Abstract
Background:
Since 2017, guidelines recommend a four-drug prophylactic antiemetic regimen—5HT3 receptor antagonist (5HT3RA), NK1 receptor antagonist (NK1RA), dexamethasone, and olanzapine—for patients receiving highly emetogenic chemotherapy (HEC). This study examines prophylactic olanzapine prescribing in patients initiating HEC and associated sociodemographic, clinical, and contextual factors.
Methods:
A retrospective cohort study was conducted using electronic health record data from an academic cancer center and affiliated community practices across the state of North Carolina, joined with geocoded community factors. Adults ≥21 years initiating cisplatin, carmustine, dacarbazine, mechlorethamine, streptozocin, or cyclophosphamide plus anthracycline (AC) in 2022 were included. Antiemetic orders placed before HEC administration were classified as three-drug (5HT3RA, NK1RA, dexamethasone) or four-drug (plus olanzapine). Late olanzapine use (within 30 days post-HEC) was also assessed. Multivariable logistic regression evaluated factors associated with regimen type.
Results:
Among 878 patients, all received orders for the three-drug regimen, but only 300 (34.2%) had olanzapine ordered within 30 days before first HEC. After adjusting for covariates, olanzapine prescribing was less likely in patients with breast (OR = 0.13, 95%CI = 0.07-0.23), gastrointestinal (OR = 0.20, 95%CI = 0.04-0.94), gynecological (OR = 0.14, 95%CI = 0.03-0.69), and head and neck (OR = 0.17, 95%CI = 0.05-0.66) compared to hematological cancers, in those receiving cisplatin (OR = 0.20, 95%CI = 0.06-0.70) and carmustine (OR = 0.02, 95%CI = 0.00-0.48) compared to AC, and in community (OR = 0.40, 95%CI = 0.27-0.61) versus academic settings. Prescribing was also lower among patients residing in low- (OR = 0.49, 95%CI = 0.25-0.93) or lower-middle income (OR = 0.50, 95%CI = 0.32-0.78) areas, but higher among those living >6 miles from treatment facilities (OR = 1.95, 95%CI = 1.08-3.58). Among patients without prophylactic olanzapine, 12.5% were prescribed it within 1 to 30 days post-HEC.
Conclusions:
Despite guideline recommendations, prophylactic olanzapine use in HEC remains suboptimal, influenced by clinical and non-clinical contextual factors. Targeted efforts at information dissemination and updated prescribing systems are needed to promote equitable, evidence-based supportive care.
Keywords
Introduction
Chemotherapy-induced nausea and vomiting are often reported among the most distressing and dreaded side effects of cancer treatment. In a 1983 study, 1 patients undergoing chemotherapy reported nausea and vomiting as the most severe side effects. Nearly four decades later in 2022, despite advancements in developing and disseminating effective prophylactic treatments, nausea remains among the top five most common and severe symptoms experienced by patients undergoing chemotherapy. 2 Beyond the immediate discomfort, chemotherapy-induced nausea and vomiting have been associated with diminished quality of life, interference with daily functioning, avoidable acute care use, substantial resource utilization and economic burden.3-6
Emetogenicity risk is usually categorized based on the regimen and those expected to result in >90% of patients experiencing nausea and vomiting within 24 hours without prophylaxis are considered as highly emetogenic chemotherapy (HEC). 7 In a pivotal randomized controlled trial by Navari, 8 prophylactic olanzapine—an atypical antipsychotic available in low-cost generic formulations 9 —demonstrated substantial improvement in nausea and vomiting symptom control and has since been recommended for use with HEC. 7 Since 2017, major evidence-based guidelines (such as those by ASCO, NCCN, and MASCC/ESMO) recommend a four-drug antiemetic prophylactic regimen before receiving HEC. This prophylactic regimen includes dexamethasone (DEX), a serotonin receptor antagonist (5-HT3 RA) such as ondansetron, a neurokinin-1 receptor antagonists (NK1 RA) such as aprepitant, and olanzapine. 10
However, despite proven efficacy, low costs, and guideline recommendations, olanzapine appears to be underused in real world clinical practice. Studies using national US data prior to 2017 have reported olanzapine use for antiemetic prophylaxis in <2% of patients initiating HEC regimens, 11 increasing to only 14% in 2021 9 —multiple years after inclusion in guideline recommendations. Evidence on uptake from recent years is limited as are the factors associated with its use in practice. In this study, we describe olanzapine prescribing for antiemetic prophylaxis among patients initiating HEC at a tertiary academic medical center or one of its seven affiliated community cancer centers across North Carolina, US. To better understand the contextual determinants of guideline adherence and olanzapine uptake, we also assess the clinical and non-clinical factors associated with patients’ receipt of a prescription for olanzapine. Understanding these prescribing patterns and their determinants is essential for informing strategies to promote appropriate use and ensure that all patients receive evidence-based, high-quality supportive care.
Methods
Study Design and Setting
We used electronic health record (EHR) data from a tertiary academic medical center and its seven community-based facilities with dedicated cancer programs to retrospectively identify a cohort of adult patients prescribed HEC. The health system services residents across North Carolina, USA, and its adjacent areas. Data from the EHR was joined with geocoded community characteristics from the American Community Survey 2020 data. 12 Figure 1 shows a schematic diagram of the study design. 13 Results are reported following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 14
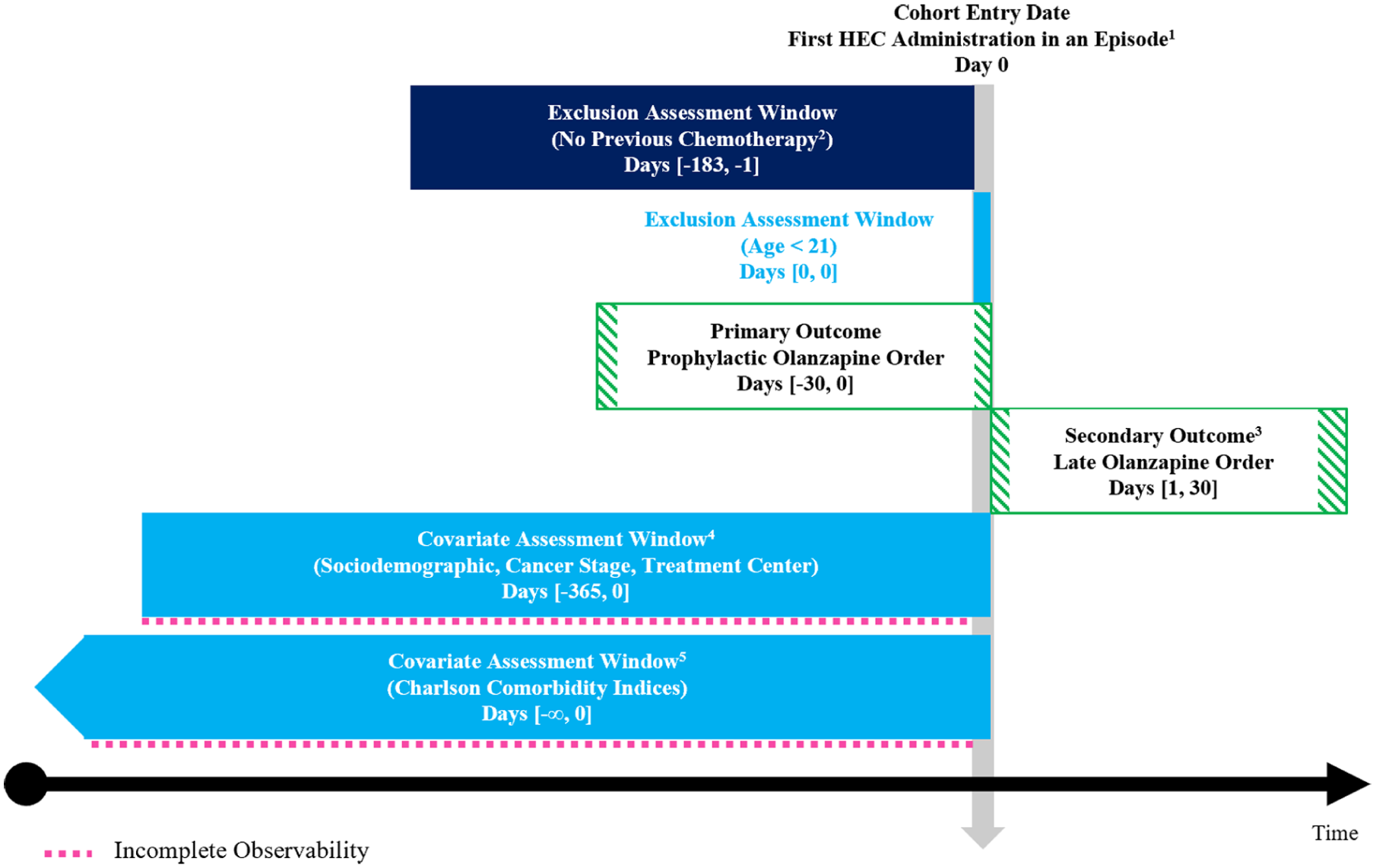
Study design diagram.
Conceptual Framework
Consistent with an ecological perspective on healthcare delivery, 15 we designed our study to consider the multilevel environment of patients, organizations, and the broader communities in which cancer care occurs. As such, we hypothesized that clinician prescribing and guideline adherence could be influenced by factors at multiple levels: (i) individual patient (eg, sociodemographics and comorbidities), (ii) organization (eg, delivery system design), and (iii) community (eg, population socioeconomic status and rurality). Recognizing multilevel influences on care quality is essential for identifying actionable targets and accelerating the translation of evidence into routine clinical practice. 16
Cohort Eligibility
Patients were included if they (i) were ≥21 years of age, (ii) initiated an intravenous or infused HEC regimen in 2022. HEC regimens included cisplatin, carmustine, dacarbazine, mechlorethamine, and streptozocin, as well as combination regimen of cyclophosphamide and an anthracycline (AC) in women in women with breast cancer (ascertained by having the same order date). 17 Patients were included if they had receive either of the regimens above regardless of accompanying supportive medications. To avoid misclassification, we excluded patients prescribed cyclophosphamide, which is considered HEC at doses ≥1500 mg/m2 and ascertaining administered dose from EHR data can be problematic. We also excluded patients prescribed carboplatin, which is considered HEC by the NCCN at area under the curve (AUC) ≥4 (mg/mL)/min, but not by other guidelines. 10 We focused on the first chemotherapy cycle to minimize confounding from prior treatment experiences and anticipatory symptoms, ensuring that prescribing decisions are primarily driven by patient characteristics at treatment initiation, rather than previous treatment experiences.
Outcomes
Since all eligible patients had orders for at least a three-drug prophylactic antiemetic regimen—that is, dexamethasone, 5-HT3 RA, and NK1 RA—the study cohort was categorized into two groups: those without (three-drug regimen) and with olanzapine (four-drug regimen). The primary outcome, evidence of prophylactic olanzapine prescribing, was ascertained from medication orders dated from 30 days prior through the date of first HEC administration (inclusive). As a secondary outcome, we also evaluated the presence of late olanzapine orders, defined as any order for olanzapine at least a day after initial HEC administration until 30 days later among those without an initial prophylactic order as a proxy for uncontrolled or breakthrough CINV symptoms. 9
Covariates
Covariates selection was informed by the multilevel conceptual framework15,16 and prior research on guideline-concordant antiemetic use. 18 Patient-level covariates included sex, age, race, ethnicity, body mass index (BMI), smoking status, health insurance, marital status, preferred language, and distance between the patient’s residence to the treatment facility, as well as clinical variables including chemotherapy regimen, cancer diagnosis, evidence of late-stage cancer, and comorbidity burden as measured by the Charlson Comorbidity Index. 19 Healthcare center-level covariates included academic versus community affiliation. Community-level contextual covariates were measured by joining the area-level information from the 2020 American Community Survey 5-year estimates 12 with the patients’ residential zip-code available through the EHR and included area demographic classification and per capita income by census tract, and rural/urban classification by county.
Statistical Analysis
Descriptive statistics were used to summarize clinical and sociodemographic characteristics of patients by prophylactic antiemetic regimen (with vs without olanzapine). Categorical variables were presented as counts and percentages. Comparisons between the two regimens were conducted using Chi-square tests. For contingency tables with expected cell counts less than five, Fisher’s exact test was applied to ensure accurate results. A multivariable logistic regression model was used to assess factors associated with the primary outcome, controlling for multi-level covariates. Adjusted odds ratios (aOR) with 95% confidence intervals (CI) were reported. Each predictor’s association with the outcome was evaluated using Wald Type II tests. Multiple imputations by chained equations using the mice package in R was used to handle missing values for patient race and ethnicity. 20 As sensitivity analysis, complete case analysis was conducted to test the robustness of findings. The results were consistent with the base model and are not reported. P-value < .05 was considered statistically significant. All analyses were conducted using R Studio.
Results
We included 878 adult patients (aged ≥21) initiating a study eligible HEC regimen in 2022. All patients had orders for at least the three-drug prophylactic antiemetic regimen—including dexamethasone, NK1 RA, and 5HT3 RA—but only 300 (34.2%) had an order for olanzapine within a month prior to their first HEC administration. This number changed to 265 (30.2%) when restricting the lookback period to 7 days.
Baseline Characteristics
Table 1 compares baseline characteristics of those who were versus were not prescribed prophylactic olanzapine. Prophylactic olanzapine prescribing rates were 36.8% (94 of 318) in patients aged <65 years and 29.6% (206 in 560) in those aged 65 years or older (P = .04). Before controlling for other factors, patients with olanzapine were significantly younger (P < .001), less likely to be White (65.7% vs 71.3%, P = .02), more likely to identify as Hispanic/Latino (8.0% vs 3.5%, P = .006), less likely to be current smokers (11.7% vs 15.4%, P = .04), and had lower comorbidity burden (38.0% vs 25.6%, P < .001). The distribution of cancer types differed significantly between groups (P < .001), with patients prescribed olanzapine more frequently diagnosed with hematological malignancies (46.0% vs 13.0%) and less frequently with head and neck (7.3% vs 26.5%) or breast cancer (19.0% vs 27.7%). Prophylactic olanzapine receipt also differed by practice setting, residential distance from the health center, and residential community-level income. Those with olanzapine orders were more likely to receive AC regimen (54.7% vs 34.9%, P < .001), be treated at an academic center (56.0% vs 39.1%, P < .001), live >32 miles from the health center (34.0% vs 22.3%, P = .001), and reside in a middle-income community ($50-99K median income per capita; 30.3% vs 19.6%, P = .004). No significant differences were observed by patient sex, primary payer, marital status, language preference, or BMI category, as well as community-level rural/urban status and racial composition.
Baseline Cohort Characteristics.
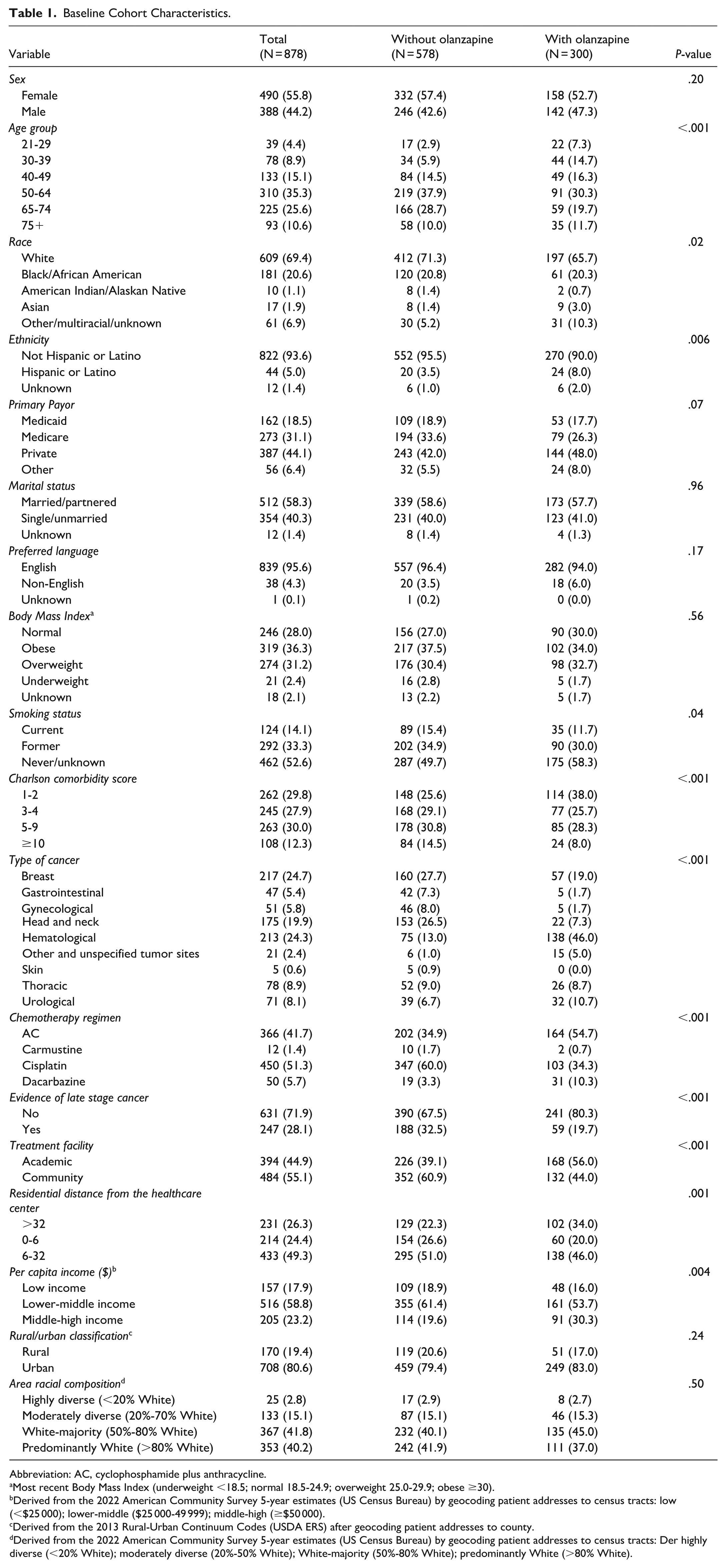
Abbreviation: AC, cyclophosphamide plus anthracycline.
Most recent Body Mass Index (underweight <18.5; normal 18.5-24.9; overweight 25.0-29.9; obese ≥30).
Derived from the 2022 American Community Survey 5-year estimates (US Census Bureau) by geocoding patient addresses to census tracts: low (<$25 000); lower-middle ($25 000-49 999); middle-high (≥$50 000).
Derived from the 2013 Rural-Urban Continuum Codes (USDA ERS) after geocoding patient addresses to county.
Derived from the 2022 American Community Survey 5-year estimates (US Census Bureau) by geocoding patient addresses to census tracts: Der highly diverse (<20% White); moderately diverse (20%-50% White); White-majority (50%-80% White); predominantly White (>80% White).
Predictors of Prophylactic Olanzapine Underuse
Table 2 presents unadjusted and adjusted associations between multilevel characteristics and prophylactic olanzapine prescribing. After adjusting for covariates by multivariable logistic regression, several factors were significantly associated with the odds of olanzapine prescribing for antiemetic prophylaxis among adult patients receiving HEC (Figure 2). Compared to patients with hematological cancers, those with breast (OR = 0.13, 95%CI: 0.07-0.23), gastrointestinal (OR = 0.20, 95%CI: 0.04-0.94), gynecological (OR = 0.14, 95%CI: 0.03-0.69), and head and neck cancers (OR = 0.17, 95%CI: 0.05-0.66) had significantly lower odds of receiving prophylactic olanzapine. Compared to the AC regimen, both cisplatin (OR = 0.20, 95%CI: 0.06-0.70) and carmustine (OR = 0.02, 95%CI: 0.00-0.48) were also associated with lower olanzapine prescribing odds. Patients treated in community settings had lower odds of olanzapine prescribing compared to those treated at the academic medical center (OR = 0.40, 95%CI: 0.27-0.61). Patients residing >32 miles from the treatment center were more likely to receive prophylactic olanzapine compared to those living within 0 to 6 miles (OR = 1.95, 95%CI: 1.08-3.58). Finally, individuals from low-income (OR = 0.49, 95%CI: 0.25-0.93) and lower-middle-income (OR = 0.50, 95%CI: 0.32-0.78) census tracts had significantly lower odds of olanzapine prescribing compared to those from middle- or high-income communities.
Unadjusted and Adjusted Associations Between Patient, Health System, and Community-Level Characteristics and Prophylactic Olanzapine Prescribing.
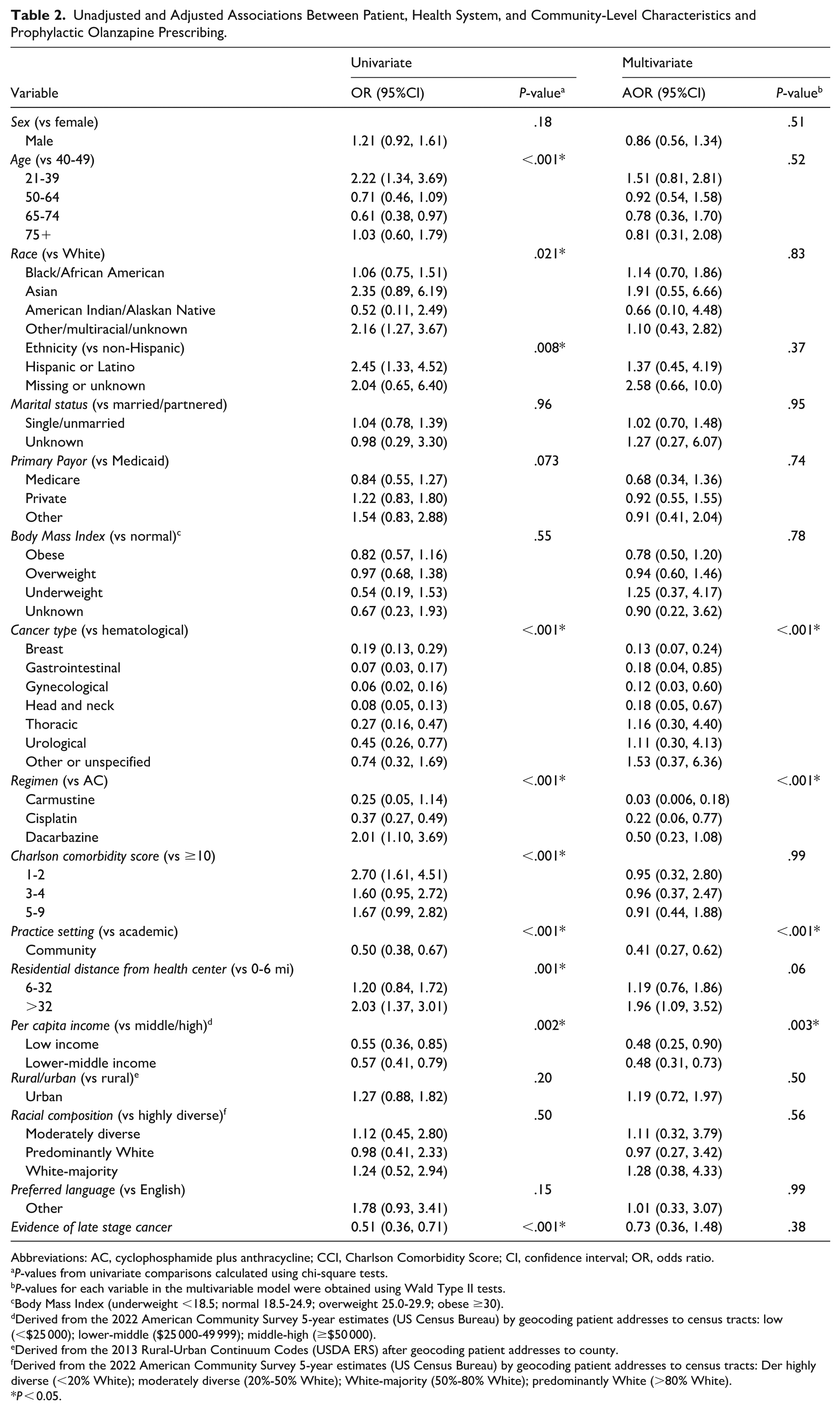
Abbreviations: AC, cyclophosphamide plus anthracycline; CCI, Charlson Comorbidity Score; CI, confidence interval; OR, odds ratio.
P-values from univariate comparisons calculated using chi-square tests.
P-values for each variable in the multivariable model were obtained using Wald Type II tests.
Body Mass Index (underweight <18.5; normal 18.5-24.9; overweight 25.0-29.9; obese ≥30).
Derived from the 2022 American Community Survey 5-year estimates (US Census Bureau) by geocoding patient addresses to census tracts: low (<$25 000); lower-middle ($25 000-49 999); middle-high (≥$50 000).
Derived from the 2013 Rural-Urban Continuum Codes (USDA ERS) after geocoding patient addresses to county.
Derived from the 2022 American Community Survey 5-year estimates (US Census Bureau) by geocoding patient addresses to census tracts: Der highly diverse (<20% White); moderately diverse (20%-50% White); White-majority (50%-80% White); predominantly White (>80% White).
P < 0.05.
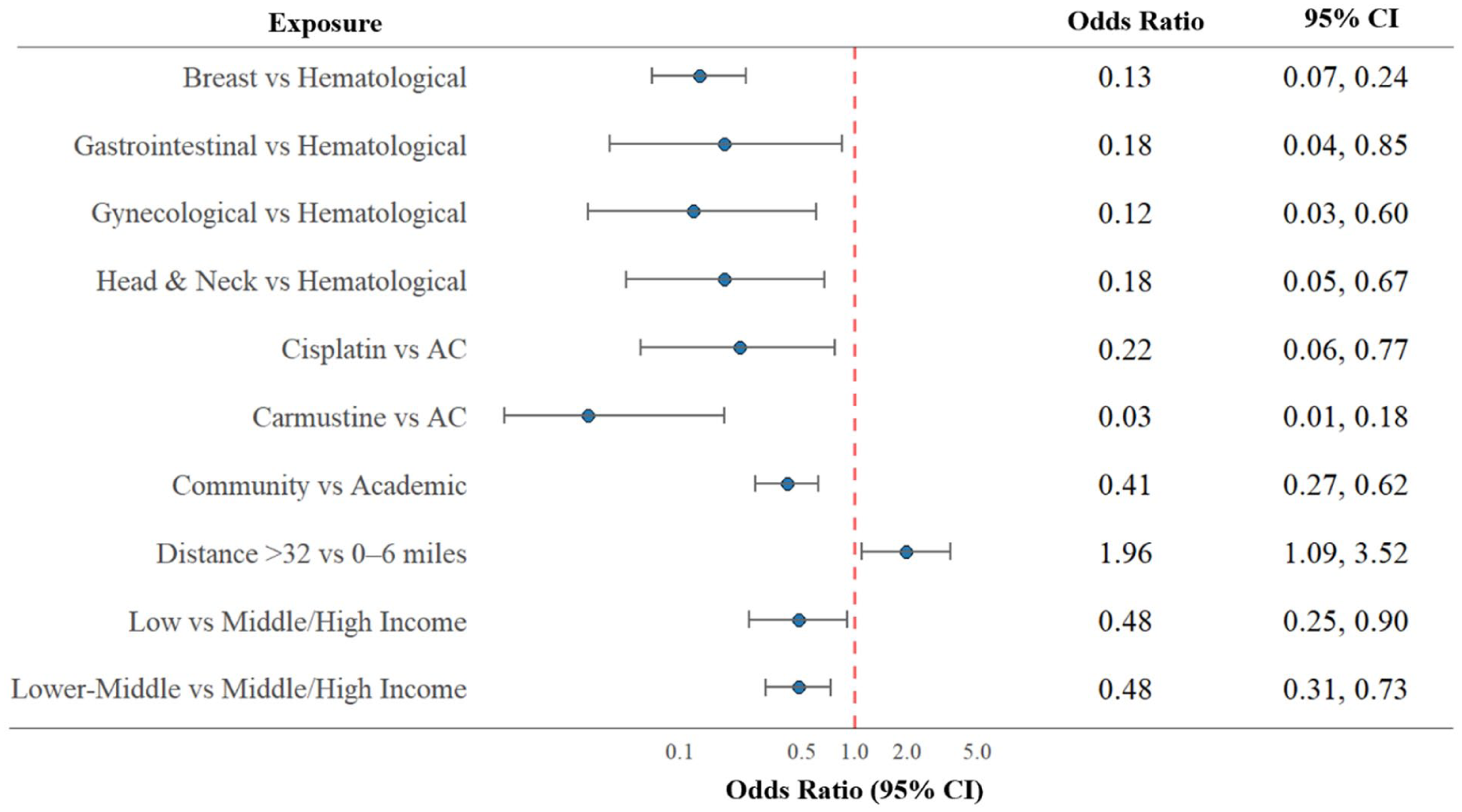
Factors significantly associated with the receipt of olanzapine for antiemetic prophylaxis in multivariable logistic regression.
Late Olanzapine Orders
Among 578 patients initiating HEC who did not a prophylactic olanzapine order, 72 (12.5%) were prescribed olanzapine within the first 30 days (median 14 days) after their initial HEC administration.
Discussion
In a 2022 real-world cohort of patients with cancer initiating intravenous HEC in a large health system comprised of an academic medical center and its community affiliates with dedicated cancer programs, all patients received orders for at least the three-drug prophylactic antiemetic regimen—consisting of dexamethasone, a 5HT3-RA, and a NK1-RA. However, olanzapine was prescribed for just over a third (34%) of patients prior to their first HEC administration. Our findings confirm prior evidence 9 and highlight the persistent underuse of olanzapine for CINV prophylaxis in HEC, despite inclusion in major antiemetic guidelines, low cost, and strong evidence supporting its effectiveness in controlling CINV symptoms across acute, delayed, and overall phases. 8 Our results also illustrate how olanzapine prescribing, similar to other prescribing behaviors, is associated with both clinical factors—such as cancer type and HEC regimen—as well as non-clinical contextual factors—such as practice setting, patients’ residential distance from their treatment center, and community income level, depriving some patients with cancer of evidence-based and guideline-recommended best supportive care during their treatment.
Importantly, over 12% of patients in our cohort without prophylactic olanzapine received it within a month after their initial HEC administration, suggesting inadequate symptom control. This pattern mirrors the late receipt of olanzapine in 6% of patients reported by Childs et al 9 and highlights a critical gap in supportive care. Despite decades of progress, substantial unmet need remains in CINV management. Studies continue to identify CINV symptoms as a major contributor to diminished quality of life,6,21 psychological distress, nutrition impairment, treatment disruption, 22 and acute care use including emergency department visits and unplanned hospitalizations. 23 Moreover, early experiences with CINV strongly predict symptom burden in subsequent treatment cycles. 24 As such, delayed prescribing of an effective prophylactic antiemetic represents a missed opportunity for symptom prevention.
Our study adds to the existing literature documenting the slow uptake of olanzapine for CINV prophylaxis in HEC. O’Sullivan et al 11 reported <1% use in a national US cohort of patients initiating HEC from 2006 to 2015. However, given their use of administrative claims data, they did not ascertain prophylactic use vs treatment of established symptoms. Roeland et al 25 used EHR data from a national US cohort and found olanzapine use in 3% of HEC courses from 2012 to 2018. Most recently, Childs et al 9 reported an increasing use in patients initiating HEC from 1.4% in 2016 to 13.9% in 2021. We report an increased (34%), although yet suboptimal, uptake of olanzapine for antiemetic prophylaxis in HEC. Moreover, all three prior studies lacked the variables to compare academic versus community settings or assess the influence of community-level contextual factors. A novel finding of the present study is the significantly lower prescribing rates for olanzapine at community cancer centers compared to the academic medical center within the same health system, despite virtually universal adherence to prescribing at least the three-drug antiemetic regimen.
These findings reflect some of the well-documented challenges in the provision of guideline-concordant best supportive care for patients in routine oncology practice. Oncologists frequently report difficulty staying current with the multitude of supportive care guidelines, largely due to the competing demands of an increasingly complex and rapidly evolving anticancer treatment landscape.26,27 Additional factors may also contribute to the gaps in supportive care. For instance, CINV symptoms are often not routinely measured and reported to the clinicians. Patients may also hesitate to disclose their symptom severity due to fears of anticancer dose reduction or a misconception that symptom severity indicates treatment effectiveness. 25 Addressing implementation challenges and minimizing underuse and disparities in supportive care prescribing may require better integration of evidence-based guidelines into routine clinical workflows. Embedding evidence-based recommendations into default EHR order sets, may be a practical way to promote consistent and guideline-concordant prescribing at the point of care.
While non-adherence to CINV prophylaxis guidelines have previously been documented,11,18,25 we highlighted the influence of non-clinical contextual factors, including practice setting (community vs academic), patient residential factors (distance from the treatment center) and community socioeconomic status (per-capita income) on receiving guideline-concordant supportive care. We found that patients living closer to treatment centers and those residing in lower-income communities were significantly less likely to receive prophylactic olanzapine. While previous studies have shown similar disparities based on community-level factors in the use of 5HT3-RAs 28 and NK1-RAs, 29 cost is unlikely to drive these differences in the case of olanzapine. Whether disparities are driven by a legacy of patient and provider cost-consciousness, concerns about side effects, or communication gaps related to lower health literacy requires further investigation. These factors contribute to inequitable symptom burden, particularly among underserved populations. Moreover, consistent with an ecological view of care delivery,15,16 our study illustrates that prescribing behaviors and guideline adherence are shaped not only by clinical factors but also by health system structures and community-level social determinants of health. These multilevel influences contribute to inequities in care quality—and likely outcomes—that are unrelated to the underlying disease and other clinical characteristics.
Limitations
Findings should be interpreted in light of several limitations: Our estimates of olanzapine underuse are likely conservative, as we relied on EHR medication orders within a relatively wide lookback period (within 30 days before HEC administration). Additionally, this approach would capture prescribing, rather than patients actually receiving the medication or their adherence. This was necessary to capture oral medications including olanzapine that may not have an administration record in the EHR. Our analysis was conducted within a single academic medical center and its affiliated community practices, which may limit the generalizability of the findings to other institutions or healthcare systems. In addition, while our study design allows for the identification of associations, it does not permit causal inferences regarding predictors of olanzapine use.
Conclusion
We highlighted persistent underuse and inequitable prescribing of olanzapine for antiemetic prophylaxis in HEC despite clinical evidence, affordability, and inclusion in major guidelines. Our findings illustrate the influence of health system structures and community-level socioeconomic status, in addition to clinical factors, on prescribing behaviors and guideline adherence. These multilevel influences contribute to disparities in care quality and likely outcomes. To close this evidence-practice gap, cancer centers and professional societies must advance efforts to integrate up-to-date supportive care guidelines across diverse care settings and address multi-level barriers to equitable antiemetic access.
Footnotes
Acknowledgements
This work was supported by the Clinical Translation Science Award (UM1TR004406) from the National Center for Advancing Translational Sciences, National Institutes of Health, and was presented in part at the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) Annual Meeting, Seattle, WA, June 2025.
Ethical Considerations
This study was conducted in accordance with the ethical standards laid down in the Declaration of Helsinki and the US Department of Health and Human Services policy for the protection of human research subjects (45 CFR 46). The research protocol was approved by the Institutional Review Board of the University of North Carolina at Chapel Hill (Approval number: 23-1149, December 2023).
Consent to Participate
Given the retrospective use of de-identified patient data, informed consent was waived by the Institutional Review Board.
Author Contributions
SF conceptualized the study. SF and JEL developed the methodology. SF and HG conducted the formal analysis. SF, JEL, KC, and JNS contributed to the analysis and interpreted the findings. SF and HG prepared the original manuscript draft, and KC, JNS, and JEL critically reviewed and edited. JEL supervised the project. All authors approved the final version for submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was received from the National Center for Advancing Translational Sciences, National Institutes of Health, through Clinical Translation Science Award Number UM1TR004406.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The de-identified dataset that supports the findings are available from the corresponding author upon reasonable request.
