Abstract
Impact on Practice
Clinical PIs has been proven to promote rational antibiotic usage and reduce toxicity, potentially improving patient care.
Antimicrobial resistance (AMR) is likely to decline by PIs because of the decrease in using broad-spectrum antibiotics, as these medications can be kept as last-resort antibiotics.
Undue antimicrobial use was reduced due to PIs and physicians’ high approval rate.
Introduction
Antimicrobial drug use must be rational to minimize antimicrobial resistance (AMR), one of the main risks to public health because of the associated morbidity, healthcare costs and mortality. For instance, it has been reported that AMR costs more than 9 billion euros annually in Europe alone. 1 In addition, the Centers for Disease Control and Prevention (CDC) estimates that AMR raises direct healthcare expenditures in the United States by an additional $20 billion annually, not including the estimated $35 billion in productivity losses. The CDC estimates that more than 2 million Americans fall ill with antibiotic-resistant illnesses each year, leading to at least 23 000 fatalities. 2 Increased antibiotic use accelerates the emergence of resistance, in which bacteria evolve and render drugs ineffective; as a result, antibiotic monitoring is critical. 3 Developing and disseminating clinical practice guidelines tailored to local needs, as well as adhering to them, is critical for preventing AMR and ensuring judicious antimicrobial use. Antimicrobial stewardship programs (ASPs) are a promising way to combat AMR through mutual participation by multidisciplinary healthcare workers such as clinical pharmacists, physicians, and other skilled healthcare professionals involved in ordering, administering, dispensing, and monitoring antibiotics. 4 It’s a continued work by a health system to rationalize antimicrobial usage by patients to improve treatment outcomes, prevent antimicrobial-related adverse events, and make sure cost-effective treatment. 5
The second most common reason for death for cancer patients is infection. 6 The immune system and other bodily systems can be adversely impact by cancer and cancer treatments in many ways. Cancer patients may be more susceptible to infections because of the cancer itself, particular cancer treatments, unhealthy diets and medications, or other health issues unrelated to cancer. Hence, infections are generally noticed in cancer patients, and effective antibiotics are needed to prevent and cure these infections. The pressing threat to the continued success of cancer therapy is the loss of antibiotic efficacy brought on by bacterial drug resistance. Antibiotic treatment failure in cancer patients raises the risk of sepsis, sepsis-related death, and sepsis-related healthcare expenses. 6 As a result, it’s no surprise that oncologists were among the first to recognize the clinical implications of rising AMR. Infections could be a problem for those receiving chemotherapy and it is well-documented that chemotherapy may become more difficult due to drug-resistant infections. Chemotherapy may promote the evolution of the microbes and the emergence of mutant bacteria in patients who become resistant to antibiotics. It indicates a new level of intricacy in the relationships between chemotherapy, gut flora, and cancer. When the white blood cell count is at its lowest, which may continue for 5 to 7 days between 7 and 12 days after each chemotherapy dose, a patient is more likely to experience side effects. AMR seeks to destroy much of the hard-won development against cancer, so improving the use of existing antibiotics and discovering new antibiotics is vital. 7 The American Cancer Society, the Centers for Disease Control and Prevention and the National Comprehensive Cancer Network offer advice to cancer patients and their healthcare teams on how to impede infections in cancer patients.8,9
The clinical pharmacist could contribute significantly in monitoring prescriptions of antimicrobials, guiding and training physicians and other healthcare personnel at small hospitals lacking a formal ASP. Small community hospitals are less likely to be able to implement an appropriate ASP, so they remain one of the last frontiers for ASPs. 10 Clinical pharmacist interventions (PIs) can help to optimize antibiotic usage, enhance outcomes, encourage rational prescription, minimize inapt use, and, in the end, possibly prevent the development of AMR. The significance of PIs in verifying medication usage has previously been well-documented.8 -10 To the best of our knowledge, there is a paucity of data regarding the clinical PIs on antimicrobials in oncological settings. Hence, the present study was conducted to determine the impact of clinical PIs on antibiotics usage in hematology-oncology set up in Karachi, Pakistan.
Methodology
Study Design and Setting
Over 4 months (from 18 June 2019 to 15 September 2019), this observational prospective study was conducted in a well-known 75-bed teaching hospital, which is a dedicated center of hematology, Bone Marrow Transplantation and allied surgical/medical specialties in Karachi, Pakistan without a structured ASP. It is the only tertiary healthcare and educational facility in the nation dedicated solely to providing complete diagnosis and treatment for individuals with blood diseases. The study comprised all those patients who were hospitalized for inpatient treatment (including pediatrics). The inclusion criteria were the patients admitted to the hospital during the study period and those having received antibacterial, antiviral, and antifungal therapies. Patients were omitted if they were transferred from another medical department, moved to the intensive care unit (ICU) for additional care, got antibiotics before admission, or were not given antibiotics while in the hospital.
Antimicrobial Interventions by Pharmacists
The primary outcome of this study was to compare antibiotic appropriateness and the secondary outcomes were to observe antibiotic utilization, therapeutic de-escalation and undue length of therapy. The following aspects of antimicrobial prescriptions were focused on by the clinical pharmacists for possible interventions: correct dosage and indication; allergies, drug adverse events, interactions and intolerance; an antibiotic shift from parenteral to oral administration; therapeutic de-escalation (a switch in antibiotic for targeting a narrower spectrum of antimicrobial activity); undue length of therapy. After reviewing the patient medication profile, PIs that necessitate a change in antimicrobial agent or treatment cessation due to prolonged therapy were communicated by the clinical pharmacist to the physician based on ASP principles either face to-face and sometimes on telephonic coordination during the rounds of a clinical team. Since there is not a formal structured ASP protocol followed in the hospital; therefore, the use of local epidemiology to help select empiric therapy, avoid drugs with overlapping activities, change antibiotics when culture findings become accessible, evaluate for toxicity, and optimize the dose, route, and duration of treatment were the principles considered by the clinical pharmacists.
Dose modification or interval adjustment with a purpose given (obesity, renal impairment, or other); drug replacement owing to unsuitability; sequential therapy; treatment de-escalation; discontinuing antibiotic if the infection was subsiding (in case if a patient had febrile neutropenia with continued neutropenia the antibiotic was discontinued with an absolute neutrophil count (ANC) of less than 500 cells per microliter who have remained afebrile for 5 to 7 days without experiencing any problems); cessation due to lack of indication to proceed; and other. PIs were categorized as being introduced to either assure adequate antimicrobial prescription or to minimize side effects for clinical advantages. Based on the existing literature, the clinical pharmacist created this classification. Replacement with a more adequate antimicrobial; treatment de-escalation; dose escalations for any rationale; frequency or dosage modifications for pharmacokinetic/pharmacodynamics purposes; a switch from IV to the oral route of administration; and rectification of a drug-drug interaction that might decrease efficacy were among the PI outcomes considered to ensure appropriate antimicrobial prescription.
Data Collection
The clinical pharmacist began performing and recording PIs in antimicrobial prescriptions on 18 June 2019, using an approach identical to a potential inspection. Every day (Monday-Friday), an automated report was sent to the clinical pharmacist detailing all hospitalized patients who had received any antimicrobial in the preceding 24 hours. The date of admission, antimicrobial agent, drug dosage, first administration date, weight and height of the patient, and location of the patient in the hospital were all included in the report. The information was gathered from patient medical histories, laboratory records, and microbiological studies. The Cockcroft-Gault equation was used to compute creatinine clearance in adult patients as necessary. Patients were monitored until they were discharged.
In the pharmacy department database, the proposal for PI, the physician’s approval or refusal of the recommendation, and the refusal reason were all noted. PIs were not documented in the medical records of the patients. The pharmacy system collected baseline characteristics and clinical data for each patient, including gender, admitting service, age, and diagnosis for admission.
Statistical Analysis
The frequency of PIs and acceptance were given as descriptive statistics. The rates of physician approval for PIs aimed at minimizing toxicity and PIs intended at assuring proper antimicrobial prescription were compared using Chi-squared analyses. The significance level for all comparisons was considered at P < .05.
Results
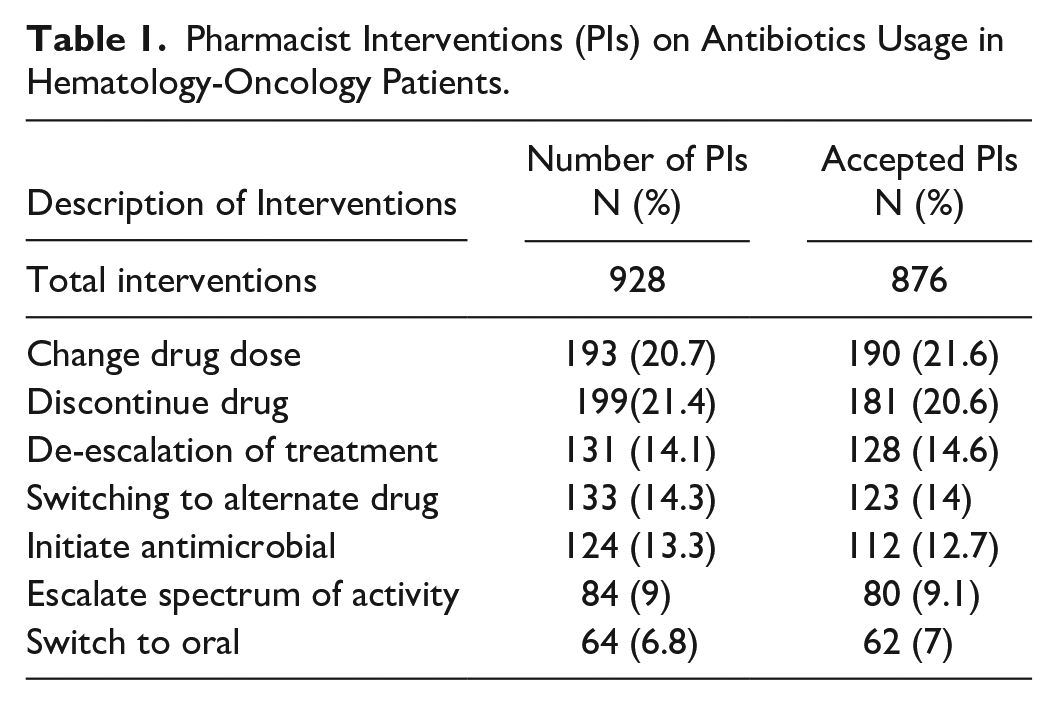
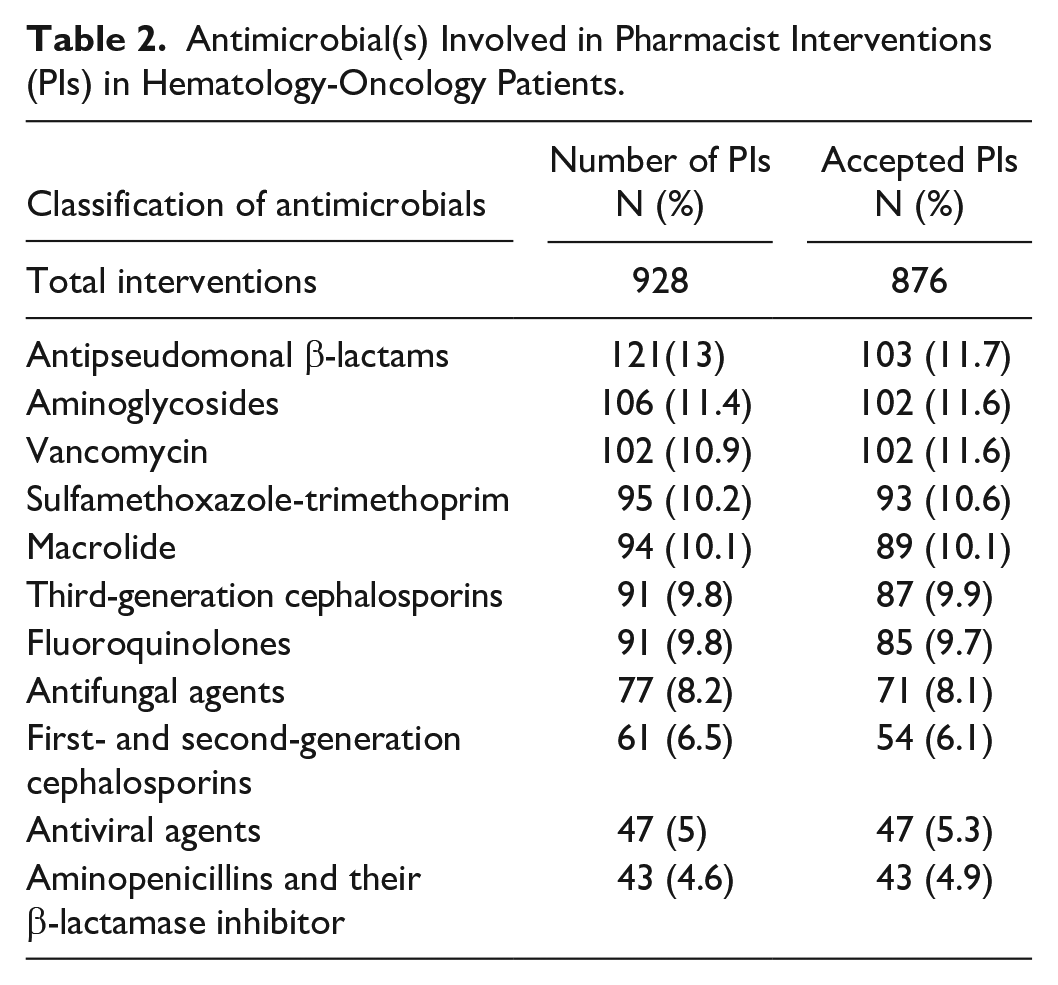
Overall 876 PIs (1-5 per patient) were implemented. Dose modifications or dosing interval changes accounted for the majority of all PIs, and the single most commonly conducted PI was dose modification or interval adjustment due to renal impairment (n = 190, 21.6%). Besides, the discontinuance due to undue long duration or lack of indication to continue necessitated most interventions (n = 181, 20.6%) followed by a de-escalation of treatment (n = 128, 14.6%) (Table 1). Over three-quarters of all recommendations were for PIs related to antipseudomonal β-lactams, aminoglycosides, sulfamethoxazole-trimethoprim and vancomycin (Table 2). Overall, 94.3% (n = 876) of the 928 implemented PIs were accepted. The most common reason for declining a PI was discharge from the hospital shortly after the PI (n = 21). The lowest acceptance percentage was for PIs for dosage adjustment or interval change owing to obesity (9.2%). Clinical benefit percentages were 71.4% for PIs that ensure adequate antibiotic prescription and 28.6% percent for PIs that reduce toxicity. The rates of acceptance for PIs relating to adequate antimicrobial treatment and toxicity were respectively 89.2% and 77.4% (P = .0001). After adopting a PI in any patient, no adverse outcomes were observed. Table 3 depicted the key areas for further interventional stewardship exploration in oncology. The major areas of PI were diagnostic stewardship, dose optimization, clinical pathways and guidelines and antibiotic prophylaxis.
Pharmacist Interventions (PIs) on Antibiotics Usage in Hematology-Oncology Patients.
Antimicrobial(s) Involved in Pharmacist Interventions (PIs) in Hematology-Oncology Patients.
Key Areas for Additional Interventional Stewardship Research in Oncology.

Discussion
We examined the impact of interventions made by clinical pharmacist in a well-known 75-bed teaching hospital, specializing in bone marrow transplantation in Karachi, Pakistan, and is illustrative of a context where more evidence on ASPs is desperately required. We found that the recommended treatments were well-received and that overall usage of special-vigilance medications decreased while the use of aminopenicillins and their β-lactamase inhibitor increased, with no associated side effects. Clinical improvements, such as improved correct antimicrobial prescribing and lower toxicity, may have resulted from the changes, as well as a decrease in total antimicrobial medication spending. 11
The most often proposed PI in our study, as in other parallel studies, was antimicrobial dose modification or interval change due to renal impairment.12,13 In light of these findings, and for the reason that PIs appear to have a direct optimistic influence on treatment outcomes in patients with renal impairment, we recommend that attending physicians get pharmacological training on dosage for patients with impaired renal function. We further recommended that due to the high prevalence of these therapies, randomized controlled clinical trials could assess the clinical and economic consequences of PIs for patients with renal impairment.
Other studies directed in small- to medium-sized hospitals applying ASP recommendations have found a higher acceptance (83.4%) for the suggested PIs, which is supported by our findings.14,15 The acceptance rate for preventive measures that could assure adequate antibiotic prescription was much greater than for interventions aimed at minimizing toxicity. It is a general practice that when there is a sign of toxicity, attending physicians are more confident in the proposed modifications. 16 The lowermost acceptance rate (9.2%) was for PIs involving drug dose changes or interval modifications related to obesity, most likely because attending physicians believed that increasing the dose would raise the chance of an adverse event. Similar findings were reported by another study. 17
Even though no antimicrobials were specifically restricted, the decline in the usage of special-vigilance medications was observed supplemented with a considerable increase in the usage of aminopenicillins and their β-lactamase inhibitor. Several studies led in small- to medium-sized hospitals with formal ASPs have found comparable results: a decrease in carbapenem use, which was linked to an increase in piperacillin-tazobactam use.16,17 The high acceptance rate described in this study could decrease the undue antimicrobial expenditure, implying savings of cost for the healthcare system but notably profiting society, as these medications can be designated as a last-resort treatment approach, decreasing the possibility of escalating drug resistance. 13
As far as we know, no intervention was risky to the patient, as evidenced by the fact that there were no adverse effects documented. Nonetheless, there are significant limitations to this research. First, we didn’t check if our recommendations had an impact on clinical outcomes like the duration of hospital stay, readmission, or mortality rates. Such results are occasionally considered in the context of ASP recommendations, and when they are, they are usually non-significant. 18
Conclusions
Clinical PIs have been demonstrated to promote the rational usage of antibiotics and reduce toxicity, potentially improving quality of care in cancer patients. Because these medications can be reserved as last-resort antibiotics, the decrease in the usage of broad-spectrum antibiotics found in the present study is likely to prevent AMR. The PIs and the higher physician acceptance rate revealed the reduction of undue antimicrobial expenses, which is particularly important in small hospitals with limited financial resources.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
