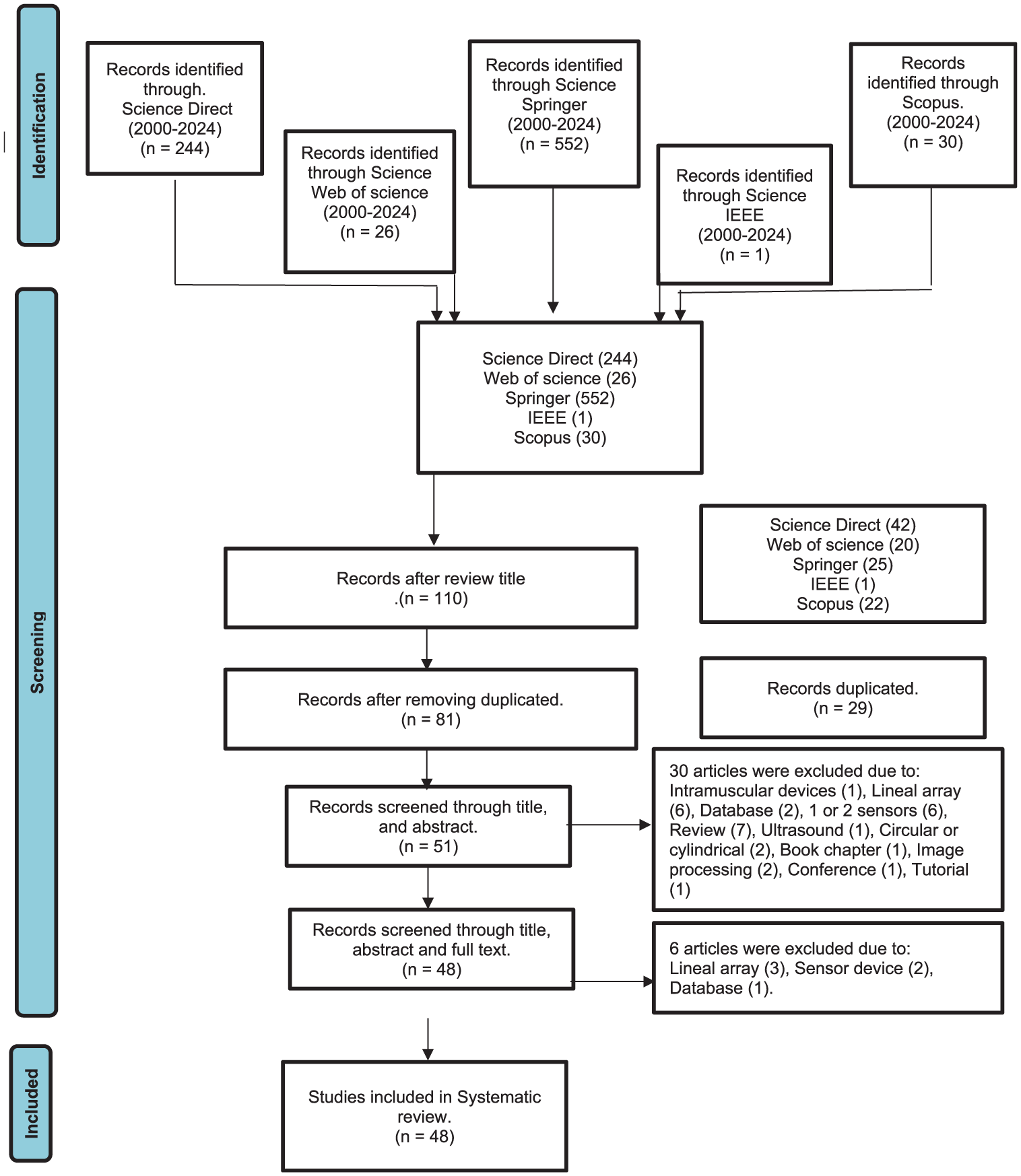
Flow chart for selecting the final items.
Step 5. Assess quality
Following a preliminary review of the full articles, six studies were excluded: three focused on linear electrode arrays, two employed a single sensor, and one utilized an electromyographic database. As a result of this screening process, a total of 48 studies were retained for analysis.
Step 6. Extract data
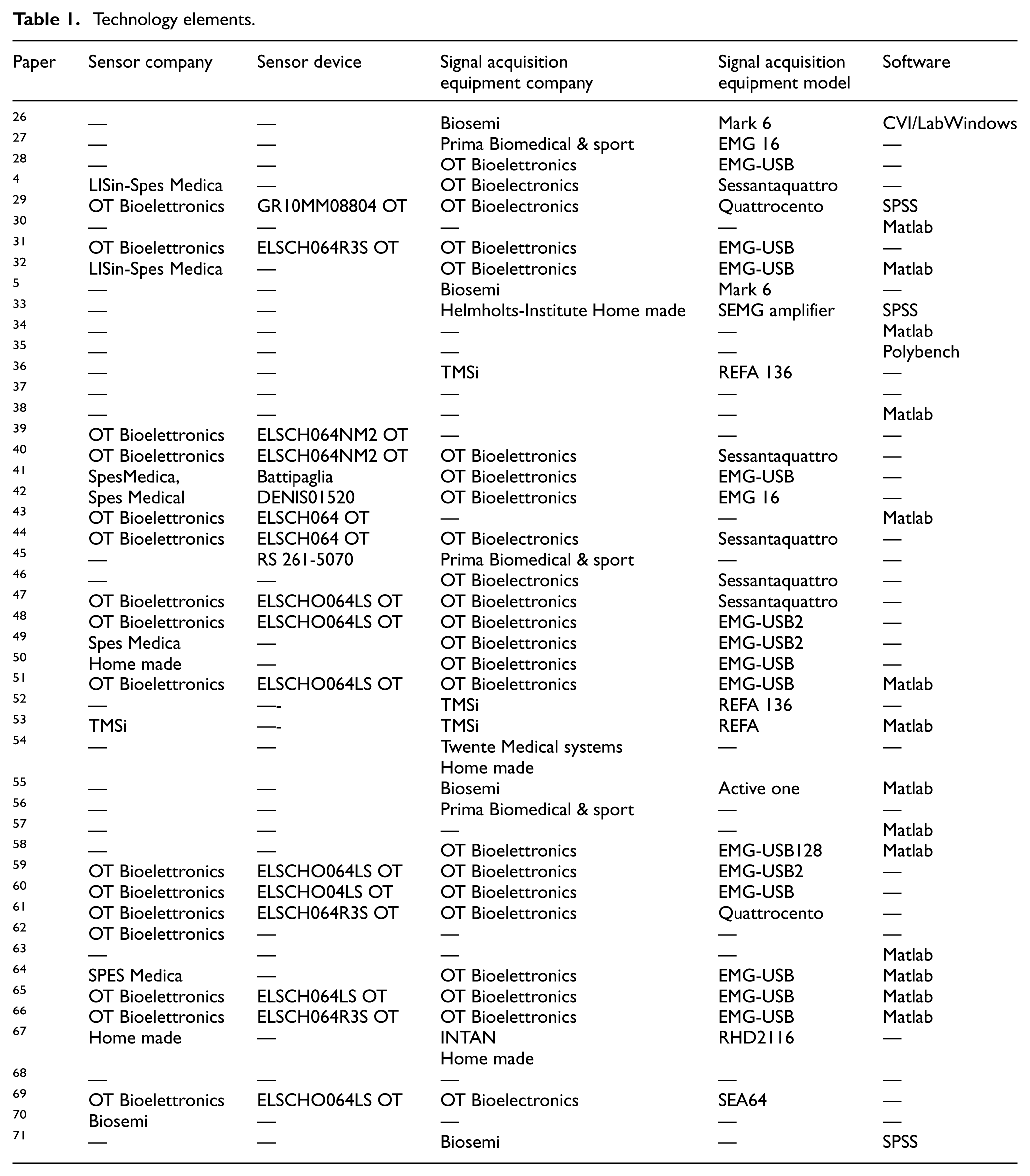
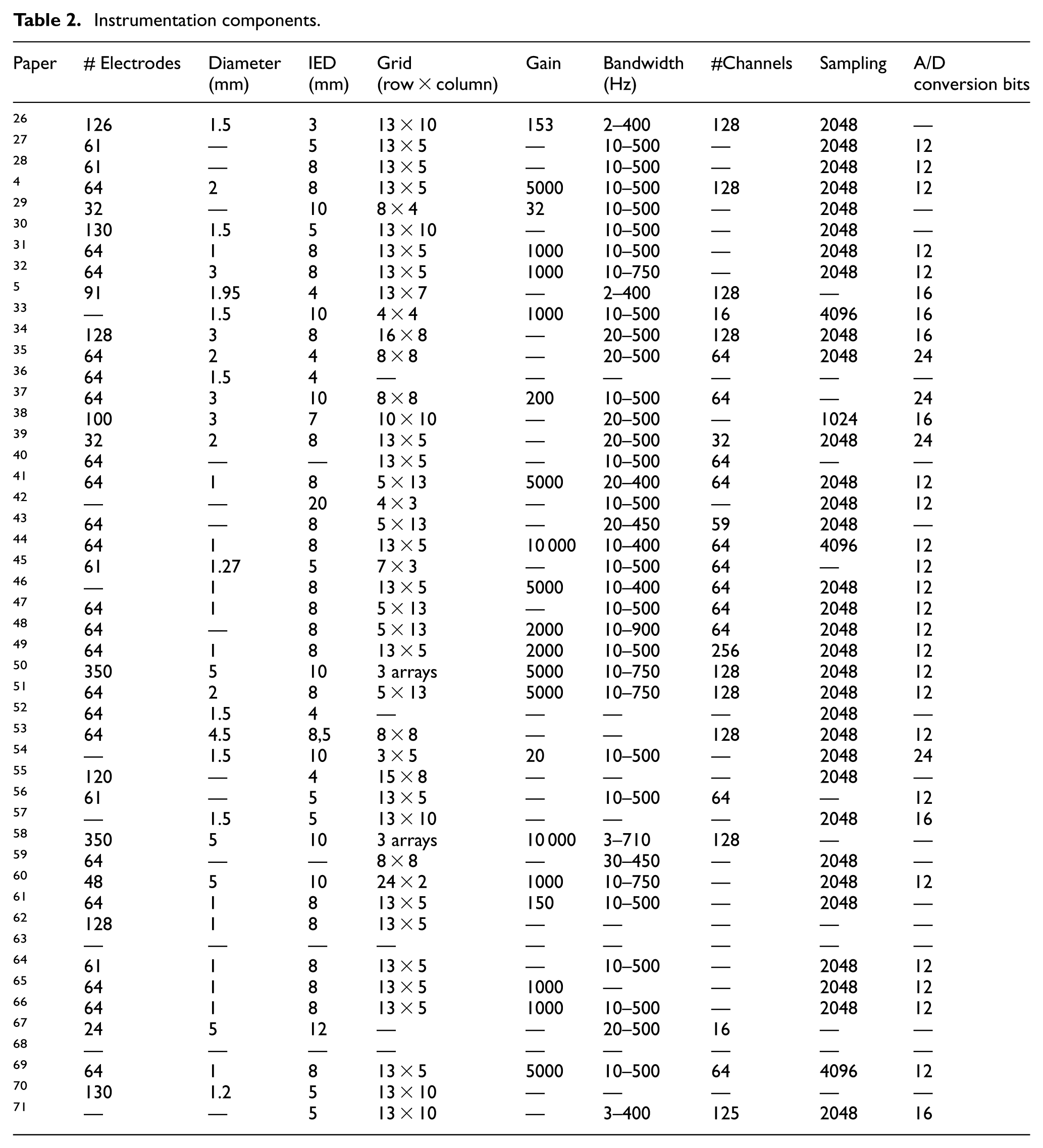
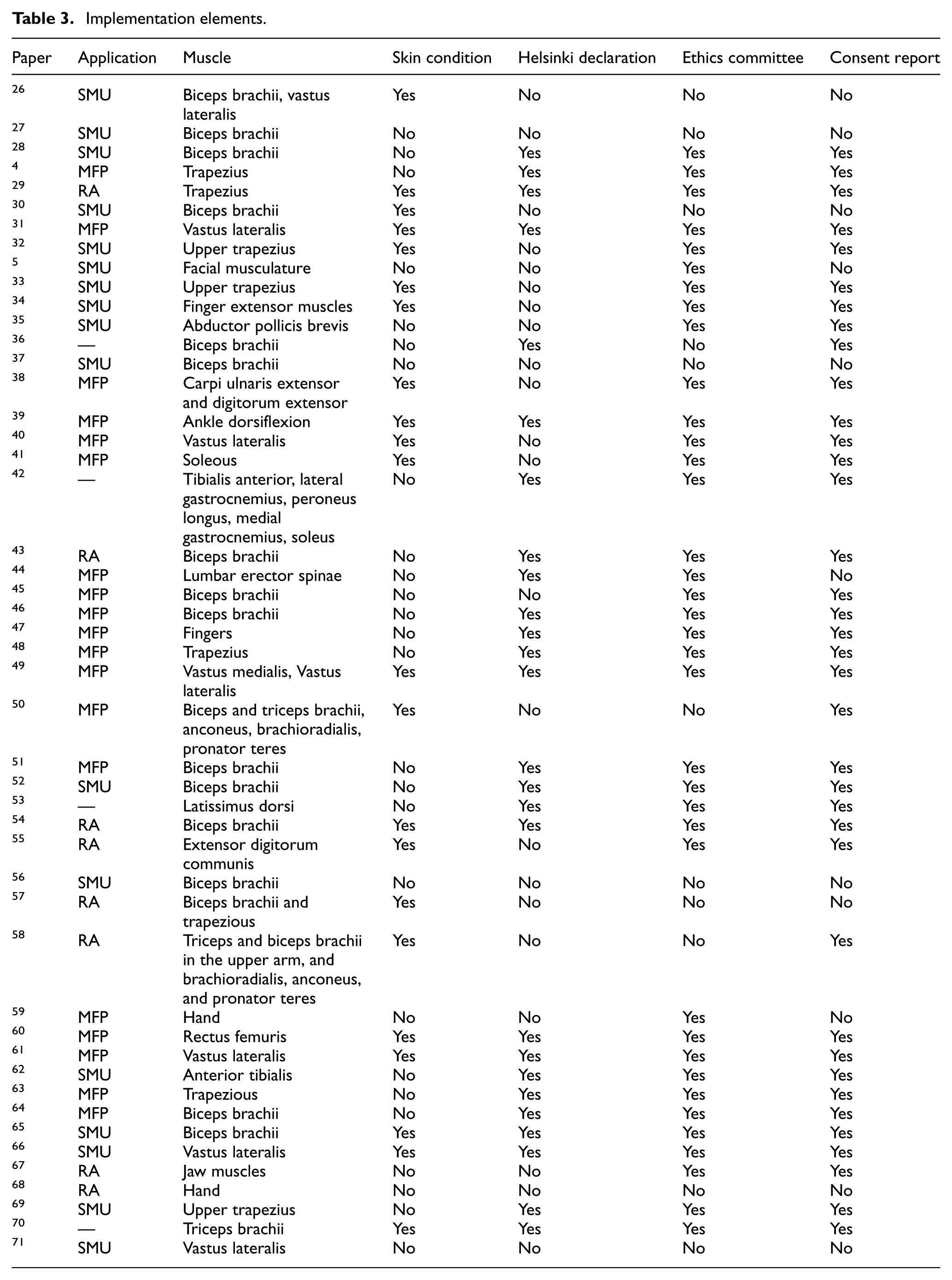
Excel spreadsheets were developed to systematically capture the components of the electromyographic signal acquisition system, as well as the protocols used for signal measurement. The information extracted is presented in Tables 1 to 3.
Technology elements.
Instrumentation components.
Implementation elements.
Step 7 and 8. Analyze and synthesize data
Tables 1 to 3 summarize the data extracted from each document, which were subsequently analyzed and synthesized to support the objectives of this review.
Results
Technology
The technological components of a system refer to the tools and elements necessary to ensure efficient operation and achieve its intended objectives. In this review, the technological elements considered include electrodes and the electromyographic (EMG) signal acquisition systems, encompassing both hardware and software aspects.
Table 1 presents the manufacturers and models of electrode equipment identified in the literature. OT Bioelettronica was the most frequently reported manufacturer, appearing in 35.4% of the studies analyzed. It was followed by Spes Medica with 6.25% and Lisin-Spes Medica with 4.16%. Additionally, homemade devices were mentioned in 4.16% of the documents, while equipment from TMSi and Biosemi Inc. was each cited in 2.08% of the cases. Notably, 45.8% of the articles did not provide information regarding the manufacturer of the electrode system used, which represents a considerable limitation in the technological traceability of the sample.
Notably, 27.04% of the 48 reviewed studies employed the ELSCH064 model in its different versions from OT Bioelettronica. Models from Spes Medica were reported in 8.32% of the studies. Additionally, low-cost, self-designed devices were mentioned in 4.16% of the papers, while equipment from the company TMSi appeared in 2.08% of the reviewed documents.
In the context of signal acquisition systems, it was found that 6.25% of the 48 reviewed documents reported the use of devices developed in university laboratories. An identical percentage (6.25%) corresponded to devices manufactured by Prima Biomedical and TMSi. Notably, 25% of the documents did not provide any information regarding the manufacturer of the equipment used. The most frequently cited company was OT Bioelettronica, appearing in 47.9% of the reviewed documents. The devices mentioned included the Mark 6, EMG 16, EMG USB, EMG USB2, EMG USB128, REFA, REFA16, Sessantaquattro, and Quattrocento models. Additionally, devices from Biosemi were reported in 8.3% of the documents.
Regarding the software used for data analysis, Matlab was the most frequently reported, appearing in 29.16% of the 48 reviewed documents. Both SVI/LabWindows and Polybench software were each mentioned in 2.08% of the studies, while SPSS software was cited in 6.25% of the cases. However, a significant proportion—60.42% of the reviewed papers—did not specify the software employed for data analysis.
Instrumentation
The instrumentation of an HD-sEMG system comprises multiple components and technical considerations aimed at ensuring accurate, efficient, and reliable operation. Among the essential elements of an HD-sEMG system are the sensors, signal conditioners, and data acquisition units. Each of these components plays a critical role and possesses specific characteristics that must be carefully evaluated to optimize system performance.
In the case of sensors, several parameters must be considered, including the number of sensors, electrode diameter, interelectrode distance (IED), and grid configuration.
Spatial resolution is a key attribute of HD-sEMG systems, as it determines the system's ability to accurately detect and differentiate muscle activity from closely spaced regions on the muscle surface. This resolution is primarily influenced by the inter-electrode distance, electrode size, and the quality of the signal detection hardware. To achieve adequate spatial resolution in HD-sEMG systems, the sampling density should exceed 200 samples per meter (Sp/m), which implies that the inter-electrode distance should be less than 5 mm. 8
Regarding the dimensions of the electrode matrix, a row-by-column format was used to describe the grid configurations. The following grid sizes—3 × 5, 4 × 3, 4 × 4, 7 × 3, 8 × 4, 10 × 10, 13 × 7, 15 × 8, 16 × 8, and 24 × 2—were each reported in 2.08% of the 48 reviewed studies. Additionally, 4.16% of the documents included two or more distinct grid configurations. The 8 × 8 grid appeared in 8.32% of the studies. A frequency of 10.41% was observed for each of the following cases: grid sizes 5 × 13 and 13 × 10, as well as for studies that did not report grid size information. The most frequently reported configuration was the 13 × 5 grid, which was present in 35.36% of the reviewed documents.
One notable aspect identified during the review was the presence of unconnected top corners in some electrode grids, a characteristic explicitly mentioned in 31.2% of the 48 analyzed studies. Additionally, 39.52% of the reviewed documents reported the use of conductive gel in conjunction with the electrode grid.
According to Merletti and Cerone, 10 amplifier gain plays a critical role in determining the amplitude of the signal before its digital conversion. This parameter is typically adjustable on the recording device and must be carefully calibrated. Insufficient gain may result in reduced resolution of the digitized signal, whereas excessive gain can lead to signal saturation, compromising data quality.
Table 2 summarizes the distribution of gain values reported across 48 reviewed studies. Gain settings of 20, 32, 150, 153, 200, and 500 were each reported in 2.08% of the documents, collectively accounting for 12.48%. Gains of 2000 and 10 000 were each cited in 4.16% of the studies, while a gain of 5000 was reported in 10.41% of the documents. The most frequently reported gain was 1000, appearing in 12.5% of the studies. Notably, 56.25% of the reviewed papers did not disclose any information regarding the gain settings employed.
Among the 48 reviewed studies, 2.08% reported using the following bandwidth ranges: 3–710, 3–400, 10–900, 20–400, 20–450, and 30–450 Hz. Bandwidths of 2–400 and 10–400 Hz were each cited in 4.16% of the documents. The bandwidth range of 10–750 Hz was reported in 8.33% of the studies, while 20–500 Hz was cited in 10.41%. Notably, 20.8% of the studies did not specify the frequency range employed. The most frequently reported bandwidth was 10–500 Hz, appearing in 39.52% of the reviewed papers.
Regarding the number of channels reported in the reviewed documentation, configurations of 32, 59, 125, and 256 channels were each mentioned in 2.08% of the studies, representing a combined total of 8.32%. A 16-channel configuration was cited in 4.16% of the documents, while 128 channels were reported in 16.6% of the cases. The most frequently reported configuration was 64 channels, appearing in 22.09% of the studies. Notably, 47.09% of the reviewed papers did not provide information regarding the number of channels used.
The most frequently reported sampling frequency was 2048 Hz, cited in 66.16% of the reviewed studies. In contrast, 24.96% of the studies did not specify the sampling frequency employed. Frequencies of 1024 and 4096 Hz were reported in 2.08 and 6.25% of the documents, respectively.
With respect to the resolution of analog-to-digital signal conversion, bit depths of 12, 16, and 24 bits were identified, corresponding to 45.83, 12.5, and 8.33% of the reviewed papers, respectively. Notably, 33.28% of the documents did not report information on the bit resolution used.
Implementation
Implementing an HD-sEMG system is a multifaceted process that requires thorough preparation, the integration of specialized technology, and precise interpretation of the acquired data. This procedure facilitates the effective diagnosis and treatment of neuromuscular disorders. The key elements involved in implementation include equipment, patient preparation, test execution, and data analysis.
Regarding the equipment, the relevant components have already been addressed in the preceding section on Technology and Instrumentation. Proper patient preparation is critical to ensuring the reliability and accuracy of HD-sEMG recordings. Signal quality is influenced not only by the technical specifications of the equipment but also by the physiological and procedural conditions under which data are collected.
Several essential factors must be considered during patient preparation: (1) Skin condition: The skin must be clean and free of lotions, creams, or perspiration that could interfere with electrode conductivity. Additionally, the integrity of the skin should be assessed to prevent artifacts or complications during signal acquisition. (2) Patient positioning: The subject should be placed in a stable and comfortable posture that permits voluntary muscle contractions without inducing movement-related artifacts. (3) Psychological readiness: Anxiety and stress can affect baseline muscle activity; therefore, providing clear instructions and maintaining a calm environment are crucial to minimizing involuntary muscle activation. (4) Clinical and pharmacological considerations: Any pre-existing neuromuscular conditions and the use of medications—such as muscle relaxants or neuroactive drugs—should be documented, as these factors may influence signal characteristics.
In addition to technical and physiological considerations, adherence to ethical and methodological standards is essential. As summarized in Table 3 of the reviewed literature, four primary guidelines were identified across experimental protocols: (1) skin condition, (2) compliance with the Declaration of Helsinki, (3) approval by an institutional ethics committee, and (4) documentation of informed consent. Among the 48 studies analyzed, 10.41% did not reference any of these guidelines, 8.33% cited only one, 14.58% referenced two, 45.8% reported adherence to three, and 20.8% demonstrated full compliance with all four. These findings underscore the variability in methodological rigor and ethical reporting within current HD-sEMG research. 72
The execution of HD-sEMG testing involves the controlled acquisition of electrical signals generated by muscle fibers during voluntary contractions. This process enables real-time observation of neuromuscular activity and facilitates the identification of pathological patterns. To ensure the reliability and diagnostic value of the recordings, several critical elements must be considered: (1) selection of the target muscle, (2) electrode placement and configuration, (3) system calibration and signal conditioning, (4) minimization of artifacts, (5) motor task design, (6) real-time monitoring and quality control, and (7) documentation of physiological variables.
Table 3 presents information related to test execution, specifically regarding target muscle selection in the 48 papers reviewed, which employed surface electromyography (sEMG) to record signals from various muscles. Among these, the Biceps Brachii was identified as one of the most frequently assessed muscles. An analysis of the number of muscles referenced per study revealed that 4.16% of the documents reported recordings from two muscles, 6.25% from three muscles, while the vast majority, 87.36%, focused exclusively on a single muscle.
Aspects such as electrode placement and configuration, system calibration and signal conditioning, real-time monitoring and quality control, and artifact minimization have already been addressed in the preceding sections on Technology and Instrumentation. In contrast, elements such as motor task design and documentation of physiological variables are clinical considerations that fall outside the scope of this investigation.
The data analysis phase is notably broad, as the specific features to be extracted depend on the intended application, which in turn determines the appropriate analytical approach. The primary applications of HD-sEMG include regional activation (RA), which enables spatial analysis of muscle engagement; muscle fiber properties (MFP), which provide insights into the physiological characteristics of muscle tissue; and single motor unit (SMU) analysis, which allows for detailed examination of individual motor unit behavior. These application domains highlight the versatility and analytical depth of HD-sEMG in neuromuscular research.
For regional activation (RA), data analysis involves calculating metrics such as root mean square (RMS) or median frequency per channel, generating heat maps, and estimating propagation vectors to represent the spatial distribution of muscle activity. In the case of muscle fiber properties (MFP), the analytical pipeline includes conduction velocity estimation and spectral analysis to infer physiological characteristics such as fiber diameter. SMU analysis employs advanced signal decomposition algorithms and spike-tracking techniques to characterize motor unit recruitment, activation frequency, and synchronization. These application areas underscore the versatility and analytical depth of HD-sEMG in neuromuscular research.
An analysis of 48 reviewed documents on the implementation of HD-sEMG systems revealed a diverse distribution of application areas; 39.5% of the studies focused on muscle fiber properties (MFP), indicating a predominant interest in the physiological characterization of muscle tissue through parameters such as conduction velocity and spectral analysis. Meanwhile, 35.41% of the studies addressed single motor unit (SMU) analysis, employing advanced decomposition algorithms to examine motor unit recruitment, activation frequency, and synchronization. Regional activation (RA) was explored in 16.6% of the studies, using metrics such as channel-wise RMS, heat maps, and propagation vectors to spatially represent muscle activity. Lastly, 8.3% of the documents did not report specific application data, highlighting opportunities to improve methodological transparency in future research.
This distribution underscores the versatility of HD-sEMG and its adaptability to various analytical approaches within the field of neuromuscular research.
The assessment of participants’
Physical characteristics of the persons in the test protocol.
Checklist for 2D HD-sEMG report
The analysis conducted through the interpretation of Tables 1 to 4 revealed that, in many research articles, the information presented regarding the HD-sEMG system was incomplete; nevertheless, these articles were still reviewed and published.
In response to this issue, key initiatives have emerged to improve technical training and promote methodological standardization within the field. Two of the most relevant are the JEK tutorials—developed by the International Society of Electrophysiology and Kinesiology (ISEK) in collaboration with the Journal of Electromyography and Kinesiology (JEK)—and the publications from the CEDE project (Consensus for Experimental Design in Electromyography).
The JEK tutorials are designed to provide specialized guidance to students and early-career researchers. Through live sessions and accessible materials, they address fundamental topics such as EMG signal analysis during movement, the use of open-source software for signal processing, and best practices in data acquisition. Their primary objective is to strengthen technical proficiency and promote reproducibility in electromyography research.
Conversely, the CEDE project represents an international collaborative effort to establish consensus-based recommendations for experimental design in electromyography. Utilizing a Delphi process, the project has developed various matrices to guide researchers on critical aspects such as electrode configuration, documentation of geometric parameters, and standardized reporting practices. These guidelines aim to enhance the quality, comparability, and transparency of published studies.
Both initiatives address a pressing need in the field: ensuring that EMG signal studies are reproducible, methodologically sound, and scientifically valuable. Their integration into research workflows and editorial standards is essential for advancing toward more robust and reliable scientific practices. It is important to note that these efforts focus on electromyographic signals broadly, encompassing both intramuscular electromyography and multichannel approaches.
As part of this review on high-density electromyography, we propose a checklist specifically focused on HD-sEMG. To develop this checklist, we examined the JEK tutorials:9,73,74,75 and, 76 each of which includes its own checklist. A similar procedure was applied to the matrices developed by the CEDE project:6,77,26 and. 78
The proposed checklist integrates elements from both the JEK tutorials and CEDE matrices, organized into four key areas that should be reported when working with HD-sEMG systems: (1) Purpose or application, (2) Type of technology used, (3) Instrumentation, and (4) System implementation.
Table 5 presents the checklist, highlighting essential components that should be documented in studies utilizing HD-sEMG. It underscores the importance of clearly specifying the intended application, as reporting requirements may vary depending on the research context.
Check list for a report of HD sEMG.
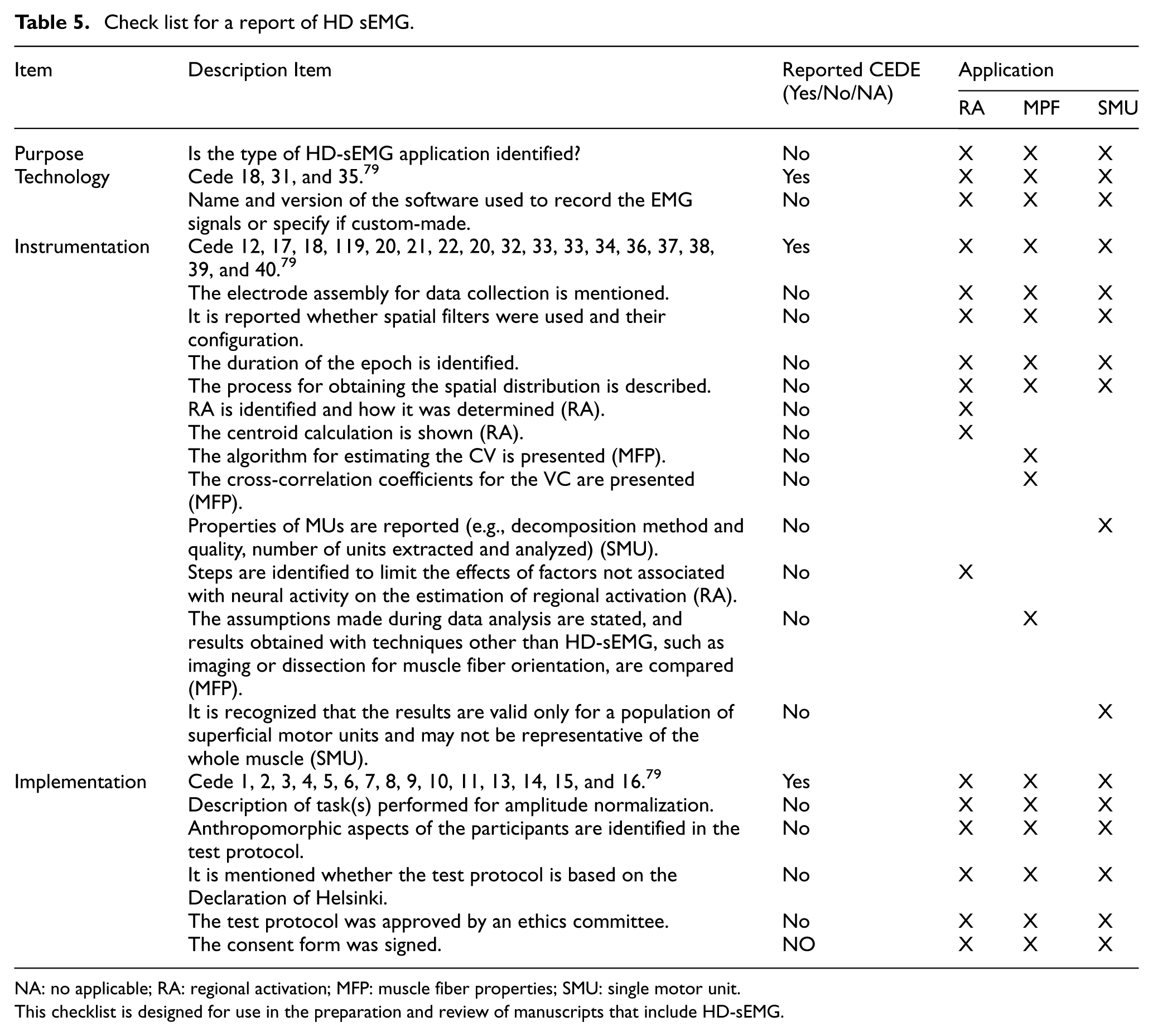
NA: no applicable; RA: regional activation; MFP: muscle fiber properties; SMU: single motor unit.
This checklist is designed for use in the preparation and review of manuscripts that include HD-sEMG.
Conclusion
This review underscores a critical gap in the reporting of HD-sEMG systems within the scientific literature, where essential methodological details are often omitted, even in peer-reviewed publications. In response to this issue, initiatives such as the JEK tutorials and the CEDE project have made substantial contributions toward enhancing technical training and promoting standardized experimental design in electromyography. Drawing on their recommendations, we propose a checklist specifically focused on HD-sEMG, structured around four key reporting domains: application, technology, instrumentation, and implementation. This checklist, presented in Table 5, is intended to improve the transparency, reproducibility, and scientific rigor of future studies involving high-density EMG systems.
Footnotes
Ethical considerations
Ethical approval was not required.
Funding
This work was supported by the Secretaría de Ciencia, Humanidades, Tecnología e Innovación (SECIHTI-México), the Instituto Politécnico Nacional (IPN), and the Universidad Autónoma del Estado de México (UAEMex).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Data availability statement
The data used to support the findings of this study are included in the article.
