Abstract
Inaccurate penicillin allergy labels (PALs) results in use of broader, less optimized antibiotics. Studies have shown challenging low-risk PALs is safe and effective. We assessed the proportion of PALs among critically ill patients after a pharmacist driven allergy de-labeling program was implemented in the medical intensive care unit (MICU) between November 2017 and March 2023. There was a notable reduction in the proportion of PALs pre-intervention (14.0%) and post-intervention (12.8%). Persistent reductions in the proportion of PALs among readmitted patients was seen in both MICU (21.4% to 15.9%) and non-MICU patients (13.8% to 11.1%). This study further emphasizes the potential for proactive surveillance and intervention on low-risk PALs by pharmacists to reduce the burden of broad-spectrum antibiotics, which may optimize antibiotic usage and possibly impact institutional antimicrobial spectrum.
Background
Penicillin allergy labels (PALs) lead to changes in antibiotic treatment, with negative impacts on antimicrobial stewardship efforts. PALs are reported by 10% of the U.S. population but only confirmed in 1% to 5% of these meaning less than 1% may have true penicillin allergy. 1 Many receive this label in childhood and frequently carry it into adulthood. As a result these patients may then receive unnecessary broader-spectrum antibiotics, further contributing to adverse reactions, more antibiotic “allergy” labels, antimicrobial resistance and increased health-care costs.2,3 These concerns have resulted in a practice shift towards proactive surveillance and de-labeling of PALs and incorporation of de-labeling into antibiotic stewardship programs. Traditionally, penicillin skin testing was considered the gold standard. However, the utility of skin testing may be limited when the pre-test probability is low, it can commonly result in a false positive. Currently emerging data in both adults and children supports direct oral penicillin challenges in low-risk patients with remote allergy histories, mild cutaneous reactions, or gastrointestinal intolerance. 4
Our pilot protocol showed that a pharmacist-driven de-labeling program was safe and feasible among critically ill patients with low-risk PALs. 5 Afterwards we characterized its effects in coronavirus-19-positive, immunocompromised, or multiple-drug allergy-labeled patients, and reported an overall very low adverse event rate at 0.5% with favorable treatment outcomes. 6 Pharmacists are integral members of the inpatient care team, and are strategically positioned to contribute to the de-labeling process for PAL due to their leading role in both medication management and antimicrobial stewardship. One systematic review of over 5000 patients reported that de-labeling campaigns by non-allergy specialists (including pharmacists) have encouraged the use of penicillin and beta-lactam antibiotics, reduced dependence on non-penicillin agents and have had a low rate (1.7%) of non-serious reactions. 7
Objective
We implemented a prospective pharmacist-led PAL de-labeling protocol in the medical intensive care unit (MICU) on March 31, 2019. The objective of this study was to evaluate the impact of the de-labeling protocol over time. The proportion of patients reporting PALs upon admission and readmission to the MICU were characterized, comparing pre- and post-protocol rates. The secondary aims were to compare these trends in other intensive care units that had not implemented such similar interventions.
Methods
The pharmacist-driven interventional protocol in the MICU at Vanderbilt University Medical Center (VUMC) included testing and de-labeling low-risk patients with PALs. Screening and clinical procedure protocol was approved under an IRB protocol that subsequently led to an approved hospital protocol. Chart reviews and data pulls were approved by IRB protocol. Patients qualified under the protocol if they were stratified to a low-risk status using a validated history-based risk assessment tool. 5 Low-risk PAL patients were designated as either: (1) >5-year history of urticaria, (2) past self-limited cutaneous rashes without systemic involvement, (3) isolated gastrointestinal symptoms, (4) limited details of remote childhood reactions, (5) only a family-based allergy history, (6) fear-based avoidance of penicillin, (7) known penicillin tolerance since original reaction, or (8) other non-allergy symptoms 5 (Supplemental Figure E1). An electronic health record (EHR) based decision support package was developed to identify PAL patients and provide assessment and documentation tools. Pregnant patients, prisoners, those on intravenous vasopressors, those on the mechanical or non-invasive ventilation, were excluded, but could be reevaluated if those statuses changed. Patients stratified as low-risk PALs were approached for informed clinical consent for a one-time 250 mg amoxicillin challenge ordered by the MICU pharmacist, followed by 1 hour of observation.
To assess the impact of the program, we compared PAL trends in the MICU and other ICUs at VUMC from November 2017 through March 2023. We evaluated the percentage of patients that re00185787251337625ported a PAL upon admission and readmission and analyzed temporal trends through an interrupted time series analysis using negative binomial regression modeling. This approach permitted us to evaluate changes in the rates of PALs before and then after this intervention.
Results
Between March 2019 and March 2023, more than 375 PALs in unique patients had been tested and removed in the MICU. Our analysis demonstrated a reduction in the percent of PALs reported amongst MICU admissions: 14.0% (605/4313) pre-protocol to 12.8% (1356/10 598) post-protocol (negative binomial regression for intervention, P < .05). The percent PALs amongst admissions in non-medical ICUs at VUMC did not change, 7.7% (1380/17 883) pre-protocol versus 7.8% (4509/57 251) post-protocol (P = .64) (Figure 1).
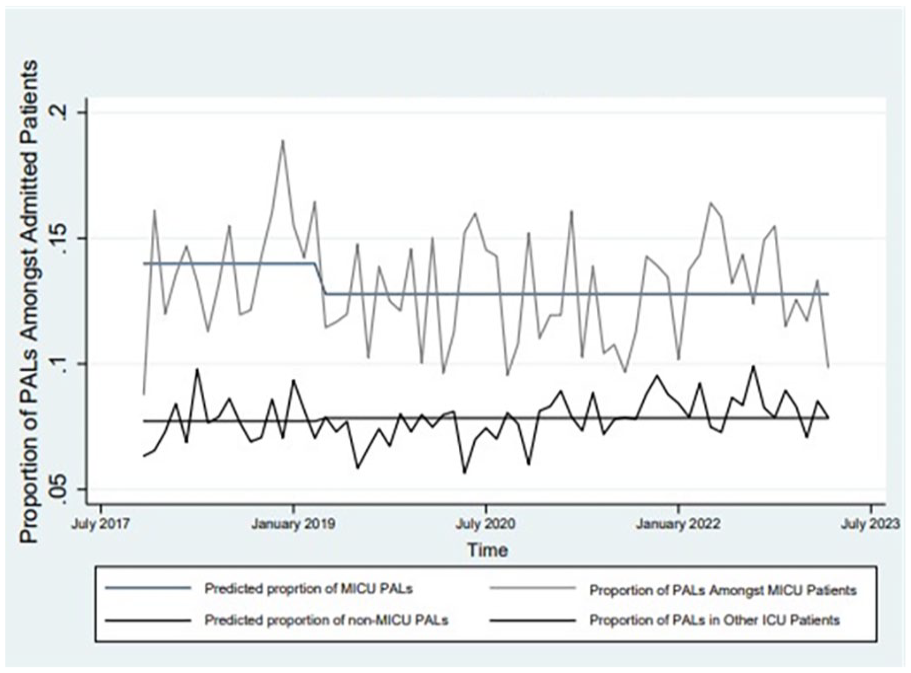
Time trend of patients admitted to the MICU or other ICUs with a penicillin allergy.
Among patients readmitted to the MICU, the percentage of patients with a PAL significantly decreased from 21.4% (120/560) pre-protocol to 15.9% (276/1734) post-protocol (P < .001). For patients readmitted to non-medical ICUs, the PAL rate fell from 13.8% (275/1991) pre-protocol to 11.1% (1196/10 780) post-protocol (P = .001) (Figure 2). During the study period nine patients were temporarily re-labeled with a PAL, but subsequently de-labeled again by another health care provider.
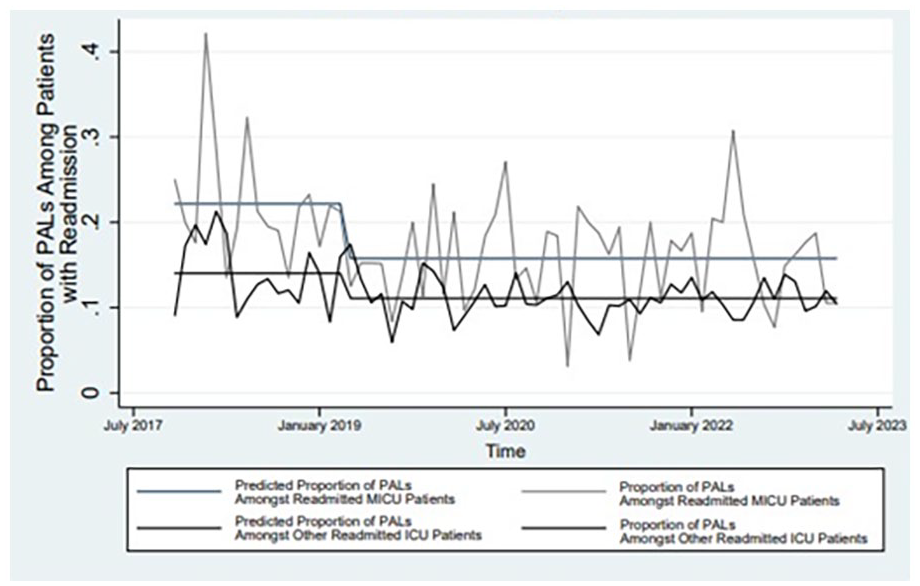
Time trend of patients readmitted to the MICU or other ICUs with a penicillin allergy.
Discussion
Our pilot pharmacist-led intervention successfully reduced the burden of PALs in the MICU, particularly in patients who were readmitted to any ICU. These findings suggest that de-labeling during a previous hospitalization was a driver of reduced rates over time. Patient crossover from the MICU may have contributed to the reduction in PALs in other ICUs; though similar protocols were initiated in other units during this time, including a randomized trial in 12 non-ICUs starting November 2021 (NCT03702270) after approval of a hospital-wide protocol. The data for these other units is available at clinicaltrials.gov and will be reported in the future. This possibility may partially account for the more diffuse reduction in the PAL rates in other ICUs. Given the protocol was carried out by the sole MICU pharmacist, medical staff crossover between ICUs likely plays a minimal or negligible role in these outcomes.
Indeed, prior research supports that active pharmacist-led de-labeling initiatives are related to significantly decreased use of broad-spectrum antibiotics such as vancomycin (33%), fluoroquinolones (36%), carbapenems (50%), clindamycin (61%), and aztreonam (68%), with a 20-fold rise in the usage of penicillin antibiotics, further augmenting antimicrobial stewardship efforts. 8 This might improve antimicrobial spectrum index and be cost neutral.
Limitations of our study include the study design being a non-controlled trial without the ability to concurrently control for contamination or overlap of patients de-labeled in the MICU and readmitted in another ICU. Contamination, overlap, or the positive impact of this intervention in the MICU could contribute to a reduction in other unit’s burden of PALs. During the study period, we think this occurred in the other ICUs in a beneficial way. Namely, that patients who were previously admitted to MICU or another unit where the PAL de-labeling protocol became active may have “crossed over” to a different ICU during a readmission, as witnessed by several study authors. Baseline period PAL rates variation amongst ICUs may be due to underlying population complexities. Our inability to track exact per-patient interventions limits our ability to definitively determine that the observed trend in reduced burden of PALs is directly related to our intervention and not a secular trend. Consequent studies should be supported by matched cohorts of patients and RCTs to give stronger evidence of the effect of the intervention. In our prior study, de-labeled PALs remained removed for >94% patients in a 2-year follow-up period. 6 Long-term follow-up in the ICU will also be required to assess durability of effects from de-labeling and potential re-labeling of PALs over time. 9
Conclusion
A pharmacist-led PALs de-labeling program for low-risk penicillin allergies significantly reduced the prevalence of PALs among critically ill patients in the MICU, especially among those with readmissions. This suggests that penicillin allergy de-labeling is beneficial not only acutely, but also in subsequent hospital encounters for optimizing antibiotic utilization and should be considered a “gift that keeps on giving.”
Supplemental Material
sj-docx-1-hpx-10.1177_00185787251337625 – Supplemental material for Impact of a Pharmacist Driven Penicillin Allergy De-labeling Protocol on Rates of Reported Allergy in the Intensive Care Unit
Supplemental material, sj-docx-1-hpx-10.1177_00185787251337625 for Impact of a Pharmacist Driven Penicillin Allergy De-labeling Protocol on Rates of Reported Allergy in the Intensive Care Unit by Mujtaba Mahmud, Shahristan R. Kokoy, Joanna L. Stollings, Allison B. McCoy, Grace Koo, Mary Lynn Dear, Todd W. Rice, Elizabeth J. Phillips and Cosby A. Stone in Hospital Pharmacy
Footnotes
Acknowledgements
Data from this paper is derived from projects previously approved by the Vanderbilt Institutional Review Board (#181180 and #181734).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was received from the Agency for Healthcare Research and Quality’s Patient-Centered Outcomes Research (PCOR) 1K12HS026395–01 and the American Academy of Allergy, Asthma and Immunology Foundation Faculty Development Award
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
