Abstract
Keywords
Introduction
Diabetes mellitus (DM) has become increasingly prevalent and a considerable health risk in the United States (U.S). In 2020, the Centers for Disease Control and Prevention (CDC) estimated that up to 34.2 million Americans, 10.5% of the U.S. population, had DM with the number of new cases increasing by 1.5 million each year. 1 When uncontrolled, hyperglycemia can lead to the deterioration of blood vessels, nerves, and other tissues, resulting in macrovascular and microvascular complications. 2 To prevent these complications, the American Diabetes Association (ADA) guidelines recommend initiation of glucose lowering medication. Specifically, the ADA guidelines recommend that most individuals with type 1 diabetes be treated with multiple daily injections of prandial and basal insulin. For type 2 diabetes (T2D), the early introduction of insulin can be used if there are signs of catabolism, symptoms of hyperglycemia, when hemoglobin A1C (A1C) levels are greater than 10%, or blood glucose levels are greater than 300 mg/dL. Additionally, the initiation of insulin therapy can be used for patients whose A1C is 2% above their target goal. 3
The role of basal insulin is to limit hepatic glucose production and lipolysis in the fasting state without impairing glucose availability for brain function. 4 With the development of long-acting insulin analogs, such as insulin glargine, limitations such as variable absorption and hypoglycemia were reduced. Insulin glargine, when compared to older insulins such as neutral protamine hagedorn (NPH) insulin or mixed insulin, offers improved conveniences and pharmacokinetics such as once-daily administration and flexibility with the timing of injection. 5 Additionally, insulins such as insulin glargine can be used successfully with other oral and parenteral agents in the treatment of DM, including in combination with prandial insulin, glucagon-like peptide-1 receptor agonists, or sodium glucose cotransporters 2 inhibitors.6-8 Given the efficacy and safety demonstrated in clinical trials, insulin glargine is supported for use by the 2023 ADA guidelines. 3
Insulin glargine was first approved by the U.S. Food and Drug Administration in 2000 under the brand name Lantus®. 9 In a 16-week multicentered clinical trial, the most common adverse effects of insulin glargine observed were severe hypoglycemia and injection site reactions. 10 A literature search in PubMed, MEDLINE, and Google Scholar using keywords and Boolean operators, namely insulin AND nausea OR emesis; resulted in a similar adverse profile in the majority of studies.11–14 The gastrointestinal adverse effects of therapy are limited in clinical trials. Insulin glargine package insert reports medication-induced diarrhea with an incidence rate of 11%. 9 However, no cases of nausea or emesis have been reported.
Collaborative Drug Therapy Management (CDTM) is a written protocol between a physician and a pharmacist which allows the pharmacist to manage medication regimens of chronic diseases including DM in an article 28 facility. In New York State, this bill allows pharmacists to modify, remove, and initiate medication therapy under the formal agreement with a physician. At an urban city-run community hospital, CDTM pharmacists are heavily utilized to manage a largely underserved DM population which includes uninsured patients or those living below the poverty level. At this institution, insulin detemir and insulin glargine (Lantus®) are formulary agents due to cost restraints.
Case Presentation
DS is a 51-year-old female with a past medical history of T2D who was referred to a CDTM pharmacist for diabetes education and management. The patient was diagnosed with T2D when she was 40 years old and did not have any previous relevant clinical history of familiar disease. At the time of first visit, DS reported weight gain of 4 kg over 3 months accompanied by polyuria and polydipsia. The patient denied any vision changes, abdominal pain, excess postprandial satiety, or any adverse reaction/allergies to pharmacologic or non-pharmacologic therapy. Additionally the patient denied any history of alcohol, marijuana, nicotine, or illicit drug use. Her weight was 143 kg with a BMI of 27.98 kg/m2. The rest of the physical examination was unremarkable. The results of blood testing 1 day prior to the visit with the CDTM pharmacist were as follows: glucose 279 mg/dL, A1C 13.9%, cholesterol 218 mg/dL, triglycerides 180 mg/dL, LDL 136 mg/dL, HDL 39 mg/dL, K+ 4.2 mEq/L, Na+ 138 mEq/L, blood urea nitrogen 13 mg/dL, serum creatinine 0.63 mg/dL, and eGFR >60 mL/min/1.73 m2. Blood pH and bicarbonate levels were within normal limits and the patient was hCG negative.
Prior to their visit with the CDTM pharmacist, DS was being treated with metformin 1000 mg by mouth twice daily and empagliflozin 25 mg by mouth daily. During the initial visit, DS was started on insulin glargine (Lantus®) 10 units subcutaneously once nightly to target elevations in blood glucose as per 2023 ADA guidelines. Within 24 hours of injecting insulin glargine (Lantus®), DS began to report excessive nausea. She remained on therapy for 9 weeks with dose adjustments up to 18 units per day made based on her self-monitored blood glucose log book. DS reported complete adherence to medication therapy and no new prescription medications were initiated while on insulin glargine (Lantus®). During this time, glycemic control improved with A1C decreasing from 13.9 to 8.4%, with no episodes of hypoglycemia. However nausea persisted and began to result in episodes of emesis especially upon waking. The patient reported rotating injection sites between her outer thigh and abdomen with no relief of symptoms. Insulin glargine (Lantus®) dose was decreased by CDTM pharmacist, with no change in nausea or emesis. Additionally, DS reported nausea could not be controlled with sodium citrate dihydrate or dextrose combined with levulose and phosphoric acid which she purchased over the counter per recommendations made by CDTM pharmacist. The case was discussed with their primary care provider and referrals were placed for gastroenterology, however no related gastrointestinal pathology were determined by specialists. As DS was being treated at an underserved urban city-run community hospital, insulin glargine (Lantus®) was switched to the alternative formulary agent which was insulin detemir. Upon switching agents, symptoms subsided over a 3 day period. The patient remained asymptomatic over the next 12 weeks, however glycemic control worsened while on insulin detemir with 2 episodes of hypoglycemia. The patient requested to re-trial insulin glargine (Lantus®) to reassess if re-initiation would produce similar adverse drug effects of nausea and emesis. Within a 1 week period, the patient reported that nausea returned within 2 hours of medication administration and continued daily with 1 episode of emesis. Insulin glargine (Lantus®) was again withdrawn and symptoms resided. DS’s A1C is now controlled on metformin, empagliflozin, and insulin detemir.
Discussion
The Naranjo Algorithm, or Adverse Drug Reaction Probability Scale, was used to assess the likelihood of a causative relationship between the administered medication and an adverse medication event. 15 The pertinent finding of the Naranjo Algorithm for the described patient in the relationship between insulin glargine (Lantus®) and emesis are described in Table 1. The patient received a score of 6, which indicates probable cause. As alternative causes such as lifestyle modification and diet could not be tracked on a day-to-day basis, confounding variables may be present. Drug-Drug and Drug-Disease interaction were not determined during the course of insulin glargine (Lantus®) therapy. Additionally, due to insurance formulary coverage, biosimilar preparations of insulin glargine could not be tested. Therefore, the Naranjo Adverse Drug Reaction Probability Scale resulted in a probable causal relationship between insulin glargine (Lantus®) and the nausea DS experienced.
Naranjo Adverse Drug Reaction Probability Scale.
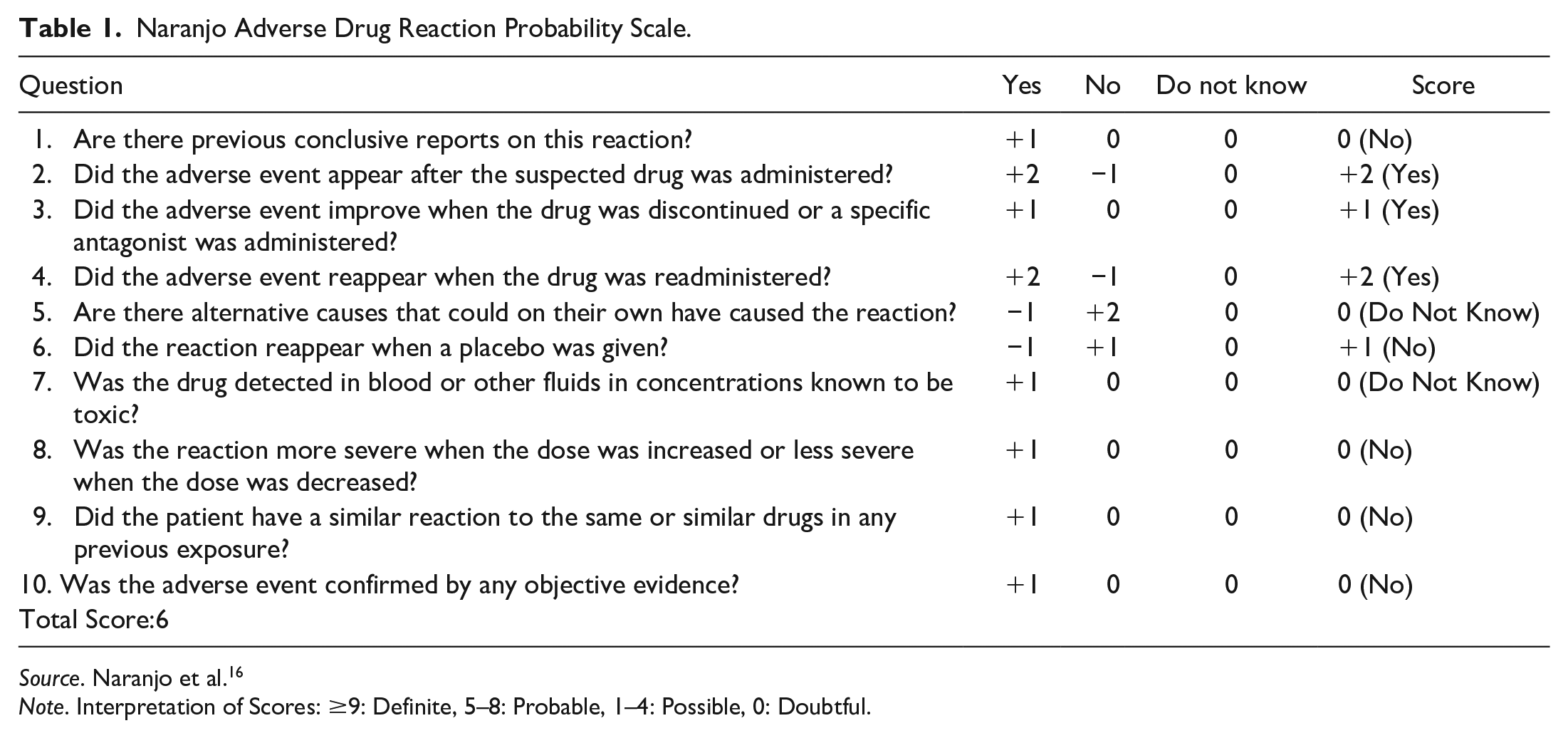
Source. Naranjo et al. 16
Note. Interpretation of Scores: ≥9: Definite, 5–8: Probable, 1–4: Possible, 0: Doubtful.
The described case report reflects the complexity of an uncommon insulin adverse effect in clinical practice. Literature and pharmacokinetic studies do not provide definitive answers as to why DS reacted to insulin glargine (Lantus®) and not insulin detemir. Furthermore, because the information obtained from the patient case was subjective, the severity and duration of symptoms are unknown. However, common causes of nausea and emesis such as gastrointestinal pathology, drug interactions or hypoglycemia were not determined. Based on the clinical picture and the available information, the possible mechanisms for her presumably medication-mediated reaction can be hypothesized.
The first major aspect to consider is the excipients used in the different insulin preparations. Both insulin glargine and insulin detemir contain zinc and metacresol. These 2 excipients have been associated with a large number of allergic reasons. 17 The 2 insulins differ in other excipients with insulin glargine containing glycerol, hydrochloric acid, and sodium hydroxide, 9 and insulin detemir preparation containing mannitol, phenol, and disodium phosphate dehydrate. 18 Both insulin glargine (Lantus®) and insulin detemir were within their expiration dates while the patient was taking them and there were no signs of adulteration when insulin pens were investigated by the CDTM pharmacist. Additionally 3 different lot numbers were used during the course of insulin glargine therapy.
Another consideration involves the molecular structure and the method of protraction between insulin glargine and insulin detemir. Insulin detemir is produced by changing the threonine amino acid at the B30 position and acylation of lysine at the B29 position with myristic acid. This results in increased reversible binding to albumin in the interstitial fluid and plasma, which is responsible for its prolonged duration of action. 19 Euglycemic clamp studies demonstrated a dose-dependent duration of action between NPH insulin and insulin glargine. 20 Insulin glargine is produced from a non-pathogenic laboratory strain of Escherichia coli. The amino acid asparagine is replaced by glycine at position A21 and 2 arginines are added to the C-terminus of the B-chain differentiating insulin glargine from human insulin. This substitution gives insulin glargine a dense crystal structure allowing for a slower dissociation rate. 9 Multiple case reports have demonstrated that structural differences in these insulin analogs may result in unique immunologic profiles and that this reaction may be due to the amino acid sequences or molecular moieties of the insulin glargine analog. 21
Conclusion
Nausea-like symptoms and their clinical relationship with insulin glargine (Lantus®) remain a topic of question. Potential mechanisms including possible sensitivity to excipients and the molecular structure of the preparation have been postulated. However, as there is currently limited literature supporting this claim, further investigation is warranted. The significance of this causation should be considered minor given the broad use of insulin glargine in the treatment of DM and the lack of reporting of this adverse effect.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
