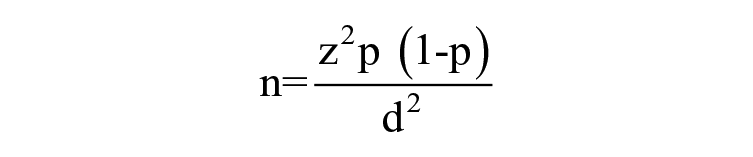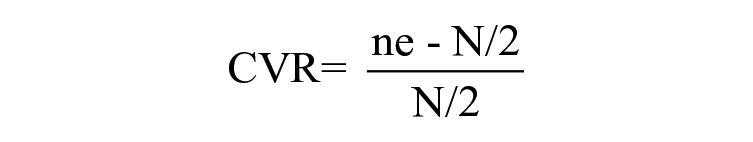Abstract
Keywords
Introduction
The national medicine regulatory structures in most countries rely on spontaneous reporting of Adverse Drug Reactions (ADR). It is a procedure that healthcare practitioners voluntarily report and submit suspected cases of ADR to a national pharmacovigilance program. According to the definition of the World Health Organization (WHO), ADR are “responses to a drug which are noxious and unintended, and which occur at doses normally used to treat a patient for diagnosis, prophylaxis, therapy or for the modification of a physiological function.” 1 ADR can range from minor discomfort to fatal complications. Most of these ADRs detected around the world were serious and unusual ones, which were not detected during the drug development process. ADR is classified as the sixth leading cause of mortality worldwide. 2 A systematic literature review conducted using 69 studies worldwide in 2002 evaluated the ADR rate, and it showed 5.5% to 7.7% of ADR rate around the world. 3 Hence, the clinical and economic impacts of ADR-related hospital admissions to the health care delivery system are extensive. Thus the spontaneous reporting of ADR plays an important role in order to ensure the standards of the safety and efficacy of the drugs.
The Uppsala Monitoring Center (UMC) is an international drug monitoring center that collaborates with the World Health Organization (WHO). 4 From the countries that had already established national structures to report ADR and who were willing to contribute their data were selected and a common reporting form was developed by UMC. It uses WHO guidelines for entering information, common terminologies, and classifications.5,6 Although many ADR reporting methods have been introduced, the main drawback is under-reporting. A systematic review conducted in 2006, evaluated the degree of under-reporting of ADR to the national reporting systems that included 37 studies from 12 different countries. The median under-reporting rate across these studies was 94%. 7 In 2002, Hughes et al conducted a study among 18 countries that showed that national spontaneous reporting schemes are different in many ways according to the country. The number of ADR reports received by each country fluctuates considerably from a few hundred each year in South Africa to over 20 000 in the USA. 8
Studies from different countries have concluded that inadequate knowledge regarding ADR among health care professionals as well as negative attitudes are associated with a great degree of under-reporting.9-11 When considering the South Asian context, the first systematic review and meta-analysis were carried out in India in 2016 regarding health Professionals’ knowledge, attitudes and practices about pharmacovigilance. It included 28 studies and overall 55.6% of the population was not aware of the existing pharmacovigilance program in India and 28.75% of them ignored the reporting of ADR and 74.5% had never reported any ADR to pharmacovigilance centers. 12 In Sri Lanka, no studies were carried out to find out the factors determining the under-reporting of ADR by healthcare professionals (HCP).
In Sri Lanka, the national pharmacovigilance center is the National Medicine Regulatory Authority (NMRA), which is an independent authority governed by the Ministry of Health and Indigenous Medical services. The NMRA has introduced a national ADR reporting form together with a set of guidelines that entails all the details concerned regarding the monitoring and reporting of ADR in Sri Lanka. 13 Further, a Google form has been introduced to report ADR, which can be filled by logging in to the website of NMRA. 14
This study aimed to describe the knowledge, attitudes, and practices of ADR reporting among HCP in the Teaching Hospital Karapitiya, a tertiary care center in Sri Lanka.
Methodology
Study Design and Setting
A descriptive cross-sectional study was conducted at the Teaching Hospital Karapitiya (THK).
Study Population
All doctors and nurses working in THK who were available during the study period and willing to participate in the study were invited to the study.
Inclusion Criteria
Doctors and nurses working in THK and who consented were enrolled in the study.
Exclusion Criteria
The HCP, who were on leave during the study period were excluded from the study. Although pharmacists have authorization to report ADRs, in Sri Lanka there are no clinical pharmacists in wards to review drugs prescribed for patients. Pharmacists’ role is limited to medicines procurement, medication supply and dispensing, inventory control and storage. 15 Hence, the pharmacists were excluded from the study.
Sample Size
The sample size was calculated using the following formula given by Lwanga and Lemeshow for a single proportion of size less than10 000. 16
n = Sample size.
d = absolute error or precision—0.05.
z = 1.96 Valve of the standard normal distribution corresponding to a significance level of 0.05.
P = Expected proportion in the study population taken as 50% (as there were no evidence of similar study in Sri Lanka and worldwide literature review shows different percentages in South Asian studies ranging from 28% to 55.6%).
n = 384.
Additional 10% is added for the non-responses = 39.
Total sample size = 423.
The minimum sample size for this phase is 423.
Sampling Technique
The total number of HCP (doctors and nurses) working in THK was found to be 2065.
Consultants—95.
Medical Officers—520.
Nurses—1450.
Assuming variations among HCP’s knowledge, attitudes and practices toward ADR reporting, study subjects were recruited using a stratified random sampling technique with a proportional allocation. Hence 19 consultants, 106 medical officers, and 297 nursing officers were recruited.
Consultants—95/2065 × 423 = 19.
Medical officers—520/2065 × 423 = 106.
Nurses—1450/2065 × 423 = 297.
Data Collection Instrument
A self-administered pre-tested questionnaire (Supplemental Annexure 1) was given to the participants. Various self-administered questionnaires used to determine knowledge, attitudes and practices (KAP) in similar studies that were carried out in different countries were reviewed.17-19 Sri Lanka remains far behind other nations in terms of pharmacovigilance. ADR reporting was not encouraged by any new initiatives, and ADR’s related practical issues have not yet been addressed. Hence, we designed a new questionnaire that meets our requirements. The new questionnaire was validated and ensured that the questions truly measure the issues of importance.
It was designed as multiple choice questions, and questions based upon the degree of agreement. The questionnaire was designed in English, Sinhala, and Tamil languages.
Questionnaire is consisted of following domains.
- Demographic data of the participants—7 questions.
- Questions about the knowledge regarding ADR-Part A: 11 questions.
- Questions related to the practice of reporting ADR-Part B: 7 questions.
- Questions regarding attitude/reason for poor reporting of ADR—Part C: 11 questions.
Knowledge and practice related questions were designed as best response questions, but more than one answer was allowed in some questions:
The validity of the questionnaire was assessed by asking a panel of content experts to review the relevance of each question on a 3 point Likert scale: 1—not essential, 2—useful but not essential, and 3—essential. Then, the content validity ratio were calculated for each question by employing Lawshe’s method. 20
CVR is the content validity ratio where ne is the number of panel members indicating “essential” and N is the total number of panel members. The final value of CVR was taken from a table including the number of panel members vs. the minimum CVR valve. 20 The questionnaire was modified according to the minimum CVR of each question. Five specialists from the fields of pharmacology, general medicine, and community medicine took part in the validation procedure. There were 8 demographic detail-related questions, of which, 7 satisfied the required CVR valve (CVR > 0.99). Hence, 1 question was eliminated (CVR = 0.2). Out of 13 questions related to ADR knowledge, 11 questions met the minimum CVR valve (CVR > 0.99). This led to the removal of 2 questions. All questions related to ADR reporting practice were included since they all met the minimal CVR valve requirements. To assess the attitude toward ADR reporting, 14 questions were included and out of those, 10 questions were finalized as they met the minimum CVR valve. Upon receiving the responses from experts, internal consistency reliability of the questionnaire was assessed by giving it to a sample of 40 randomly selected doctors and nurses in 2 different hospitals. After calculating the Cronbach’s alpha, which was 0.73, no adjustments were made.
Data Collection Procedure
The proposal was submitted to the Ethical Review Committee of the Faculty of Medicine, University of Ruhuna and approval was obtained. Administrative approval was obtained from the THK.
Data Analysis
After collection, data were entered into the SPSS for analysis. Checking, clearing and coding of the data were done before starting the analysis process. The attitude-related questions were analyzed based upon the participant’s degree of agreement using a Likert scale. The degrees of the agreement changed from strongly disagree to strongly agree.
Numbers with percentages for categorical variables were used where appropriate.
Results
Demographic Characteristics
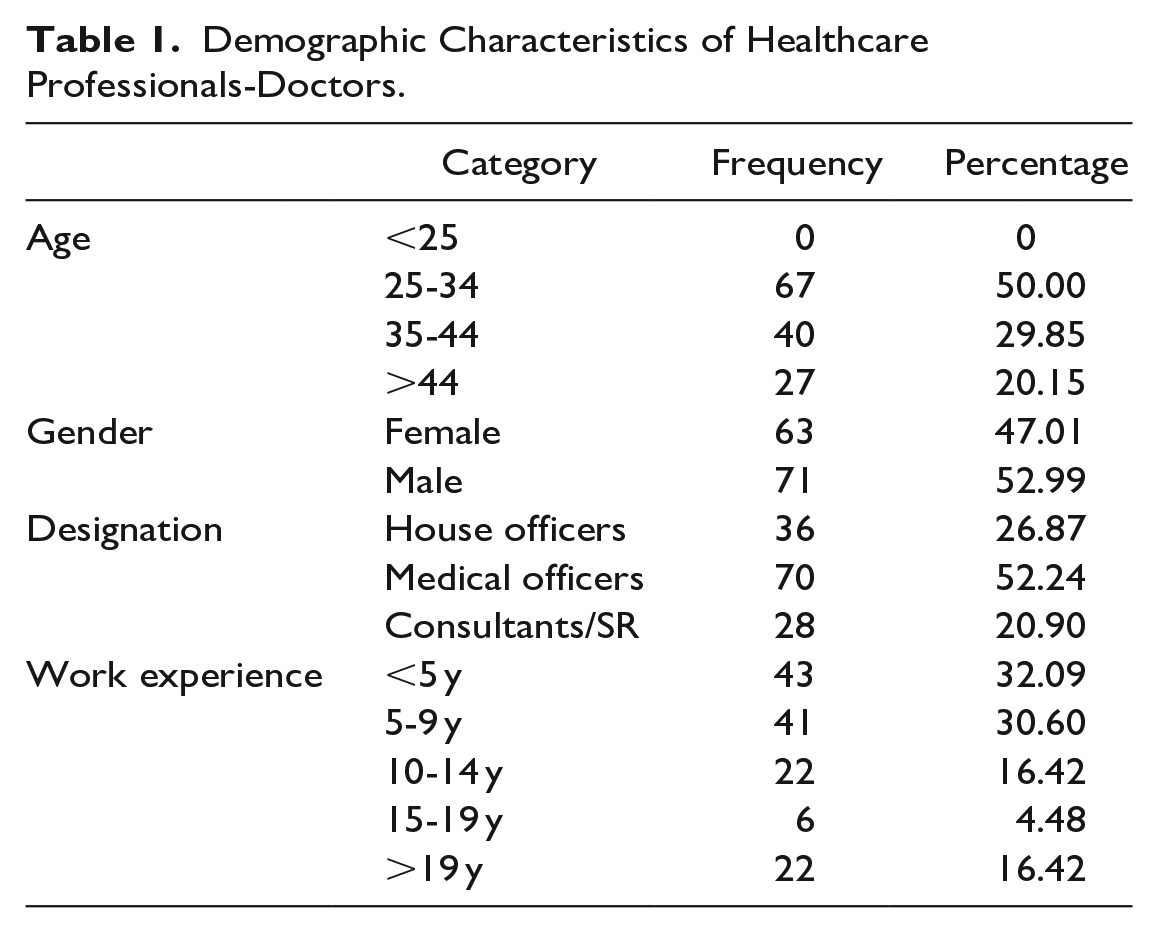
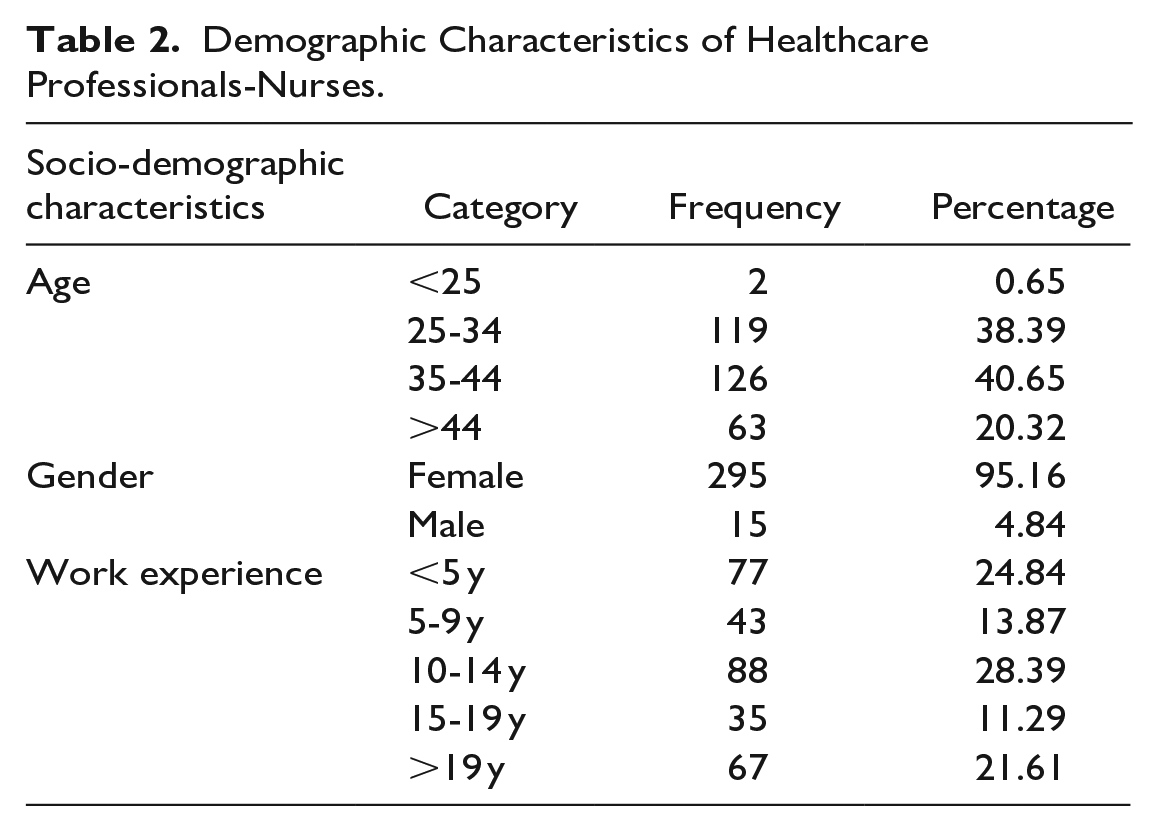
In total, 467 questionnaires were distributed among doctors and nurses in THK. Out of 467 questionnaires, 444 were duly filled, giving a response rate of 95.07%. Of the total 444 respondents, 134 (31%) were doctors and 310 (69%) were nurses. Among them 358 (80.6%) HCPs were female and 86 (19.4%) were male. The mean age of all respondents was 37.6 years (±8.3). The majority of participants (41%) were in the 25 to 34 age groups.
Demographic characteristics of participants who participated in this study are presented in Tables 1 and 2.
Demographic Characteristics of Healthcare Professionals-Doctors.
Demographic Characteristics of Healthcare Professionals-Nurses.
Description of Knowledge Regarding ADR
Regarding the knowledge of ADR, as shown in the following table (Table 3), 62.8% (n = 279) participants were aware about the term pharmacovigilance, while 47.3% (n = 210) could correctly define the term. Majority of respondents (90%, n = 400) were aware of the term ADR, while 64.8% (n = 288) could correctly define it. Among the respondents, 30.8% (n = 137) knew about the types of ADR and only 15.5% (n = 70) were able to correctly mention a drug that is banned due to ADR. However, 38.7% (n = 172) respondents have marked ADR reporting centers correctly. Most of the respondents 61.7% (n = 274) mentioned that they used several sources to gather ADR information, while 9% (n = 40) of them used textbooks and 10.3% (n = 46) of them used the internet to gather ADR information.
Healthcare Professionals’ Knowledge Regarding ADR.
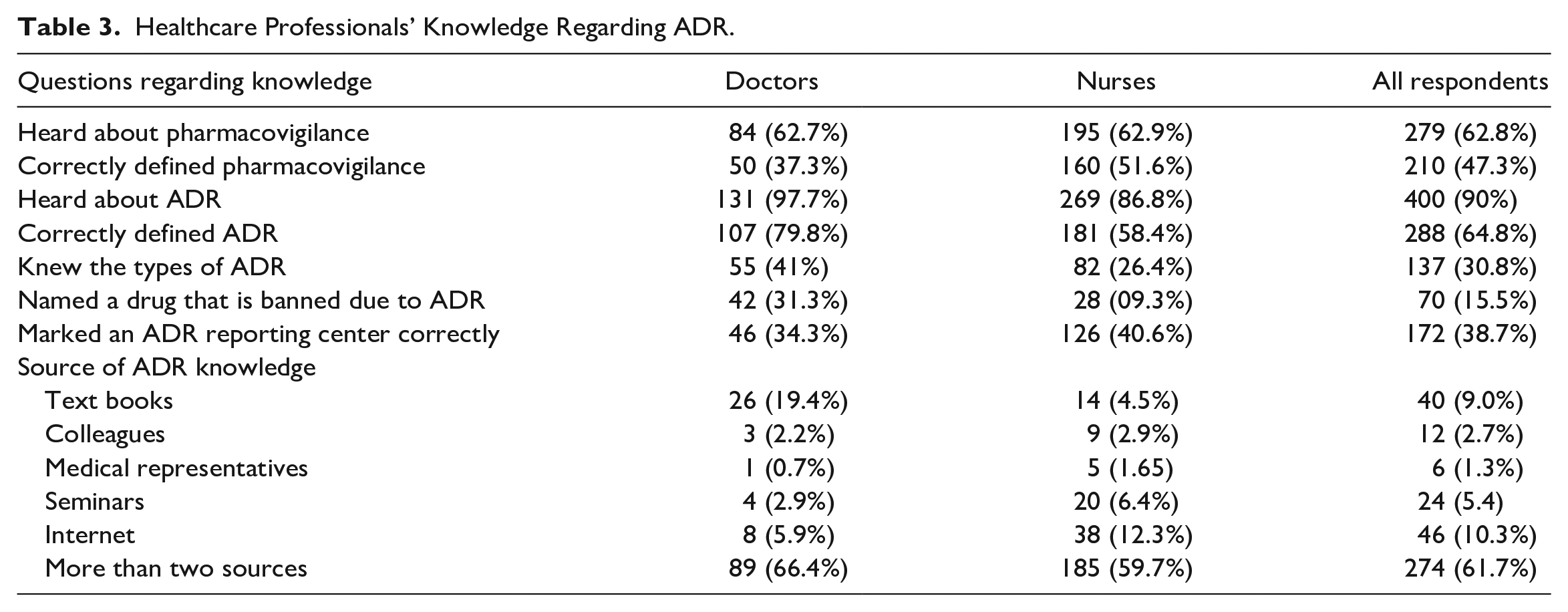
An attempt was made to determine the overall knowledge regarding ADR and reporting procedures. One mark was given for each expected answer and if multiple answers were present, marks were allocated accordingly. Marks obtained for the knowledge related questions and the number of health professionals are shown in Table 4. The median score, obtained from those health professionals was 4 (IQR: 5-3) and the maximum marks they obtained was 7 out of 9.
Healthcare Professionals’ Overall Knowledge Regarding ADR.
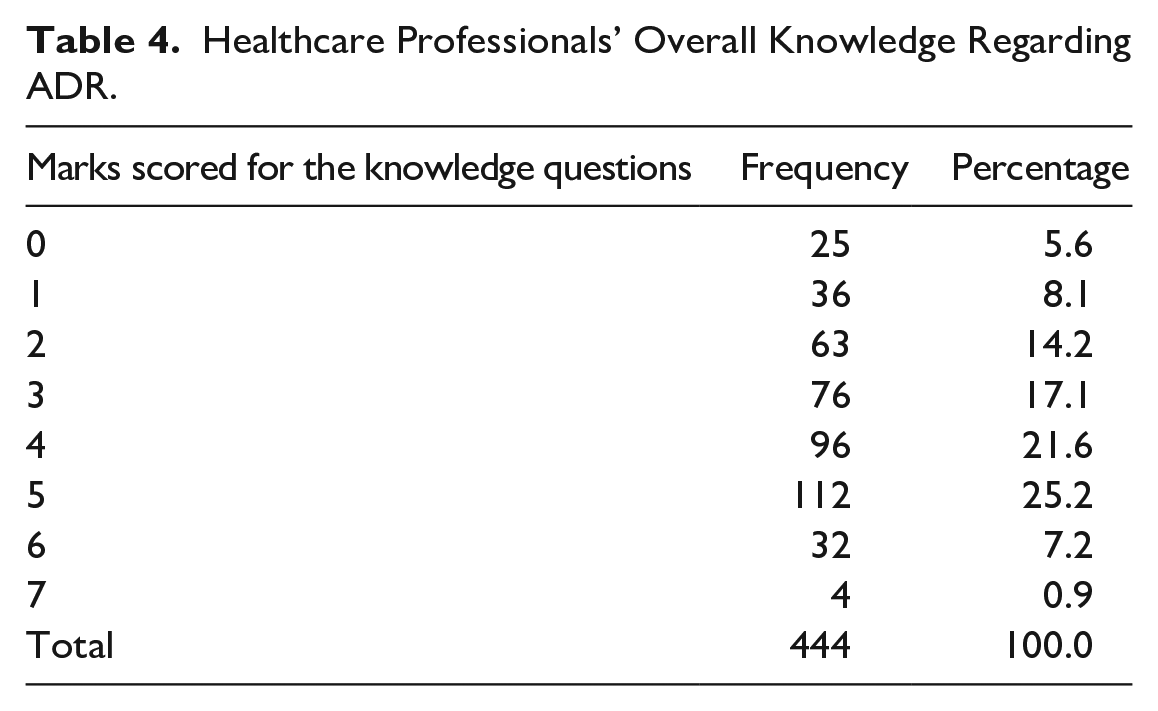
Further, the maximum number of ADRs that a single HCP could identify during clinical practice was 10 ADR each month (n = 4, 0.9%). Participants who had come across ADR during clinical practice performed well on knowledge-related questions. A Pearson correlation coefficient was computed to assess the linear relationship between the number of ADR recognized during clinical practice and the score obtained for the knowledge-related questions. The study found that, in comparison to those with insufficient knowledge, health professionals with considerably greater knowledge of ADR are more likely to recognize ADR. Pearson correlation coefficient r = .16 (P < .001).
Description of Practices Regarding ADR
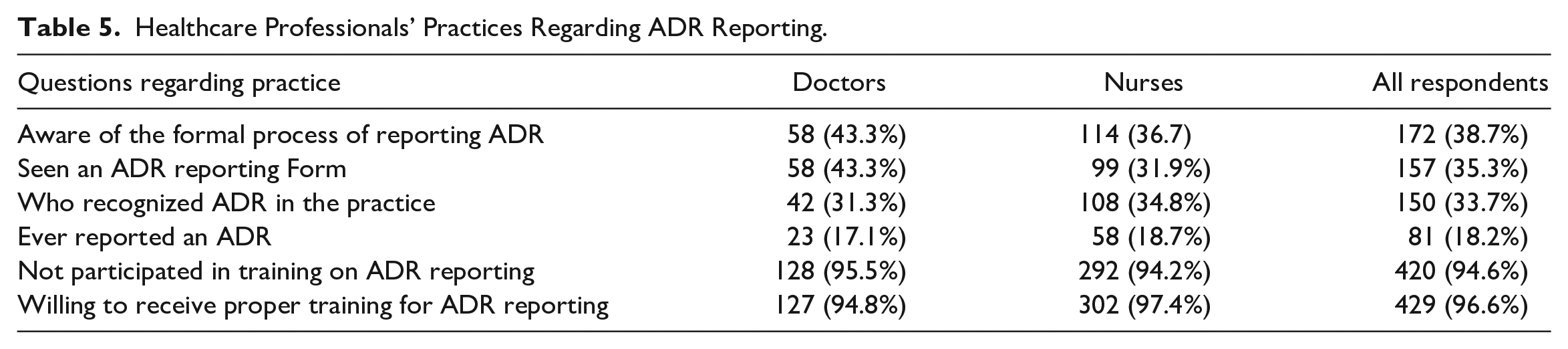
Among the respondents, only 38.7% (n = 172) were aware of the formal process of reporting ADR and, only 35.3% (n = 157) stated that they had seen the ADR reporting form. Further, as shown in Table 5, only 33.7% (n = 150) respondents have recognized ADR during their practice and only a small proportion 18.2% (n = 81) have ever reported an ADR during their practice. A majority of respondents 94.4% (n = 420) stated that they had not received training on ADR reporting. However, most of them 96.6% (n = 429) were willing to receive proper training on ADR reporting.
Healthcare Professionals’ Practices Regarding ADR Reporting.
Description of Attitudes Regarding ADR
There were 8 questions regarding the attitude of HCP with regard to ADR reporting. Responses were analyzed based upon the participant’s degree of agreement using a Likert scale.
The degrees of agreement changed from strongly disagree to strongly agree. As the questions were negatively worded, strongly disagreed and disagreed responses were considered as positive attitude, strongly agreed and agreed responses were taken as poor attitude while the respondents who stayed neutral were considered as neutral attitude. The answer of strongly disagreed was assigned a score of 5, disagreed with 4, agreed with 2 and strongly agreed with 1 mark.
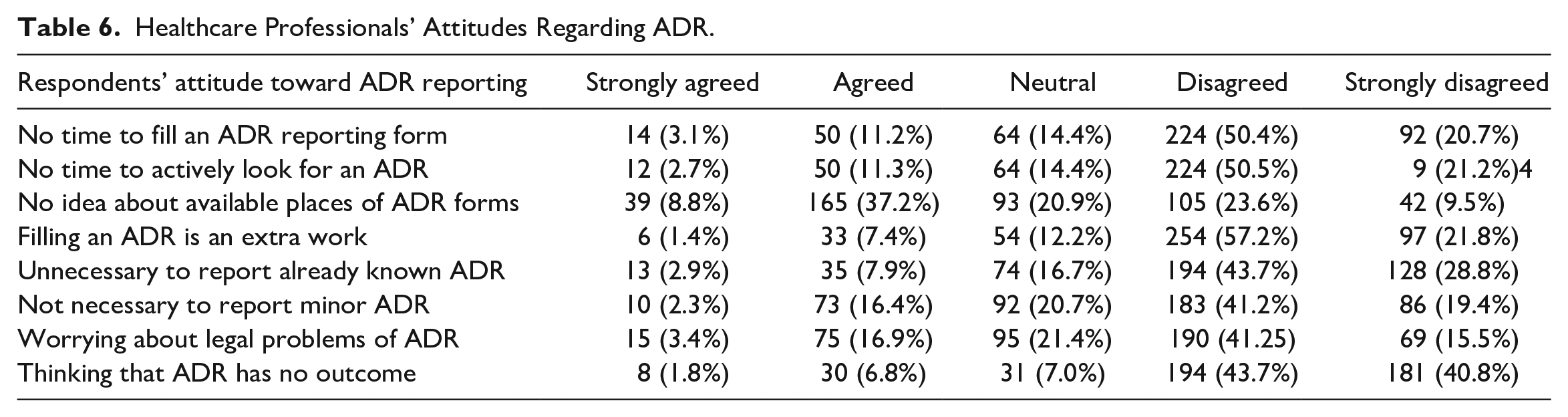
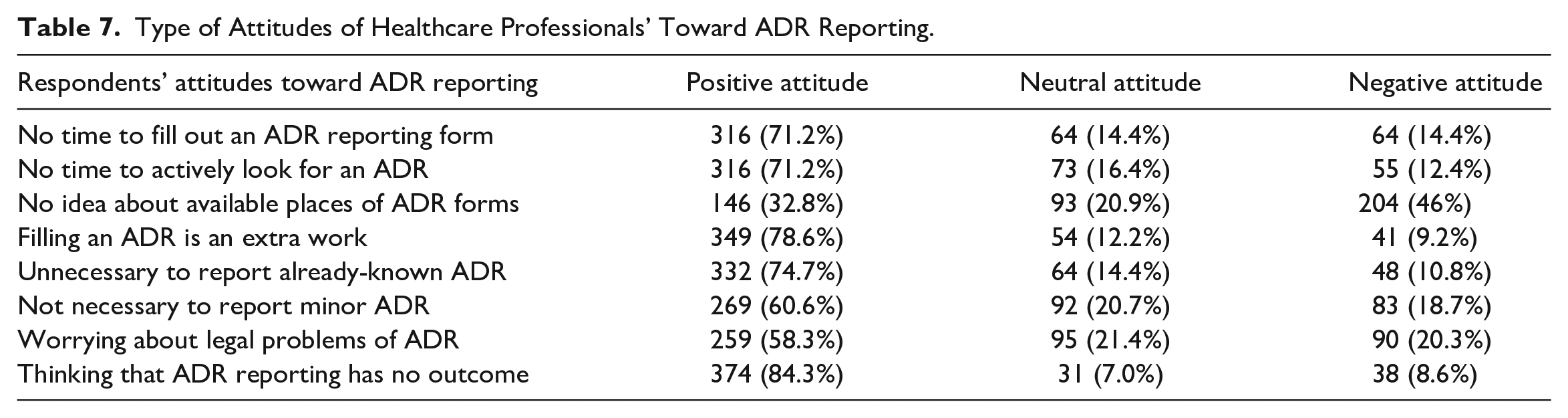
As shown in Table 6, the majority of them disagreed and strongly disagreed with the statement that there is no adequate time to fill out an ADR form. This means 71.7% of respondents (n = 316) have adequate time to complete an ADR report. Further, they have adequate time to actively look for an ADR during clinical practice. Of the total, 46% (n = 204) have no idea about the available places of ADR forms. Among the respondents, 78.6% (n = 349) of them do not consider filling an ADR form as an additional work. Further, 74.7% (n = 332) HCP stated it is necessary to report already known ADR and 60.6% (n = 269) knew the importance of reporting minor ADR. Regarding the legal aspect of ADR reporting, 58.3% (n = 259) of HCP were not worried about the legal aspect of ADR reporting, and the majority of them (84.3%, n = 374) accepted ADR has an outcome.
Healthcare Professionals’ Attitudes Regarding ADR.
In general, the respondents had good attitudes toward ADR reporting. As shown in Table 7, overall 84.1% (n = 373) had a positive attitude toward ADR reporting, while 13.54% (n = 60) of them stayed neutral and 2.25% (n = 10) had a negative attitude toward ADR reporting.
Type of Attitudes of Healthcare Professionals’ Toward ADR Reporting.
There were 2 more questions regarding factors contributing to reporting ADR. Among the respondents, 61% (n = 271) mentioned that they had no idea about the ADR reporting procedure while 39% (n = 173) mentioned that the unavailability of the ADR reporting forms is the reason for underreporting.
Discussion
This study was conducted in Sri Lanka to find out the knowledge, attitudes and practices of ADR reporting among healthcare professionals. Although most respondents could select the correct definitions of ADR and pharmacovigilance, the majority of HCP did not aware of the types of ADR, banned drugs due to ADR and ADR reporting centers. Further, this study showed that HCP with relatively better knowledge of ADR are more likely to identify ADR compared with those with insufficient knowledge. These findings are in line with a similar study, a systematic review carried out in India. 12 Another systematic review carried out in Europe, which included 17 different publications showed that under-reporting of ADRs was directly co-related with healthcare professionals’ low knowledge about pharmacovigilance activities and drug safety. 21 This implied the importance of healthcare professionals’ knowledge to improve ADR reporting. Worldwide various interventions were introduced to improve ADR reporting while educational interventions were the commonest among them. A cluster randomized controlled trial carried out in Portugal in 2007, showed that the ADR reporting rate among physicians had been increased by 10-fold (95% CI, 3.8-7.5) in the year following an hour-long educational intervention. 22 As the majority of respondents in the current study (97%) were willing to receive proper training regarding ADR reporting, there is a potential to promote ADR reporting among HCP in Sri Lanka by educational interventions.
Among the respondents, only smaller proportions had seen an ADR reporting form (35.3%) and had recognized ADR during their clinical practice (33.7%). Additionally, only 18.2% of HCP reported at least one ADR. Respondents have stated that unaware about the reporting procedure and the unavailability of the ADR reporting forms are the major contributory factors for poor ADR reporting. Hence, raising awareness among HCP and convincing the magnitude of the drug safety problems would be good options to improve ADR reporting, which have not been addressed so far in Sri Lanka.
The attitudes of HCP toward ADR reporting are interesting. This study disclosed that the majority of HCP believed that they have adequate time to fill an ADR reporting and filling an ADR reporting form is not considered as extra work. Those findings can be considered as positive impression which helpful to improve future ADR reporting. The study findings are compatible with many studies conducted regarding the topic, which showed the positive attitude strongly related to the ADR reporting.23-25
The following recommendations can be suggested based on the current study results.
- Raise awareness of the magnitude of drug safety problems by conducting workshops.
- Convince the health professionals that reporting ADRs is their professional obligation.
- Make ADR reports freely available in hospitals including emergency treatment centers and out patients departments.
- Introduce more user-friendly ADR reporting methods to report ADR.
Further, creating a hospital-based ADR database that will be maintained by a national pharmacovigilance center and establishment of further regional centers to coordinate ADR reporting and pharmacovigilance activities could be taken into consideration in enhancing national pharmacovigilance. Finally, we recommended further studies at the national level to implement policies to increase ADR reporting in Sri Lanka.
Strengths and Limitations
Strengths of the Study
The data collection instrument was newly developed and validated according to the expert opinion and pre-tested in similar setting. Data collection was carried out by the principle investigator.
The excellent level of participant response (95.07%) in compared to studies conducted in developing countries.19,26,27
Both quantitative data and qualitative data were used to explore the results and yield to potential solutions.
Limitation of the Study
As data were collected based on self-reported information, the possibility of reporting errors and recall biases could not be ruled out.
Only 1 hospital was involved in the study. Therefore, knowledge and practices elsewhere may differ. Additionally, some health professionals, such as pharmacists, were not included in the study.
Conclusions
The collective results of this study disclose that the healthcare professionals’ knowledge and practices regarding ADR reporting procedures are not satisfactory. However, they have positive attitudes toward ADR reporting which is a favorable fact to improve ADR reporting. It is essential to have a system that is easily accessible and efficient for health care professionals to report ADR. Considerable attention should be paid for executing a national program in order to develop the concept and practice of pharmacovigilance in the country.
Supplemental Material
sj-docx-1-hpx-10.1177_00185787231194988 – Supplemental material for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study
Supplemental material, sj-docx-1-hpx-10.1177_00185787231194988 for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study by Menikpurage Thilini Madhushika, Sudheera Sammanthi Jayasinghe, Polwaththa Gayani Chandima Liyanage, Wellappuli Arachchige Dilan Malinda and Palitha Abeykoon in Hospital Pharmacy
Supplemental Material
sj-docx-2-hpx-10.1177_00185787231194988 – Supplemental material for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study
Supplemental material, sj-docx-2-hpx-10.1177_00185787231194988 for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study by Menikpurage Thilini Madhushika, Sudheera Sammanthi Jayasinghe, Polwaththa Gayani Chandima Liyanage, Wellappuli Arachchige Dilan Malinda and Palitha Abeykoon in Hospital Pharmacy
Supplemental Material
sj-docx-3-hpx-10.1177_00185787231194988 – Supplemental material for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study
Supplemental material, sj-docx-3-hpx-10.1177_00185787231194988 for Knowledge, Attitudes, and Practices of Adverse Drug Reaction Reporting Among Healthcare Professionals in Sri Lanka- A Cross Sectional Study by Menikpurage Thilini Madhushika, Sudheera Sammanthi Jayasinghe, Polwaththa Gayani Chandima Liyanage, Wellappuli Arachchige Dilan Malinda and Palitha Abeykoon in Hospital Pharmacy
Footnotes
Acknowledgements
The authors are thankful to the staff of THK for their unreserved cooperation in making this study a fruitful work.
Authors’ Contributions
M.T. Madhushika conceived the idea. P.L.G.C Liyanage and S.S. Jayasinghe helped to develop the proposal and questionnaire. M.T. Madhushika and WAD Malinda conducted the study. M.T. Madhushika wrote the first and subsequent drafts. P.L.G.C Liyanage, S.S. Jayasinghe and P Abeykoon developed the ideas. All authors read and approved the final article.
Availability of Data and Materials
Personal identification data were not collected. Privacy and confidentiality were maintained and data were used only for scientific publications. Gathered data were kept confidential with the principal investigator without disclosing to any third party and all data will be destroyed 5 years after the final analysis has been completed. Soft copies of the data are kept with a password protected whereas hard copies are kept in a locked cabinet. The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Faculty Research Grant, Faculty of Medicine, University of Ruhuna, Sri Lanka in 2021. (FoM/RG/2021/01).
Ethical Approval and Consent to Participate
Ethical clearance was obtained from the Ethical Review Committee, Faculty of Medicine, University of Ruhuna. All the steps were performed in accordance with the guidelines and regulations of the Ethical Review Committee, Faculty of Medicine, University of Ruhuna. Administrative approval will be obtained from the THK. All the participants who enrolled in this study were given an information sheet (Supplemental Annexure 2) before getting consent and informed written consents (Supplemental Annexure 3) were obtained prior to the study. They were allowed to withdraw from the study at any point without stating reason for withdrawal.
Consent to Publish
Not Applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.