Abstract
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) is one of the most widespread and dangerous pathogens in healthcare settings. 1 Global antimicrobial surveillance trends indicate that 40% of all S. aureus isolates are resistant to methicillin, with the highest prevalence of MRSA (54.7%) in the East South Central portion of the United States. 2 Vancomycin remains the drug of choice to treat severe MRSA infections. 3 Available data consistently demonstrate reduced time to target concentrations with use of vancomycin loading doses.4-7 As a result, the administration of vancomycin loading doses are commonly used in clinical practice. However, routine use of vancomycin loading doses is not supported by evidence from large randomized trials, and few studies have assessed the impact of loading doses on patient-centered clinical outcomes. Vancomycin has an established dose-response and dose-toxicity correlation and has been associated with nephrotoxicity, particularly in the setting of vancomycin area under the curve (AUC) ≥600 mg/L■hour and trough concentrations ≥15 μg/mL.8-10 While safety data surrounding loading doses remains inconsistent, avoiding vancomycin loading doses when not indicated has been proposed as a strategy to help mitigate the risk of nephrotoxicity when vancomycin and other nephrotoxic agents are used in combination. 11
Consensus guidelines for vancomycin therapy were published in March of 2020 by the American Society of Health-System Pharmacists (ASHP), the Infectious Diseases Society of America (IDSA), the Pediatric Infectious Diseases Society (PIDS), and the Society of Infectious Diseases Pharmacists (SIDP). 12 These guidelines state that a loading dose of 20 to 35 mg/kg can be considered for critically ill patients with suspected or documented serious MRSA infections in order to achieve rapid attainment of target vancomycin concentrations. In effort to align with evidence-based recommendations and improve the quality and safety of vancomycin use within our institution, our institution’s Antimicrobial Stewardship and Pharmacy and Therapeutics Committees approved changes in April 2020 related to use of vancomycin loading doses within the parameters of our pharmacist-driven vancomycin dosing and monitoring protocol. These changes reserved vancomycin loading doses for patients with hemodynamic instability evidenced by one of the following criteria: systolic blood pressure (SBP) <90 mmHg, mean arterial pressure (MAP) <60 mmHg, or requirement of vasopressor support. Prior to this revision, all patients treated with vancomycin within our institution received an initial weight-based loading dose. The purpose of this study is to assess clinical efficacy and safety outcomes related to the use of vancomycin loading doses.
Methods
Study Design
This was a single center, retrospective, quasi-experimental study of critically and non-critically ill patients treated with vancomycin from December 1, 2018 to August 1, 2021 within a large academic medical center. Patients were included in this study if they were ≥18 years of age, had culture-proven MRSA infection from any source, and received vancomycin therapy for a minimum of 5 days. Exclusion criteria were as follows: requirement for renal replacement therapy before or during hospitalization, receipt of vancomycin prior to inpatient admission, presence of polymicrobial infection, or transition to comfort care during infectious treatment period. Patients who received vancomycin therapy prior to our institution’s vancomycin dosing protocol revisions (pre-intervention) were compared to patients who received vancomycin after the revisions (post-intervention). During the entire study period, vancomycin maintenance dosing was based on an institution-specific nomogram based on patient weight and renal function. The nomogram was designed to target steady-state trough concentrations of 10 to 20 μg/mL, which was previously the target therapeutic range for vancomycin based on consensus guidelines prior to March 2020. 13 In the pre-intervention group, all patients were eligible to receive a weight-based vancomycin loading dose prior to initiation of a maintenance dose. Patients in the post-intervention group were started directly on a vancomycin maintenance dose according to the institution-specific nomogram if they did not meet criteria for a loading dose (ie, SBP < 90 mmHg, MAP < 60 mmHg, or requirement of vasopressor support). In both groups, loading doses were weight-based, with a target range of 20 to 30 mg/kg/dose, without exceeding a maximum of 3000 mg. Front-line pharmacists were responsible for vancomycin dosing and monitoring during the entire study period.
Outcomes
The primary outcome was all-cause, inpatient mortality. Secondary outcomes included persistent systemic signs of infection at ≥5 days after vancomycin initiation (ie, white blood cell (WBC) count ≥12 × 103 mm3 and temperature ≥100.4°F), switch to alternative anti-MRSA therapy, and nephrotoxicity determined by presence of acute kidney injury (AKI) while receiving vancomycin and up to 72 hours after treatment discontinuation. The presence of AKI was determined by the serum creatinine (SCr) component of the Kidney Disease Improving Global Outcomes (KDIGO) criteria defined as a rise in SCr ≥ 0.3 mg/dL within 48 hours (during the course of vancomycin therapy). 14 Switch to alternative anti-MRSA therapy was classified by a change in anti-MRSA therapy due to lack of efficacy or patient intolerability, in which rationale was provided in documentation. Antibiotic de-escalation was not included as a switch to alternative anti-MRSA therapy. Baseline demographics were collected to assess comparability of disease severity and comorbidity burden between the two cohorts including: need for mechanical ventilation, receipt of vasopressor support, intensive care unit (ICU) admission, history of chronic kidney disease, and immunocompromising conditions. Vancomycin therapeutic drug monitoring was assessed by comparing initial trough concentrations and total percentage of trough concentrations within the pre-specified target range of 10 to 20 μg/mL. 15 All trough concentrations included for analysis were obtained at steady-state. Receipt of concurrent nephrotoxins while on vancomycin therapy was classified as receipt of the following: aminoglycosides, amphotericin B, angiotensin converting enzyme inhibitors, angiotensin receptor blockers, cidofovir, ganciclovir, loop diuretics, non-steroidal anti-inflammatory drugs, piperacillin/tazobactam, sulfamethoxazole/trimethoprim, or tacrolimus (a full list of extracted data is provided in Table S1 in the Supplemental Appendix). This study was approved by the Institutional Review Board at the University of Alabama at Birmingham with a waiver of informed consent due to the study design.
Statistical Analysis
Descriptive statistics were used to summarize categorical variables as frequencies and percentages, and continuous variables as means and standard deviations. Categorical variables were compared using the chi-square test, or Fisher’s exact test if the assumptions for the chi-square test were not tenable. Distributions of continuous variables were assessed using graphical techniques and tests of normality. All continuous variables except for hospital length of stay were normally distributed. As such, all continuous variables except for hospital length of stay were compared using the two-group t-test; hospital length of stay was compared using the Wilcoxon rank-sum test. Unadjusted logistic regression analyses were performed to estimate odds ratios (OR), 95% confidence intervals (CI), and P-values for potential predictors of outcomes included in these models. Multivariable logistic regression models for the secondary outcomes were assessed using the following pre-specified variables identified by the investigation team with the potential to influence the outcome(s) of interest during the treatment period: initial trough levels <10 μg/mL, any therapeutic trough value while on therapy, and presence of bacteremia (for temperature ≥100.4°F and WBC ≥12 × 103 mm3 at ≥5 days after treatment initiation); concomitant steroid use and immunocompromising conditions were also evaluated as a predictor of prolonged leukocytosis. In addition, BMI ≥ 30 kg/m2, chronic kidney disease, vasopressor support, receipt of loading dose, trough values ≥15 μg/mL, and number of concomitant nephrotoxins were assessed for their influence on nephrotoxicity. Multivariable logistic regression analyses were not performed for the primary outcome due to the low incidence of mortality in each group [n = 3 (4.8%) for the pre-intervention group, n = 4 (6.8%) for the post-intervention group]. Additional analyses were performed to compare the primary outcome, secondary outcomes, and selected patient characteristics between pre-intervention patients who received a loading dose and post-intervention patients who did not receive a loading dose. These analyses were performed using the same statistical methods (ie, logistic regression analyses and chi-square tests) as previously described. All tests of significance were two-tailed and a P-value of ≤.05 was considered statistically significant. All statistical analyses were performed using SAS (SAS Institute Inc., Cary, NC, USA), version 9.4.
Results
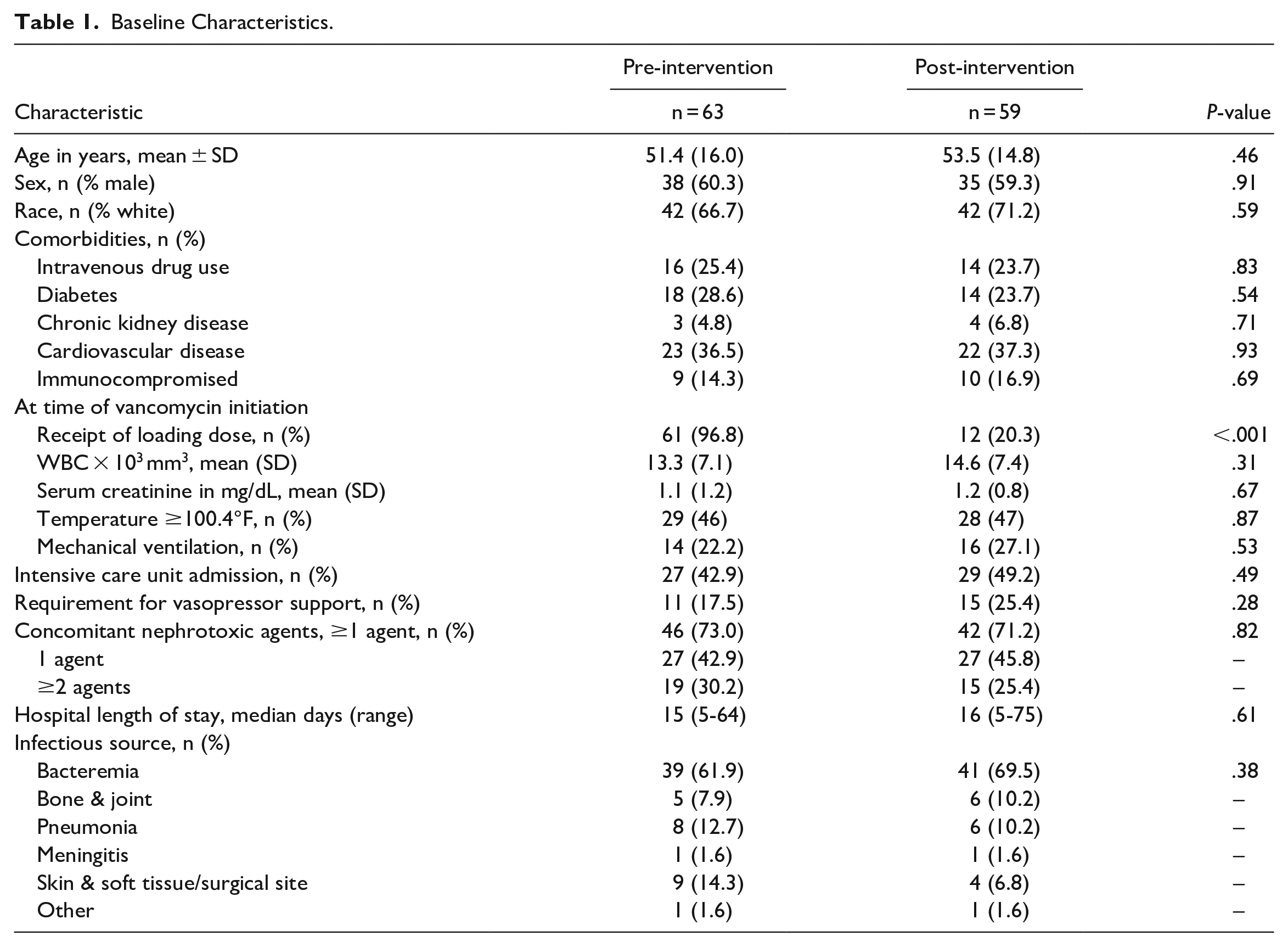
A total of 443 patients were identified as having culture-proven MRSA infection between December 1, 2018 and August 1, 2021. After applying exclusion criteria, 122 patients were available for final analysis (Figure 1). Of these patients, 63 (52%) received vancomycin prior to our institution’s vancomycin protocol revision (pre-intervention) while 59 (48%) received vancomycin after our protocol revision (post-intervention). Baseline characteristics were well-matched between the two groups (Table 1). As anticipated, there was a significantly higher prevalence of loading dose recipients in the pre-intervention group compared to the post-intervention group (97% vs 20%, respectively; P < .001). In each group, the most common site of infection was bacteremia (additional details regarding infectious source can be found in Table S2 in the Supplemental Appendix). Percent of patients requiring vasopressor support, mechanical ventilation, and admission to an ICU were similar in both groups. Of the patients who received a loading dose in the pre-intervention group, only 9 (14%) would have met at least one of our revised protocol’s loading dose criteria. In the post-intervention group, 8 (14%) patients met criteria for a loading dose, thus reinforcing similar severity of illness between patients in each group.
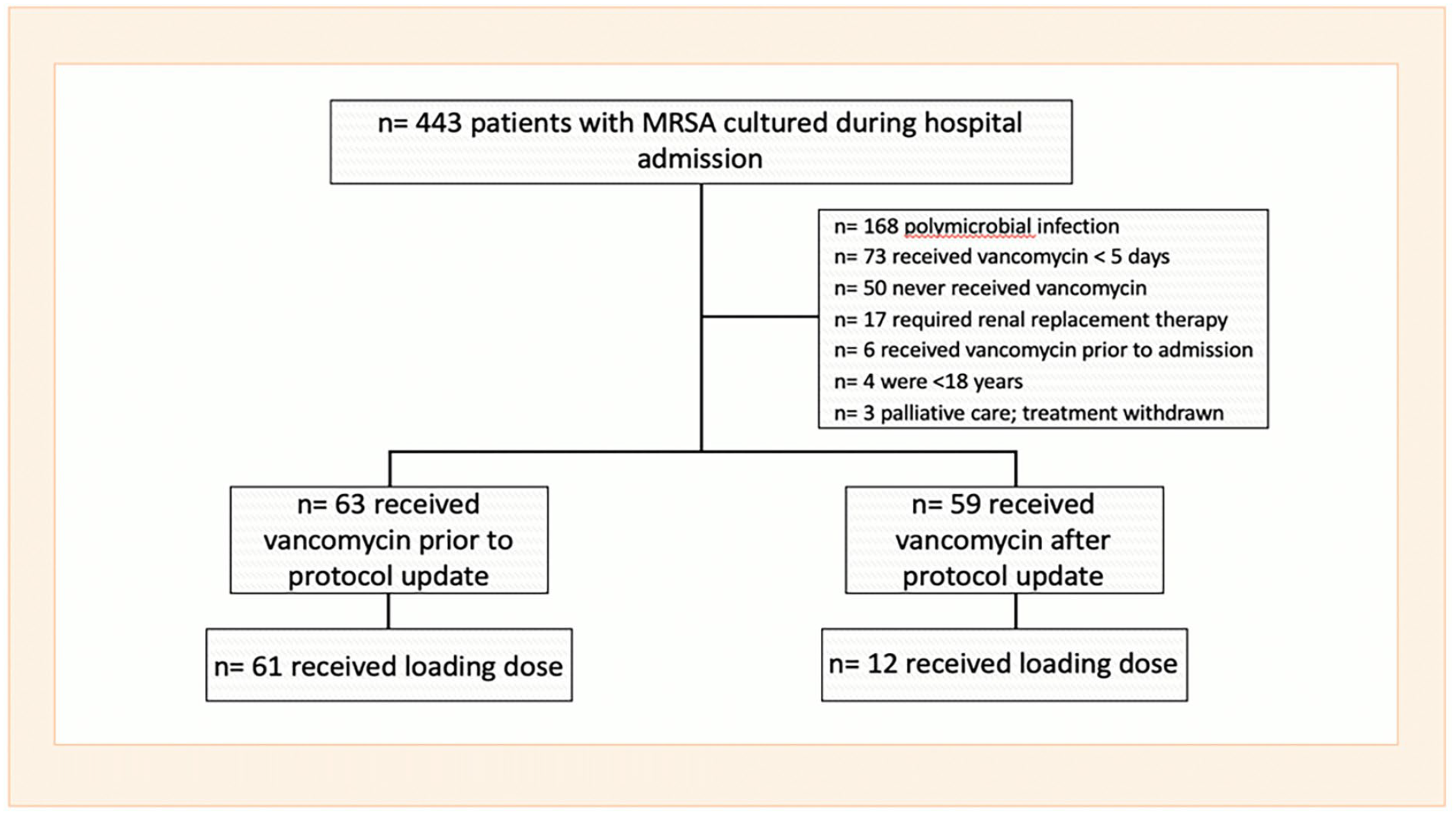
Application of inclusion and exclusion criteria.
Baseline Characteristics.
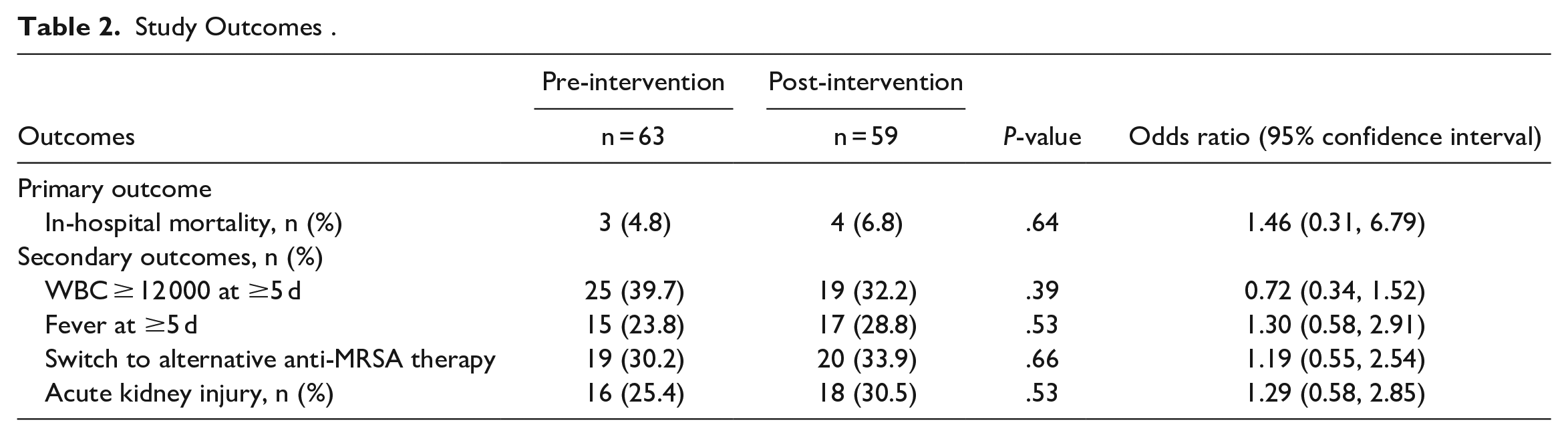
There was no difference in all-cause, inpatient mortality between the pre-intervention and post-intervention groups (4.8%vs 6.8%, respectively; P = .64), though overall mortality was low (Table 2). No differences were noted in any of the secondary outcomes, including presence of fever or leukocytosis at ≥5 days after vancomycin initiation, switch to alternative anti-MRSA therapy, or incidence of nephrotoxicity. For patients who received a loading dose, the average weight-based loading dose received by patients was 25.1 mg/kg in the pre-intervention group and 24.7 mg/kg in the post-intervention group. Initial trough values within the target range were achieved by 28.6% of patients in the pre-intervention group and 42.4% in the post-intervention group.
Study Outcomes .
Data from a multivariable logistic regression analysis identified the presence of bacteremia as a positive predictor for our secondary outcome of persistent leukocytosis; whereas, any trough concentration of 10 to 20 μg/mL was associated with faster resolution of leukocytosis. Factors associated with nephrotoxicity were BMI ≥ 30 kg/m2, any trough concentrations ≥15 μg/mL, and the presence of 2 or more concomitant nephrotoxic agents (Table 3). Patients in the pre-intervention group had a higher percentage of trough concentrations ≥15 μg/mL and ≥2 concomitant nephrotoxic agents, while more patients in the post-intervention group had a baseline BMI ≥ 30 kg/m2. Despite the numerical differences, no significant difference was detected within these variables when compared to each group (Additional results from the multivariable analysis can be found in Tables S3 and S4 in the Supplemental Appendix).
Factors Influencing AKI Identified by Multivariable Logistic Regression Analysis.
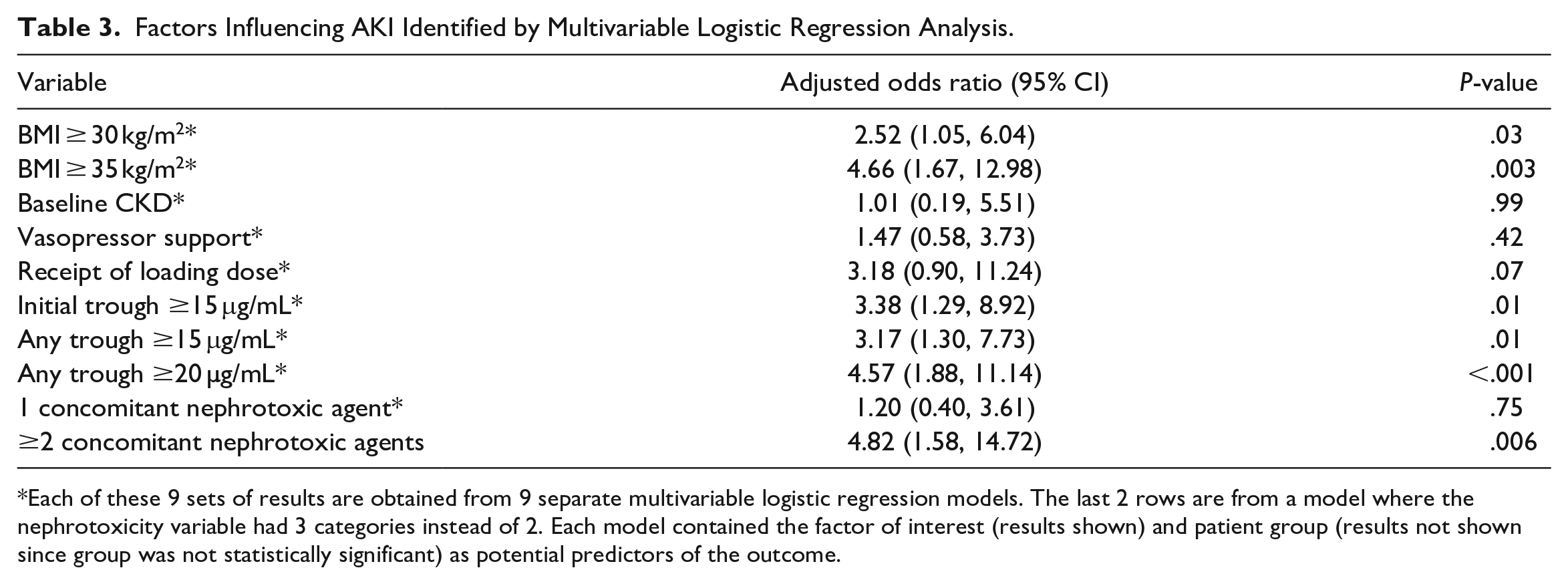
Each of these 9 sets of results are obtained from 9 separate multivariable logistic regression models. The last 2 rows are from a model where the nephrotoxicity variable had 3 categories instead of 2. Each model contained the factor of interest (results shown) and patient group (results not shown since group was not statistically significant) as potential predictors of the outcome.
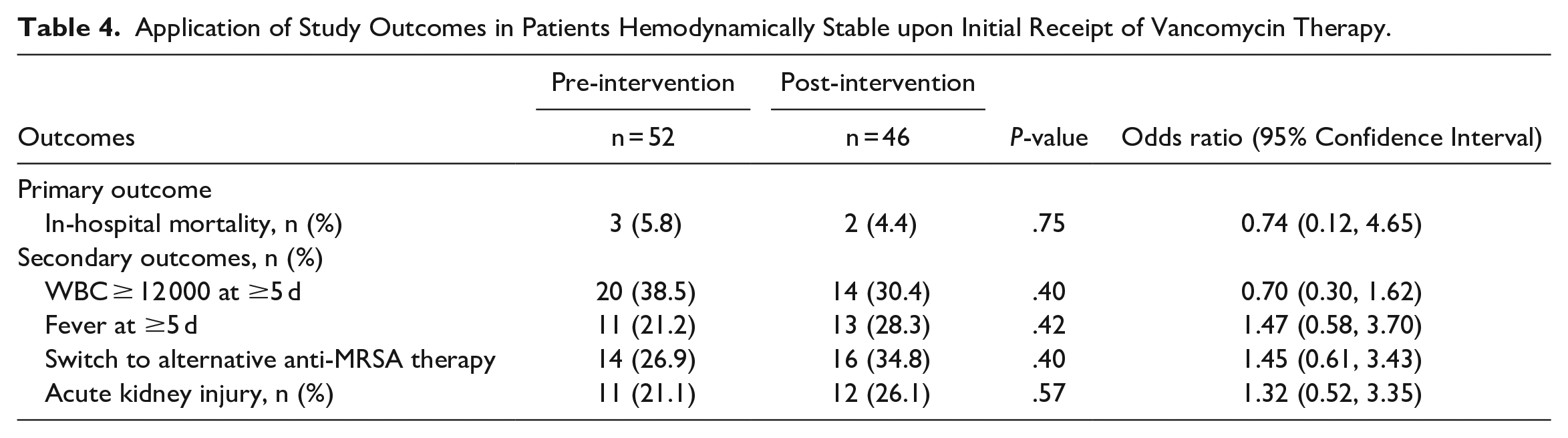
To directly evaluate the clinical impact of loading doses in a subgroup of patients who were hemodynamically stable upon initiation of vancomycin therapy, primary and secondary outcomes were compared between pre-intervention patients who received a loading dose (loading dose group) and post-intervention patients who did not receive a loading dose (non-loading dose group). This excluded 11 patients from the pre-intervention group and 13 patients from the post-intervention group for this analysis. After application of exclusion parameters, the total number of pre- and post-intervention patients included for analysis were n = 52 and 46, respectively (see Figure S3 in the Supplemental Appendix for application of exclusion criteria). In the pre-intervention group, 18 (34.6%) patients required ICU admission compared to 20 (43.5%) patients in the post-intervention group (P = .37). Thirty-three (63.5%) patients in the pre-intervention group had the presence of bacteremia compared to 31 (67%) patients in the post-intervention group (P = .68). In the pre-intervention group, 37 (71.2%) patients had ≥1 concomitant nephrotoxic agent [15 (28.8%) had ≥2 concomitant nephrotoxic agents] compared to 32 (69.6%) patients in the post-intervention group [10 (21.7%) had ≥2 concomitant nephrotoxic agents] (P = .86; for comparison of ≥1 concomitant nephrotoxic agent). The primary outcome of all-cause, inpatient mortality was no different among hemodynamically stable patients who received a loading dose compared to hemodynamically stable patients who did not receive a loading dose (5.8% vs 4.4%, respectively, P = .75). Additionally, no differences were noted in any secondary outcomes among this subgroup of patients (Table 4).
Application of Study Outcomes in Patients Hemodynamically Stable upon Initial Receipt of Vancomycin Therapy.
Discussion
This study represents one of few to assess clinical outcomes associated with vancomycin loading doses in patients with MRSA infection. Previous studies that evaluated clinical outcomes associated with vancomycin loading doses are limited to patients with MRSA pneumonia and bacteremia.16-18 This study included patients with culture-proven MRSA from any site of infection, making it the most inclusive study to date in terms of infectious etiology. Consensus guidelines currently recommend consideration of a loading dose in critically ill patients with suspected or documented serious MRSA infections for rapid attainment of target concentrations. 12 However, the lack of clinical outcomes data and potential for nephrotoxicity has resulted in clinicians deviating from these recommendations, particularly with routine use of a loading dose. 19
In a single-center, pre-post intervention study of loading doses in mixed-acuity patients, Ueda and colleagues found that a 25 mg/kg loading dose provided improved early clinical response compared to no loading dose. 20 Despite this finding, no differences were identified in clinical or microbiological success at end of therapy nor in 28-day mortality. A larger cohort study of vancomycin loading doses in critically ill patients showed that a loading dose of ≥20 mg/kg versus no loading dose provides no difference in clinical failure, 30-day mortality, resolution of systemic symptoms, or substitution of an alternative agent in patients with MRSA bacteremia or MRSA pneumonia. 18 However, obese patients qualifying for a weight-based dose exceeding 2000 mg were excluded from this study, thus limiting the application of these findings to individuals <100 kg. Our institution’s vancomycin dosing protocol recommends maximum loading doses of 3000 mg, removing the weight limitation from our study. Our study included patient populations omitted from similar studies, and our results are consistent with previous data suggesting loading doses are not associated with improved clinical patient outcomes or reduction in mortality.
Nephrotoxicity is one of the major adverse effects of vancomycin therapy, especially in the setting of host-related factors such as increased weight, pre-existing renal dysfunction, critical illness, and concomitant nephrotoxic agents.12,21 However, data evaluating the association between vancomycin loading doses and nephrotoxicity are conflicting. Data from a large retrospective cohort study found that receipt of a vancomycin loading dose was an independent predictor of nephrotoxicity in mixed-acuity patients receiving concomitant piperacillin/tazobactam, supporting an increased prevalence of AKI in the setting of additional nephrotoxins. 22 Flannery et al 18 found no difference in AKI associated with loading doses in their critically ill cohort study, but exclusion of patients ≥125 kg served as a limitation for this finding. A large cohort study of patients with severe renal impairment treated with vancomycin therapy in the emergency department found a lower rate of nephrotoxicity with initial doses >20 mg/kg compared to initial doses ≤20 mg/kg. 23 Our study accounts for host-factors associated with nephrotoxicity and suggests that loading doses alone do not confer a higher incidence of AKI. Numerically, more patients in the post-intervention cohort developed nephrotoxicity despite a lower number of concurrent nephrotoxins compared to the pre-intervention group. Our multivariable logistic regression analysis found that any trough concentration ≥15 μg/mL, ≥2 concomitant nephrotoxic agents, and BMI ≥ 30 kg/m2 are independent predictors for AKI in patients receiving vancomycin.
This study was designed to provide insight into the efficacy and safety surrounding our institution’s revised vancomycin loading dose criteria, and our findings are applicable to other healthcare organizations. We expanded upon previous literature by studying both critically and non-critically ill patients with culture-proven MRSA infection in addition to evaluating clinical outcomes associated with all sources of infection. By not limiting our patient population to a specific infectious source, we were able to evaluate any clinically relevant MRSA infection. MRSA bacteremia was the most common infectious etiology in our patient population; however, identifying an individual source-derived response was difficult considering the varying etiologies of infection and limited sample size. To be consistent with previous literature, the outcomes of this study were aimed to investigate mortality and morbidity, utilizing components of other studies’ composite endpoints that can be assessed through objective data. The two treatment groups studied were similar in terms of severity of illness, comorbidities, and other treatment characteristics that might have predisposed to receipt of a loading dose or clinical outcome, further strengthening this study. Additionally, real-time deviations from protocol resulted in the omission or addition of a loading dose in the pre- and post-intervention groups when a loading dose was or was not indicated, respectively. To account for those deviations, additional analyses were conducted to provide a more accurate comparison of “loading dose versus no loading dose” between patients among the two groups who were hemodynamically stable at initiation of vancomycin therapy (ie, patients in which initial therapy would have changed as a result to the protocol revision). The consistency of our results increases the validity of our findings, and further supports our overall conclusion.
Notably, our study has many limitations including the retrospective, non-randomized, single-center design. Additionally, our study size was small resulting in insufficient statistical power to determine a difference in mortality, thus our findings cannot exclude beta error. Consequently, we did not run multivariable logistic regression analyses for the primary outcome due to the low incidence of mortality in each group. These small numbers would not allow us to obtain statistically or scientifically valid results for adjusted analyses for mortality. During the study period, our institution utilized a vancomycin dosing nomogram and monitored trough concentrations, rather than area-under-the-curve (AUC), as the target for efficacy and safety. While observational data suggests that the use of validated dosing nomograms lead to reliable attainment of target vancomycin concentrations, we are not able to make inferences regarding the clinical benefits of loading doses in the setting of AUC-guided dosing. 7 Furthermore, evidence suggests that AUC provides a toxicodynamic benefit with a lower incidence of AKI compared to traditional trough monitoring, potentially inflating the overall incidence of AKI found in our study. 12 While source control was obtained by the primary investigator during the data collection process, the subjectivity of assessment and incomplete documentation regarding the time to- and extent of- adequate source control made it difficult for our study team to fully assess this variable. To avoid potential detection bias, this information was not evaluated in our statistical analyses. Notably, 20% of patients in the post-intervention cohort received a loading dose as our institution’s protocol was revised to limit loading doses to patients with evidence of hemodynamic instability. Therefore, our study is unable to determine if a clinical benefit, or lack thereof, exists among this subgroup of patients. Lastly, the presence of the SARS-CoV-2 pandemic may have influenced outcomes among our post-intervention group. However, detection of SARS-CoV-2 was minimal among the post-intervention group.
Conclusion
In this cohort of critically and non-critically ill adult patients with culture-proven MRSA infections, routine use of a 20 to 30 mg/kg vancomycin loading dose was not associated with any difference in mortality, resolution of signs of infection, switch to alternative anti-MRSA therapy, or nephrotoxicity. Further research is needed to determine the impact of vancomycin loading doses on outcomes of hemodynamically unstable patients with MRSA infections.
Supplemental Material
sj-docx-1-hpx-10.1177_00185787231196435 – Supplemental material for Impact of Limiting Vancomycin Loading Doses in Patients With Methicillin-resistant Staphylococcus aureus Infections After Hospital Protocol Revision
Supplemental material, sj-docx-1-hpx-10.1177_00185787231196435 for Impact of Limiting Vancomycin Loading Doses in Patients With Methicillin-resistant Staphylococcus aureus Infections After Hospital Protocol Revision by Alec R. Raley, Matthew L. Brown, Morgan Frawley, Robert A. Oster and William Seth Edwards in Hospital Pharmacy
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the following grant from the National Institutes of Health: UL1 TR003096.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
