Abstract
The sole intention of this investigation is to control and protect the environment by low emissions. An experiment was conducted in a DI CI engine by fueling with 20% of rubber seed biodiesel blended with diesel (B20). Similar experiments were performed with the three different proportions of combined additives (pentanol and Mentha piperita leaves extract), namely, B20A1, B20A2, and B20A3. The performance and emission characteristics were compared with baseline fuel (diesel). Compared with B20, B20A2 exhibited higher brake thermal efficiency of 27.98% and the brake-specific fuel consumption is less by 4.83%. However, the engine exhaust temperature was slightly higher. All the additive mixed blends emitted lesser CO, hydrocarbon, and smoke, especially with B20A2. In addition, B20A2 showed 12% reduction of NOx than B20. Experimental results proved that B20A2 blend was found to be a novel environmentally friendly fuel with most favorable emissions.
I. Introduction
Energy demand in future can be fulfilled only by alternative fuel source due to the fast depletion of the fossil fuels. All over the world, several researches are being carried out for the replacement of conventional diesel fuel.1,2 Biodiesel has been considered as an alternative source for the replacement of fossil fuels due to its various merits. 2 Vegetable oils can be obtained from edible and non-edible oilseed which have physical characteristics close to those of diesel fuel and therefore behave like similar fuels. Behaving equally well is rubber seed oil (RSO), which is classified as non-edible and is widely grown in Asian countries.
Experimental investigations prove that petro-diesel can be partially replaced only by 20%–30% of biodiesel, without affecting the performance.3,4 It is also very clear that biodiesel can reduce the greenhouse gas emissions and soot formation.5,6 It is a renewable one too. However, biodiesel exhibits elevated NOx emission due to higher oxygen content, which was considered to be the major task of the researcher. 7 Considering the harmful effects of engine exhaust, researchers derived several solutions like fuel reformulation, additive method, exhaust gas recirculation, oxidation catalysts, antioxidation catalysts, and diesel particulate filters. 8 Nano-sized particulate matters are carcinogenic and inhaling them leads to severe health issues for human beings. 9 Particulate matters basically affect the human brain by general critical stress response. 10 NOx emission is considered one of the root causes of formation of ground-level ozone and acid rain. Long drawn-out exposure to NO can be the reason for aggressive coughing, breathing problem, cyanosis, and pulmonary edema. 11
Blending biodiesel with oxygenated fuel additives can enhance the combustion while NOx emission cannot be reduced. 12 Pentanol is one of the next-generation biofuels having five carbons in its molecular structure and is produced from renewable feedstock, which may help to improve the challenging problems of energy security and environmental issues. Physical properties of pentanol exhibit lower viscosity and high volatility, which could improve the atomizing quality. Although pentanol contains higher oxygen, due to its low cetane number, it cannot maintain the ignition quality. 13 In order to improve the cold flow property and trim down the viscosity of biodiesel, alcohol is preferred as fuel additive. 14 Alcohol when blended with biodiesel attains the character of suppressing the smoke emission. 15 Usage of antioxidant additives in biodiesel will significantly reduce NOx emission by increasing the antioxidant stability. 16 Carotenoid and chlorophyll seem to be good antioxidants, normally found in the plant leaf, which has the ability to control the photo oxidation. 17 Oxygen structure present in chlorophyll and carotenoid possesses the ability of enhancing the combustion of biodiesel in better means.
The main objective of this experimental investigation is to assess the favorable emission characteristics of a natural aspirated diesel engine fueled with 20% of rubber seed biodiesel blended with diesel along with addition of a novel antioxidant extracted from the Mentha piperita leaves (MPL), with pentanol as an additive.
II. Materials and Methods
RSO was purchased from Virdhunagar, Tamil Nadu, India. Chemicals required for transesterification, such as potassium hydroxide (KOH), sulfuric acid, methanol, and ethanol of 99% purity, were procured from Nice Chemicals, Kerala, India. The presence of higher amount of unsaturated fatty acid in RSO can be normalized by transesterification process. KOH and sulfuric acid were used as catalyst and methanol (CH3OH) has been chosen as alcohol for esterification processes. 18 Raw RSO and methanol in the presence of 1.5% of concentrated sulfuric acid was heated up to 60 °C and stirred continuously for 1.5 h. The products of the esterification processes were allowed to settle for 8 h in a separating funnel to separate biodiesel from the other by-products. The products of the first stage were used as the input of the alkaline esterification process. The products of the first stage and methanol in the presence of KOH were heated at 55 °C 19 for an hour with constant stirring. Finally, the products were allowed to settle down in a separating funnel for about 5–6 h. Then, the biodiesel was washed using distilled water to remove the excess alcohol and catalyst. The fatty acid profile of the raw RSO and methyl ester of RSO is presented and compared in Table 1 .
Fatty acid profile of rubber seed oil (RSO) and rubber seed methyl ester (RSME).

A. Preparation of antioxidant
The chopped MPL was treated with warm water at a temperature of 50 °C for 10 min as shown in Figure 1 . The cleaned leaves were put into the hot air oven to remove moisture content present in the leaf, at a temperature of 70 °C for 15 min. Then, it was ground into small pieces of 1–2 mm, for the purpose of breaking the cell walls and to increase the surface area. The ground leaf (100 g) was mixed with 20 mL of solvent containing 15 mL of ethanol and 5 mL of acetone. The mixture was stirred continuously for an hour, until a pale green opalescent liquid on top appeared. 20 Then, the solution was kept undisturbed for 1 h, and the sediments were removed from the solution. The extract was filtered by Whatman no. 1 filter paper. 21 The filtrate was concentrated in a rotary evaporator at 40 °C. The concentrated extract was oven dried at 40 °C for 3 days and freeze dried for 48 h. The concentrated extract was subjected to chemical analysis to confirm the presence of carotenoid, chlorophyll, proteins, and fatty acids. 22 The final product thus prepared can be straightly used as an antioxidant additive. The additional solvent present in the antioxidant additive can be taken back by storing it in a dark place at room temperature. In order to elevate its miscibility with biodiesel, the additive can be diluted with ethanol.
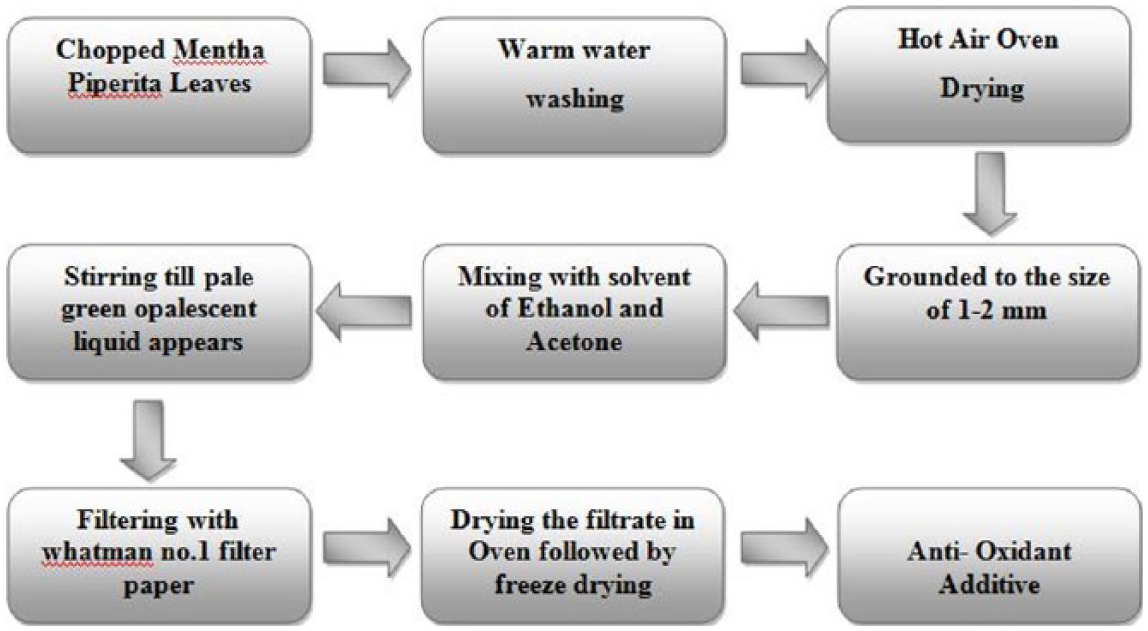
Block diagram describing the extraction of antioxidant from Mentha piperita leaves
Estimation of chlorophyll content
The chlorophyll content was estimated adopting spectrophotometric methodology. From 0.1 g of finely ground MPL, a little amount of chloroplast pigments was extracted. A 10 mL of ethanol of 80% purity and the extracted chloroplast pigments were taken in the test tube in such a way that chloroplast pigments completely immersed in ethanol. It was then kept in a water bath for 15 min at 80 °C. One milliliter of sample was taken from this prepared mixture and was diluted with 10 mL of ethanol of 80% purity, and this mixture was made to undergo spectrophotometric analysis (Spectronic Instruments, model Spectronic 20 Genesys) at two wavelengths of 645 and 663 nm. The complete determination was carried out in triplicate. Equation (1) was used to calculate the total chlorophyll content
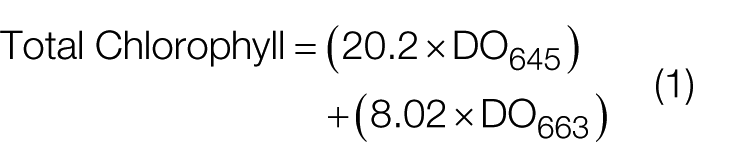
Estimation of antioxidant
With minor modifications based on the method described by Ou et al., 23 the extraction of the antioxidant compounds was performed. MPL powder of 1 g was dissolved in 10 mL mix of acetone and water in equal ratio. The solution was stirred for an hour in an orbital shaker and then centrifuged at 4000 r/min for 15 min. The supernatant was filtered through a Whatman no. 1 filter paper. The residue was dried in an evaporator at 40 °C. It was reconstituted with a phosphate buffer pH 7.4 and brought to a final volume of 15 mL. The ORAC (oxygen radical absorbance capacity) assay was performed and the results confirmed that the solution contained carotenoid (α-carotene and β-carotene).
B. Test procedure and engine setup
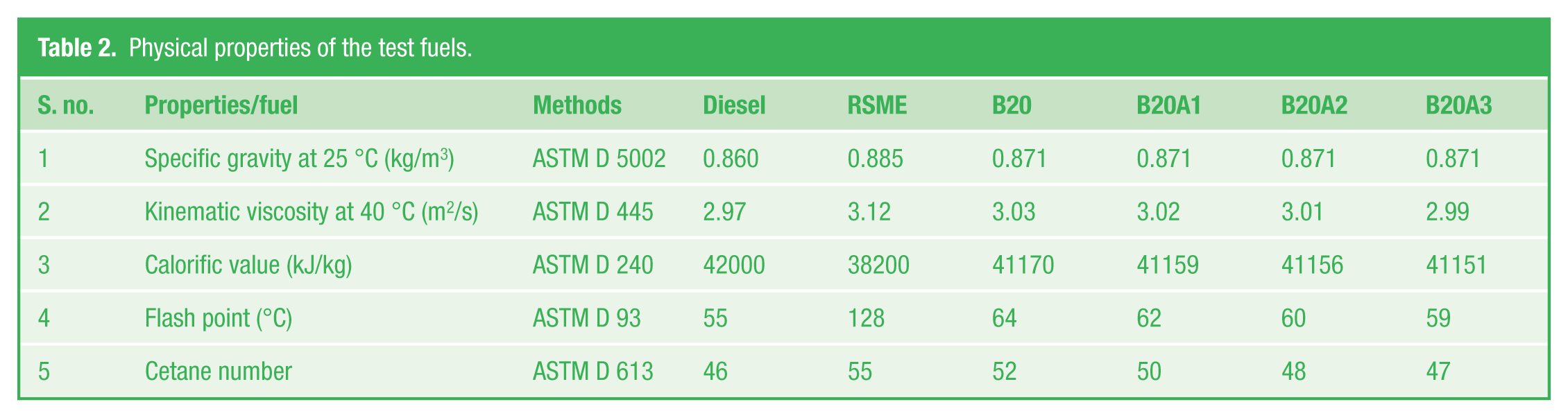
Using diesel, B20 (20% of rubber seed biodiesel and 80% diesel), B20A1 (B20 + 0.25% pentanol + 0.25% MPL extract), B20A2 (B20 + 0.5% pentanol + 0.5% MPL extract), and B20A15 (B20 + 0.75% pentanol + 0.75% MPL extract) as test fuels, experiments were conducted in a four-stroke, direct-injection, naturally-aspirated, constant-speed (1500 r/min), single-cylinder diesel engine as shown in Figure 2 . Physical properties of all the test fuels were determined and are tabulated in Table 2 .
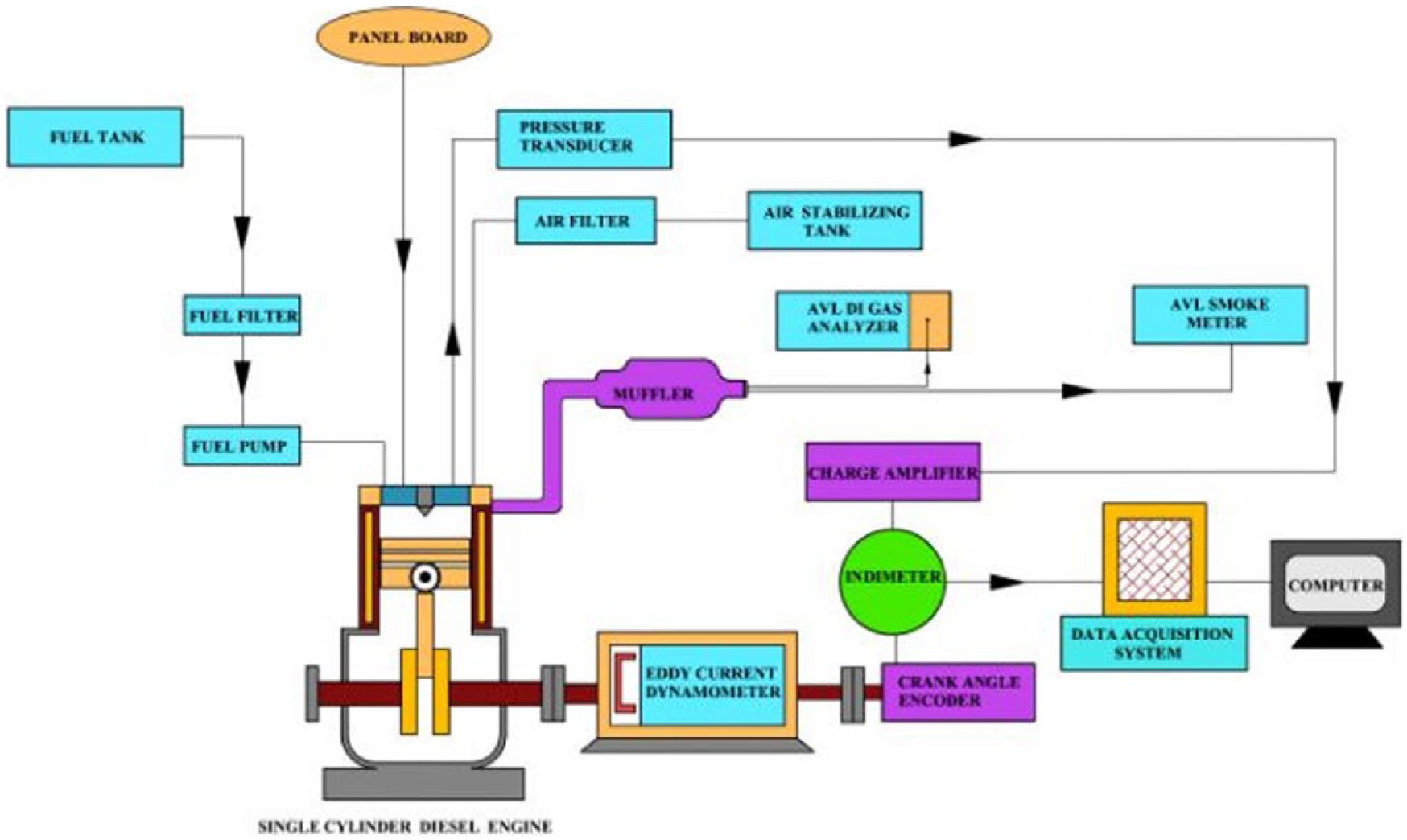
Schematic layout of test engine with measuring accessories
Physical properties of the test fuels.
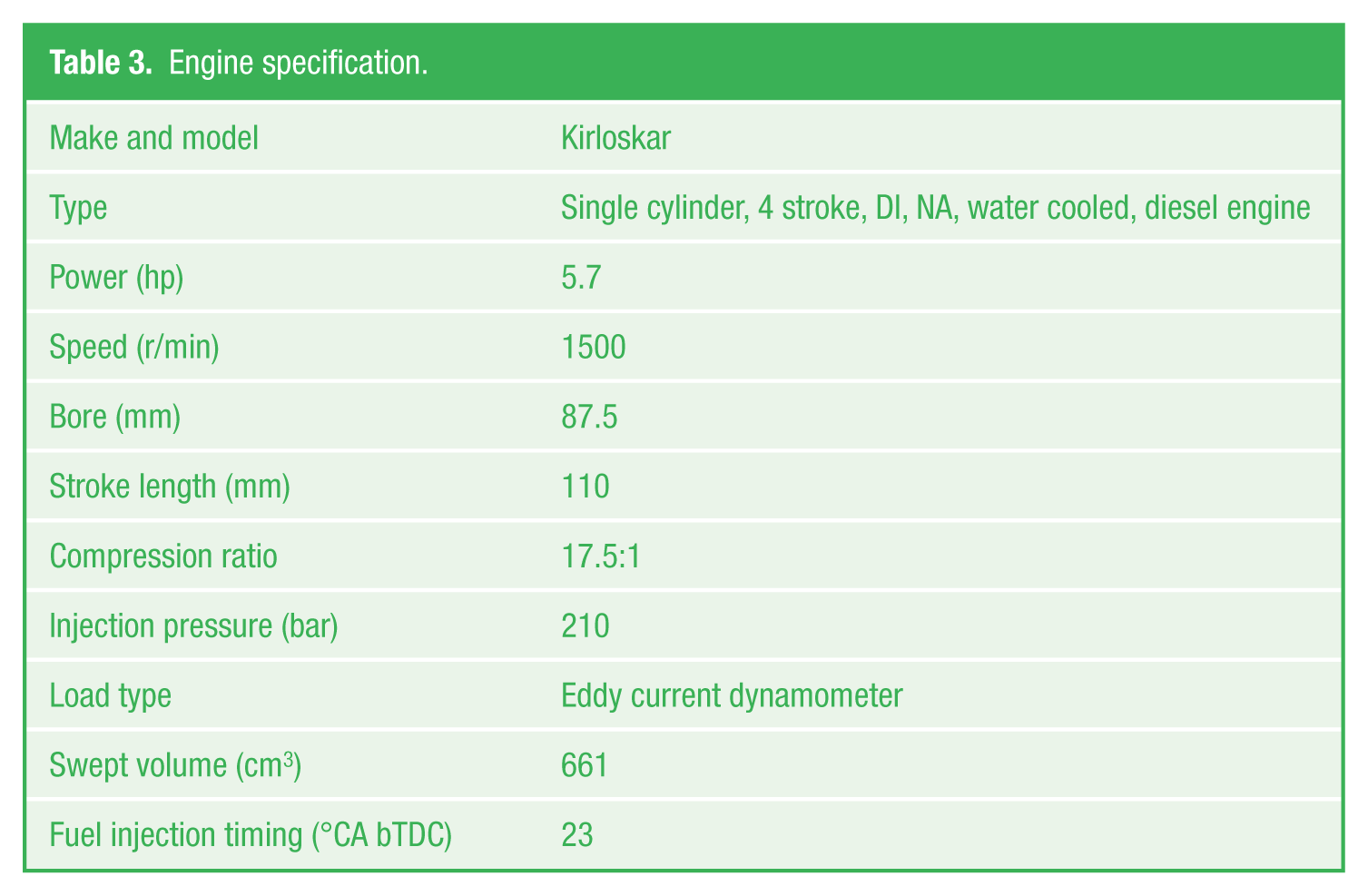
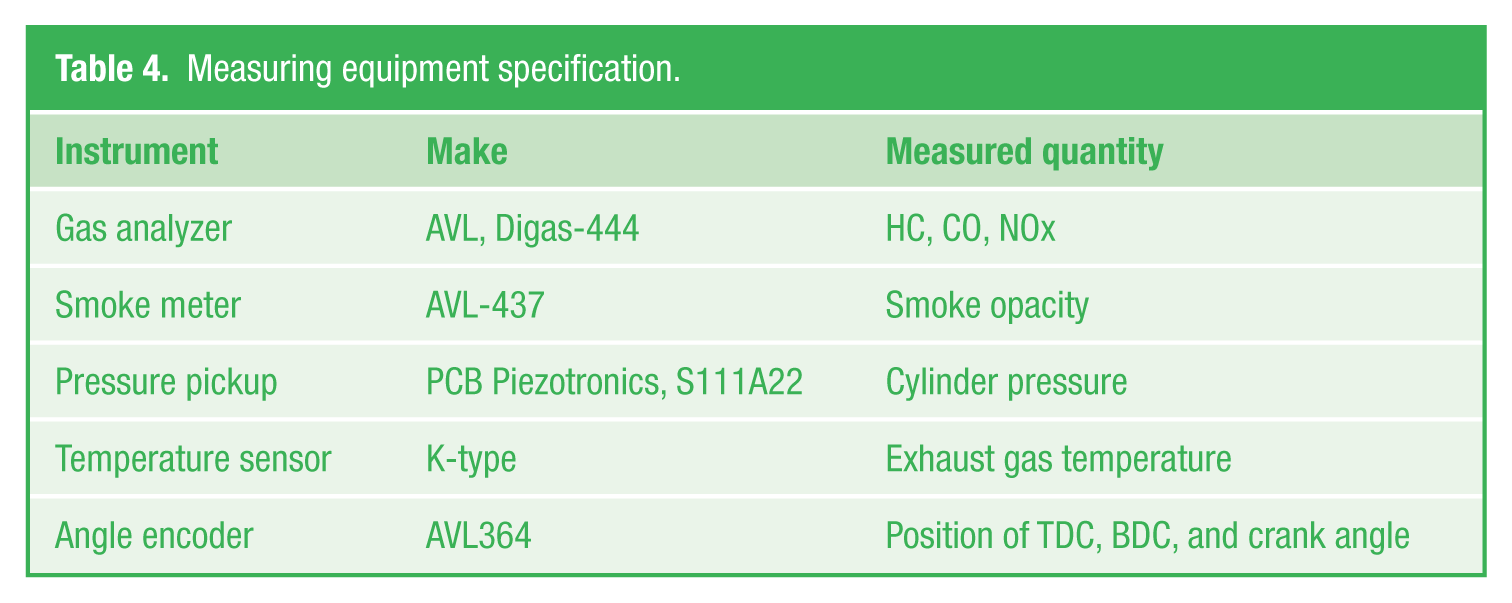
Performance and emission parameters of the various test fuels were measured at 0%, 25%, 50%, 75%, and 100% of the engine load. Engine was allowed to run to attain steady state for each loading and readings were taken. 24 Average data were considered for computation for all readings to minimize errors and to reduce the uncertainties in measurement. Specification of engine and various measuring equipments like temperature sensor, smoke meter, and gas analyzer are given in Tables 3 and 4.
Engine specification.
Measuring equipment specification.
III. Results and Discussion
A. Performance characteristics
Brake-specific fuel consumption
The brake-specific fuel consumption (BSFC) for all the test fuels at different engine loads is represented in Figure 3 . It can be noticed that BSFC of rubber seed biodiesel was always higher compared with diesel. BSFC normally decreases for all the test fuels with increase in the engine load, due to higher percentage increase in brake power. However, while analyzing the case of the variation in additives to the blended fuel, it shows that B20A2 (20% rubber seed biodiesel + 80% diesel + 0.5% pentanol + 0.5% MPL extract) exhibited closer result with diesel, especially at 75% of the engine load, compared to B20, B20A1, and B20A3. By investigation, it is very clear that lower calorific value (CV) of biodiesel can be balanced by injecting additional fuel into the combustion chamber in order to deliver the same power. The physical property of the pentanol and the antioxidant capability of MPL extract compensated the flaw and also helped in fuel efficiency.
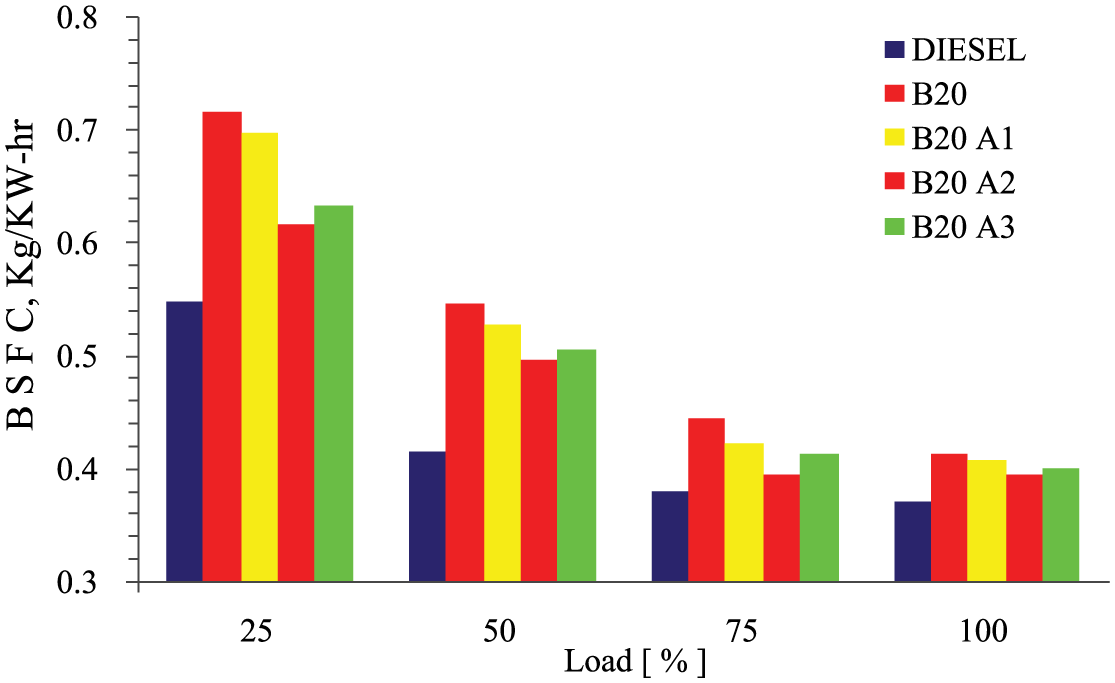
Variations of BSFC with load for different test fuels
Brake thermal efficiency
The deviation of brake thermal efficiency (BTE) against load for various test fuels is presented in Figure 4 . Reduction in heat loss, increase in power, and conversion of fuel energy into mechanical output were the indication of BTE. From the experimentation, it has been found that BTE increased with increase in engine load for all the test fuels. The maximum BTE obtained was about 27.98% for B20A2, which was quite higher than that for diesel (27.64%). The maximum BTE obtained for the other test fuels were 27.64% for diesel, 25.98% for B20, 26.96% for B20A1, and 27.24% for B20A3. Additional lubricity of the optimum level in the biodiesel improved the efficiency by converting all the fuel energy into possible mechanical output, minimizing the loss. Compared with the B20 blend, the entire additive mixed test fuels showed the elevated result which revealed good thermal efficiencies.
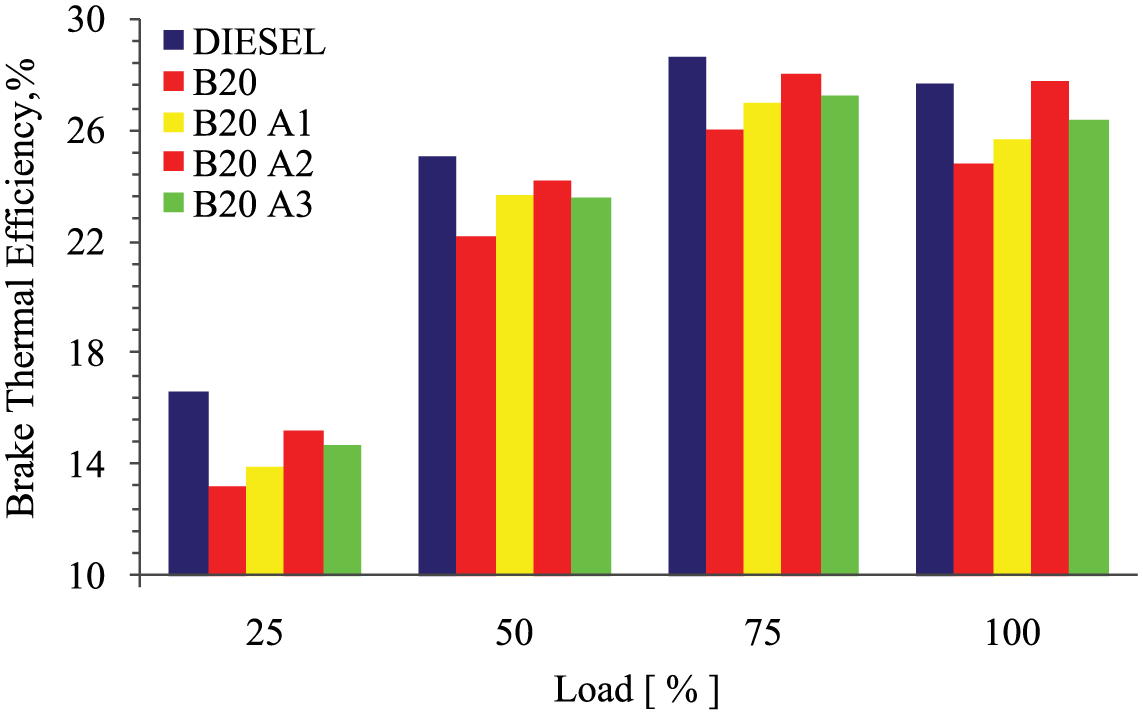
Variations of BTE with load for different test fuels
Exhaust gas temperature
Changes of exhaust gas temperature (EGT) with brake power for different test blends are portrayed in Figure 5 . EGT plays a key role in thermal NOx formation and also the indication of in-cylinder temperature, considering high oxygen concentrations in the fuel. Investigation states that EGT is always low for biodiesel blend and additive mixed biodiesel blend, irrespective of increase in the engine load. Compared with diesel, B20 showed higher reduction in EGT of 5.26%, followed by B20A2 of about 4.22%, B20A1 by 3.18%, and B20A3 by 2.26%. Deep-rooted analysis confirmed that oxygen content in the biodiesel led to complete combustion and the addition of pentanol, a high volatile fuel, prevented the ignition delay and the antioxidant additive from MPL ignored the combustion delay. Higher latent heat of vaporization of additive generated a cooling effect in the combustion period and therefore the exhaust temperature was minimized.
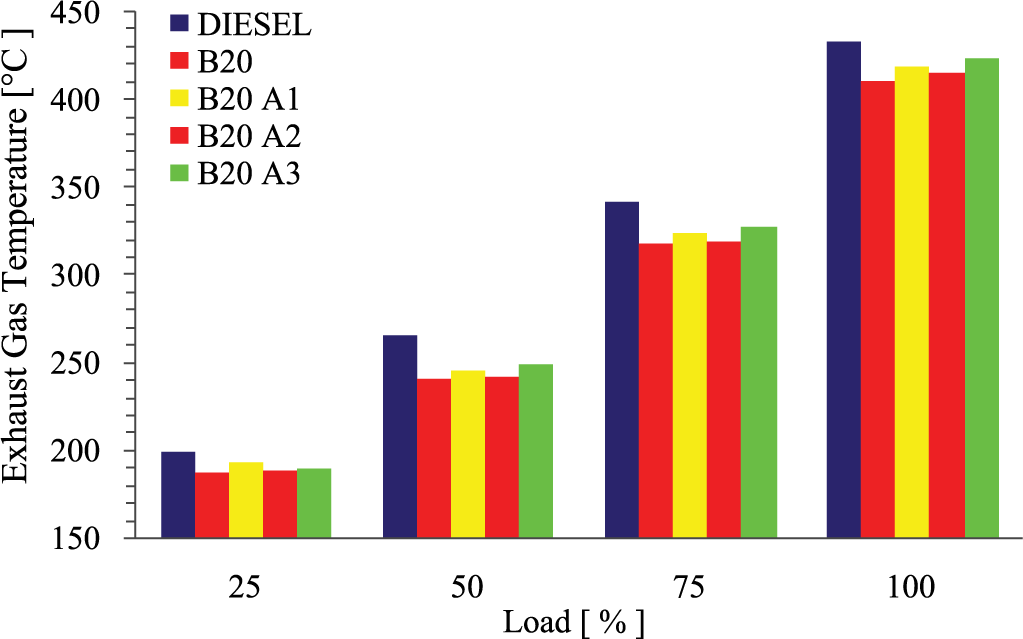
Variations of EGT with load for different test fuels
B. Emission characteristics
Carbon monoxide emission
Figure 6 represents the plots of carbon monoxide against the various loads for the test fuel in comparison with the baseline fuel. CO emission is the ideal emission product assessor. Elevated air–fuel ratio normally enhances the CO emission, as per the result obtained from the test. It is observed that the CO emission was significantly low for all the test fuels, because it depends on oxygen content, carbon content, and combustion efficiency of the fuel. In the present test fuels, pentanol contains long carbon chain, the biodiesel normally possesses higher oxygen content, and the antioxidant additives exhibit higher combustion efficiency. However, B20A2 showed higher reduction of CO emission of 16% than that of diesel, 3.5% less than B20, 2% less than B20A1, and 6.5% less than B20A3.
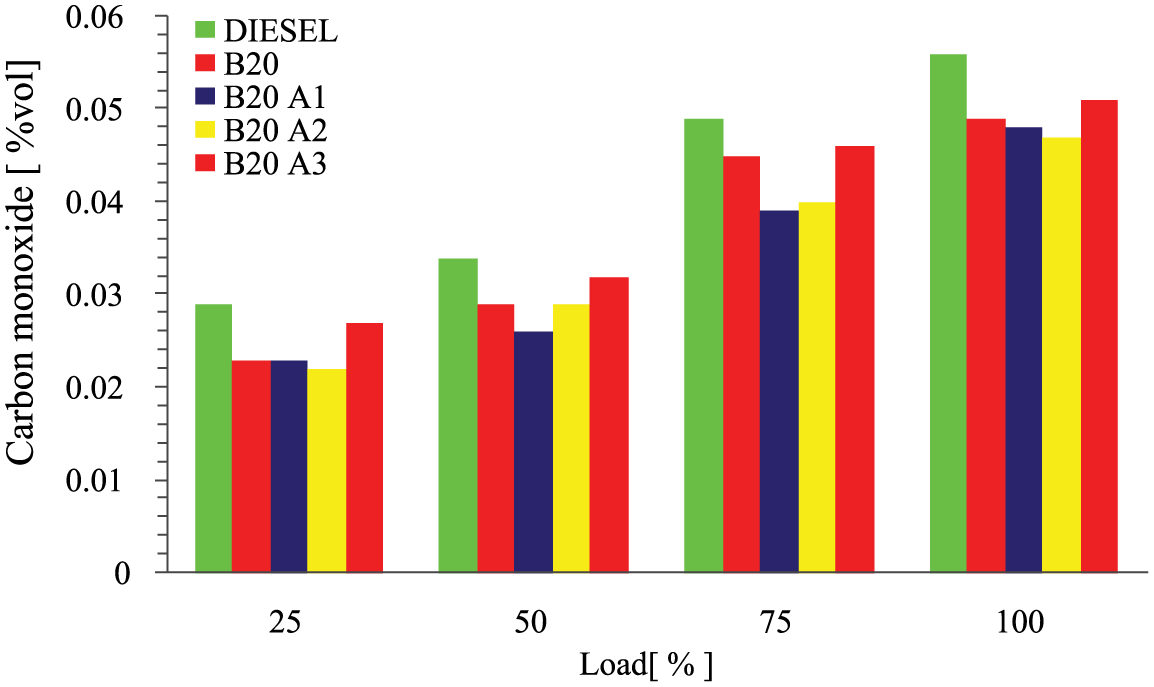
Variations of carbon monoxide emissions with load for different test fuels
Unburned hydrocarbon emission
The effect of hydrocarbon (HC) emission against the engine load for the various test fuel taken for analysis is demonstrated in Figure 7 . Over mixing of fuel and air beyond flammability limits leads to incomplete combustion, which is considered the root cause of HC emission. HC emission is affected by engine operating condition, fuel properties, and fuel spray characteristics. 25 The study of test fuels showed that the HC emission of the biodiesel was slightly higher due to viscous characteristics. However, addition of antioxidants lowered the viscous nature of the fuel and brought to light higher oxygen content and higher cetane number of biodiesel to perform an environmentally favorable emission. At medium load, HC emission was minimum when compared with low and high load. Considering all the test fuels, B20A2 exhibited lower emission in the order of B20A2 < B20A3 < B20A1 < B20 < diesel.
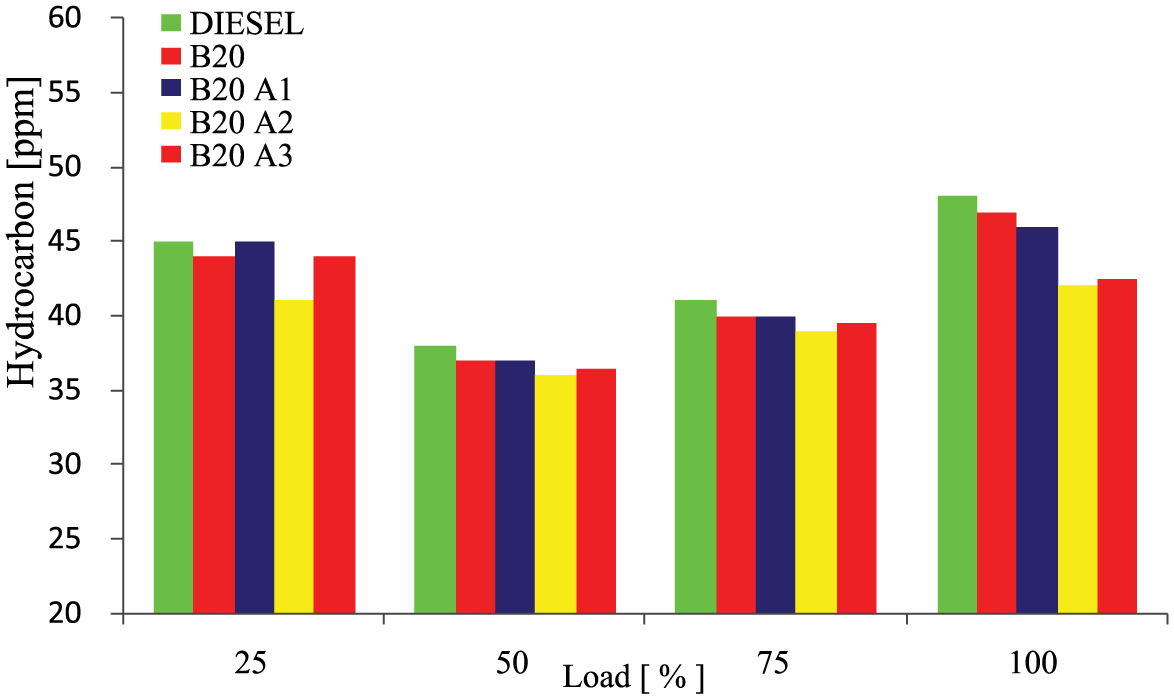
Variations of hydrocarbon emissions with load for different test fuels
NOx emission
Increase in the engine temperature is one of the key factors of oxides of nitrogen emission. Higher content of oxygen improves the emission of NOx. Higher combustion temperature promotes the NOx emission. Free radicals can also generate NOx formation reactions throughout the combustion of biodiesel within the flame, whereas mixing of antioxidants can probably hold back the contribution of these free radicals in the progression of NOx forming reactions. Figure 8 confirms the variation of NOx emission against the engine load for the various test fuels. NOx emission was found to be higher for all the test fuels when compared with base fuel. By mixing the additives with biodiesel, it supported in reducing the harmful NOx emission. Comparing with B20, the NOx was reduced when adding the antioxidant of MPL to biodiesel blend, of about 12% for B20A2 test fuel, 11.5% for B20A1 blend, and 4.5% for B20A3 blend. The adiabatic flame temperature was suppressed by the presence of ethanol in the additive and free radical slaking effect of carotenoid. When compared with diesel, B20A2 test fuel emitted 8% higher NOx.
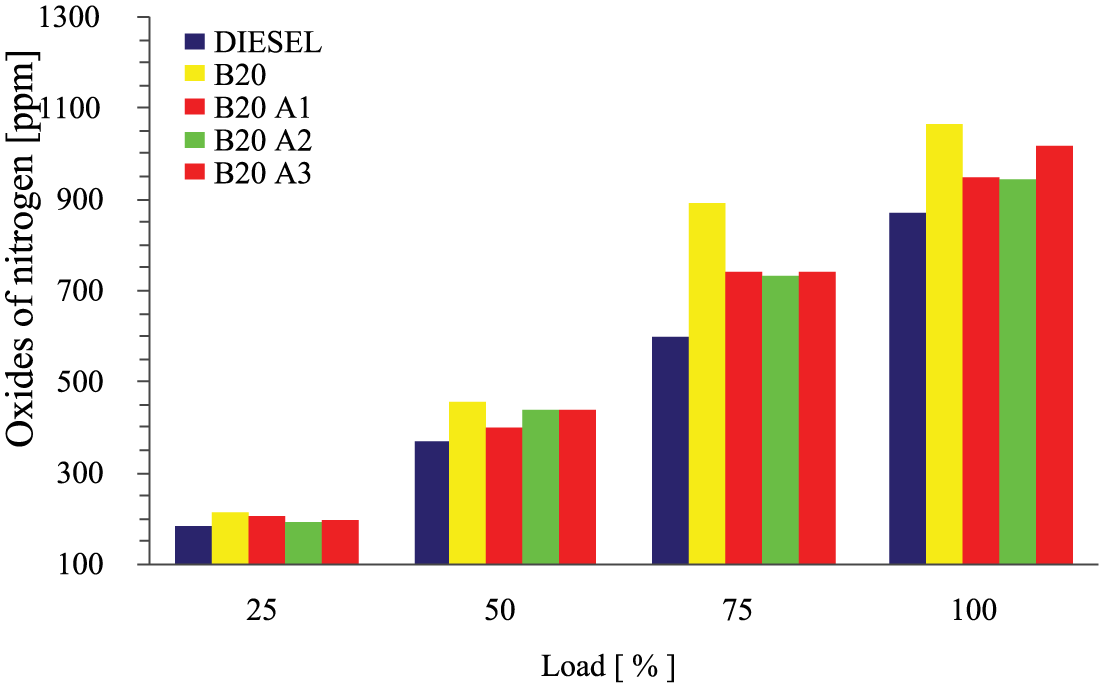
Variations of oxides of nitrogen emission with load for different test fuels
Smoke opacity
Figure 9 represents the effect of smoke emission against different engine load conditions for the various test blend fuels. Results ensure that for the entire test blend, smoke emission was found to increase when the engine loading was increased. The cause of increase in smoke emission was due to oversupply of fuel which could not be completely burnt in short duration during the high load period. The introduction of oxygenated fuel additives can overcome this issue. It is very clear from the experimental analysis that smoke emission diminishes with the boost in additive fraction in the fuel and stands in the order of B20A3 < B20A2 < B20A1 < B20 < diesel. Also it is found that the reduction of smoke emission was better in the 75% load compared with the full load condition.
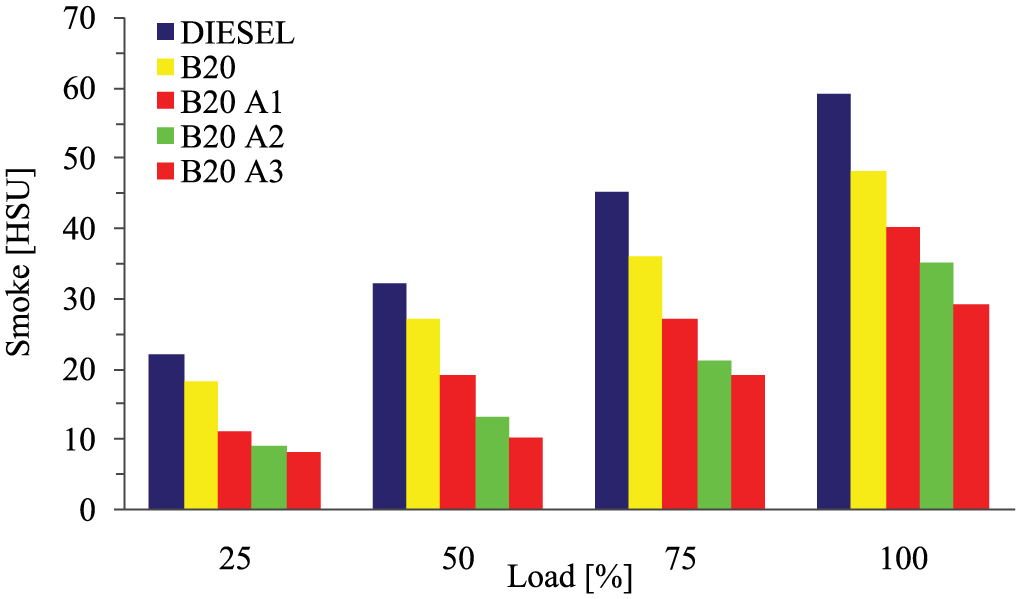
Variations of smoke emission with load for different test fuels
IV. Conclusion
The following conclusions are made based on the experimental analysis:
Antioxidants present in the additives support the oxidation stability, due to the availability of carotenoid and chlorophyll derivative in the MPL extract.
CO, HC, and smoke emission are significantly reduced when compared with diesel, in all load conditions.
Addition of the antioxidant reduces NOx emission of about 12% for B20A2 test fuel, when compared with B20 blend. But considering baseline fuel, NOx emission is slightly higher, of about 8%.
Investigation states that EGT is always low for biodiesel blend and additive mixed biodiesel blend in comparison with diesel. B20 shows higher reduction in EGT of 5.26%, followed by B20A2 of about 4.22%. The main reason behind is that B20 and the B20 with additives contain higher oxygen content that supports complete combustion.
BSFC is higher for all the test fuels other than diesel, and it is very clear that glutinous nature of the biodiesel blend and additive mixed biodiesel blends can be balanced by injecting more fuel into the combustion chamber.
The maximum BTE obtained is about 27.98% for B20A2, which is quite higher than that of diesel (27.64%). The maximum BTE obtained for the other test fuels are 27.64% for diesel, 25.98% for B20, 26.96% for B20A1, and 27.24% for B20A3.
Finally, it is concluded that the antioxidant derived from MPL extract and pentanol is jointly utilized as a prominent novel additive. It replaces conventional synthetic fuel additives whenever the biodiesel blends are used as fuel in diesel engines without any engine modification. This novel additive directly helps the CI engine to protect the environment by favorable emissions.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
