Abstract
Objective
To quantify the impact of performing challenging cognitive, physical and psychological tasks on subsequent cognitive performance, and whether differences in performance are predicted by psychological variables.
Background
Successful performance in many occupations depends on resilient cognition: the degree to which cognitive functions can withstand, or are resilient to, the effects of stress. Several studies have examined the effect of individual stressors on cognition; however, the capacity to compare different types of stress across studies is limited.
Method
Fifty-eight participants completed cognitive, physical, psychological and control interventions, immediately preceded, and followed, by a battery of cognitive tasks. Self-efficacy and cognitive appraisal were reported at baseline. Perceived stress was recorded post-intervention. Subjective workload was recorded for each cognitive battery and intervention.
Results
Cognitive performance was impaired by the cognitive, physical and psychological interventions, with the greatest effect following the cognitive intervention. The subjective workload reported for the post-intervention cognitive battery was higher following the cognitive and physical interventions. Neither self-efficacy, cognitive appraisal, perceived stress nor subjective workload of the intervention strongly predicted post-intervention performance.
Conclusion
Given the differences among interventions and cognitive domains, it appears that challenges to resilient cognition are broad and varied, and the mechanism(s) by which impairment occurs is complex.
Application
Considering the increase in subjective workload for the post-intervention cognitive battery, a combination of subjective and objective measures of cognitive performance monitoring should be considered.
In occupational settings such as emergency services, the military, construction and even sport, the stressors faced can be diverse and substantial. While physical adaptations to training and stress are well-understood, the responses of cognitive performance to acute stress are less established; yet cognition plays a substantial role in regulating many aspects of performance in these settings. Stressors that impair cognition include combinations of physical and mental exertion (Fogt et al., 2010), anxiety and stress (Oei et al., 2006), and unpredictable environments (Campbell & Nobel, 2009). While several studies have examined the effect of individual stressors on cognitive performance (Browne et al., 2017; Mazlomi et al., 2017; Ruf et al., 2017), the capacity to directly compare how different types of stress affect cognition across studies is limited, and a consequence of different participants and study protocols.
One avenue for understanding the common mechanism(s) by which different stressors influence subsequent cognitive performance relates to the concept of cognitive control. Cognitive control is used to describe the processes, or capacity, by which individuals manage goal-orientated behaviours against habitual tendencies or in the face of many choices (Badre & Nee, 2018; Norman & Shallice, 1986). Although often referred to as a single construct, cognitive control encompasses a broad range of behaviours and mental activity (Mackie et al., 2013). Importantly, cognitive control is required when attentional demands are greater than what can be maintained by an automatic process (Umemoto et al., 2019). Such tasks give rise to the sensation of effort and will feel difficult to perform continuously over time (Ackerman, 2011). When task demands are low, or when a task has been practised to the point at which it is considered an automatic behaviour, brain areas related to cognitive control are less engaged (Erickson et al., 2007; Weissman et al., 2002) and these tasks can be sustained, with little reduction in performance or a conscious sensation of effort (Ackerman, 2011).
The capacity to exert cognitive control is also thought to be limited, in the sense that performance on tasks requiring cognitive control is worse when such tasks are performed simultaneously or sequentially (Baumeister & Vohs, 2018). This effect holds whether or not the tasks are in the same domain. For example, performance on a cognitive task requiring response inhibition is impaired by walking on a treadmill for two hours, but only when participants are required to carry a heavy load and the task is sufficiently effortful (Eddy et al., 2015). Performing a computerised cognitive task for 30 min impairs maximal 5 km running performance (Pageaux et al., 2014) and the effortful act of suppressing one’s emotions reduces performance on an anagram solving task (Baumeister et al., 1998). The worse performance on the secondary task has been explained by theories that tasks of cognitive control deplete a common resource (Muraven & Baumeister, 2000) or that prior cognitive control exertion reduces motivation to allocate the resources required to meet task demands (Hagger et al., 2010; Robinson et al., 2010). Debate of such theory is not the focus of the current study; however, it remains important to understand the impact of tasks requiring cognitive control on subsequent cognitive performance, especially in situations such as occupational contexts where impaired cognition can have dire consequences.
Exploring cognitive control, this capacity is influenced by many factors, including psychological variables that underpin how our capabilities and experiences are appraised. Self-efficacy for example, is defined as a personal judgement of ‘how well one can execute courses of action required to deal with prospective situations’ (Bandura, 2010). Across various scenarios, self-efficacy can predict behaviour, including exerting cognitive control (Oman & King, 1998) and cognitive performance (Bouffard-Bouchard, 1990). Self-efficacy relating to the management of stressful situations appears to buffer negative consequences resulting from occupational stress, such as physiological reactivity (Schaubroeck & Merritt, 1997) and burnout (Salanova et al., 2002). Like self-efficacy, cognitive appraisal of a situation can impact both an individual’s performance (White, 2008) and their physiological response to a situation (Gaab et al., 2005b). Appraisal can be broadly divided into an initial assessment of the situation as a threat or challenge, and a secondary appraisal of the extent of coping options and perceived resources (Folkman & Lazarus, 1984).
The primary aim of this study was to quantify the impact of performing challenging cognitive, physical and psychological tasks – all requiring cognitive control – on subsequent cognitive performance. Second, we examined whether differences in cognitive performance between these interventions or individuals were predicted by self-efficacy towards, and/or cognitive appraisal of the intervention to be completed, or the perceived stress and/or subjective workload experienced during the intervention. We expected that cognitive performance would decrease following each of the experimental interventions, compared to the control, and given the common requirement of cognitive control, that impairment would be similar across the experimental interventions. When considering individual responses, we expected that those who reported higher perceived stress, or greater subjective workload or effort, would experience greater performance decline. We further hypothesised that greater self-efficacy, and a more favourable – low threat, high self-competence and control – cognitive appraisal of the intervention would attenuate the expected decrease in cognitive performance following the experimental interventions.
Method
Participants
Sixty participants volunteered to participate in the randomised cross-over study. This sample size was based on the assumption that for regression equations using six or more predictors (baseline cognitive performance, self-efficacy, primary appraisal, secondary appraisal, perceived stress and subjective workload), a minimum of 10 participants per predictor variable is required (VanVoorhis & Morgan, 2007). Fifty-eight participants (27 males and 31 females; 25 ± 5 years, maximal oxygen consumption 45.6 ± 10.9 mL/kg/min) completed the entire study protocol. Two participants withdrew following their second and third sessions, respectively, due to scheduling issues. This resulting sample size was supported by evidence that linear regression models require a minimum of two participants per variable for adequate estimation of regression coefficients, standard errors, confidence intervals and adjusted R2 (Austin & Steyerberg, 2015). This research complied with the tenets of the Declaration of Helsinki and was approved by University of Canberra Human Research Ethics Committee (Project Number 1504). Volunteers were eligible to participate if they were aged between 18 and 40 years, and free from injury, neurological condition or mental illness which could impact performance on any of the tasks. To control for the detrimental effect of sleep deprivation on cognitive performance, volunteers were excluded if they were night-shift workers or had a diagnosed sleep disorder (Alhola & Polo-Kantola, 2007). To ensure safety, only those who were deemed suitable based on a pre-exercise screening questionnaire (Exercise and Sport Science Australia Adult Pre-Exercise Screening Tool) were admitted to the study.
Experimental Design
Participants were required to attend the laboratory on five occasions. During Session 1, participants completed an incremental treadmill test to determine cardiorespiratory fitness (maximal oxygen consumption), followed by familiarisation of the cognitive battery, questionnaires and scales (Figure 1(a)). The incremental treadmill protocol used was a ramped Bruce protocol (Will & Walter, 1999). This test begins at a slow walking pace and increases in speed and/or incline every 15 seconds until the participant can no longer continue. Cardiorespiratory fitness was assessed due to the reported associations between fitness and cognitive performance (Fortune et al., 2019; Zhu et al., 2014), and fitness and the impact of physical exertion on cognitive performance (Chang et al., 2012). Familiarisation of the cognitive battery included written and verbal instructions and a 5 min rehearsal of each task. Schematic of experimental protocol. PVT = psychomotor vigilance task, SART = sustained attention to response task, n-Back = n-back task, Shift = set-shifting task.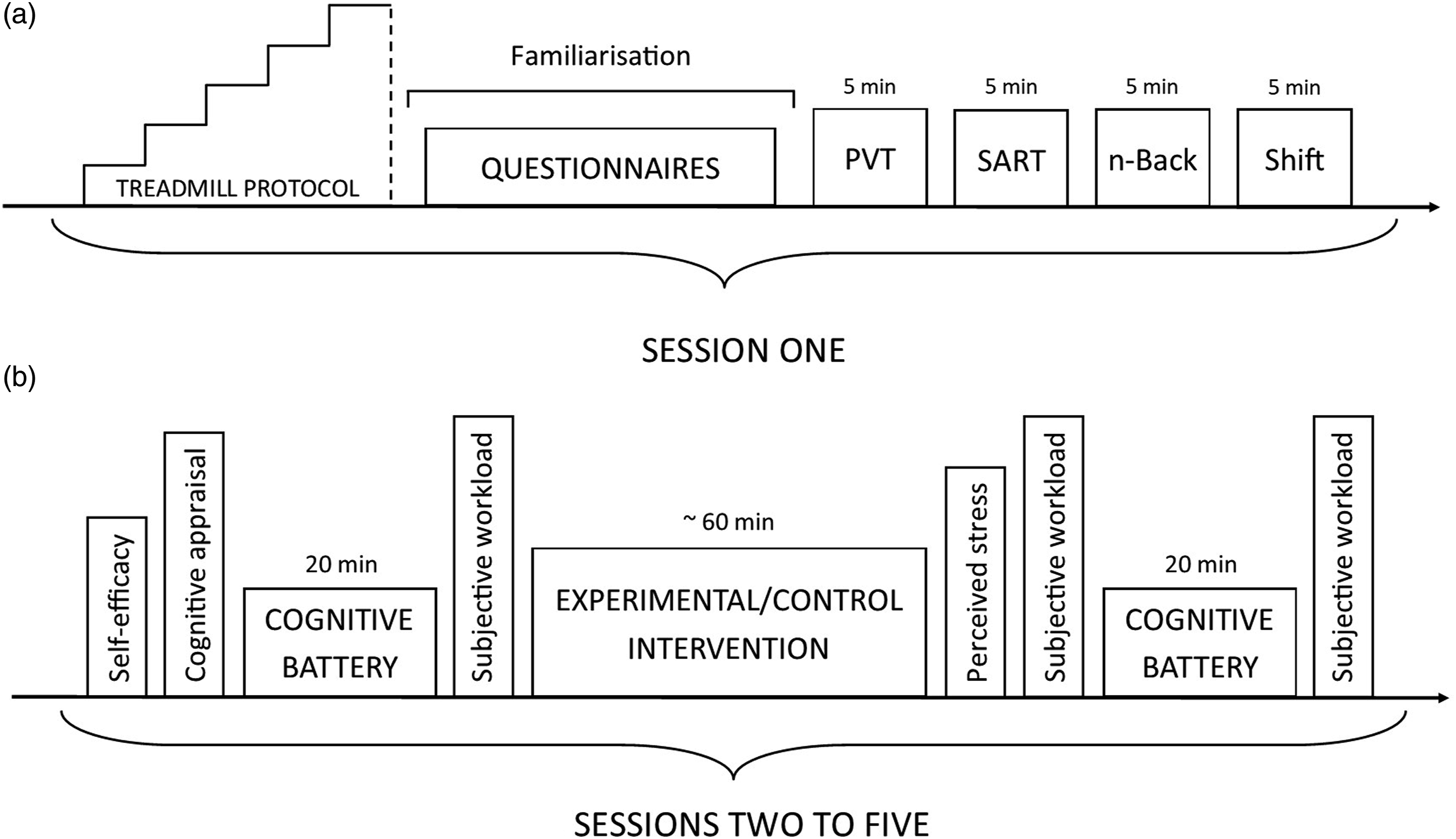
Sessions 2–5 were completed in a randomised order. During these sessions, participants completed either a cognitive, physical, psychological or control intervention lasting ∼60 min, immediately preceded and followed by a 20 min cognitive battery. Prior to the baseline cognitive battery, participants completed scales designed to assess self-efficacy and cognitive appraisal of the upcoming intervention. Upon completion, but prior to the post-intervention cognitive battery, subjective workload and stress experienced during the intervention were recorded (Figure 1(b)). Participants did not have access to their ratings of previous interventions.
Prior to each session, participants were given instructions to arrive at the laboratory hydrated and having eaten, to have slept for at least 7 hr, and refrained from the consumption of alcohol and vigorous exercise in the 24 hr prior, and caffeine in the 3 hr prior. Each participant completed the sessions individually and at the same time of day (within a ∼1 hr period). At the completion of the five testing sessions, participants were compensated for their time and travel costs with a $200 gift card.
Experimental and Control Interventions
Cognitive Intervention
The cognitive intervention consisted of participants performing a modified version of the Stroop colour-word task for 60 min. In this task, the four words ‘yellow’, ‘blue’, ‘red’ and ‘green’ were presented serially on a computer screen. The word remained on the screen until the participant recorded a response, followed by a 1.5 s rest interval. Participants were instructed to press one of four coloured buttons on the keyboard with the correct response being the button corresponding to the ink colour, except for when the ink colour was red, and the button to be pressed was the button linked to the written word. The word presented, and its ink colour, were randomly selected and all trials were incongruent. Participants were instructed to respond as quickly and accurately as possible and feedback was given after each response in relation to response speed and accuracy. This task has proved effective in inducing a state of mental fatigue (Martin et al., 2019), and versions of the task have been used to manipulate cognitive control demands (Englert & Wolff, 2015). Engagement in this intervention was monitored by accuracy. Mean accuracy was 97 ± 3% (mean ± SD).
Physical Intervention
The physical intervention consisted of walking on a treadmill at 6 km.hr−1 and 0% gradient for 60 min, wearing a weighted vest standardised to 30% of body mass. Persisting with an effortful physical task requires cognitive control to continue despite increasing discomfort and can result in subsequent impairments in cognitive control (Bray et al., 2008; Muraven et al., 1998). Throughout the treadmill task, heart rate was monitored continuously, and rating of perceived exertion (RPE) recorded using the Borg 6–20 scale (Borg, 1970) every 10 min. To standardise workload, the weight of the vest and/or the speed of the treadmill was adjusted, if required, to maintain a heart rate <130 beats.min−1 and a RPE between ‘somewhat hard’ (RPE 13) and ‘hard’ (RPE 15) throughout the entire intervention. Mean weight of the vest was 22 ± 5 kg (mean ± SD).
Psychological Intervention
The psychological intervention was adapted from Vansteenwegen et al., 2008, and designed to induce the negative emotional state of fear (Rachman, 1977) via a percutaneous muscle stimulation protocol. This emotional state will initiate a process of emotional regulation, known to draw on cognitive control resources (Ochsner & Gross, 2005). Fear induced through conditioning protocols have also been shown to require attention (Yates et al., 2010) and involve executive functioning (Carter et al., 2003). In this study, a neutral stimulus was paired with an aversive stimulus and participants learnt that the neutral stimulus pre-empted the aversive stimulus, as the stimuli were repeatedly paired. Once understood, the presentation of the neutral stimulus alone provokes the expression of a conditional fear response, reducing the need to expose the participant to the aversive stimulus (Knight et al., 2004). For this intervention, participants were seated upright with hips and knees flexed at 90° and straps placed across the chest, hips, and right ankle to prevent extraneous movement. A high-voltage constant current stimulator (DS7AH; Digitimer, Hertfordshire, UK) set to a maximum voltage of 400 V was used to provide muscle stimulation through two carbon rubber electrodes placed on the quadriceps of the right leg. During the first phase, participants received 20 x 100 Hz stimulations each lasting 250 ms, followed by 30 s rest. Following each stimulation, participants rated the pain experienced on a numerical rating scale anchored by no pain (0) to worst pain imaginable (10). Stimulation began at 20 mA and was increased gradually until the participant rated the stimulation at a pain level of 5. The stimulation intensity rated as a 5 was then multiplied by 1.5 and remained constant (113 ± 69 mA) throughout the remainder of the intervention. During the second phase (conditioning phase), a computer screen was placed in front of the participant upon which a black cross was presented on a white background (neutral stimulus) for a period of 30 s, followed by 15 s of a green screen (aversive stimulus). At the change of the screen from the aversive stimulus back to the neutral stimulus, participants received the 250 ms stimulation determined in phase 1, a process which was repeated 20 times. During the final phase (deconditioning phase), participants were presented with the same pattern of stimuli, however, electrical stimulation occurred pseudo-randomly. On 2 of the following 12 occasions stimulation occurred as per the conditioning phase, while on the other occasions stimulation did not occur.
Control Intervention
The control intervention consisted of watching Earth, a documentary film following the migration paths of animal families for 60 min (Fothergill & Linfield, 2007). This documentary was selected based on its engaging, yet emotionally neutral content, and its previous use as an activity with low cognitive control demand (Pageaux et al., 2013).
Cognitive Battery
During sessions 2–5, immediately preceding and following the intervention, participants completed a 20 min cognitive battery. The battery consisted of four 5 min tasks assessing the cognitive domains vigilance, response inhibition, working memory, and task-switching. The order of the tasks was randomised between participants, but consistent across the four sessions. For all tasks, participants were instructed to respond as quickly and accurately as possible. Responses faster than 100 ms were recorded as a false alarm and excluded from analysis. This value was chosen as the minimum reaction time needed for the processes of stimulus perception and motor response to occur (Whelan, 2008).
Vigilance and Sustained Attention
The Psychomotor Vigilance Task (PVT) was used to assess vigilance and sustained attention (Basner & Dinges, 2011). In this task, participants were instructed to monitor a red rectangular box on a black background for a yellow stimulus counter which would appear within the rectangle. As soon as the stimulus counter appeared, participants were required to press a response key that stopped the counter and displayed the reaction time in milliseconds for a period of 1 s. The inter-stimulus interval, defined as the period between the last response and the appearance of the next stimulus, varied randomly from 2 to 10 s. Mean reaction time was averaged across the task, excluding false alarms and lapses (responses slower than 500 ms (Basner & Dinges, 2011)).
Response Inhibition
The Sustained Attention to Response Task (SART) was used to assess sustained attention and response inhibition (Carter et al., 2013). In this task, participants were presented with a black cross in the centre of the screen for 900 ms, followed by presentation of a black number (1–9) for 250 ms. All stimuli were presented on a white background. Participants were instructed to press a response key as soon as the number, presented in a random order, appeared on screen, but withhold a response to the number 3. The number 3 target stimuli occurred with a probability of 1 out of 9. Errors of commission were defined as responding to a stimulus when a response was not required. Errors of omission were defined as a participant withholding a response when a response was required. Total accuracy and the percentage of errors of commission and omission were recorded across the task. Mean reaction time was recorded for correct responses.
Working Memory
A modified version of the n-Back Task was used to assess working memory (Scheibe & Blanchard-Fields, 2000). In this task, participants were presented with a black number (0–9) in the centre of a white screen for 500 ms, followed by a blank screen for 2.5 s before the next number was presented. Participants were instructed to press a response key on the left if the number on screen matched the number presented two numbers previously (target). Participants were instructed to press a response key on the right when the number on screen did not match the number presented two numbers previously (non-target). The numbers presented were randomly generated with 33% of the stimuli targets and 67% non-targets. Total accuracy and mean reaction time for correct responses were recorded.
Task-Switching
A set-shifting task was used to assess the cognitive construct task-switching and cognitive flexibility (Fong et al., 2014). In the set-shifting task (Fong et al., 2014), participants were presented with a cue stimulus in the centre of the screen for 500 ms, followed by a target stimulus for 1000 ms. One cue was a yellow triangle followed by a randomly generated target stimulus (either 1, 2, 3, 4, 6, 7, 8 or 9). For this cue and target combination, participants were instructed to press a response key on the left if the target stimulus was less than 5, and a response key on the right if the target stimulus was greater than 5. The other cue was a blue circle followed by a randomly generated target stimulus (1–9). For this cue and target combination, participants were instructed to press a response key on the left if the target stimulus was odd, and a response key on the right if the target stimulus was even. The presentation of the cues alternated randomly, although each task consisted of an equal number of the cue stimuli. Switch cost was calculated by subtracting the mean reaction time of responses with the same cue stimulus as the trial before from the mean reaction time of responses with a different cue to the response previous. Total accuracy, mean reaction time for correct responses, and switch cost were recorded across the task.
Subjective Measures
Self-Efficacy
The New General Self-Efficacy scale was used to quantify self-efficacy (Chen et al., 2001). For this measure, a single score was calculated by taking the average of the eight items, with a higher score indicating greater task self-efficacy. Cronbach’s reliability coefficients for this measure have been reported to range from 0.86 to 0.90 (Chen et al., 2001).
Cognitive Appraisal
The Primary Appraisal Secondary Appraisal scale was used to assess anticipatory cognitive appraisal (Gaab, 2009). Participants completed this scale following the New General Self-Efficacy scale, prior to the baseline cognitive battery. Participants were instructed to respond to the statements considering the upcoming intervention. Cronbach’s reliability coefficients range from 0.61 to 0.83 for primary appraisal and 0.74 to 0.80 for secondary appraisal (Gaab et al., 2005a).
Perceived Stress
A visual analogue scale was used to quantify perceived stress. Immediately following each of the interventions, participants were instructed to mark along a 100 mm horizontal line the level of stress experienced during the intervention anchored by the statements ‘not stressful at all’ on the far left and, ‘extremely stressful’ on the far right. Distance from the beginning of the line on the far left was recorded in millimetres. Perceived stress measured via a visual analogue scale has been shown to correlate with physiological indicators of stress including changes in heart rate and cortisol during a psychological stressor (Hellhammer & Schubert, 2012).
Subjective Workload
The National Aeronautics and Space Administration-Task Load Index rating scale (NASA-TLX) was used to quantify the subjective workload associated with performing each of the interventions and cognitive battery (Hart & Staveland, 1988). Participants were instructed to score each of the items on a scale divided into 20 equal intervals anchored by the descriptors very high and very low. This score was multiplied by five, resulting in a final score between 0 and 100 for each of the subscales, which was then averaged. Cronbach’s reliability coefficient for overall workload using this scale has been reported as 0.71 (Hoonakker et al., 2011).
Further detail regarding the self-efficacy and cognitive appraisal scales are found in the Supplementary Material.
Statistical Analysis
To investigate the impact of the interventions on subsequent cognitive performance, a two-way repeated measures analysis of variance (ANOVA) was used for the cognitive outcomes for each cognitive task, using
To investigate whether self-efficacy and cognitive appraisal, and perceived stress and subjective workload predicted post-intervention cognitive performance, we used separate hierarchical multiple regressions for each of the interventions. From the analysis conducted for our first aim, the PVT was determined as the most sensitive to the cognitive, physical, and psychological interventions, and thus, performance on this task was used as the dependent variable. Sex, age, and performance in the baseline cognitive battery were entered into the model first for the control, cognitive and psychological interventions. For the physical intervention, cardiorespiratory fitness was also included in the first step given the established relationship between fitness and the impact of physical exertion on cognitive performance (Chang et al., 2012). Self-efficacy, primary appraisal and secondary appraisal, and perceived stress and subjective workload of the intervention were then entered into the models, first individually and then combined, to examine their independent and additive contribution to the change in cognitive performance, beyond the variables entered in step 1. Significance was set at
Results
Impact of Interventions on Cognitive Performance
Repeated measures analysis of variance for the cognitive outcome’s reaction time, percentage of commission errors and switch cost.
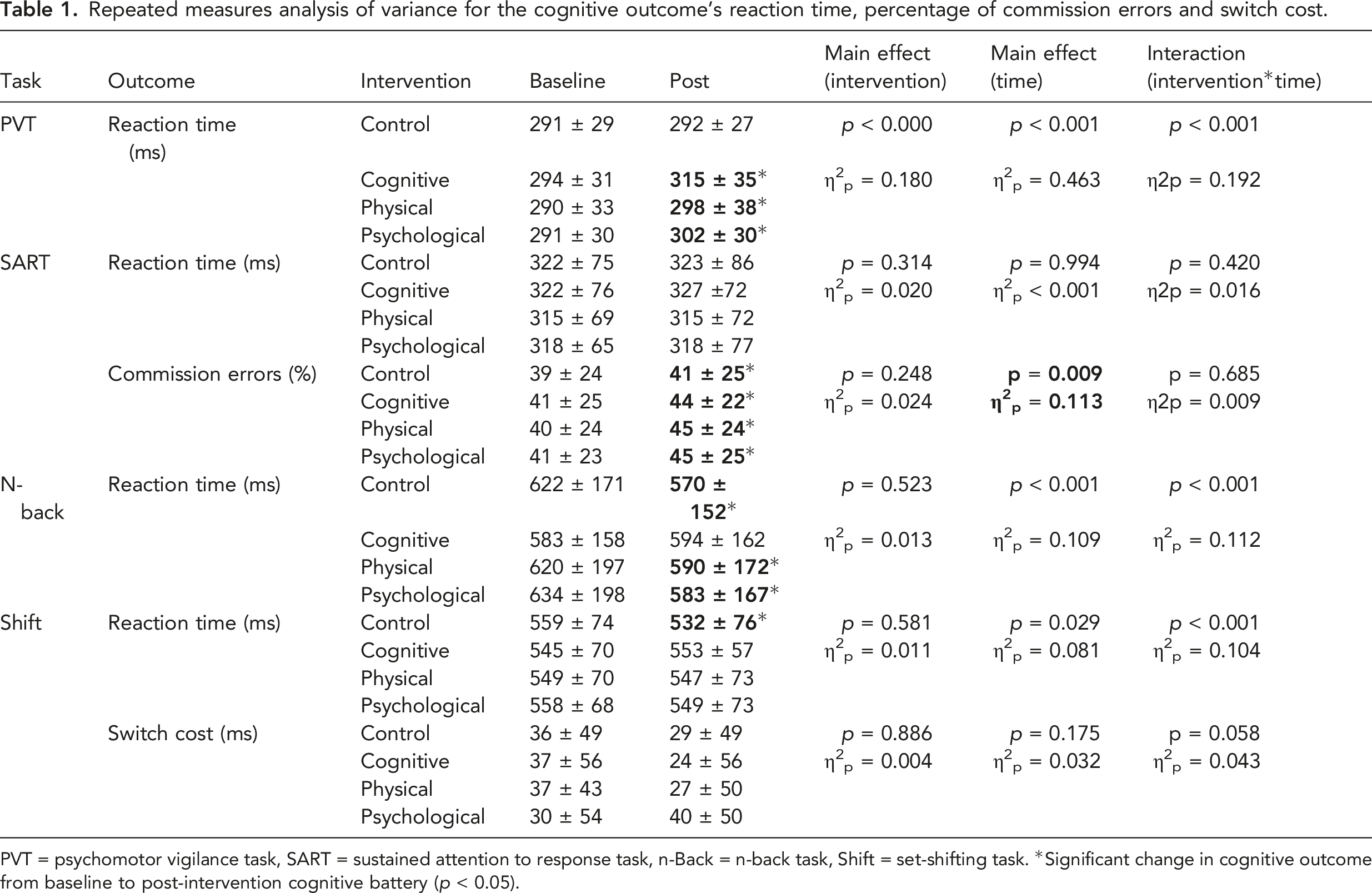
PVT = psychomotor vigilance task, SART = sustained attention to response task, n-Back = n-back task, Shift = set-shifting task. *Significant change in cognitive outcome from baseline to post-intervention cognitive battery (
Friedman’s analysis of variance for the cognitive outcome’s accuracy and percentage of omission errors.
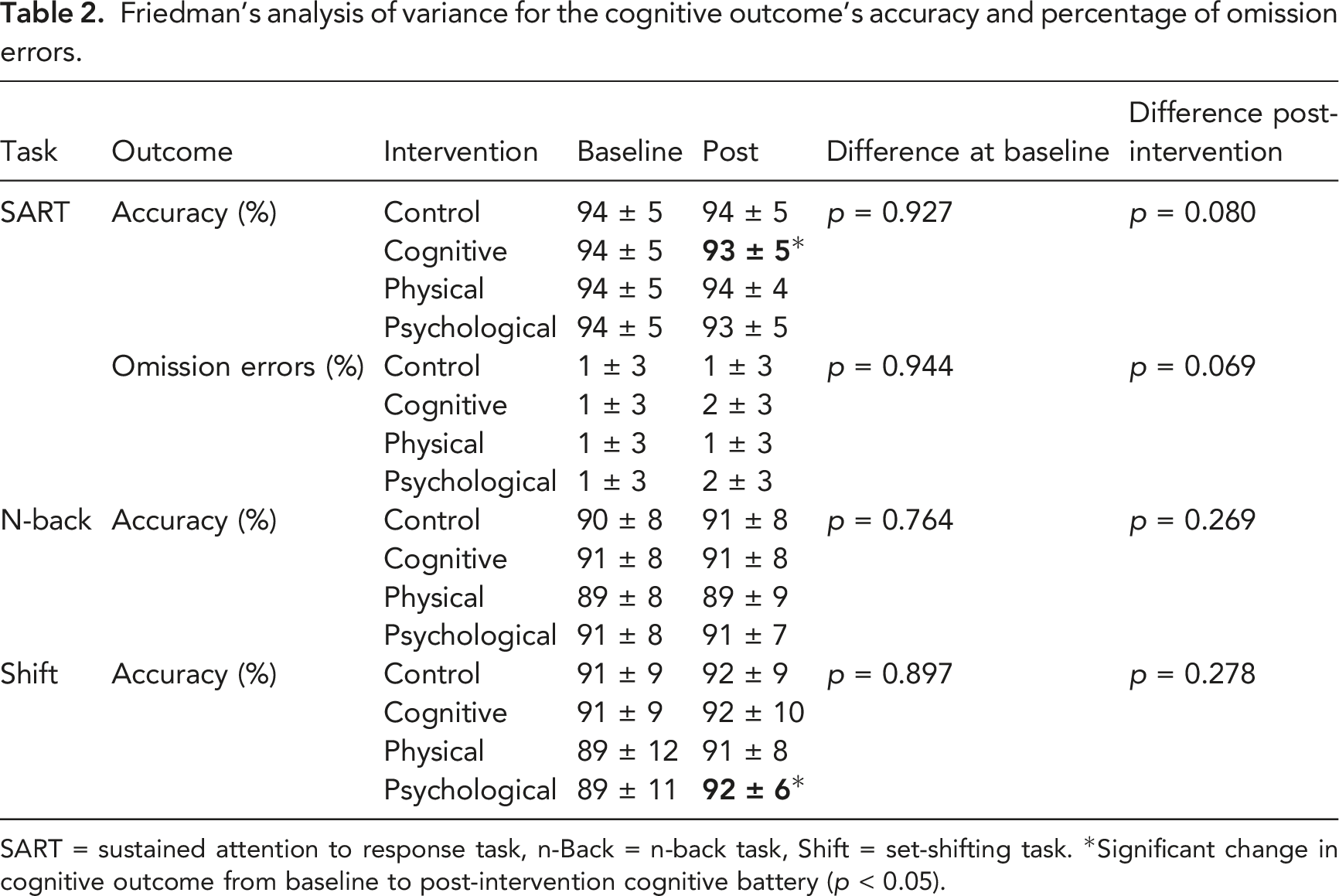
SART = sustained attention to response task, n-Back = n-back task, Shift = set-shifting task. *Significant change in cognitive outcome from baseline to post-intervention cognitive battery (
Performance on the n-Back task improved over time for all interventions except for the cognitive intervention. Accuracy was not different among interventions at baseline or post-intervention (Table 2). Accuracy was similar over time for the cognitive (
For the set-shifting task, accuracy was similar among interventions at both baseline and post-intervention (Table 2). Accuracy improved over
Impact of Intervention and Time on Subjective Workload of Cognitive Battery
The subjective workload for each of the cognitive batteries was influenced by the interventions. There was an interaction between Subjective workload for the baseline and post-intervention cognitive battery for each intervention. *Significant difference between baseline and post-intervention cognitive battery (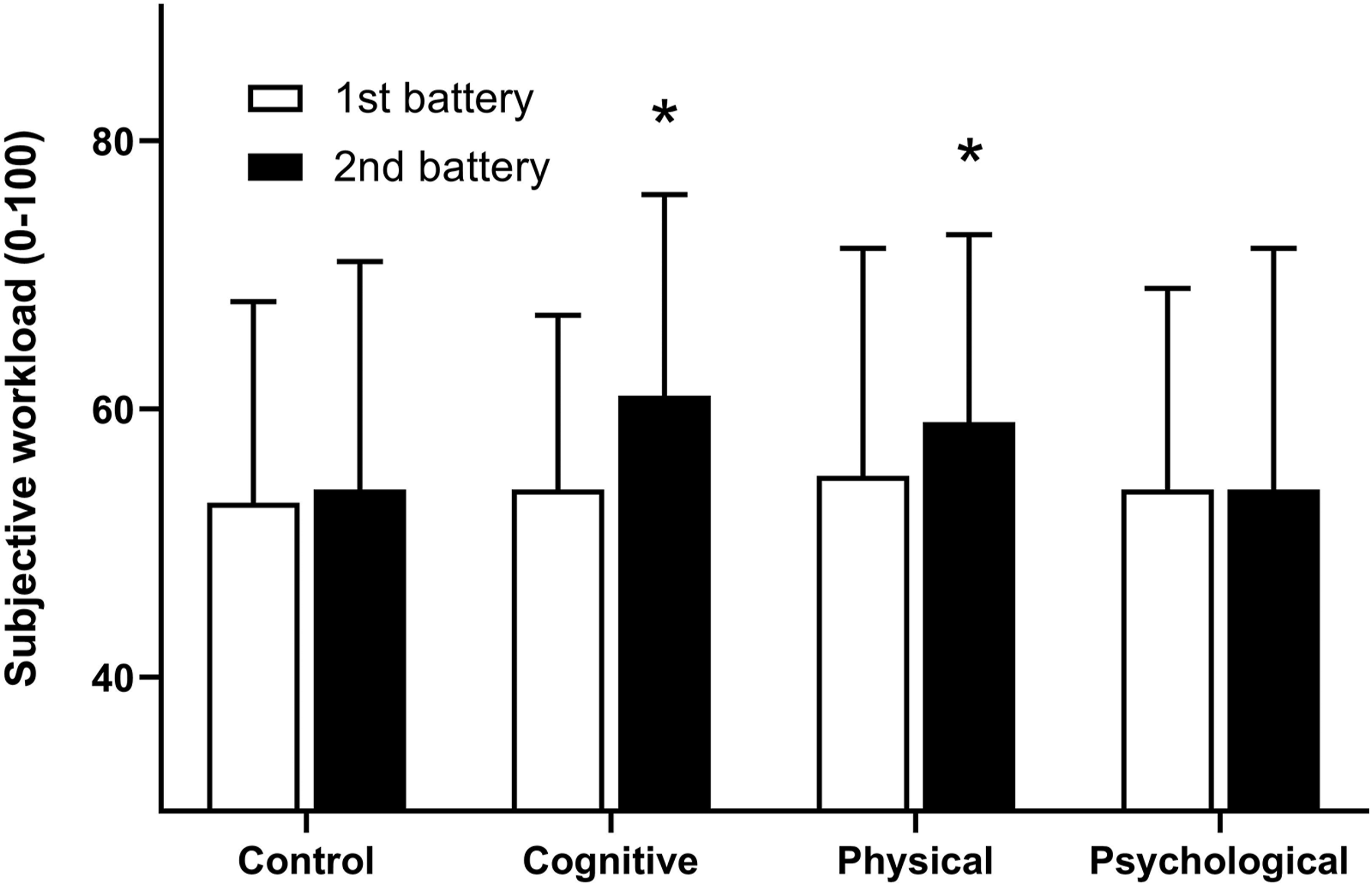
Influence of Self-Efficacy and Cognitive Appraisal on Post-Intervention Cognitive Performance
Hierarchical regression predicting mean reaction time for the psychomotor vigilance task in the post-intervention cognitive battery.
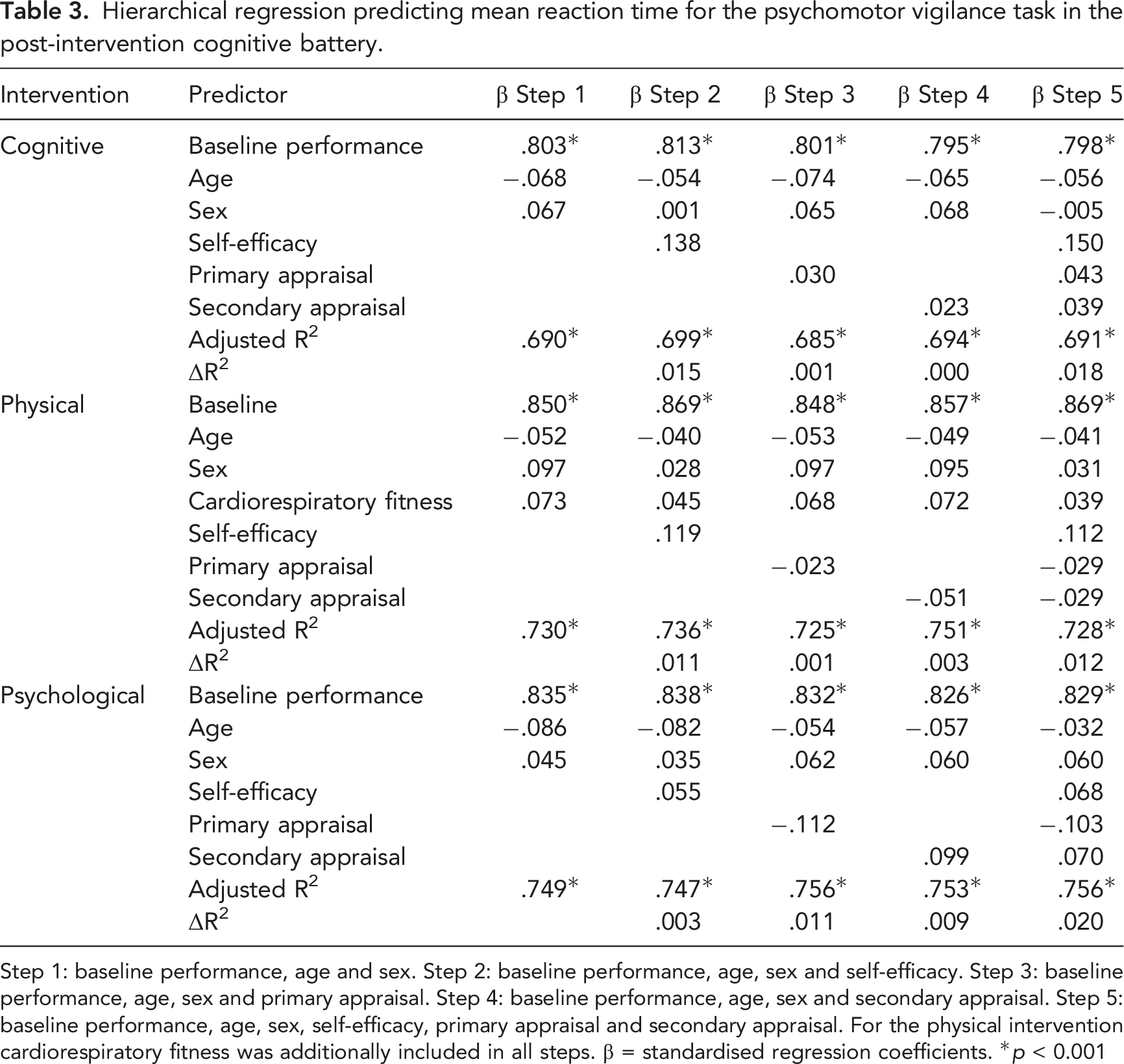
Step 1: baseline performance, age and sex. Step 2: baseline performance, age, sex and self-efficacy. Step 3: baseline performance, age, sex and primary appraisal. Step 4: baseline performance, age, sex and secondary appraisal. Step 5: baseline performance, age, sex, self-efficacy, primary appraisal and secondary appraisal. For the physical intervention cardiorespiratory fitness was additionally included in all steps. β = standardised regression coefficients. *
Influence of Perceived Stress and Subjective Workload of Intervention on Post-Intervention Cognitive Performance
Hierarchical regression predicting mean reaction time for the psychomotor vigilance task in the post-intervention cognitive battery.
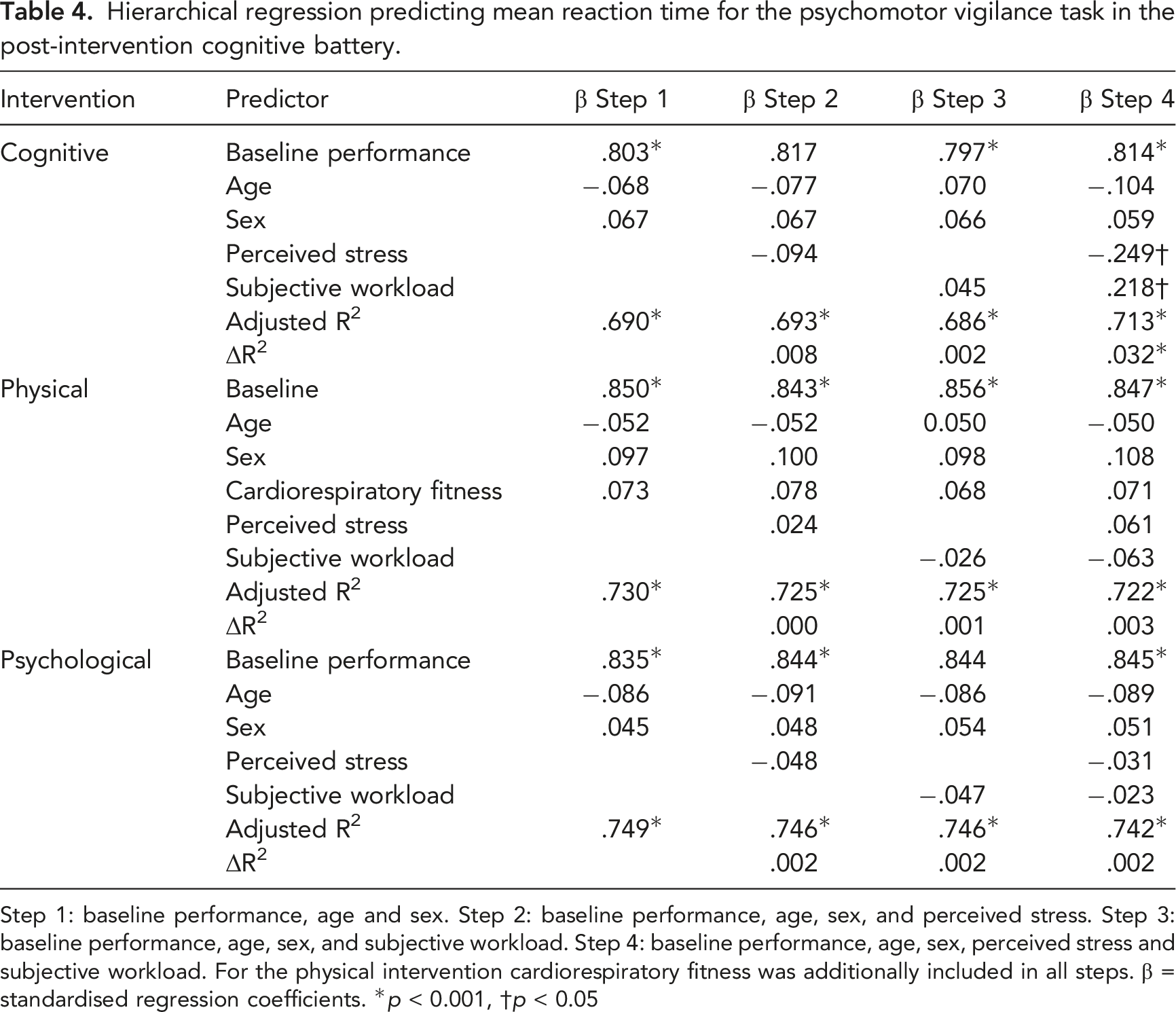
Step 1: baseline performance, age and sex. Step 2: baseline performance, age, sex, and perceived stress. Step 3: baseline performance, age, sex, and subjective workload. Step 4: baseline performance, age, sex, perceived stress and subjective workload. For the physical intervention cardiorespiratory fitness was additionally included in all steps. β = standardised regression coefficients. *
Discussion
In this study, we demonstrated that performing a task requiring cognitive control impairs subsequent cognitive performance, with impaired cognition most evident following the cognitive intervention. Following the cognitive intervention, mean reaction time on the PVT was 7% slower, compared to 3% and 4% slower following the physical and psychological interventions, respectively. Furthermore, performing the cognitive intervention was detrimental to accuracy on the SART, and reaction time on the n-Back Task, where a practise effect was observed, and reaction time was faster post-intervention for all interventions except the cognitive intervention. It appears that the impact of prior task performance is strongest when both tasks are similar in their domains (i.e., a cognitive task followed by another cognitive task), although some cross-domain effects, hypothetically related to the common requirement of cognitive control, were observed. The impaired performance of the PVT across all the experimental interventions indicates that we must broaden our consideration of potential challenges to cognition to include both physical and psychological stressors.
Alongside changes in cognition, subjective workload following the cognitive and physical interventions was higher compared to baseline, with no change over time for the control or psychological intervention. A higher perceived workload for the same objective workload may indicate greater effort or cognitive resources are required to overcome the effects of prior exertion. While increased effort may be maintained for short periods, when the duration of these tasks is extended, or the difficulty increased, the capacity to maintain performance at an acceptable level is likely reduced (Engle-Friedman, 2014). Indeed, the susceptibility of an individual to fatigue or performance decrements should not only be assessed via changes in performance but in combination with subjective measures. The observation of increased perceived workload, often described as compensatory effort, has been reported in sleep deprivation (Hockey et al., 1998) and mental fatigue (Matthews & Desmond, 2002). Compensatory effort is thought to protect tasks or activities important to the individual, or have implications for safety, with greater increases in effort associated with higher levels of performance protection (Hockey et al., 1998). However, when cognitive demands are low, performance may be more susceptible to decrements (Matthews & Desmond, 2002), an outcome predicted by the effort regulation hypothesis (Matthews et al., 1996). This hypothesis suggests that when task demands are low, the need for active performance regulation is less apparent and performance is often impaired. In this study, this theory may explain the greatest performance decrements on the simple PVT, compared to the tasks of response inhibition, working memory and task-switching. Given the inherent increased engagement required by these tasks, with moderate levels of increased effort such tasks may be less vulnerable to performance impairment because of the already high levels of engagement. In any case, the capacity of an individual to maintain increased effort should be considered when tasks become prolonged, more demanding or unpredictable.
After controlling for age, sex and performance on the baseline cognitive battery, self-efficacy towards and cognitive appraisal of the interventions were unable to predict subsequent cognitive performance. Under various conditions, self-efficacy is predictive of performance (Bouffard-Bouchard, 1990) and behaviour (Oman & King, 1998) and may mediate an individual’s ability to manage a stressful situation (Schaubroeck & Merritt, 1997). Self-efficacy is associated with task persistence (Bouffard-Bouchard, 1990) and thought to encompass a motivational component that determines the duration and intensity of an individual’s engagement in certain behaviours to achieve a desired outcome (Bandura, 1977). In this study, participants were required to evaluate self-efficacy towards the interventions, not performance on the post-intervention cognitive battery. We expected that self-efficacy towards the intervention might influence the cognitive control required by the intervention, and hence, performance in the post-intervention cognitive battery. Similarly, we expected that appraising the intervention as a challenge, as opposed to a threat, would minimise the impact on performance. This does not appear to be the case and the distinction between a challenge and a threat may be crucial in determining the usefulness of similar measures in future. Measurement of self-efficacy at another timepoint, such as after the intervention and prior to the post-intervention cognitive battery, may have elicited different results. It also appears that as participants were unfamiliar with the interventions, the lack of experience with the interventions and the perception of ability and resources required to cope with these interventions, may have reduced the variability among interventions and individuals. Indeed, increased experience across different contexts tends to increase self-efficacy (Fagan et al., 2004; Prieto & Altmaier, 1994). Such measures may be more useful in occupational contexts where task requirements are better understood.
In addition to self-efficacy and cognitive appraisal, we expected that perceived stress and/or subjective workload of the interventions would predict changes in cognitive performance. Subjective workload was reported on 3 occasions during each intervention, following performance of the cognitive batteries, as well as the intervention itself. Tasks which require cognitive control are described as effortful (Umemoto et al., 2019), however, effort is subjective and depend on a person’s self-efficacy, appraisal, and ability. The perceived stress and subjective workload of the tasks likely differs among individuals, and its impact on subsequent cognitive performance. In the current study, the combination of perceived stress and subjective workload for the intervention predicted performance on the post-intervention cognitive battery, beyond baseline and demographic variables. However, this additional variance explained was only for the cognitive intervention, and only accounted for a small effect. The explanation for why these perceptual measures were only predictive for the cognitive intervention is unclear but may relate to this intervention having the greatest impact on subsequent performance. Further, the inability to identify the hypothesised stronger relationship among perceived stress, subjective workload and post-intervention cognitive performance may relate to the small magnitude of impairment in the experimental interventions. This effect may have been partially accounted for by the concept of compensatory effort.
Conclusion
We examined the impact of performing cognitive, physical, and psychological tasks – all requiring cognitive control – on subsequent cognitive performance, and whether performance could be predicted by self-efficacy, cognitive appraisal, perceived stress and subjective workload of the tasks. The effects of prior task performance were apparent across the interventions, with the impact most apparent on tasks with similar demands. Apart from the impact on performance, the cognitive and physical interventions increased subjective workload on the post-intervention cognitive battery, indicating an increase in effort or cognitive resources required to overcome the negative effects of these interventions. For application in the real-world, we suggest that in occupations where the challenges to cognition are multi-faceted, both subjective and objective measures of performance monitoring should be included to identify the risk of impending decrements in performance. Although psychological measures contributed little to the explained variance in performance across the interventions, small adjustments in methodology (such as the timing of questionnaires), more demanding experimental interventions and longer cognitive tasks may yield different results. Using more prolonged experimental or cognitive tasks, or multiple concurrent tasks requiring cognitive control, may better replicate real life, stress the individual, and challenge subsequent cognition.
Key Points
Performing a challenging cognitive, physical or psychological task can impair subsequent cognitive performance and should be avoided when optimal cognition is required. A preceding challenging task can increase the effort required by a subsequent cognitive task, without influencing performance. Monitoring subjective workload may identify fatigue and precede a decline in performance.
Supplemental Material
sj-pdf-1-hfs-10.1177_00187208211065548 - Supplemental material for The Impact of Cognitive, Physical, and Psychological Stressors on Subsequent Cognitive Performance
Supplemental material, sj-pdf-1-hfs-10.1177_00187208211065548, for The Impact of Cognitive, Physical, and Psychological Stressors on Subsequent Cognitive Performance by Kristy Martin, Andrew Flood, David B. Pyne, Julien D. Périard, Richard Keegan, and Ben Rattray in Human Factors: The Journal of Human Factors and Ergonomics Society
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Defence Science and Technology Group, in collaboration with the Australian Army, Human Performance Research Network (201941).
Supplementary Material
The online supplemental material is available with the manuscript on the
Kristy Martin is an assistant professor in the Research Institute for Sport and Exercise at the University of Canberra. She received her PhD in sport and exercise science from the University of Canberra in 2017.
Andrew Flood is a lecturer in the Discipline of Psychology at the University of Canberra. He received his PhD in sport and exercise psychology from the University of Canberra in 2018.
David B. Pyne is a professor in the Research Institute for Sport and Exercise at the University of Canberra. He received his PhD in biochemistry and molecular biology from the Australian National University in 1995.
Julien Périard is an associate professor in the Research Institute for Sport and Exercise at the University of Canberra. He received his PhD in exercise physiology from the University of Sydney in 2011.
Richard Keegan is an associate professor at the Research Institute for Sport and Exercise, University of Canberra. He received his PhD in sport and exercise psychology from Loughborough University in 2010.
Ben Rattray is an associate professor in the Research Institute for Sport and Exercise at the University of Canberra. He received his PhD in exercise physiology from the University of Sydney in 2009.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
