Abstract
Hospitalized patients with asthma or COPD typically receive respiratory medications via inhalers (pressurized metered dose inhalers, dry powder inhalers, or soft mist inhalers) or nebulizers. Because in-hospital use of inhalers may be complicated by required interchanges from nonformulary home inhalers to inpatient formulary inhalers, clinician or patient errors in administration, unclear dosage received, wasted drug, misplaced devices, and increased costs, switching from the use of inhalers to an all-nebulization strategy may streamline processes and improve outcomes. As respiratory therapists and hospital pharmacists, we have had experience with making this switch in different types and sizes of hospital systems. Despite the challenges we encountered, each of our approaches to implement an all-nebulization protocol was ultimately successful. In this article, we summarize our learnings during the operationalization of all-nebulization protocols, describe the benefits we observed post-implementation, and provide recommendations including detailed guidance for how to implement this type of switch successfully.
Keywords
Introduction
In-hospital administration of respiratory medications typically involves the use of inhalers (pressurized metered dose inhalers [MDIs], dry powder inhalers [DPIs], or soft mist inhalers [SMIs]) or nebulizers for patients with asthma or COPD. 1 As almost 40 different inhalers are currently marketed in the US, 2 hospitals cannot realistically carry all of them, and streamlining those available may lead to additional complications. Several concerns exist regarding the use of MDIs and DPIs, such as improper use and difficulties in correct use resulting from patient impairment (cognitive, respiratory, or neuromuscular) or clinician error.3-6 For example, a cross-sectional study in a specialized respiratory inpatient unit found that nurses demonstrated <50% competence with the usage of eight different inhaler devices, and none were able to demonstrate correct usage for all of the inhalers. 3 These findings highlight the need for better training of nurses on the numerous inhalers being used, which is further complicated by high turnover rates among nursing professionals.
Cost of wasted drug represents another concern associated with inhaler use, as most hospital stays are not long enough for patients to use the entire amount of drug in the device, especially as many inhalers are not produced in sizes suitable for institutional use. 7 Several studies have illustrated the significance of this waste, which has been reported to be up to 87% of dispensed doses, with associated costs.7-10
Inhaler use is also hampered by varied lung deposition of drug/dose delivered, as many patients could have inadequate respiratory muscle strength, leading to lowered peak inspiratory flow rates (PIFRs) that compromise the amount of drug inhaled.4,11 Furthermore, many hospitals do not routinely monitor PIFR, or may still dispense DPIs despite suboptimal PIFR, particularly upon patient discharge. 12 Notably, most inhalers deposit <20% of the dose into the lungs, even with the correct technique. 13 In addition to the noted clinical limitations with inhaler use, the presence of hydrofluorocarbon propellants in MDIs contributes to global warming/climate change, which may be of increased consideration for some health systems.14-16
Support for the Implementation of an All-Nebulization Strategy
In this article, our focus is to discuss best strategies and practices for switching to an all-nebulization approach in different types and sizes of hospital systems based on our firsthand experiences as respiratory therapists (RTs) and hospital pharmacists working in a variety of clinical settings. The objectives of this article are to summarize our learnings regarding the operationalization of all-nebulization protocols, to describe the benefits we have observed post-implementation, and to provide detailed recommendations for how to switch to an all-nebulization protocol.
Published literature from both small and large hospitals/hospital systems supports the benefit of switching from a combination of nebulizers and inhalers to an all-nebulization strategy.1,8,9,17,18 We recommend reviewing these papers in detail as they illustrate the successful implementation of these protocols, which have yielded improvements in therapeutic duplications, discharge discrepancies, and significant cost savings without negatively impacting length of stay, readmissions, patient satisfaction or RT visits per inpatient stay. Cost savings will vary by size of facility and the specific protocol implemented, but annual savings up to $1.6 million in a large health system have been reported. 17
Clinical Perspective
Our pharmacy and respiratory therapy experiences consist of work in clinical settings ranging from a small 130-bed standalone hospital to a large 31-hospital, multi-state health system which includes small critical access hospitals up to large academic centers. Challenges were present in each setting, and implementation of all-nebulization protocols occurred via a mixture of approaches. Each approach was ultimately successful.
Inhaler-to-nebulization therapy switches were initiated both prior to and during the SARS coronavirus 2 (SARS-CoV-2) 2019 pandemic and as early as 2016. Reasons for switching were primarily economic, but several additional clinical drivers were identified to justify switching to an all-nebulization protocol. These factors included various issues with inhalers, such as missing inhalers, improper drug administration, and treatments not being tracked appropriately. The excessive time RTs spent looking for and administering inhalers that were part of the formulary was cited as additional justification for switching to all nebulization; for instance, inhalers are lost when patients move from one nursing unit to another, and failure to clean out locked medication drawers with patient turnover can result in several patients’ medications being mixed together in the drawer, raising additional concerns regarding safety and infection control. Furthermore, in the medium-sized hospital setting, reduction in duplications in physician orders (ie, crossover coverage of oral inhaler orders with nebulizer orders) as a result of the interchange allowed RTs to visit patient rooms at the same frequency as before the interchange despite the new requirements for all respiratory medications to be administered by RTs. 1
The switching strategy implemented across hospital settings included both all-at-once and phased approaches. In the large, multicenter health system, 17 the switch was first piloted at a large facility using an all-at-once approach and was thereafter phased into additional facilities. Mixed approaches were also taken to handle therapeutic interchanges, with interchanges being carried out manually in some instances but automatically via the electronic health record (EHR) in others. In the small standalone hospital, the interchange was entirely automatic. In the case of the large multicenter health system, in which a larger facility was chosen as the pilot site, interchanges were manual during the pilot phase but then carried out automatically as additional facilities underwent the switch.
Notable advantages were observed following the employment of an all-nebulization protocol across systems. In the medium-sized hospital prior to switching to all nebulization, nurses were administering inhaler doses while RTs were administering nebulizer doses, and overtreatment of patients was suspected. 1 By switching to an all-nebulization strategy, RTs were responsible for all respiratory medications, therefore decreasing the likelihood of duplications in therapy. Additionally, prior to switching, physicians were generally not aware of the contents of their standing orders. Switching helped to remedy these issues and, notably, did not cause an increase in RT workload. 1
Like inhalers, nebulizers may present some limitations; however, many existing concerns are based on limited evidence and may be dissipated with emerging clinical data. The longer time required for drug administration with some nebulizers and the potential for aerosol generation with standard jet nebulizers (compared with breath-actuated nebulizers) may be issues of concern with older nebulizers. 4 Furthermore, sources of microbial contaminants identified in microbiome profiles of nebulizers used by hospital inpatients are predominantly nonhuman (environmental), demonstrating that human-associated microbiota are not a primary source of nebulizer contamination. 19
The risk of SARS-CoV-2 and other viral transmission with the use of nebulizers cannot be ruled out; however, little direct evidence exists based upon newer guidance and SARS-CoV-2 nebulization clinical studies.20-22 Early in the pandemic, concerns about administration of respiratory medications were largely related to shortages of personal protective equipment (PPE); having adequate PPE alleviates issues surrounding risk of viral transmission to healthcare workers. A systematic review of studies evaluating risk of transmission of respiratory viruses including SARS-CoV-2 during aerosol-generating medical procedures demonstrated that risk of transmission was increased primarily among healthcare workers who did not wear masks or showed low compliance for masking. 23 Anecdotal evidence from the pandemic suggests that nebulizers could be safely used with regard to disease transmission, and newer published data appear to support this.21,22 A study evaluating factors associated with airborne SARS-CoV-2 demonstrated no association with the use of potential aerosol-generating procedures (which included nebulization for the purposes of the study) and positivity of air samplings. 24 However, no direct comparison of the risk of transmission between nebulizers and inhalers has been reported beyond one study.22,25 In summary, many early concerns about nebulizers, based on limited evidence, are being challenged by emerging clinical data and did not preclude the development and execution of an all-nebulization strategy as outlined in greater detail below.
Process and Recommendations for the Development and Execution of an All-Nebulization Strategy
Key steps that we have identified in the successful switch to an all-nebulization strategy at small and large hospitals/hospital systems alike are (1) assessment of current practice, (2) identification and engagement of stakeholders, (3) writing of the protocol and developing the formulary, (4) stakeholder education, (5) program implementation, and (6) consideration of post-implementation outcomes validation. We provide considerations surrounding each of these topics as well as our specific recommendations and example scenarios from our collective professional experience. A workflow algorithm for implementing the switch is outlined in Figure 1 and Supplemental Appendix 1, which may be used as a reference guide. Key responsibilities of each of the stakeholders are summarized in Figure 2. A class therapeutic interchange example is shown in Figure 3 and a detailed interchange example is illustrated in Supplemental Appendix 2.
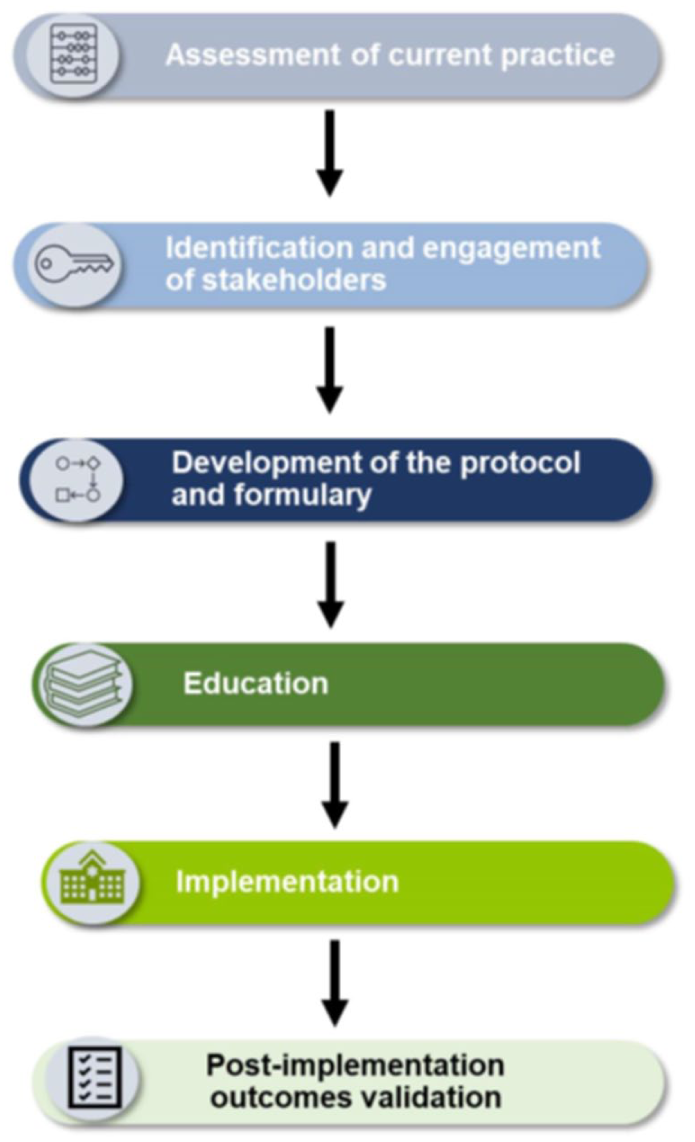
Summary workflow algorithm for implementing the switch to an all-nebulization strategy.
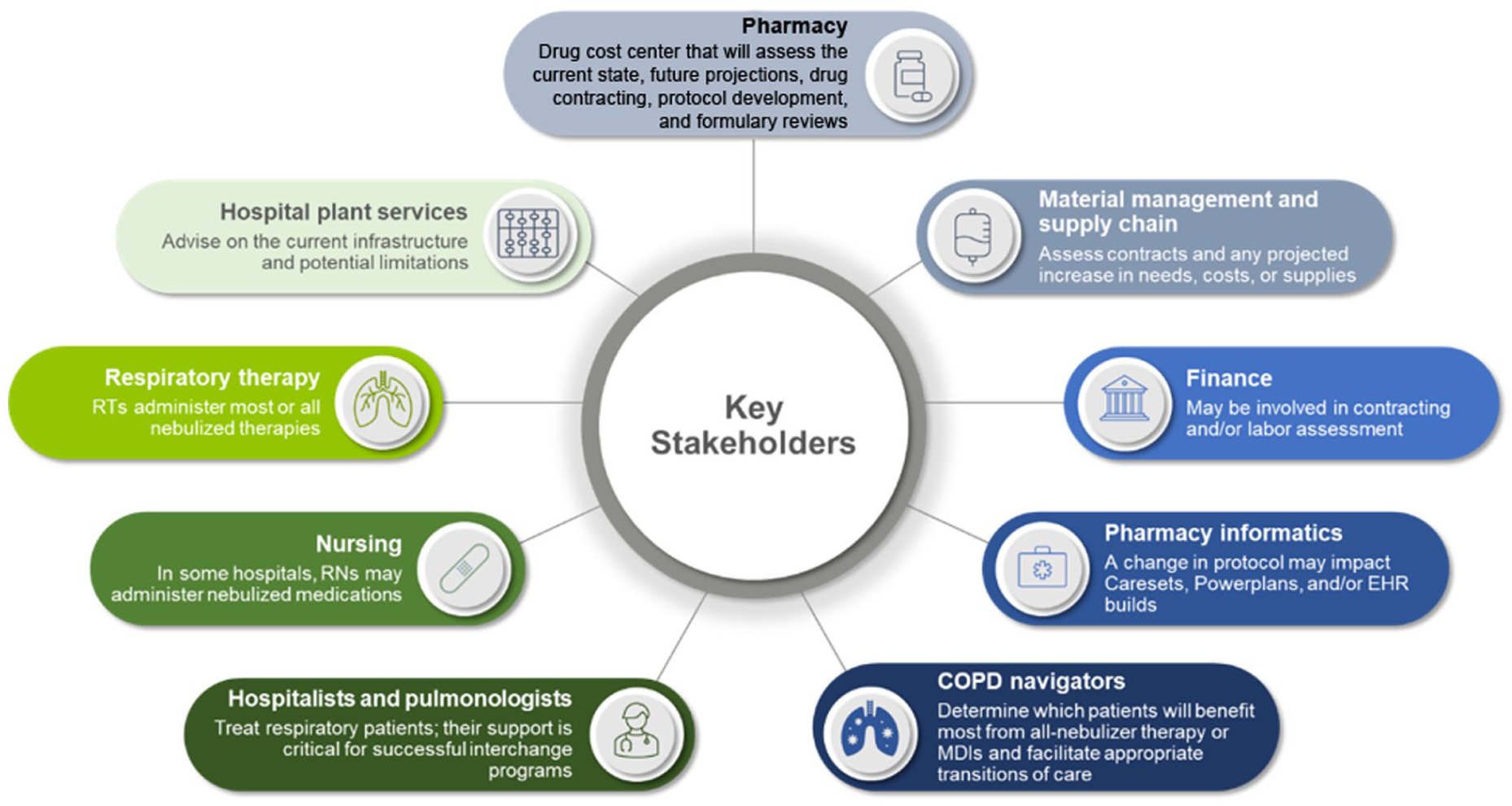
Potential key stakeholders involved in the implementation of an all-nebulization strategy.
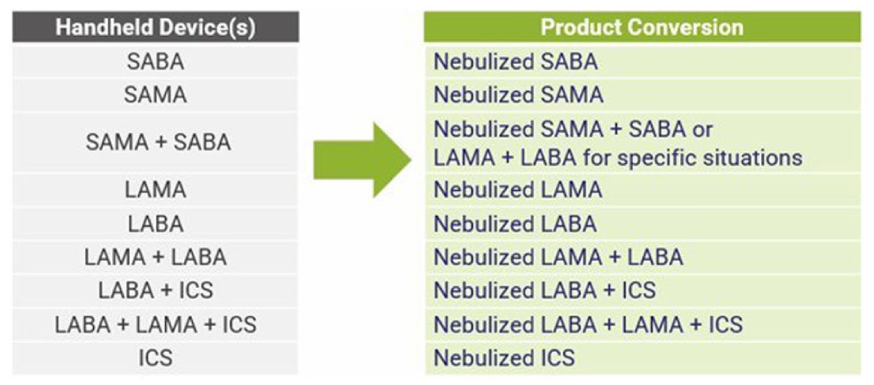
Example of class therapeutic interchange.
Assessment of Current Practice
Recommendation: Determine total annual costs of both inhalers and nebulized medications and any associated supplies used in the medication delivery process.
Evaluating total annual costs of respiratory medications and associated supplies (ie, spacers and nebulizer tubing) is essential to understanding current practice. Drug purchase costs often underestimate true expenses due to wasted inhalers and unused doses. Tracking inhaler vs. nebulizer dose counts helps establish a baseline; in one large system, inhalers accounted for most costs despite nebulizers making up 80% of doses. 17 This assessment is important for establishing a baseline of nebulizer vs. inhaler use and associated overall drug costs.
Identification and Engagement of Stakeholders
At small and large hospitals alike, identifying and securing the engagement of key stakeholders is critically important for implementing the switch to an all-nebulization protocol. Key responsibilities for each stakeholder party are described below and in Figure 2.
Pharmacy
Recommendation: Once all drug and supply chain contracts, financials, protocol development, drug monograph requirements (if applicable), and key stakeholder involvement are finalized, engage with pharmacy leadership to schedule P&T committee meeting review and approval.
The pharmacy serves as the central authority for assessing current and projected drug costs and plays a key role in developing therapeutic interchange protocols that support both clinical outcomes and procurement strategy. Pharmacy and therapeutics (P&T) committees serve as the primary link of communication between pharmacy and medical staff and are responsible for approval and maintenance of the formulary. 26 The P&T committee carefully evaluates any medications for formulary consideration, with decisions based on improvement of patient care. If a nonformulary medication is being requested for inclusion in the protocol, a specific drug review monograph will likely need to be created to appropriately vet the clinical and economic value necessary to support its addition to the hospital or health system formulary. P&T leadership can provide the specific requirements to meet this expectation.
In smaller hospitals or when non-pharmacy staff lead implementation, early engagement of pharmacy leadership is critical, while in larger systems, pharmacy often leads the transition to all-nebulization. After initial assessments, key elements—including contracts, financials, protocols, and stakeholder input—should be finalized before seeking P&T approval. Teams must be ready to address concerns such as patient inhaler education post-discharge, though evidence shows patients often lack proper inhaler training regardless of inpatient administration method. Given the expanded availability of respiratory medications, shifting to all-nebulization now offers a timely opportunity to reduce costs and enhance care.
Respiratory Therapy
Recommendation: Assess RT involvement in delivery of nebulized medications vs. inhalers including any potential differences in practice that may exist among different hospitals in a health system.
RTs are critical to the hospital staff and play a vital role in implementing an all-nebulization strategy. The ability to provide therapies and the labor/workload of RTs should be assessed, as they have many competing priorities. In many hospital systems, RTs administer most or all nebulized therapies while nurses may administer inhalers. Therefore, RTs must be assured that their overall workload will not be negatively impacted by taking on additional patients as would likely be required by an all-nebulization protocol.
Workload mapping can help evaluate the impact of shifting all respiratory therapy to RT-administered nebulization. In our experience, RTs were generally receptive of the decision to switch to an all-nebulization protocol. Many RTs expressed that nebulizers may be more appropriate in cases of shortness of breath or a patients’ inability to use an MDI, but in the small community hospital setting they did request to use Misty Fast™ nebulizers to speed up the administration process and help offset their demanding workloads. Understanding RT shifts and adjusting EHR medication timing may further optimize efficiency.
Nursing
Recommendation: Assess nursing involvement in delivery of medications via nebulizers vs. inhalers, including any potential differences in practice that may exist among different hospitals in a health system.
Nursing involvement in the administration of respiratory therapies varies by hospital. In most hospitals, registered nurses (RNs) are responsible for administering inhalers; however, some hospitals may have RNs administer nebulized medications as well due to lack of 24-hour pharmacy and RT-support. For hospitals in which RNs participate in administration of nebulized therapies, thorough training and explanation of changes is required to ensure alignment and maintain optimal patient care. At hospitals in which only RTs administer nebulized therapies, nurses should be advised of the change away from inhaler therapy and educated on any changes in their role in the care of these patients. In our experience, nursing staff was amenable to the interchange and expressed that removal of the therapy administration aspect of the overall patient care process made their role less confusing.
Physicians, Hospitalists, and Pulmonologists
Recommendation: Engage and acquire support from key providers.
The opinions of these stakeholders factor heavily into any P&T committee decisions. Therefore, it is critical to identify any concerns or potential resistance from these providers and address them prior to P&T committee meetings. Highlighting factors of particular interest to these stakeholders that can be improved by an all-nebulization strategy can foster their support; examples to cover include standardization of processes, poor inhaler technique and inadequate training of hospital personnel who administer inhalers, clinical care of patients with suboptimal PIFR, current standard of care (ie, hospitals are likely already administering a high percentage of respiratory medications via nebulization), transition to home care (including inability to carry all marketed inhalers), patient exacerbations on inhalers post-discharge, and inhaler costs/wasted doses. In our experience, hospitalists and pulmonologists generally agreed overall that nebulizers provide better therapy in acute exacerbations. In some hospitals, RT case managers/RT navigators who work directly with the patients being discharged are utilized to provide seamless continuity of care by training on various inhalers and facilitating the process of getting patients onto home nebulized medications if needed. In our experience, this approach established confidence with transitions of care.
Material Management and Supply Chain
Recommendation: Assess current usage of nebulizers and all associated supplies (eg, tubing, spacers) that are used in the respiratory medication delivery process as well as any variances that may exist among different hospitals.
Before switching to an all-nebulization strategy, each hospital should assess their current nebulizer use and estimate costs associated with a potential change, including: (1) any projected increases in needs, costs, or supplies; (2) any potential for new volume-based contracts. The hospital should also reassess its current nebulizer, and whether an alternative should be used (eg, converting to a nebulizer that can deliver the medication more quickly such as Misty Max to Misty Fast) based on future needs (see table in Supplemental Appendix 1 for examples of common commercially available nebulizers).
Hospital Plant Services Operations
Recommendation: Assess all sites of care in which inhalers are being utilized to ensure that infrastructure is in place to allow for the complete transition to an all-nebulization process. Hospital plant services may need to be consulted in collaboration with the clinical team.
Depending on the hospital system, nebulizers likely utilize compressed air piping out of the walls in patient care settings. It is essential to ensure there are no gaps in the ability to deliver care by nebulizer if inhalers are removed from the formulary. In our experience in the larger health system, this evaluation found no major issues regarding infrastructure, however, it did reveal that certain areas—like operating rooms, medical imaging, and other specialty departments—needed to maintain albuterol inhalers in remote dispensing units to allow rapid response for patients who may experience adverse reactions/allergies to drugs or contrast agents. In this specific context, nebulizer use was viewed negatively due to prolonged set-up and potential negative patient outcomes. As a result, albuterol inhalers are maintained on formulary for these sites. Behavioral health units are also key specialty sites of care where MDIs may be delivered more safely compared with nebulizers and could be maintained on the formulary. While nebulizer tubing in behavioral health units may pose a perceived patient self-harm risk, safe storage protocols can allow for continued nebulizer use.
Finance
Recommendation: Consult finance for assessment of contracts (if involved) and any discussions that may relate to labor adjustments.
The finance department may or may not be involved in contracting and other considerations for switching to all nebulization, depending upon hospital size and resources. Typically, the finance department is involved in smaller hospital systems’ contracting and therefore would be involved in implementing an all-nebulization protocol. In our experience in a large hospital system, finance assisted in evaluating the potential transition from inhalers to common canister vs. an all-nebulization approach as it related to costs per dose and labor but was not involved in contracting. Finance will likely need to be involved if there are labor-centric discussions; however, available evidence demonstrates that conversion to an all-nebulization process can be done without increasing labor.1,17 In the small hospital setting, discussions with finance stakeholders were largely driven by pharmacy and drug representatives.
Pharmacy Informatics
Recommendation: Determine pharmacy informatics availability and bandwidth for EHR modifications to support an inhaler to all-nebulization conversion.
The change to nebulization may impact medication orderables in the EHR. Inhalers in existing caresets/powerplans or standalone medication synonyms/sentences may need to be removed and updated with nebulized regimens. If a new protocol needs to be built using an automatic therapeutic interchange in which the EHR automatically converts inhaler orders to corresponding nebulization regimens, pharmacy informatics will be needed to write and test/authenticate the logic. Further, auto-interchange logic should be addressed for medication reconciliation during physician orders. Converting dosages from inhalers to nebulizers is not standard knowledge for all physicians; therefore, leaving orders for the inhalers with the dose and frequency information is of value to generate the best corresponding nebulization regimen. Standard medication administration times for specific nebulized medications may also need to be adjusted to improve RT workflow. Depending on hospital size and resources, a manual interchange at pharmacy order entry can be utilized during the verification process if pharmacy informatics is not available.
COPD Navigators
Recommendation: Collaborate with COPD navigators and/or others involved in transitions of care to modify or improve processes related to respiratory medications.
COPD navigators are a vital resource in determining which patients will benefit most from nebulizer therapy vs. handheld inhalers post-discharge. They may instruct patients on proper inhaler use, ensure that patients have sufficient PIFR for prescribed inhalers, and assist with appropriate referrals to durable medical equipment (DME) companies that specialize in transitions of care related to nebulized medications for those patients that may require nebulization therapy at discharge. COPD navigators and case managers, along with pharmacists, can assist with transitions of care processes to ensure appropriate therapy is prescribed at discharge based on the patients’ needs and insurance coverage. Not all hospitals or health systems have transitions of care specialists that are centric to these patients, as this often represents an area of limited resources. It should be clearly stated that the majority of patients will be discharged from the hospital on an inhaler regimen, but for a subset of patients with advanced disease, low PIFRs, or issues with dexterity or coordination, the consideration of nebulized maintenance therapy options (long-acting beta-agonists [LABA]/long-acting muscarinic antagonists [LAMA] ± inhaled corticosteroids [ICS]) at home may be warranted. SMIs could be options for low PIFR patients, but not all will have suitable or affordable insurance coverage. Medicare Part B access to nebulized LAMA/LABAs ± ICS could, therefore, be a viable option for outpatient maintenance. In this subset of patients, it is beneficial for case managers/COPD navigators to utilize DME companies that have a sound understanding of nebulized medications in terms of utilizing Medicare Part B vs. Medicare Part D, as it improves access and affordability of guideline directed therapies, which could potentially improve outcomes. As a result, education of this group of health professionals on this specific conversion is recommended to improve processes and understanding as it relates to respiratory patients.
Development of the Protocol and Formulary
Recommendation: Collaborate with pharmacy and key providers to develop an agreed-upon inhaler-to-nebulizer protocol that can deliver the expected clinical treatment options deemed necessary for all respiratory patients.
When developing an all-nebulization protocol, consider existing differences between adult and pediatric care pathways that may be in place. For instance, in one author’s experience, the pediatric ERs utilized a pre-existing albuterol MDI/spacer same-day discharge protocol, whereas the adult ER defaulted to nebulizations as the standard. This highlights the importance of cross-department communication, as pharmacy may not be aware of unique workflow or clinical needs in specific units.
Formulary decisions should include a range of nebulized options—short-acting beta-agonists (SABAs), short-acting muscarinic antagonists (SAMAs), LABAs, LAMAs, and ICS—to support diverse clinical needs. Favoring only inexpensive short-acting agents may reduce costs but risks limited provider adoption. Long-acting options can improve RT workflow by reducing dosing frequency and may enhance protocol acceptance, especially when aligned with GINA/GOLD guidelines.27,28 Utilization of only low-cost short-acting agents during hospital admission as a substitution for long-acting inhalers at home may yield less than optimal outcomes with an increase in missed treatments, as well as increased RT labor costs often offsetting the medication treatment cost savings. 29 If high costs are a barrier, hospitals should engage manufacturers to negotiate more favorable contract terms such as sub-WAC or sub-GPO pricing.
Protocols should incorporate subprotocols to minimize duplicative therapies, like combining LAMAs with SAMAs or ICS with systemic steroids, which add cost and workload without clear benefit. These combinations are often clinically unnecessary and may increase adverse event risk.30-32 Although not all providers may agree, implementing such measures can improve efficiency and safety. Once finalized, the protocol and any formulary additions should be submitted for P&T committee approval. A class sample interchange can be found in Figure 3 and a detailed inhaler interchange can be found in Supplemental Appendix 2.
Education
Recommendation: Educate all key stakeholders that may be applicable including pharmacists, pharmacy buyers, RTs, nurses, providers, COPD navigators, pharmacy informatics, and procurement supply chain teams.
Education of all involved stakeholders and patients is critical to a successful hospital interchange to all nebulization. RNs, RTs, all pharmacy personnel, supply chain, patient educators, and providers must all be informed regarding specific go-live dates with enough advance warning to ensure the process is carried out appropriately. Creation of documents that describe available data surrounding mixing of various nebulized therapies may foster improved workflow for RTs.33,34 For example, in our experience in a large hospital system, such documents were provided to all RT directors at each facility so that they could be posted to local staff as appropriate.
Implementation
Recommendation: Once all key stakeholders are appropriately educated, implement the protocol based on an agreed-upon approach with specific dates.
Implementation can follow a phased or all-at-once approach. For departments concerned about labor, starting with specific drug classes (eg, LABA or LABA/ICS) and gradually expanding to ICS, LAMAs, SAMAs, and SABAs can ease the transition. This staged rollout allows RT teams to adjust workflows incrementally, minimizing disruption and improving operational adoption of the new protocol.
In large hospital systems, initiating a pilot site before system-wide implementation offers valuable insights. A phased rollout following the pilot allows other hospitals to adopt the protocol based on observed successes and lessons learned, promoting broader buy-in and smoother adoption across the network.
Pharmacy informatics capabilities must be assessed to determine whether implementation should begin with manual processes or full automated therapeutic interchange. Additional key steps include allowing time to deplete existing inhaler stock, ensuring adequate inventory of nebulized medications, and preparing remote dispensing units. New formulary drugs must be evaluated for storage needs and BCMA scan compatibility. Pharmacy buyers should be educated early, with clear timelines, to ensure readiness and prevent supply chain disruptions before go-live dates.
Post-Implementation Outcomes Validation
Recommendation: After enough time has passed to adequately assess the impact of the all-nebulization protocol post-implementation, conduct an assessment on the effects of switching with regard to costs, RT labor, readmissions, and any other preidentified areas of interest or concern to key stakeholders.
Some hospitals/hospital systems may require that a post-implementation outcomes validation step is built into the program for sign off. Key outcomes for evaluation should be identified prospectively (ie, costs, RT labor, readmissions) and reflect the topics most important to involved stakeholders. Publishing results of these outcomes studies can garner additional support from the scientific community for all-nebulization strategies should findings be generally consistent and favorable similar to those already published.1,8,9,17,18
Conclusions
Hospitals may benefit when converting from an inhaler or mixed inhaler/nebulizer approach to an all-nebulization approach for the administration of respiratory medications. We describe and offer our recommendations based on the successful implementation of all-nebulization protocols initiated by RTs and pharmacists for use in a variety of hospital settings that were using a combination of nebulizers and inhalers. Use of an all-nebulization strategy has the potential advantages of improved economics without increasing RT workload when implemented appropriately. Furthermore, the strategy has the potential to optimize patient care by providing a more standardized approach for treating respiratory patients.
Supplemental Material
sj-docx-1-hpx-10.1177_00185787251403051 – Supplemental material for Rationale and Strategy for Switching to an All-Nebulization Protocol for In-Hospital Administration of Respiratory Medications
Supplemental material, sj-docx-1-hpx-10.1177_00185787251403051 for Rationale and Strategy for Switching to an All-Nebulization Protocol for In-Hospital Administration of Respiratory Medications by Trent Larson, Lexie Caraway, Bobby Chasteen and Jason Glick in Hospital Pharmacy
Footnotes
Acknowledgements
Medical writing and editorial support were provided by Hilary Durbano, PhD, of AlphaBioCom, a Red Nucleus company, and were funded by Theravance Biopharma US, Inc.
Author Contributions
All authors conform to the ICMJE recommendations for the conduct, reporting, editing, and publication of this manuscript. All authors have given consent to the submission and publication of this work, have read and approved the manuscript, and are able to publicly discuss and defend the manuscript’s content. All authors performed literature review, manuscript preparation, and manuscript review.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for the development of this manuscript was funded by Theravance Biopharma US, Inc., San Francisco, CA, USA. The authors received no financial support for the research or authorship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
