Abstract
Introduction
Bloodstream infections are associated with significant morbidity and mortality which have persisted despite the introduction of new antibiotic therapies, optimized dosing regimens, and increased collaborative decision-making across disciplines. 1 Previous studies have demonstrated that time to directed antibiotic therapy is a key predictor of mortality in patients with bacteremia. 2 In addition, inappropriate antibiotic use contributes to adverse outcomes such as the development of antibiotic resistance, adverse drug reactions, secondary infections, and excess healthcare costs. For these reasons, it is imperative that patients with bacteremia receive effective antibiotics as quickly as possible, with de-escalation occurring as appropriate thereafter.
Integration of pharmacists and use of decision support within the electronic medical record (EMR) (Epic) have been shown to have a significant improvement in the care of patients with bacteremia. The implementation of an automated, pharmacist-driven, scoring system within the EMR has been shown to improve patient care in patients with Staphylococcus aureus bacteremia by increasing the adherence to disease specific quality-of-care measures. 3 In addition, multiple studies have shown that reporting rapid molecular diagnostic test results on blood cultures to a pharmacist rather than a nurse decreases the time to directed antibiotics.4,5 These studies show the impact that pharmacist interventions on blood culture results can have on improving patient care. However, there is a lack of data to date evaluating a system to incorporate review of all blood culture results into standard pharmacist workflow.
The purpose of this study was to evaluate the implementation of an automated, pharmacist-driven, antimicrobial scoring system within the EMR that incorporates the review of blood culture results into the standard pharmacist workflow.
Materials and Methods
Study Design
This was a retrospective, single-center, quasi-experimental study of hospitalized adult (18-89 years of age) patients with bacteremia between July 6, 2018 and July 5, 2019 (pre-implementation group) and September 6, 2019 and September 5, 2020 (post-implementation group) at a 1882 bed tertiary care academic medical center. The study was approved by the institution’s Office of Responsible Research Practices Institutional Review Board. Patients were identified by an electronic report of positive blood cultures within the study period. The report was then randomized, and patients were screened until the desired sample size of 100 patients in each group was obtained. Patients had to receive at least one antibiotic dose in addition to a positive blood culture to be included in this study. Patients meeting any of the following criteria were excluded: incarcerated, pregnant, intensive care unit admission, S. aureus bacteremia or fungemia, positive blood culture that was deemed a contaminant by the treatment team, positive blood culture at an outside hospital prior to transfer to our institution, expired or discharged before the blood culture results were available.
Antimicrobial Scoring System
This automated system was developed within the EMR (EPIC, Verona, WI) by informatics pharmacists in collaboration with clinical pharmacy specialists and is designed to generate a score in the Antimicrobial Score patient list column when a patient has a positive culture from a blood, cerebrospinal fluid (CSF), or respiratory source. The antimicrobial score was expanded from the existing framework of a score for S. aureus bacteremia established by Wang et al. 6 This antimicrobial score was part of a larger set of clinical scores that includes pharmacokinetic monitoring, renal function monitoring, and anticoagulation monitoring, which were an established component of the pharmacist clinical monitoring workflow. Various clinical rules were developed, with each generating a different score value, and corresponding to a higher urgency of clinical review need. Each clinical rule would be evaluated by the EHR independently, and the scores added together. The pharmacist could then use the score column in their patient list roster to sort their patients by the highest scores. Additional columns assist with this workflow, and allow the pharmacist to mark a score as reviewed for the day, or see how long ago a score has been reviewed. A score of 5 points indicates that there is an abnormal culture result without any susceptibilities reported and a score of 50 points indicates that there are susceptibilities reported with the abnormal culture result (see Appendix 1). A score of 500 points is reserved specifically for patients with S. aureus bacteremia or fungemia which were excluded in this study.
In the pre-implementation period, pharmacists were reviewing S. aureus bacteremia results but there was no standardized workflow for pharmacist review of other blood culture results. On August 6, 2019, the institution’s Department of Pharmacy launched the antimicrobial scoring system following required pharmacist education. The system allowed for real-time pharmacist notification of new abnormal blood, CSF, and respiratory culture results, which then allowed them to use clinical judgment to make recommendations to the primary team provider with the goal of ensuring appropriate antimicrobial coverage based on the culture results.
There are three 8-hour shifts within the pharmacy department, with the third shift being an overnight shift. Pharmacists were required to evaluate the antimicrobial scoring system results at least once per shift on first and second shift to identify any patients with a score assigned. It is part of standard pharmacist responsibilities to check if new scores have fired for their patients and address them in a timely manner, as the antimicrobial score does not create a pop-up alert. Both 5- and 50-point scores that resulted on third shift were generally not addressed until first shift as third shift only responded to 500-point scores which were excluded from this study. Clinical generalist pharmacists are staffing on each shift while clinical pharmacy specialists are only available during the first shift. These pharmacists evaluate all patients with new culture results and make recommendations to the treating provider as necessary to direct the antibiotic regimen to the culture results.
Positive blood cultures are first reported by gram stain. An update to the antimicrobial score is triggered once the gram stain and initial culture result are reported within the EMR. These results are also reported to the patient’s nurse who then alerts the treatment team of the positive culture. Molecular diagnostic testing of the blood cultures was then performed using Verigene© gram-positive and gram-negative systems (Luminex) to determine speciation and the presence of select resistance markers. The Verigene© ran at all times of the day and was run on every positive blood culture. Once susceptibilities were reported in the EMR, the score was triggered to update again.
Data and Outcomes
Data collected included age, sex, race, hospital service, admission and discharge dates, source of bacteremia (pulmonary, urinary, line associated, skin or wound, intra-abdominal, or unknown), whether source control was achieved if necessary (defined as removal of central venous catheters, drainage of abscesses, removal of infected hardware or tissue, or debridement of osteomyelitis or wounds), documentation of Clostridioides difficile infection in the EMR within 3 months following discharge, whether the patient was immunosuppressed (defined as receiving any cytotoxic agents, corticosteroids ≥20mg of prednisone equivalence daily for more than 2 weeks, antirejection medications within the past 2 weeks, HIV patients with a CD4 cell count <200 cells/mm3, or patients with an absolute neutrophil count <500 cells/mm3), the date and time of the Verigene© or other molecular diagnostic test results (this time was used as a proxy for the exact time the antimicrobial scoring would generate a score and alert a pharmacist to the blood culture results), the start and stop dates for each antibiotic ordered while inpatient, the date and time of first negative blood culture (if repeat blood cultures were collected), and death within 30 days of the blood culture results.
The primary outcome was time to directed antibiotic therapy, which was defined as the difference from the time of the Verigene© or other molecular diagnostic test results and the time to therapy administration that is guided by culture susceptibilities, which can include either escalations or de-escalations in antibiotic spectrum (eg, de-escalating from piperacillin/tazobactam to ceftriaxone for an E. coli bacteremia; or escalating from cefepime to meropenem for an extended spectrum beta-lactamase positive K. pneumoniae bacteremia). Secondary outcomes included hospital length-of-stay, days of therapy (DOT, defined as the total number of antibiotic days received by a patient; eg, if a patient received 2 antibiotics for 7 days then that equaled a DOT of 14), time to effective therapy (defined as the difference from the time of Verigene© or other molecular diagnostic test results and the administration of antibiotic therapy that includes one antibiotic that has activity against the bacteria that was identified on culture), rate of C. difficile infections in the 3 months following discharge, and time to bacteremia clearance (defined as time from positive culture collection to time of collection of first negative culture).
Statistical Analysis
Demographic and clinical information was analyzed using descriptive statistics. Given a paucity of comparable pre-existing literature, no sample size calculation was able to be completed. Comparisons of categorical data were performed using χ2 or Fisher’s exact test, as appropriate. Comparisons of continuous data were performed using a Student’s t test or Mann-Whitney U test, as appropriate. Normally distributed data were reported as mean ± standard deviation (SD) while non-normally distributed data were reported as median [IQR]. A 2-tailed significance of <.05 was considered statistically significant. All statistical analyses were completed using SPSS (Version 28, Chicago, Illinois).
Results
A total of 527 patients were randomly screened until the desired sample of 100 patients in each group was reached; 327 patients were excluded with some patients meeting more than one exclusion criteria (Figure 1). The most common reasons for exclusion were Staphylococcus aureus bacteremia and organisms identified on blood culture that were deemed a contaminant by the treatment team.
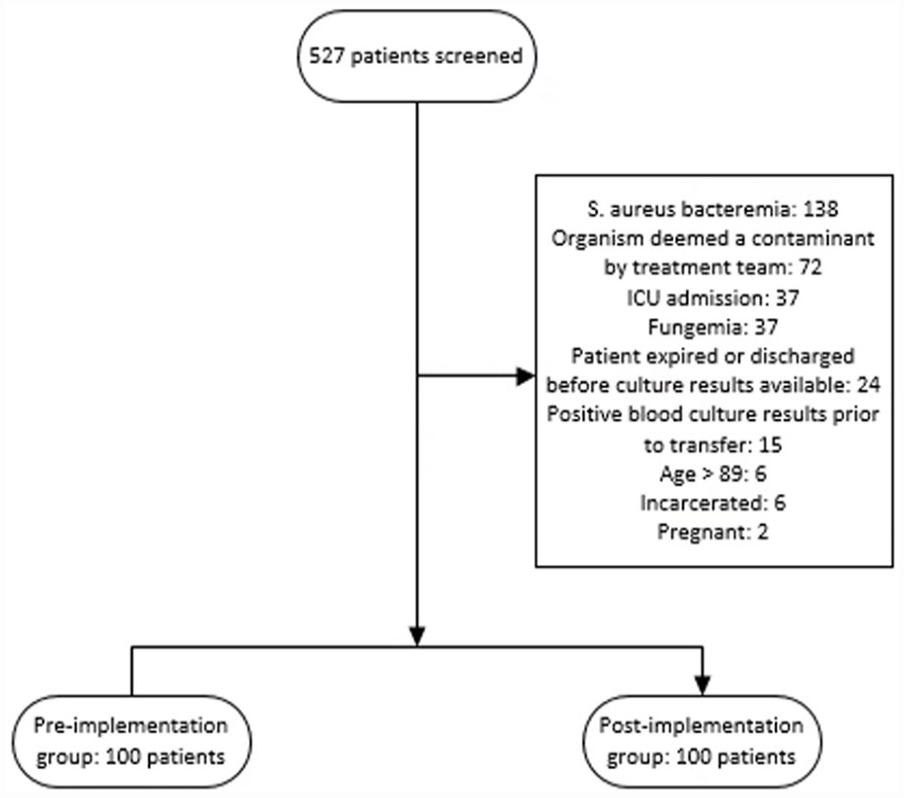
Flowchart of inclusion and exclusion criteria.
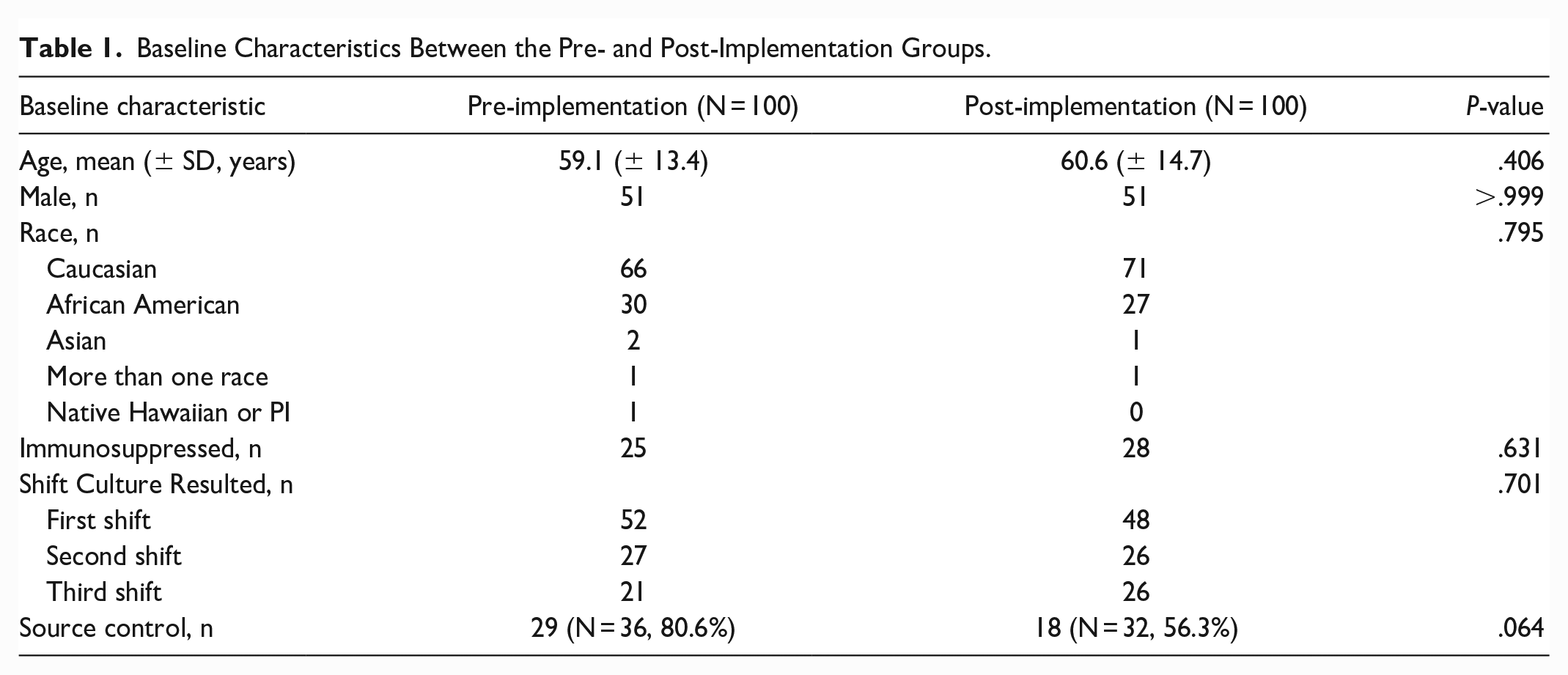
The baseline characteristics for each group are summarized in Table 1. No statistically significant differences were noted between the pre- and post-implementation group for demographic information. There was also no difference noted between groups in terms of patient characteristics such as number of immunocompromised patients and patients who achieved source control, although numerically the post-implementation group did have a lower percentage of source control than the pre-implementation group (56.3%vs 80.6%; P = .064).
Baseline Characteristics Between the Pre- and Post-Implementation Groups.
The most common bacteria identified on blood culture were Escherichia coli, Klebsiella spp., Streptococcus spp., and Enterococcus faecalis (Table 2), and no significant difference was noted in isolated bacteria between the 2 groups (P = .686). No patients in this study had a polymicrobial bacteremia. There was no difference observed in the source of bacteremia, with the most common sources being urinary, line associated, and intra-abdominal.
Infection Characteristics Between the Pre- and Post-Implementation Groups.
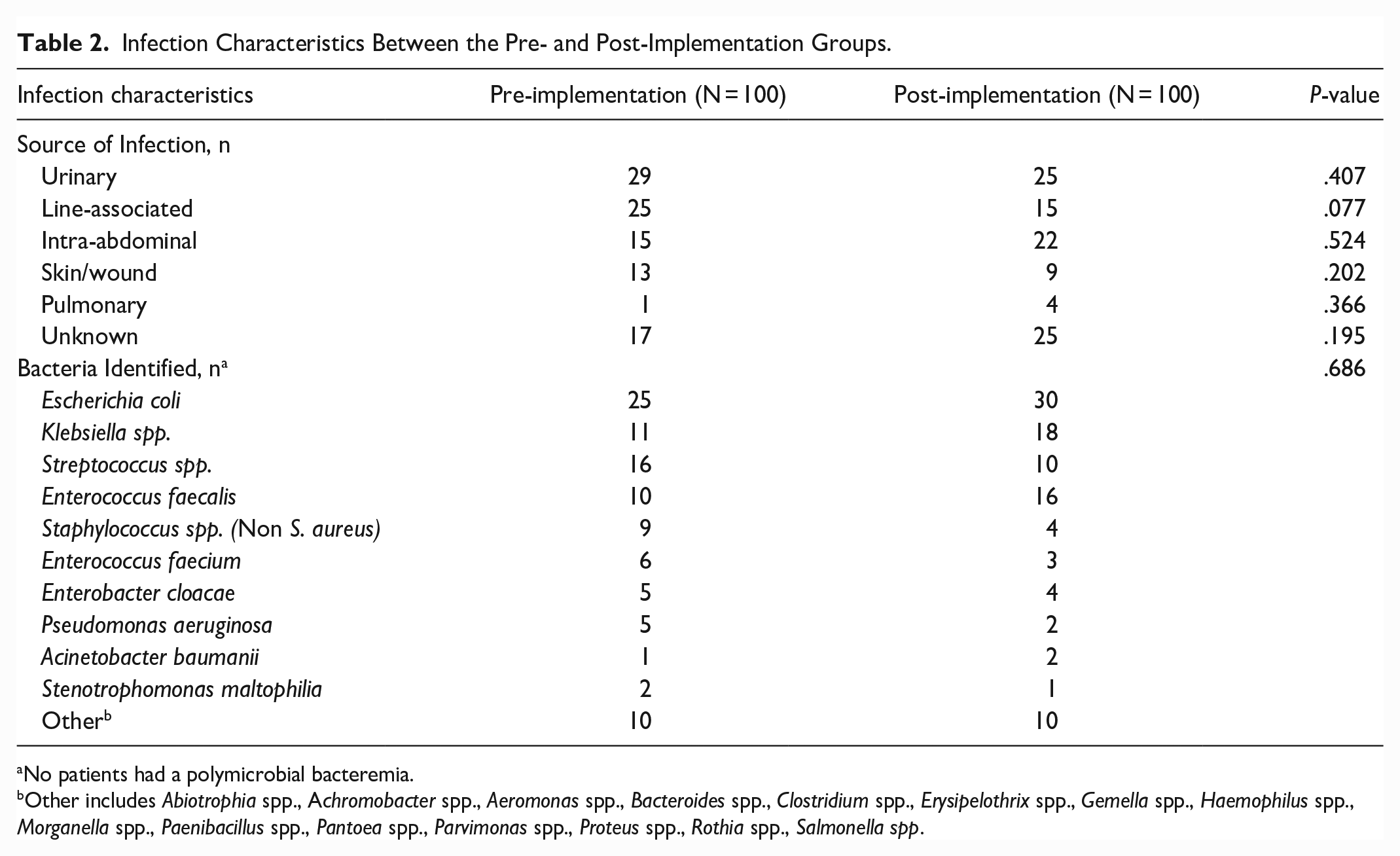
No patients had a polymicrobial bacteremia.
Other includes Abiotrophia spp., Achromobacter spp., Aeromonas spp., Bacteroides spp., Clostridium spp., Erysipelothrix spp., Gemella spp., Haemophilus spp., Morganella spp., Paenibacillus spp., Pantoea spp., Parvimonas spp., Proteus spp., Rothia spp., Salmonella spp.
There was no difference in time to directed antibiotic therapy between the pre- and post-implementation of the antimicrobial scoring system groups (median 32.5 hours [10.4-64.5] vs 37.4 hours [13.5-56.6]; P = .757). There was also no difference in time to effective antibiotic therapy (−12.6 hours [−23.4 to 1.1] vs −14.2 [−20.1 to −0.6]; P = .905; negative numbers indicating that effective antibiotic therapy was started prior to culture results becoming available). Of note, 74.5% of patients were on effective therapy prior to culture results. The lack of significance between groups was maintained when analyzed what shift the blood culture resulted (Table 3). No differences in other secondary outcomes including 30-day all-cause mortality, hospital length-of-stay, inpatient days of therapy, and rate of C. difficile infections within 3 months of culture results were observed (Table 3).
Outcomes Between the Pre- and Post-Implementation Groups.
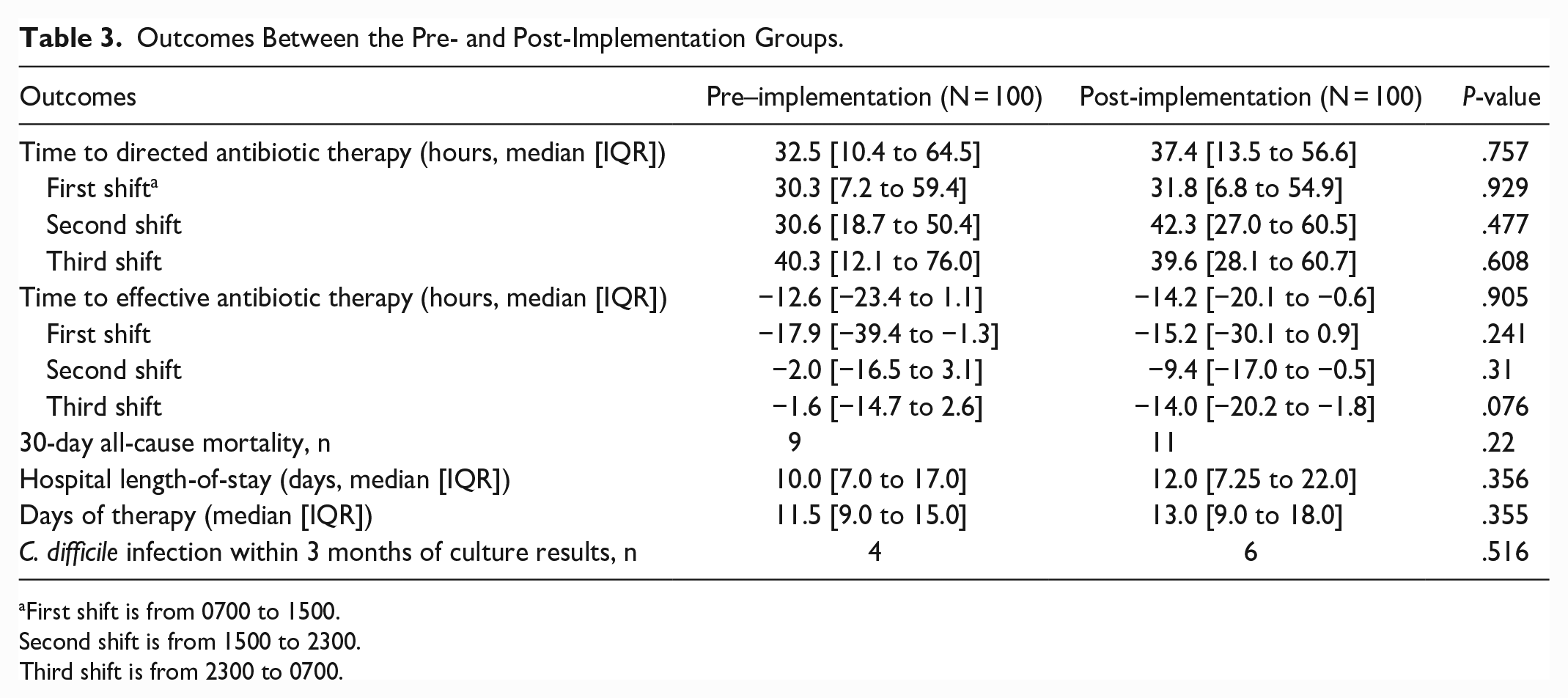
First shift is from 0700 to 1500.
Second shift is from 1500 to 2300.
Third shift is from 2300 to 0700.
Discussion
The implementation of an automated, pharmacist-driven, antimicrobial, patient acuity scoring system had no effect on time to directed antibiotic therapy in the included study population. This is comparable to the study by Wenzler et al 3 which did not find any difference in time to targeted antibiotic therapy after implementation of an automated scoring system for S. aureus bacteremia. Wenzler et al did however find an improvement in other outcomes related to the management S. aureus bacteremia (rate of ID consultation, repeat blood cultures, echocardiogram, and targeted antimicrobial therapy). Unlike our results, McCarthy et al 4 and Mahrous et al 5 both reported significant decreases in time to directed therapy after pharmacist interventions response to positive blood cultures. However, the methodology for both differed significantly from our scoring system as they included active notification of pharmacists by the microbiology lab followed by intervention rather than the approach of our scoring system. The active and direct notification of positive cultures may explain why these 2 studies found a significant improvement in time to directed therapy while our study and Wenzler et al did not.
The implementation of our antimicrobial scoring system also had no effect on time to effective antibiotic therapy. Kinn et al 7 showed that 24-hour pharmacist review of rapid diagnostic blood culture results decreased the incidence of patients receiving ineffective therapy, which our study did not find. However, in our study 74.5% of patients were receiving effective therapy at the time the culture resulted, limiting the possibility of pharmacist intervention. In addition, there was not 24-hour pharmacist review during the study period, as pharmacist were only required to review on first- and second-shift. There was also no difference observed on the secondary outcomes of 30-day all-cause mortality, hospital length-of-stay, days of antibiotic therapy, and rate of C. difficile infections within 3 months of culture results. These findings are consistent with what was found in the study by Wenzler et al 3 as they also found no improvements in hospital length-of-stay, 30-day mortality, and inpatient DOT.
There are a few potential explanations for why the implementation of this antimicrobial scoring tool did not show a difference in treatment related or patient outcomes. As mentioned by Dilworth et al 8 in a comment on the study by Wenzler et al, 3 interpretation of the impact of time to antibiotic endpoints is fraught with confounding clinical and administrative factors, as well as measurement errors, making the actual impact of antimicrobial stewardship initiatives very difficult to determine. At our institution, pharmacists were already well integrated into most rounding services prior to the implementation date, meaning that they would already be reviewing blood culture results and making recommended changes to the treatment team as part of their standard patient care workflow. This study did not specifically evaluate patients admitted by services without a dedicated rounding pharmacist. Additionally, patients with bacteremia will commonly receive an infectious disease consult, in which case a pharmacist reviewing the antimicrobial score may be less likely to make a recommendation and instead defer to the infectious disease team. Furthermore, second shift pharmacists are not in direct contact with the treatment team, and the covering providers on second and third shift often defer decisions related to antibiotic de-escalation to the primary team the following day. These examples would reduce the effectiveness of the antimicrobial scoring tool in a large population. In addition, during the pre- and post- implementation study periods, an antimicrobial stewardship on-call program for specific resistant organisms was already in place, in which the antimicrobial stewardship pharmacist or provider was directly notified real time for cultures with those specific organisms. The incidence of organisms that would fall under the scope of this program was not collected, so the effect of this program on the results of this study is unknown. Finally, while not statistically significant, a lower percentage of patients in the post-implementation group achieved source control compared to the pre-implementation group. This could potentially have led to our findings as a lack of source control may cause some providers to wait to de-escalate antibiotics until source control can be obtained.
There were several limitations present in this study. Data related to pharmacist interventions from reviewing the antimicrobial scores was not documented as pharmacists are not required to write notes on interventions made from non-500-point antimicrobial scores. As all pharmacist recommendations related to the antimicrobial scores are provided verbally to the treatment team, assessment of the timing of pharmacist intervention is not possible. The antimicrobial scoring system also does not include a bug-drug mismatch component which could more readily indicate that a change in antibiotic therapy is needed. In addition, there was a large variety of organisms identified in this study meaning it may not be powered enough to detect differences in organisms that may more commonly necessitate changing antibiotic therapy. In our study, 74.5% of patients were receiving effective therapy at the time the culture resulted, reducing the opportunities for intervention and likely means the study was underpowered to detect a difference in time to effective therapy. In addition, our institution has numerous resources available that guide selection of appropriate empiric antimicrobial regimens and educate staff on interpreting blood culture results, including for multi-drug resistant organisms. Due to these resources, there may be less room for possible interventions as empiric therapy choices may be more likely to be appropriate as evidenced by the high rate of effective antibiotics at the time of positive culture in this study. Lastly, the second half of the post-implementation group took place at the beginning of the COVID-19 pandemic. While this likely would not have influenced the primary outcome of time to directed antibiotic therapy, it may have had an impact on secondary outcomes such as hospital length-of-stay and days of therapy.
Further research should focus on identifying patient populations and/or specific bacterial pathogens that would benefit from the antimicrobial scoring system. Specifically targeting patients admitted to hospital services without a dedicated rounding pharmacist may demonstrate a greater effectiveness of the scoring system. Without the antimicrobial scoring tool, this population would likely not have a pharmacist review their blood culture results which could increase the likelihood of an intervention being made for these patients. For this reason, a system like the one studied here may be most beneficial at an institution without resources for dedicated rounding pharmacists. Alternatively, future studies could focus on a narrower group of pathogens that may less likely be covered by typical empiric antibiotic regimens.
Conclusion
The implementation of an automated, pharmacist-driven, antimicrobial, patient acuity scoring tool within the electronic medical record did not improve time to directed antibiotic therapy in hospitalized bacteremic patients. In addition to the overall difficulty in identifying the impact of antimicrobial stewardship tools, there were several confounding factors present which could have altered outcomes data and limited the effectiveness of this tool. More research is needed to identify a specific population or institutional structure that would benefit from the antimicrobial scoring tool.
Footnotes
Appendix 1
Antimicrobial Scoring System Criteria.

| 5 points | Indicates a positive blood, respiratory, or CSF culture within the last 48 hours with no susceptibilities listed |
| 50 points | Indicates susceptibilities have been reported for a positive blood, respiratory, or CSF culture within the last 48 hours |
| 500 points | Indicates Staphylococcus aureus bacteremia or fungemia |
Scores are additive. For example, a score of 55 is possible if both criteria for 5 and 50 points are met concurrently.
Acknowledgements
None
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
