Abstract
Objective
Surgeon tremor was measured during vitreoretinal microscopic surgeries under different hand support conditions.
Background
While the ophthalmic surgeon’s forearm is supported using a standard symmetric wrist rest when operating on the patient’s same side as the dominant hand (SSD), the surgeon’s hand is placed directly on the patient’s forehead when operating on the contralateral side of the dominant hand (CSD). It was hypothesized that more tremor is associated with CSD surgeries than SSD surgeries and that, using an experimental asymmetric wrist rest where the contralateral wrist bar gradually rises and curves toward the patient’s operative eye, there is no difference in tremor associated with CSD and SSD surgeries.
Methods
Seventy-six microscope videos, recorded from three surgeons performing macular membrane peeling operations, were analyzed using marker-less motion tracking, and movement data (instrument path length and acceleration) were recorded. Tremor acceleration frequency and magnitude were measured using spectral analysis. Following 47 surgeries using a conventional symmetric wrist support, surgeons incorporated the experimental asymmetric wrist rest into their surgical routine.
Results
There was 0.11 mm/s2 (22%) greater (p = .05) average tremor acceleration magnitude for CSD surgeries (0.62 mm/s2, SD = 0.08) than SSD surgeries (0.51 mm/s2, SD = 0.09) for the symmetric wrist rest, while no significant (p > .05) differences were observed (0.57 mm, SD = 0.13 for SSD and 0.58 mm, SD = 0.11 for CSD surgeries) for the experimental asymmetric wrist rest.
Conclusion
The asymmetric wrist support reduced the difference in tremor acceleration between CSD and SSD surgeries.
Background
Hand tremor is a physiological phenomenon where rhythmic, oscillating movements occur when antagonistic muscles co-contract (Morrison & Newell, 1999; Smaga, 2003; Wade et al., 1982). These oscillations arise from a larger mechanical-reflex and smaller central-neurogenic origins (Burne et al., 1984; Elble, 2003; Elble & Randall, 1976; Lippold, 1970). Normal elbow tremor falls between 3 and 5 Hz, wrist tremor between 7 and 10 Hz, and metacarpal joint tremor between 17 and 30 Hz (Elble, 2003).
Tremor can significantly impact performance in microscopic surgery where high levels of fine motor dexterity are required (Mürbe et al., 2001). Surgeon tremor can greatly influence the outcome of certain, precise, microsurgical operations (Coulson et al., 2010). Tremor can impede the performance of challenging microsurgical tasks (Elble, 2003), has been deemed a major limitation (Vasilakos et al., 1998), and is called “the enemy” of delicate surgery (MacDonald, 2005) and “the root cause for human imprecision during microsurgery” (Verrelli et al., 2016).
Upper extremity support has been shown to reduce surgeon hand tremor (Arnold et al., 1993; Coulson et al., 2010; Evans, 1978; Hara et al., 2015; Nissenbaum et al., 1979). The effects of wrist support location on tremor have also been previously studied. Placing support proximal to the wrist (i.e., forearm) has been considered superior than elbow support alone. Direct support at the level of the hand paradoxically produces increased finger tremor compared to the forearm. There is a compensatory antitremor effect of having increased degrees of freedom at the wrist compared to when the hand is supported (Safwat et al., 2009). Morrison and Newell (1999) also observed the greatest decrease in finger tremor when the finger and hand were free to move. Unrestricted movement about the wrist joint resulted in significantly less tremor in the index finger. This suggests a compensatory synergy around the wrist joint that is critical in the upper limb during postural tasks. The wrist may act as a fulcrum and the addition of specific joint space degrees of freedom there in tremor tasks reduces the degree of oscillation of the index finger (Morrison & Newell, 2000). Forearm support has been demonstrated to have biomechanical advantages to hand or wrist support in computer operators (Aarås et al., 1998, Aarås et al., 2001; Cook et al., 2004a, 2004b; Lee & Huang, 2006). Support at the level of the forearm rather than just hand support may offer advantages for the surgeon and also computer operators.
In vitreoretinal surgery, surgeon’s both hands may be supported by just resting them on the patient’s forehead without even using a wrist rest. Alternatively, symmetric U-shaped wrist rests (Varitronics Inc., 2018; Western Ophthalmics, 2018) are commonly used. The operative eye is not at the geometric center of the U-shaped symmetric wrist rest (SWR), like the nose or mouth is. The average interpupillary distance is 61.7 mm for women and 64.0 mm for men (Dodgson, 2004; Smith & Atchison, 1997). Simply put, the average center of the eyes is approximately 31.5 mm lateral to the midline. This places the operative eye closer to the adjacent wrist rest bar than the opposite bar as illustrated in Figure 1. When operating right handed on the left eye, the distance from the surgeon’s right hand to the left eye increases (the white dashed line represents the additional distance away from the eye). Due to this horizontal asymmetry and the additional requirement for the hand opposite the operative eye to elevate over the nasal bridge, the SWR does not support both hands simultaneously. If the SWR was elevated enough to permit right hand support for left eye surgery, the left hand would be too high limiting hand mobility (too steep of an angle to the eye) for surgery.
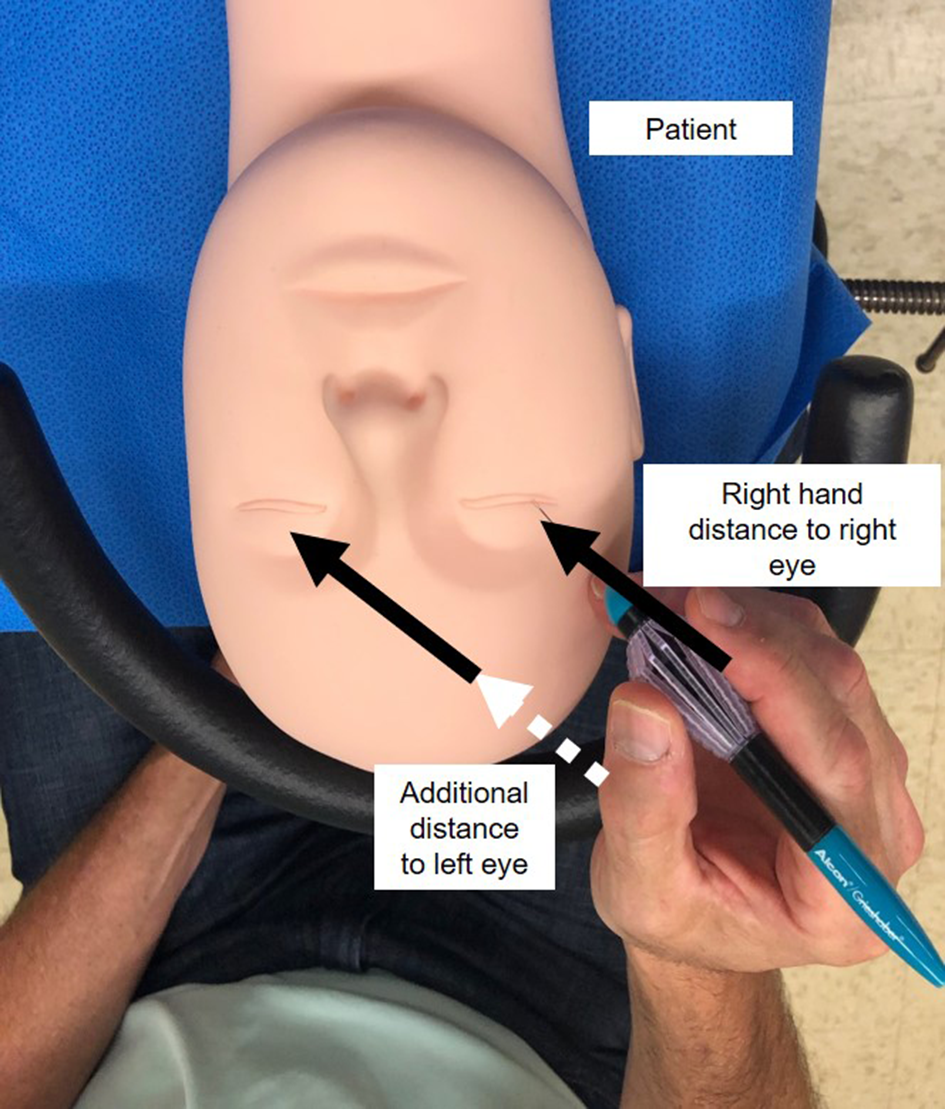
Wrist rest for the right hand is much closer to the right eye than the left eye and the surgeon’s right hand must elevate over the nasal bridge for access to the left eye.
The SWR can therefore provide good support for the right hand in right eye surgery, particularly helpful for the right-hand dominant surgeon. The left hand for surgery on the right eye simply rests on the patient’s forehead and may not be required to perform the most dexterous parts of microsurgery. Conversely, for left eye surgery, the left hand is well supported with the SWR, but the dominant right hand must rely upon the patient’s forehead for support (Figure 2). The principles are just reversed for the left-hand dominant surgeon, with the dominant hand being well supported by the SWR for left eye surgery but forehead support is required for the right eye.
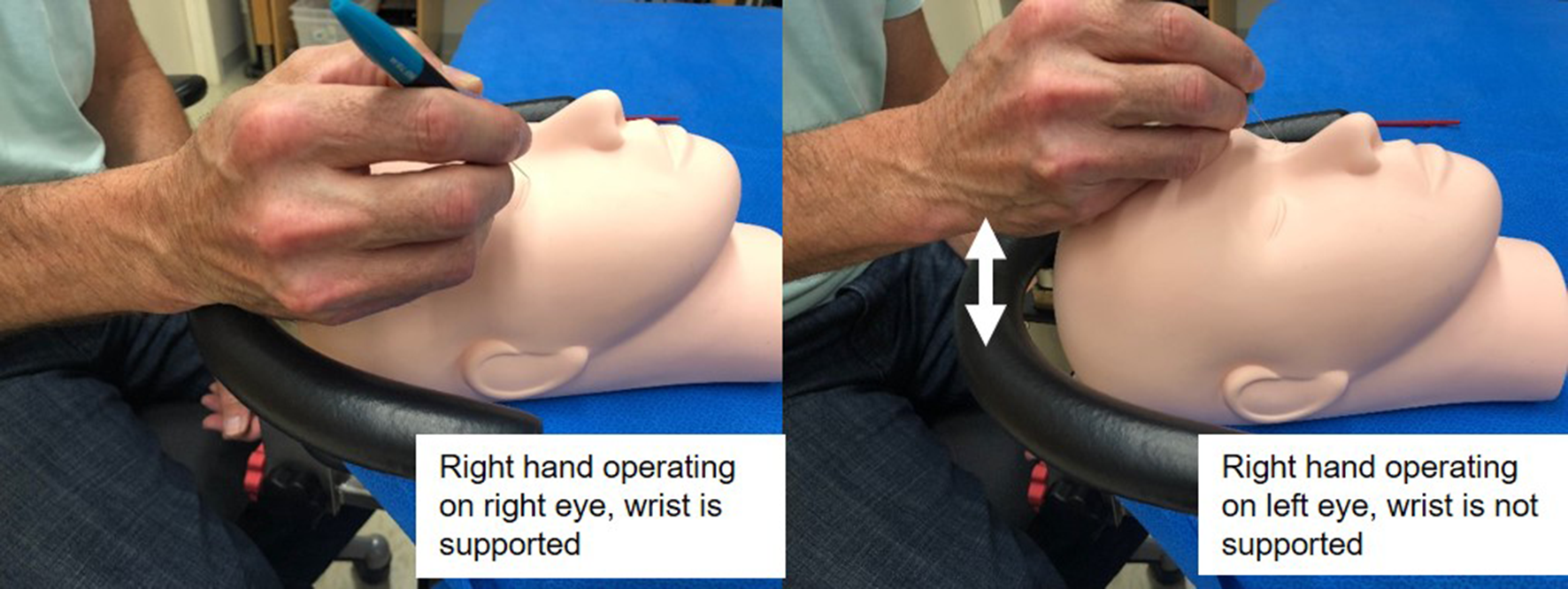
Left: A right-handed surgeon has a fully supported wrist when operating on the right eye with a SWR. Right: A right-handed surgeon has no wrist support when operating on the left eye with a SWR. SWR, symmetric wrist rest.
A novel experimental asymmetric wrist rest (AWR) has been designed where the contralateral wrist bar gradually rises and curves toward the patient’s operative eye, keeping the surgeon’s hands equidistant to the operative eye and overcoming the nasal bridge (Figure 3). The AWR allows both hands to have the favorable biomechanical forearm support rather than only direct hand support of the patient’s forehead (Figure 4). The current SWR only provides forearm support when surgery is performed on same side as the dominant hand (SSD). Conversely, the surgeon’s hand is placed directly on the patient’s forehead when operating on the contralateral side of the dominant hand (CSD). In addition, the AWR was designed to make the fine horizontal adjustments required from natural variation of patient head size, head position, and surgeon hand size to optimize support of surgeon’s hands while increasing degrees of freedom (by moving support from the hand to the wrist). The SWR can only be adjusted vertically.

Prototype asymmetric wrist rest.
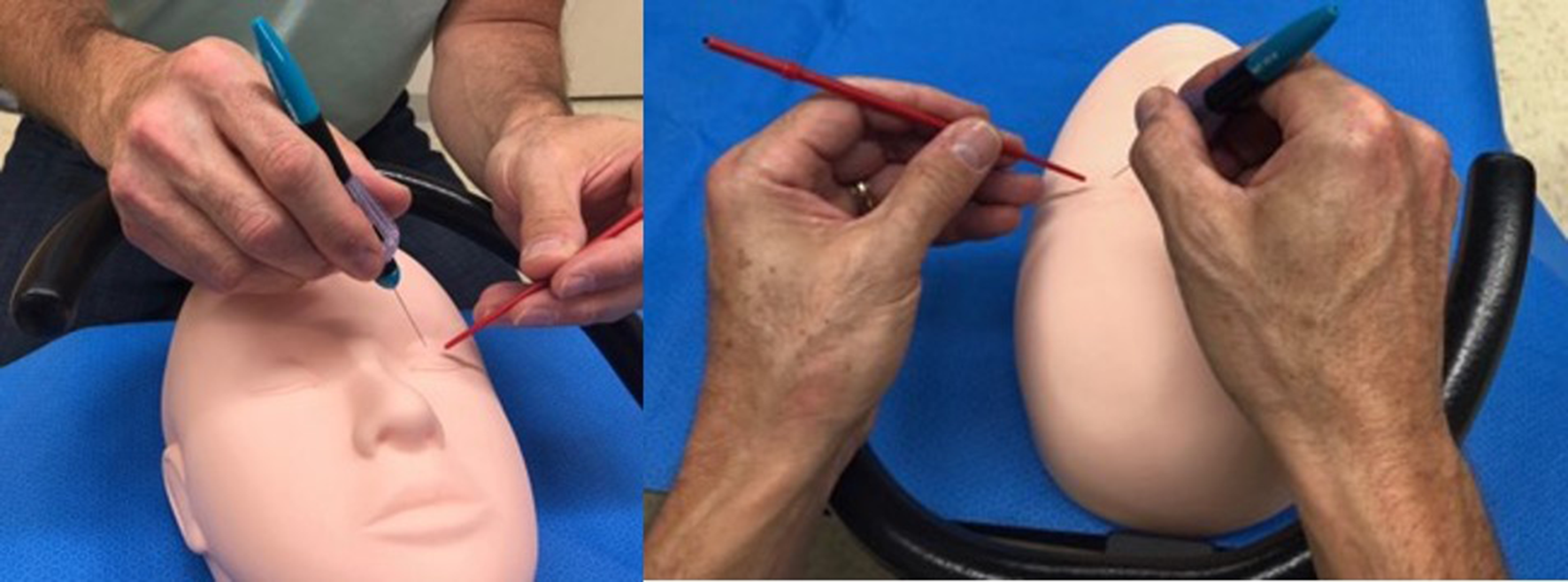
Left: Experimental AWR allows the right hand to be supported at the wrist even when operating on a left eye. Right: Surgeon’s hands being supported by the AWR from various perspectives. AWR, asymmetric wrist rest.
It was hypothesized that more tremor is associated with CSD surgeries than SSD surgeries due to the relative advantage of forearm versus hand support. The main objective of the current study was to measure the tremor magnitude and frequency of the surgeon’s instrument in situ during vitreoretinal membrane peeling surgery with a focus on differences between the SSD and CSD surgeries. In situ comparison with the SWR would confirm whether forearm support (SSD) is superior to hand support (CSD) as suggested biomechanically (Morrison & Newell, 1999; Safwat et al., 2009). In addition to comparing hand tremor when using a conventional SWR, the AWR was evaluated to determine whether improved forearm support for the contralateral eye would eliminate the difference in SSD versus CSD tremor, suggesting that forearm support is superior to hand support in microsurgery.
Methods
Participants and Data Sources
The participants were three collaborating surgeons utilizing wrist rests for vitreoretinal surgery. All were right-hand dominant male attending surgeons with multiple years of surgical experience. The co-inventor (author MDW) of the AWR did not participate in the cases studied or in the data analysis. None of the collaborating surgeons contributing cases had monitary conflicts of interest and since tremor is considered an involuntary physiological phenomenon, we did not consider the authors to be biased.
Macular surgery requires the greatest dexterity and all study cases were macular hole surgeries with peeling of the internal limiting membrane, or epiretinal membrane surgeries with or without peeling of the internal limiting membrane. The type of surgery performed, the eye of the surgery, and the mechanism of wrist support were recorded. All data were obtained under approval from the University of Wisconsin-Madison IRB.
Video Recordings
Vitreoretinal surgeries were routinely recorded for educational and research purposes directly from a Zeiss Lumera 700 surgical microscope in high definition (either 1,080 or 720 pixels) video (Zeiss, 2018) over the course of 15 months. The videos were all captured at 30 fps. As this study focuses on surgical tremor arising from the wrist joint (frequencies ranging from 7 to 10 Hz), based on the Nyquist sampling theorem, a sampling rate of 30 Hz was sufficient. All videos were de-identified by removing meta data information with respect to time and date created, clinic, and surgeon and identifying patient information before leaving the operating room.
Video segments were serially selected and screened for focus, brightness, and on-screen time. The minimum focus and brightness were defined such that the instrument must be easily distinguishable from the background. The minimum on-screen time was that the instrument was completely in the field of view of the video for at least 10 s. If a video segment met all the above criteria, it was included. Of 83 surgery videos available, 76 fit the selection criteria and were used for video tracking.
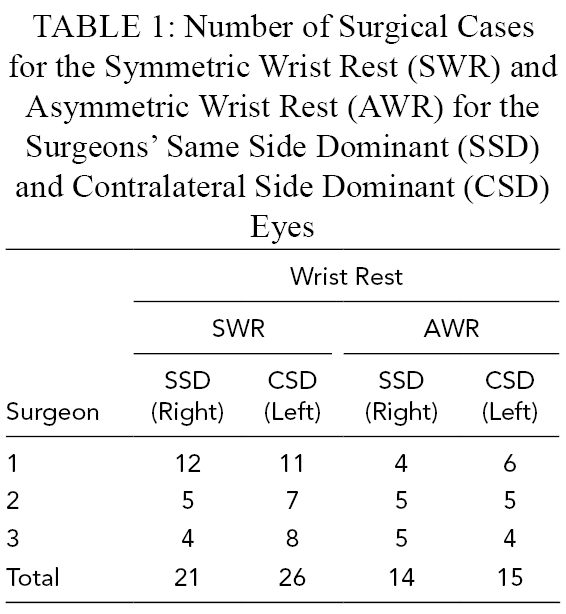
The University of Wisconsin Hospitals and Clinics and SSM Health (Madison, WI) were the sources of the surgical case videos. Surgery video recorded in the first 6 months utilized the SWR. Subsequently an AWR was introduced and the remaining surgeries were completed using either wrist rests. Microscope videos were recorded for surgeries performed on both sides and both types of wrist rests. The number of videos recorded for each surgeon using the SWR and AWR is shown in Table 1.
Number of Surgical Cases for the Symmetric Wrist Rest (SWR) and Asymmetric Wrist Rest (AWR) for the Surgeons’ Same Side Dominant (SSD) and Contralateral Side Dominant (CSD) Eyes
Video Tracking
To quantify tremor during retinal surgery, marker-less video tracking of the surgical instrument was conducted. The video tracking methodology and software used was developed at the University of Wisconsin-Madison and is described in Chen et al. (2013) and Chen et al. (2014). This method has previously been used in our laboratory for studying clinical breast exams (Azari et al., 2014) and open surgeries (Azari et al., 2019). Tracking was performed by focusing a small rectangular region of interest (ROI) manually on the tips of the forceps used during surgery as shown in Figure 5. The surgery video segment lengths tracked ranged from 10 s to 2 min. The data collected (in pixels) included every frame x-coordinate location, y-coordinate location, velocity, and acceleration of the ROI (Chen et al., 2014). Distances in millimeters were obtained by calibrating the number of pixels against the known dimensions of the forceps. Due to the differing amount of zoom depending on surgical video, the range of mm/pixel falls between 15 and 22 μm/pixel.
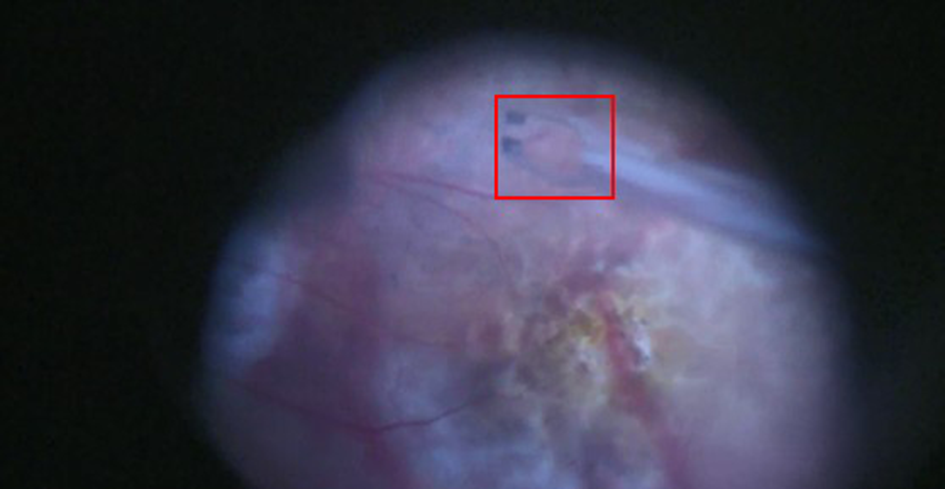
Video image of tracked ROI for retinal surgery videos. The ROI is initially located at the forceps tip and the tracking algorithm records the frame-by-frame forceps movements. ROI, region of interest.
Data Processing
The main points of interest from the video tracked data were the magnitude of tremor, the tremor frequency, and the distance the instrument traveled during the retinal peeling procedure (path length). The ROI’s x-coordinate location, y-coordinate location, speed, and acceleration were normalized using the dimensions of the forceps, the most commonly used being 23G (outer diameter = 0.64 mm) or 25G (outer diameter = 0.51 mm). The tracked data were plotted and compared against the video frame-by-frame using Multimedia Video Task Analysis (MVTA) software developed at the University of Wisconsin-Madison.
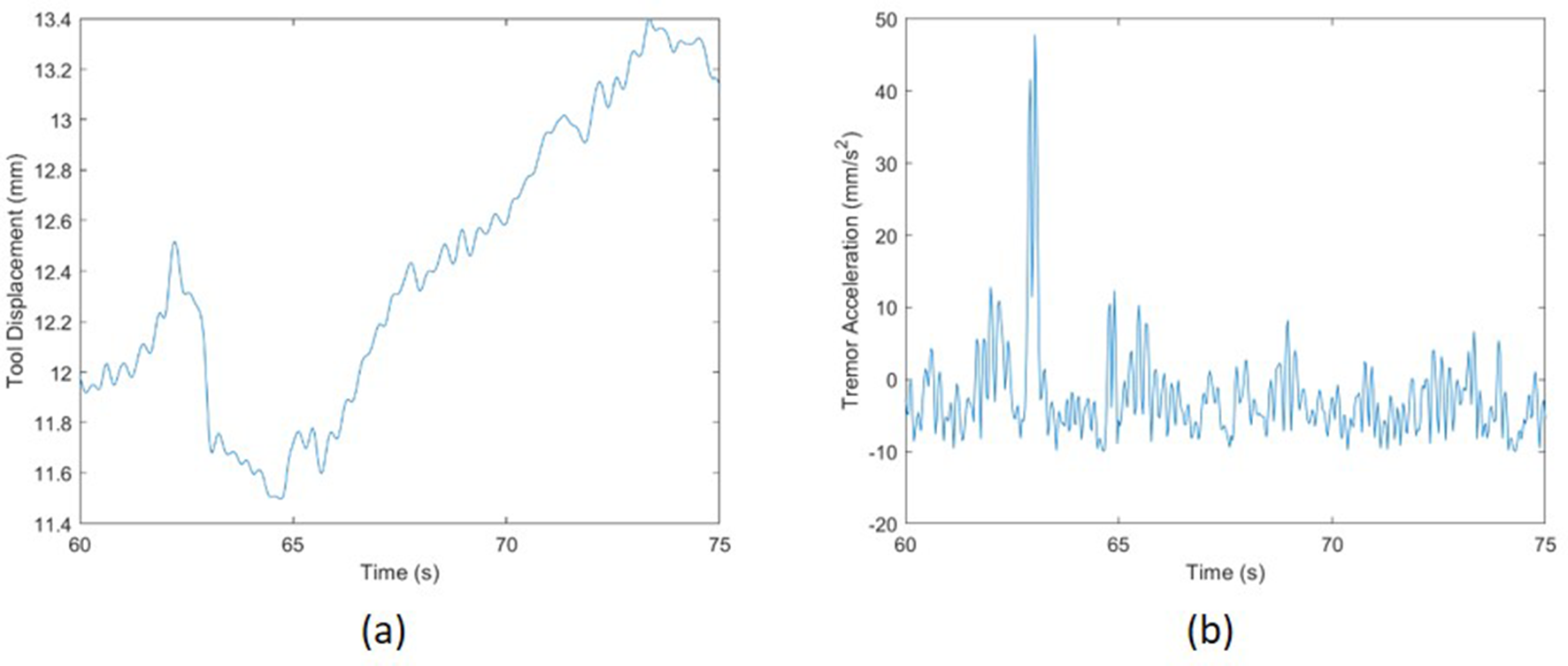
Representative displacement and acceleration data are plotted against time in Figure 6. The peak tremor magnitude and associated frequencies were measured using a single-sided fast Fourier transform (FFT). Due to the varying length of video clips, the window size of the FFT was chosen to match the shortest video clips used (10 s) and averaged. Because the video sampling frequency is 30 Hz, the frequency range was between 0 and 15 Hz based on the Nyquist sampling theorem. This sufficiently spans the desired frequency range of wrist tremor. A sample FFT plot is shown in Figure 7 with peak tremor acceleration frequency and magnitude for involuntary movements labeled (peak tremor acceleration magnitude).
(
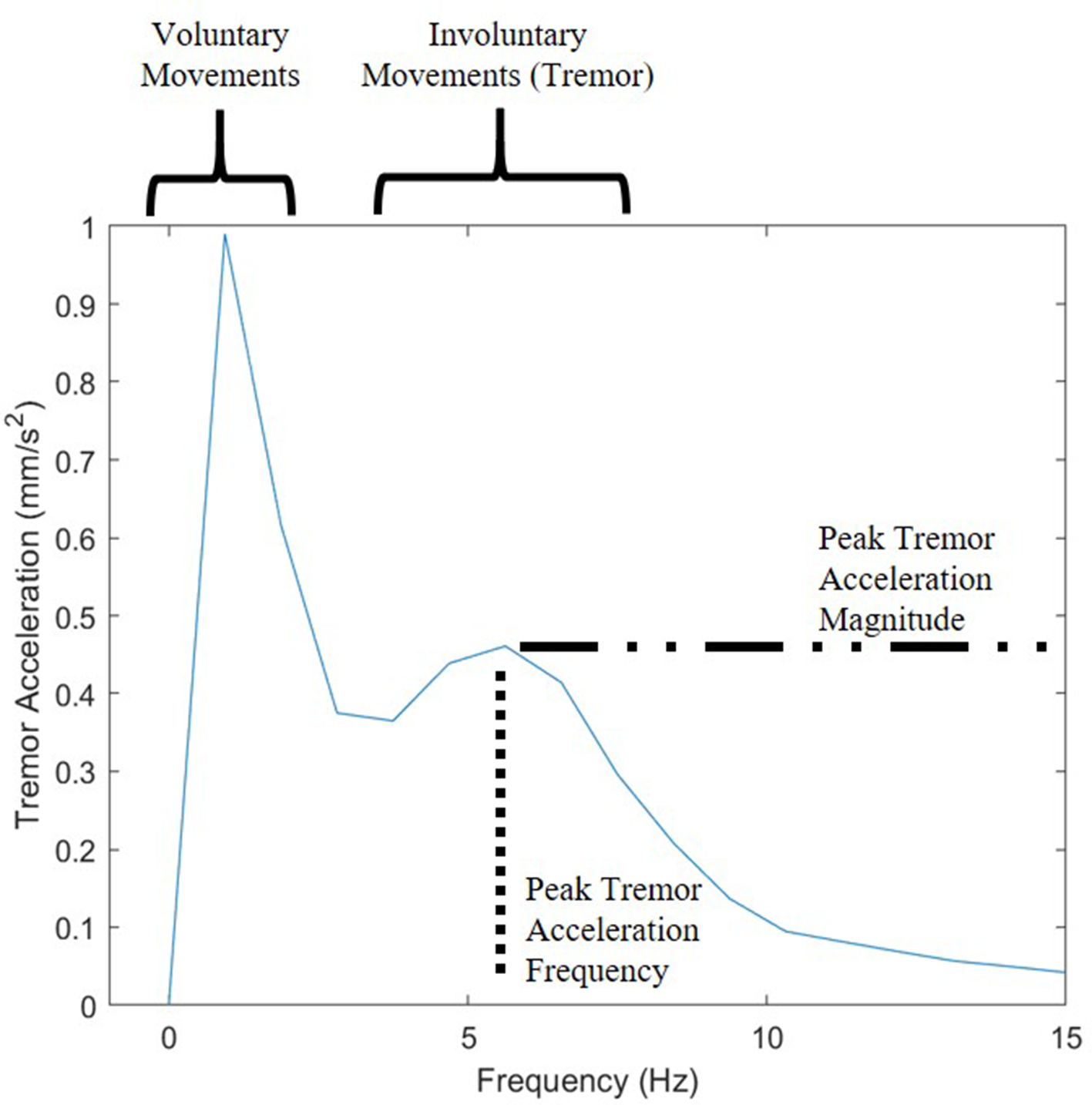
Single-sided FFT with tremor frequency in the 4–6 Hz range. FFT, fast Fourier transform.
Instrument path length was obtained from plotting the location of the forceps throughout the video clip. A line integral was then used to find the total distance traveled by the forceps. Instrument speed was obtained by dividing the path length against time. An example of a path length plot is shown in Figure 8. Speed and acceleration were calculated by the combined forceps travel distance and digital differentiation of path length.
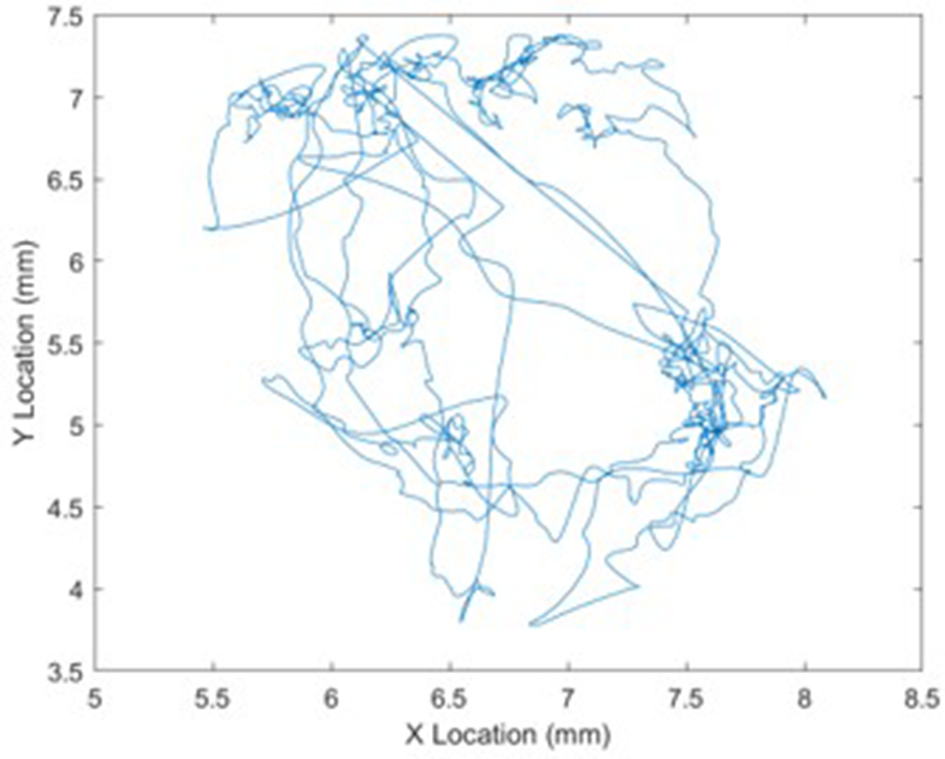
Path traced out by instrument during operation. The instrument speed was found by dividing total path length by time.
Results
Tremor Quantification Using Marker-Less Tracking
Since there were no previous reports found of instrument tremor measurements while operating in the eye, as a benchmark, tremor was measured when a surgeon held the forceps out of the eye while assuming a similar posture as operating in order to compare against published tremor measurements in a laboratory setting. The out-of-eye tremor was consistently in the 5–8 Hz range. In comparison to out-of-eye tremor, hand tremor measured while the instrument was in the eye displayed a narrower frequency range, between 5 and 6 Hz. This may be due to the attenuation caused by trocars in the eye or the vitreous humor of the eye. Another distinguishing characteristic between in-eye and out-of-eye motion data was the amount of movement at the sub-one hertz range. The laboratory out-of-eye tremor measurement was taken when the surgeon was not actively performing surgical tasks while the in situ in-eye tremor measurements were taken during active surgical motions. As a result, there was a much greater magnitude of sub-one hertz movement during actual surgery videos.
Tremor for SSD and CSD Surgeries Using Different Wrist Rests
Peak tremor acceleration, frequency, and instrument speed measured during surgeries for both eyes and both wrist rests are summarized in Table 2. The normality assumption was tested by a quantile–quantile plot for all three outcomes (acceleration, frequency, and speed) and no evidence of violation was observed as there were very few points deviating from the diagonal line. Linear mixed effect models were built for each dependent variable using surgical side (SSD vs. CSD) and wrist rest (AWR v. SWR) and their interaction as the fixed effects components, and the surgeon-specific random intercepts as the random effects component in order to take the within-surgeon correlation into account.
Average ±SD for Tremor Acceleration with Both Wrist Rests and Both Surgeries
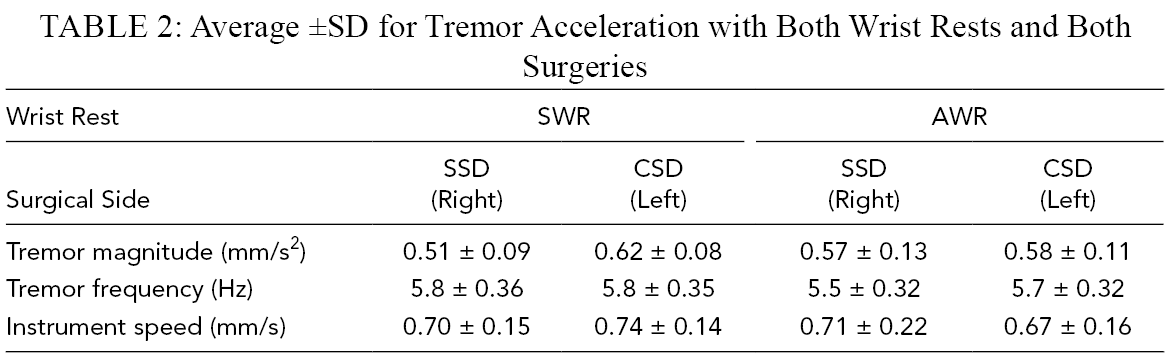
The interaction between surgical side and wrist rest acceleration was statistically significant, F(1, 72) = 4.31, p = .042. Pairwise comparisons indicate that the acceleration difference between CSD and SSD surgeries using the SWR was statistically significant (p = .001), while acceleration differences between CSD and SSD surgeries using the AWR were not statistically significant (p = .84). Tremor acceleration was 0.11 mm/s2 greater (22%) for the CSD surgeries than the SSD surgeries when using the SWR. The main effect of surgical side, F(1, 72) = 6.405, p = .014. There was a 0.07 mm/s2 greater (12%) tremor acceleration magnitude for CSD surgeries than the SSD surgeries for both wrist rests. The main effect of wrist rest, F(1,72) = 0.029, p = .864, for tremor acceleration magnitude was not statistically significant (Table 2).
No statistically significant tremor frequency effects were observed for the main effects of surgical side, F(1,72) = 2.861, p = .095, and interaction of wrist rest and surgical side, F(1,72) = 1.306, p = .256. However, a statistically significant difference was found for the main effect of wrist rest, F(1,72) = 5.444, p = .022. No statistically significant instrument speed effects were observed for the main effects of surgical side, F(1,72) = 0.001, p = .976, wrist rest, F(1,72) = .732, p = .395, or their interaction, F(1,72) = 1.237, p = .270.
Discussion
Riviere and Jensen (2000) measured hand tremor during vitreoretinal microsurgery using an inertial sensing device attached to the instrument handle in the 3–5 Hz range and reported a maximum acceleration of 30.1 mm/s2. The current study measured tremor directly at the instrument tip during microscopic surgery in situ. The rhythmic, oscillating movements in tremor occur when antagonistic muscles co-contract (Morrison & Newell, 1999; Wade et al., 1982). These oscillations arise from a larger mechanical-reflex and smaller central-neurogenic origins (Elble, 2003). Normal elbow tremor falls between 3 and 5 Hz, wrist tremor 7 and 10 Hz, and metacarpal joint tremor 17 and 30 Hz (Elble, 2003). Low frequencies less than 3 Hz were attributed to the voluntary movements of the surgeon’s hand (Figure 7). Movements not related to surgeon movements, such as patient breathing, eye movements from inadequate anesthesia/akinesia, and vibrations of the microscope or patient, can also contribute to this frequency range.
The tremor frequencies found using the marker-less video tracking techniques in this paper reflect findings from previous studies. Although metacarpal joint tremor ranges from 17 to 30 Hz, there is no metacarpal joint tremor during vitreoretinal surgeries because the surgical instrument is grasped between the fingers, so metacarpal motion is restricted. Consequently, we are only concerned with tremor arising from the wrist joint in the 7–10 Hz range. This frequency range was verified by Safwat et al. (2009) who observed tremor frequencies between 2 and 8 Hz in microsurgery manipulations when sampling at 1000 Hz, so the video sample rate of 30 Hz was sufficient for preventing aliasing. When holding an instrument out of the eye, surgeons displayed tremor in the 5–8 Hz range at the tip of the instrument. When operating in the eye, the tremor frequency dropped to the 5–6 Hz range; this decrease in tremor frequency may possibly be due to the added mass of the trochar compared to air or even friction on the forceps from the trochars that allow entrance into the eye.
Physiologic tremor may be influenced by numerous factors (Belykh et al., 2018; Fargen et al., 2016; Rui et al., 2018). Effects known to increase surgical hand tremor include caffeine (Humayun et al., 1997; Wharrad et al., 1985), fatigue, previous alcohol consumption, recent exercise, sleep deprivation, lack of experience, and fasting (Belykh et al., 2018; Fargen et al., 2016; Gizicki et al., 2017; Rui et al., 2018; Slack et al., 2008a). Methods used previously for tremor reduction include relaxation techniques (Comby et al., 1992), β-blockers (Arnold et al., 1993; Elman et al., 1998; Franke et al., 2013; Humayun et al., 1997; Rui et al., 2018), and wrist support (Arnold et al., 1993; Belykh et al., 2018; Coulson et al., 2010; Evans, 1978; Hara et al., 2015; Rui et al., 2018).
Tremor has been mitigated by low doses (10–40 mg) of the β-blocker propranolol (Jefferson et al., 1979; Wasielewski et al., 1998). Some surgeons realizing that their tremor is problematic, self-medicated using oral β-blockers for reducing tremor or improving coordination (Elman et al., 1998; Franke et al., 2013; Humayun et al., 1997). β-blockers blunt the tremor increasing the impact of caffeine (Arnold et al., 1993). A randomized study found that administering 40 mg of propranolol to surgeons 1 hr before surgery significantly improved surgical performance and decreased anxiety and hand tremor without unintended effects for the surgeon or patient (Elman et al., 1998). Recent recommendations to enhance surgical performance include the use of β-blocking agents (10–40 mg of propranolol 1 hr prior to surgery) for reducing physiologic hand tremor and improving overall surgical performance (Franke et al., 2013; Rui et al., 2018).
In the current study, increased tremor acceleration was observed for the CSD than the SSD surgeries when using the SWR. These findings confirm the concept that hand support on the patient’s forehead when operating on the CSD is inferior to support at the level of the forearm and results in more tremor. Right-hand dominant surgeons would gain sufficient support from the SWR only for right eye surgery. This difference in surgical tremor reflects the findings in Morrison and Newell (2000). The current study demonstrates that an AWR can help reduce instrument tremor, measured in situ. Since no significant effects for tremor frequency or speed were observed with the use of an AWR, it is anticipated that the AWR attenuated tremor magnitude.
The deviations from normality observed in the tracked kinematics data should be acknowledged. Data transformations did not significantly change the normality of the data; but since analysis of variance is considered robust, statistical analysis was performed using the nontransformed data.
The location of optimal wrist support has also been examined in previous research (Morrison & Newell, 2000; Safwat et al., 2009). Support located anywhere on the arm was better than no support at all (Arnold et al., 1993; Belykh et al., 2018; Coulson et al., 2010; Evans, 1978; Hara et al., 2015; Morrison & Newell, 2000; Nissenbaum et al., 1979; Safwat et al., 2009). Placing support proximal to the wrist was superior than elbow support alone (Morrison & Newell, 2000; Safwat et al., 2009). Direct support at the level of the hand paradoxically produces increased finger tremor (Morrison & Newell, 2000; Safwat et al., 2009).
There is a compensatory antitremor effect of having increased degrees of freedom at the wrist compared to the hand being supported (Safwat et al., 2009). Morrison and Newell also found that the greatest decrease in finger tremor was observed when the finger and hand were free to move. The unrestricted movement about the wrist joint resulted in significantly less tremor in the index finger. This suggests a compensatory synergy around the wrist joint that is critical in the upper limb during postural tasks. The wrist may act as a fulcrum and the addition of specific joint space degrees of freedom there in tremor tasks reduces the degree of oscillation of the index finger (Morrison & Newell, 2000).
Forearm supports compared to hand supports have demonstrated ergonomic advantages for computer operators in passive and dynamic activities (Aarås et al., 2001; Cook et al., 2004a, 2004b; Lee & Huang, 2006). Forearm supports proximal to the wrist have demonstrated desirable effects including decreased discomfort and decreased muscular load of the neck, shoulders, forearm, and upper arm muscles for computer operators (Aarås et al., 1998, Aarås et al., 2001; Cook et al., 2004a, 2004b; Lee & Huang, 2006). Forearm support, rather than just hand support, may provide the surgeon ergonomic advantages. During ophthalmic procedures, prolonged static positioning with cervical extension and unsupported arms produced excessive loading in the cervicobrachial region, which may accelerate disk degeneration (Chakrabarti et al., 2007), although differences in loading due to the precise location of surgeon support during retinal surgery have not been studied.
Wrist freedom of movement has demonstrated additional benefits besides tremor reduction (Safwat et al., 2009). Unconstrained wrist movement allowed for greater hand dexterity and greater surgeon comfort without producing an increase in tremor. Wrist support proximal to the wrist contributes to tremor reduction without impingement of the freedom of movement of the wrist. An AWR design allows bilateral support just proximal to the wrist where tremor can be minimized and wrist movement unimpaired. The SWR has proximal wrist support with the ipsilateral hand and hypothenar support or hand support in the contralateral hand. Hypothenar or hand support with both hands is present for those not using either method of wrist support. Although hypothenar support may be inferior to proximal wrist support, it is likely superior to no support or support at the level of the elbow only.
It is unsurprising that surgeon hand tremor can be a problem in microsurgery. The displacement magnitude of a typical vitreoretinal surgeon hand tremor is 156 µm (Peral-Gutierrez et al., 2004). Vitreoretinal surgery on the macula involves removing epiretinal membranes or peeling of the internal limiting membrane 3.6 µm thick (Kalvoda et al., 2009). The tremor displacement magnitude therefore is 40 times greater than the tissue that is being carefully removed from the surface of the retina. Surgeon muscular fatigue increases with operating time, and surgeon hand tremor is reported to increase 6.67% for every hour of operation (Slack et al., 2008a, Slack et al., 2008b). This is an 8.4-fold increase in tremor compared to one performing normal desk work in a work day. This increase was a similar occurrence in both novice and experienced surgeons (Slack et al., 2008a). Consequently the 22% difference observed in the current study between CSD and SSD surgeries is an important finding.
Evidence that surgeon tremor is a significant problem is a common justification for robot-assisted surgery (Channa et al., 2017; Gudeloglu et al., 2014; Nuzzi & Brusasco, 2018; Riviere et al., 2006; Roizenblatt et al., 2018). Specifically, for one of the most technically demanding microsurgical fields, vitreoretinal surgery, a main technical limitation is imprecise maneuver during micromanipulation of tissues due to natural hand tremor (Nuzzi & Brusasco, 2018). In a recent review of robot-assisted vitreoretinal surgery, chief among the challenges faced by ultra-microsurgeons is physiologic hand tremor (Roizenblatt et al., 2018). A high number of surgical procedures have been performed with robot assistance without enough evidence to support full implementation. At this point, it is not clear if the significantly higher costs and longer operative times associated with robotic-assisted surgery are justified for the majority of surgical procedures (Broholm et al., 2016).
The intelligent arm holder EXPERT performs as a robot-controlled robot to reduce fatigue and improve performance during microneurosurgery (Goto et al., 2013). It is limited by its design for the right hand only, tight feeling during transposition of the surgeon’s arm, and a large footprint in the operating room. It does underscore the need to reduce surgeon fatigue and improve performance. The iArmS, another intelligent arm-support system used in endoscopic endonasal transsphenoidal surgery, did reduce shaking of the camera image suggesting improved surgeon stability. It is made for both arms but has a very high cost and a large footprint (Ogiwara et al., 2017). The development of the microsurgical robot or robotic arm support reflects a desire for surgeon tremor reduction, decreased fatigue, and improved performance.
Another potential benefit of improved wrist support for reducing physiologic tremor may be that it helps reduce muscle fatigue and provides greater freedom of the wrist. There is a consistent relationship between psychological status and tremor (Rui et al., 2018). Relaxation therapy reduced tremor (Comby et al., 1992). Listening to surgeon-selected pleasant music before and during surgery improved microsurgical performance (Belykh et al., 2018). Anxiety, including prior to public speaking, increased tremor (Rui et al., 2018). Enhanced surgeon comfort and confidence in membrane peeling may permit tremor reducing relaxation along with the actual reduction of postural tremor reducing surgeon tremor. Hand stabilization proximal to the wrist allows maximal range of motion of the wrist with reduced tremor compared to hand placement on the patient’s forehead with subsequently more limited degrees of freedom.
Despite the identification of surgeon tremor being problematic in microsurgery, actual evidence of tremor reduction and improving surgical performance is lacking. Arm support improved accuracy in micromanipulation tasks and reduced tremor intensity (Safwat et al., 2009). Future study will include determining if microsurgical outcomes can be improved with this tremor reduction. Additionally, it is unclear if a 22% reduction of tremor acceleration is of sufficient magnitude to have a clinical benefit. Interestingly, the Micron, an actively stabilized surgical robot, claims reduced surgeon tremor by 50% ex vivo in a stapedotomy procedure (Vendrametto et al., 2015). Since physiologic tremor has been identified as a major limitation of microsurgery and is a major justification for RAS, a 22% difference may be clinically significant.
Limitations of the current study include a limited number of surgeons (3) and AWR cases (29) and a noncontrol of other factors that may influence surgeon tremor (caffeine use, exercise, fatigue, etc.). A total of 47 cases with the SWR showing a 22% difference in CSD versus SSD tremor suggest that wrist support alone is an important factor. Future study will control for other factors impacting surgeon tremor. Another weakness is the lack of accounting for learning for the AWR. The current study did not control for learning effects or surgeon experience using an AWR, as surgeons were allowed to use either wrist rest after the initial period working with the SWR. Future studies will investigate potential learning effects.
Conclusions
Surgeon tremor was successfully measured from routinely collected surgical video in situ without any interference with the actual surgery. With the current SWR, a 22% greater tremor acceleration was found with hand support compared to the preferred distal forearm support location. Our analysis suggests that the AWR, which permits support proximal to the wrist, eliminates the tremor acceleration difference between eyes. Reduction of surgeon tremor with subsequent improvement in surgeon dexterity, without robot assistance, has the potential of improving surgical outcomes in a cost-effective manner.
Key Points
In situ tremor was recorded and quantified during vitreoretinal surgeries.
Surgeries were performed under different wrist support conditions.
The study found that tremor acceleration was reduced by 0.11 mm/s2 when the forearm is supported.
Initial asymmetric wrist rest results suggest that there is no difference in tremor acceleration for left and right eye conditions.
An experimental AWR that provided forearm support for both eyes will continue to be evaluated.
Footnotes
Acknowledgments
We thank David Azari with assistance in the preliminary analysis as well as Hao Zhou with assistance in video tracking. Jonathan Chang is supported in part by an unrestricted grant from Research to Prevent Blindness and a Core Grant for Vision Research from the NIH to the University of Wisconsin-Madison (P30 EY016665).
Author Biographies
Yifan Li received his BS (2016) and MS (2017) degrees from the University of Wisconsin-Madison in biomedical engineering. He is currently a PhD student in industrial and operations engineering at the University of Michigan (Ann Arbor, MI).
Mitchell D. Wolf is a clinical adjunct associate professor at the University of Wisconsin Department of Ophthalmology and Visual Sciences. He received both his undergraduate and medical degrees at the University of Michigan (Ann Arbor, MI).
Amol D. Kulkarni is a clinical adjunct assistant professor at the University of Wisconsin Department of Ophthalmology and Visual Sciences. He received his medical degree at the University of Mumbai (India).
James Bell is a clinical adjunct assistant professor at the University of Wisconsin Department of Ophthalmology and Visual Sciences and received his medical degree at the University of Louisville in Kentucky.
Jonathan S. Chang received his MD degree from Columbia University in 2008. He is currently an assistant professor in the Department of Ophthalmology and Visual Sciences at the University of Wisconsin School of Medicine and Public Health (Madison, WI).
Amit Nimunkar received his PhD from the University of Wisconsin-Madison and is currently a faculty associate in the department of biomedical engineering.
Robert G. Radwin is Duane H. and Dorothy M. Bluemke Professor in the College of Engineering at the University of Wisconsin-Madison, where he advances new methods for research and practice of human factors engineering and occupational ergonomics. He has a BS degree from New York University Polytechnic School of Engineering, and MS and PhD degrees from the University of Michigan (Ann Arbor, MI).
