Abstract
Keywords
Introduction
Venous thromboembolism (VTE) results in poor patient outcomes such as increased morbidity, mortality, length of stay (LOS); as well as increased costs to the health system. 1 As VTEs are a leading cause of preventable death in Australia, the Australian Commission on Safety and Quality in Health Care mandates hospitals to implement measures that reduce VTEs.1,2 VTE risk assessments for hospitalized patients play a key role in preventing VTE complications, and mortality, by providing thromboprophylaxis guidelines.3 -6
Local and international guidelines recommend using tools such as the Caprini score to determine VTE risk.7,8 The Caprini score is a risk tool for surgical patients that uses 39 risk factors, weighted and summed, to a achieve an overall VTE risk score; higher scores indicate greater VTE risk. 9 In addition to VTE risk, bleeding risk should also be considered prior to prescribing anticoagulation for thromboprophylaxis. Currently, there are no formalized risk calculators for approximating bleeding risk secondary to thromboprophylaxis. However, the HASBLED score has been developed to estimate bleeding risk for anticoagulation treatment for atrial fibrillation. 10 Although not specific to prescribing thromboprophylaxis, the HASBLED may have utility in other patient cohorts.11,12
If thromboprophylaxis is required according to VTE risk assessment, guidelines can be used to determine the most appropriate regimen. However, various guidelines have different thromboprophylaxis recommendations for low molecular weight heparin (LMWH) and unfractionated heparin (UFH) at varying doses and frequencies.3 -5,8,13 Compared to UFH, LMWH has a lower risk of heparin induced thrombocytopenia, with equal or greater efficacy.8,13,14 As a result, LMWH is often preferred over UFH. However, UFH is often used in patients with renal impairment or high bleeding risk due to hepatic metabolism. It has a shorter half-life, and its actions are reversible with protamine sulfate.8,13,14
VTE guidelines often have thromboprophylaxis recommendations for specific patient cohorts. Currently, there are limited recommendations for gastroenterology patients and the Caprini score has not been validated in this cohort. This is problematic due to their high risk of both VTEs and bleeding. With the lack of thromboprophylaxis guidelines in this high-risk cohort, the documentation of VTE risk assessments and the appropriateness of thromboprophylaxis regimens within this clinical setting is unknown, particularly in an Australian setting. This study investigated the completion of VTE risk assessments, and the appropriateness of VTE prophylaxis regimens, in accordance with Queensland Health guidelines for gastroenterology patients. The quality and safety of prescribed thromboprophylaxis regimens was assessed based on their VTE risk and bleeding risk.
Method
A retrospective observational study of gastroenterology patients who were prescribed VTE thromboprophylaxis in a major tertiary hospital in Queensland, Australia was conducted. A list of all patients admitted under the gastroenterology team between 1st May 2019 and 1st May 2020 was generated, and a random subset of patients were selected. The electronic medical records of these patients were then manually reviewed for inclusion. Patients were excluded if their LOS was <24 hours, if they were not being treated by a gastroenterology specialty, if they were using therapeutic anticoagulation prior to admission, or if there was missing documentation that made it impossible to calculate their VTE risk.
Data collected included patient socio-demographic characteristics such as age, gender, height, and weight; patient admission details, such as principal diagnosis, LOS, and specialty; and clinical characteristics such as concurrent use of antiplatelets and non-steroidal anti-inflammatory drugs, VTE history (ie, past VTE or VTE during the current admission) and laboratory tests (Appendix A). Laboratory test results for glomerular filtration rate (eGFR), platelet count, and hemoglobin (Hb) level were collected both at baseline (at admission) and at discharge. Any documentation of a re-admission to the same hospital within 30 to 60 days was collected. This timeframe was chosen because the risk of developing a VTE is highest in the first 2 months post-discharge. 6
Data Analysis
The VTE risk and bleeding risk for each patient, according to applicable risk factors, was calculated using a modified Caprini score, and the HASBLED score (Appendix B).7,9 The modified Caprini score was used, instead of the original Caprini score, because all 31 specific variables required for the original Caprini score were not documented. Each patient was allocated a modified Caprini score out of 13; ≤3 indicated low VTE risk, 3 to 4 indicated moderate VTE risk, and ≥5 indicated high VTE risk. 8 The HASBLED score is only validated for bleeding risk for atrial fibrillation; however, it was used as a surrogate marker of bleeding risk for gastroenterology patients as there is no formalized risk-assessment tool for these patients. 10 Each patient was allocated a HASBLED score out of 9; higher scores were indicative of higher bleeding risk. 10 Thromboprophylaxis-related complications were determined by changes in hemoglobin level and platelet count from baseline to discharge. A reduction in platelets of 50%, and a decrease in Hb levels of 20 g/L, were chosen as clinically significant markers for medication harm secondary to thromboprophylaxis.15,16
Descriptive statistics were presented for clinical and socio-demographic characteristics; and thromboprophylaxis prescription according to Caprini and HASBLED scores. Chi-squared (
Results
A random sample of 271 patients were reviewed and 177 were excluded: Eighty-seven due to LOS <24 hours, 81 not under gastroenterology, 6 due to therapeutic anticoagulation prior to admission, and 3 due to missing documentation. This resulted in a final sample of 94 patients that met the final inclusion criteria. Patients had a mean (SD) age of 58.3 (±20) years and a mean (SD) body-mass-index (BMI) of 26.9 (±7.1) kg/m2, while 43 (45.7%) were female (Table 1). The LOS was 5.4 (±3.9)days and 72 (77%) of patients had surgery during their admission. The most common presenting complaints were upper gastrointestinal disorders (29%) and gastrointestinal bleeds (27%).
Clinical and Socio-Demographic Patient Characteristics (n = 94).

Note. GI = gastrointestinal, UGIB = Upper gastrointestinal bleed, PUD = peptic ulcer disease.
One patient did not have height recorded
Two patient did not have weight recorded
Overall VTE Risk, Bleeding Risk, and Thromboprophylaxis Prescription
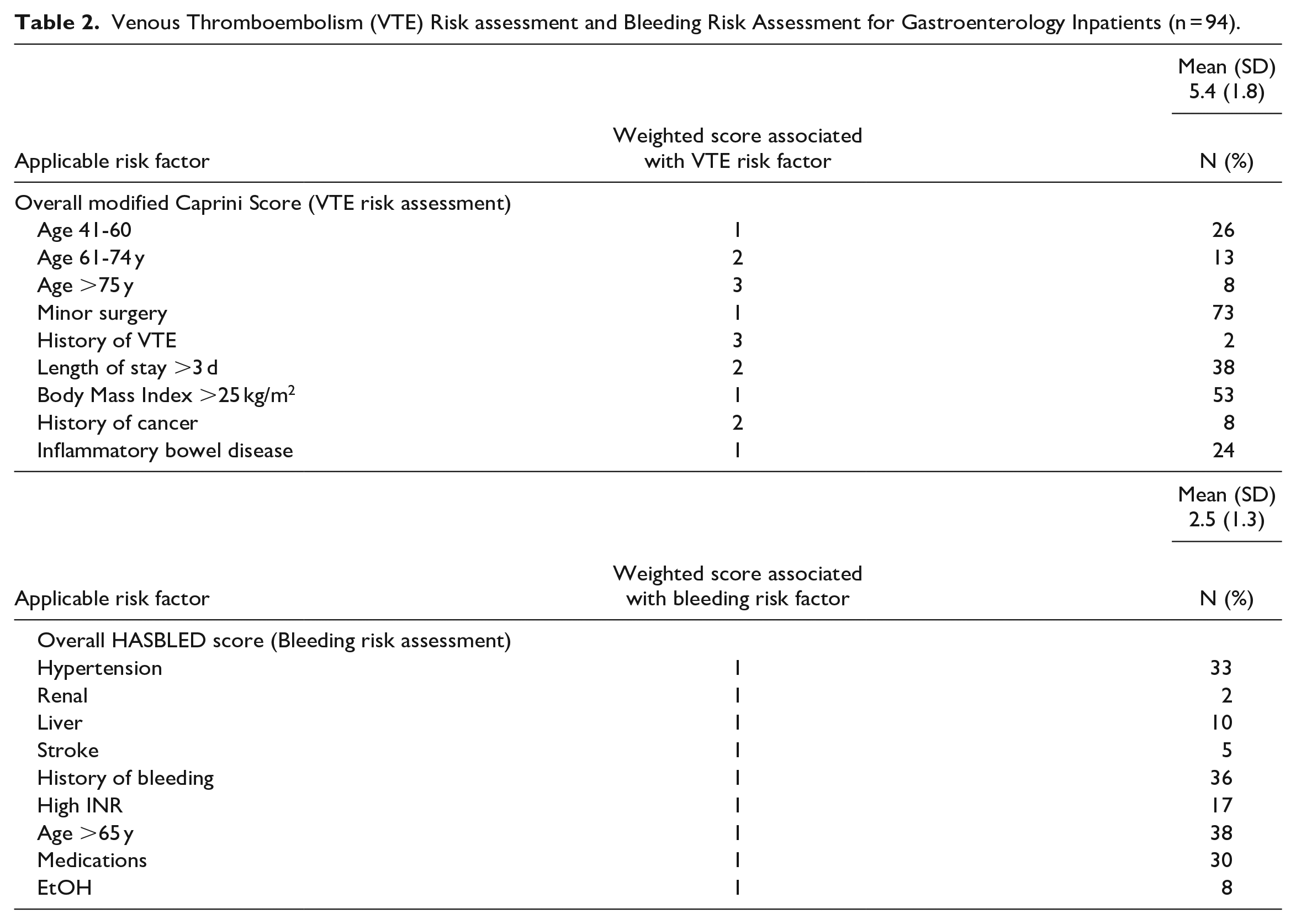
Of the 94 inpatients, 26 (28%) had documented contraindications for prescribed thromboprophylaxis because of active bleeding. Therefore, 68 patients had with no contraindications to thromboprophylaxis. Of these 68 patients, 32 (47%) were not prescribed thromboprophylaxis and did not have documentation of VTE risk assessment recorded. Overall, patients had a high VTE risk with a mean (±SD) modified Caprini score of 5.4 (±1.8), and a lower bleeding risk with a mean (±SD) HASBLED score of 2.5 (±1.3) (Table 2).
Venous Thromboembolism (VTE) Risk assessment and Bleeding Risk Assessment for Gastroenterology Inpatients (n = 94).
The Use of VTE Prophylaxis Based on Thrombotic Risk
The use of VTE prophylaxis based on Caprini scores is shown (Figure 1). There were 13 patients with a low VTE risk, of which 4 (31%) were prescribed thromboprophylaxis. There were 55 patients with moderate to high VTE risk and 32 (58%) were prescribed thromboprophylaxis. When comparing the proportion of thromboprophylaxis prescribing between patients with low VTE risk and moderate-high VTE risk, no significant difference between thromboprophylaxis prescribing according to VTE risk was found (P = .075) (Table 3).
Comparison of Thromboprophylaxis Prescribing by VTE Risk and Bleeding Risk for Gastroenterology Inpatients (n = 68).
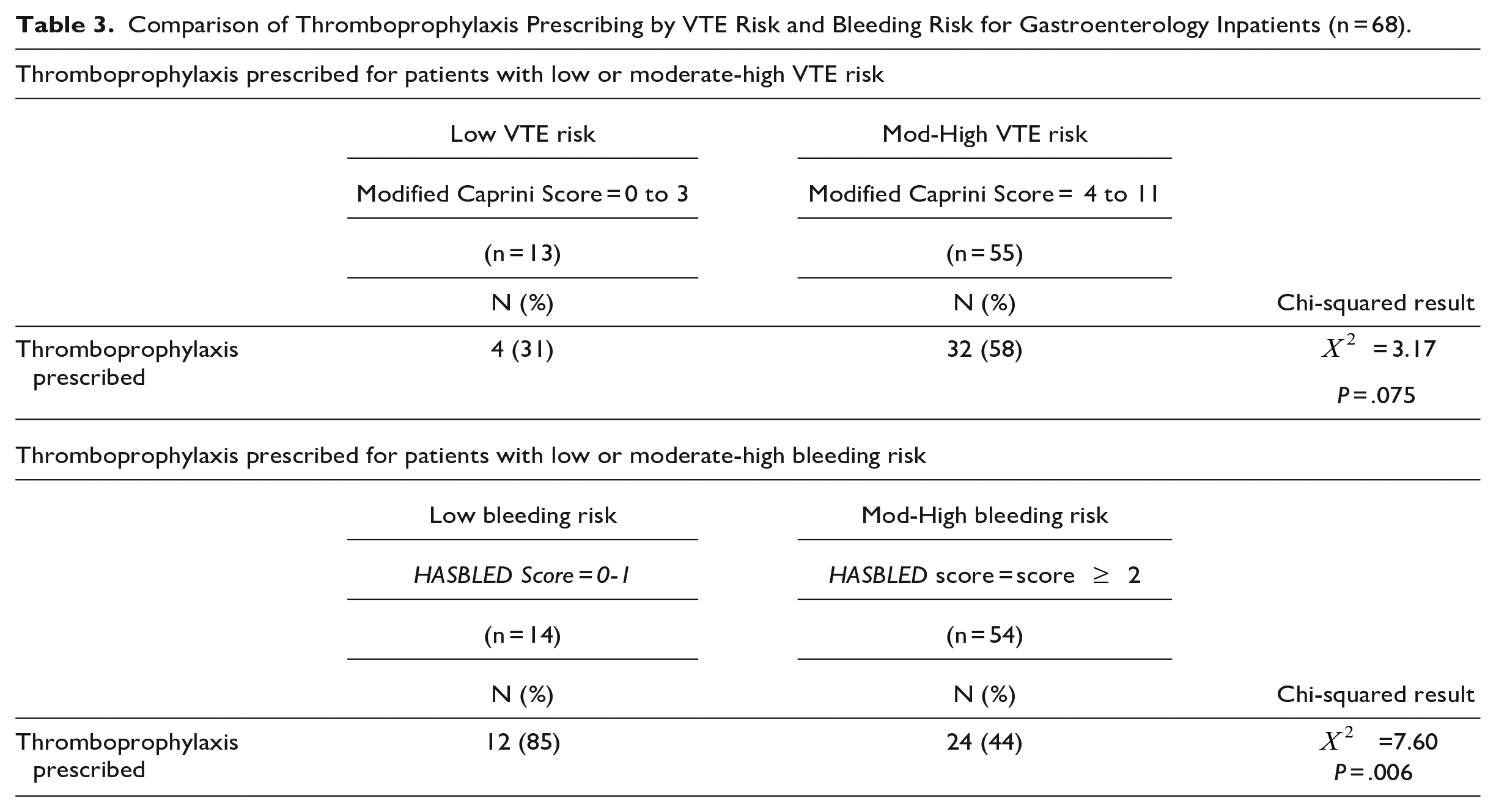
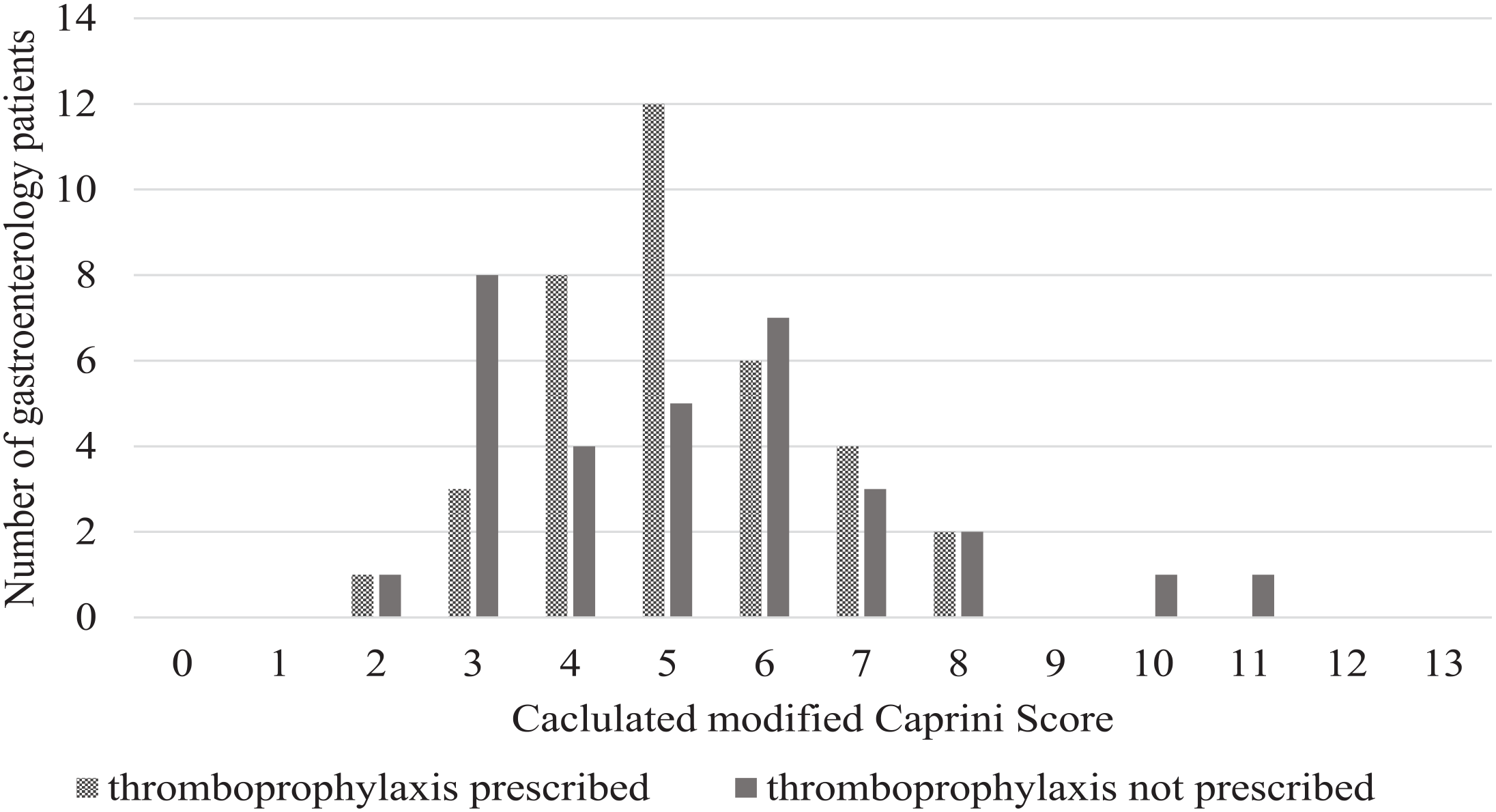
Thromboprophylaxis prescription according to calculated modified Caprini scores for gastroenterology patients with no contraindications to thromboprophylaxis (n = 68).
The Use of VTE Prophylaxis Based on Bleeding Risk
The use of VTE prophylaxis based on HASBLED scores had a trend for decreased thromboprophylaxis prescribing as bleeding risk increased (Figure 2). There were 14 patients with a low bleeding risk, of which 12 (85%) were prescribed thromboprophylaxis. Of the 54 patients with moderate to high bleeding risk, 24 (44%) were prescribed thromboprophylaxis. When comparing the proportion of thromboprophylaxis prescribing between patients with low versus moderate-high bleeding risk, there was a significant difference between the groups (P = .006); patients with moderate-high bleeding risk were less likely to be prescribed thromboprophylaxis (Table 3).
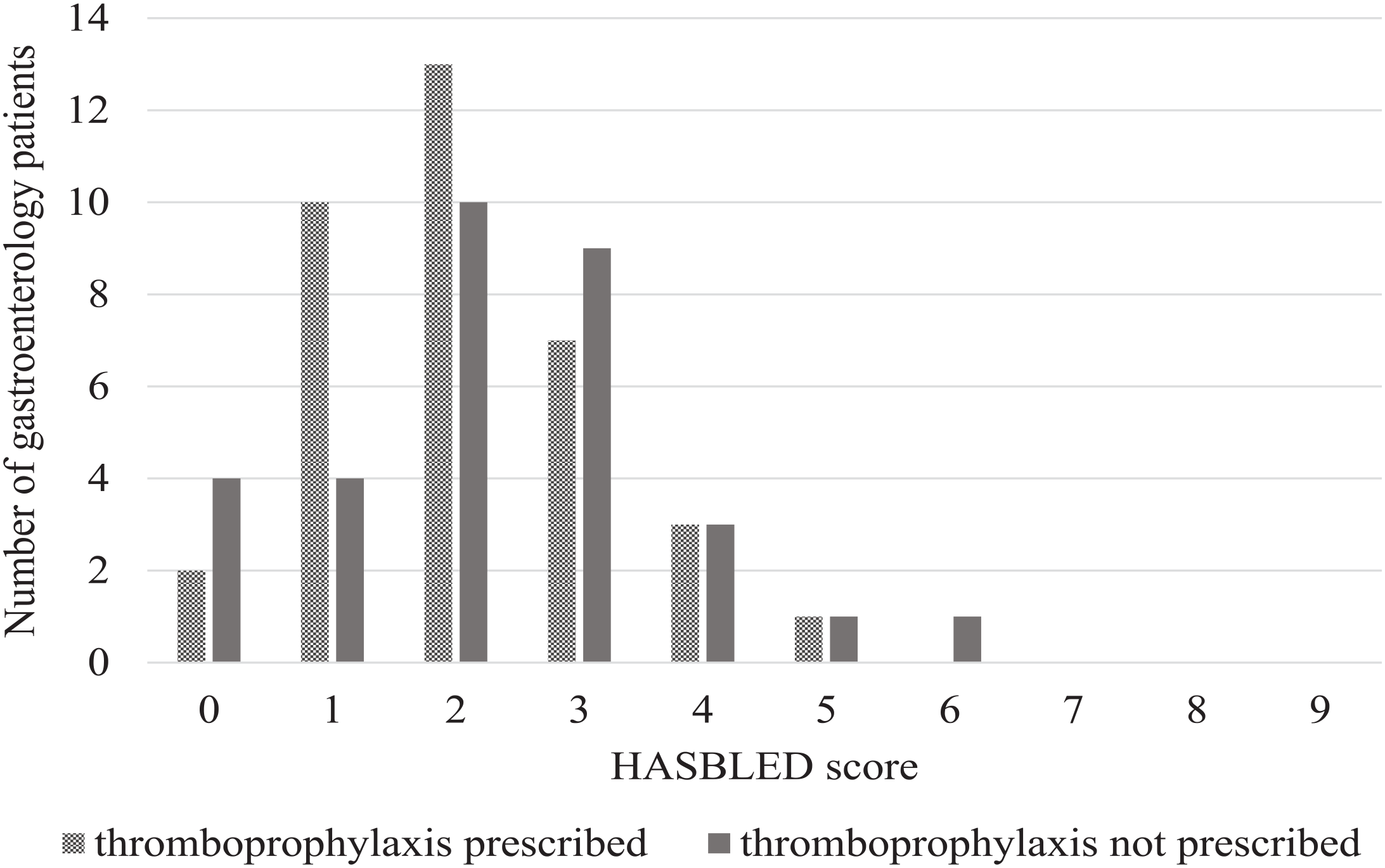
Thromboprophylaxis prescription according to calculated HASBLED scores for gastroenterology patients with no contraindications to thromboprophylaxis (n = 68).
Type of Thromboprophylaxis Prescribing and Associated Complications
Patients were 11.5 times more likely to receive LMWH (Dalteparin) than UFH, with 33 (92%), and 3 (8%) patients prescribed Dalteparin and UFH, respectively. For all gastroenterology patients, appropriate doses of UFH, and dalteparin were prescribed in accordance with VTE guidelines. 8 A review of all re-admissions within 30 to 60 days did not show any VTE thromboprophylaxis complications. The changes in mean values of Hb and platelet levels from admission to discharge according to prescribed thromboprophylaxis is shown (Table 4). There was no significant difference in the mean Hb level change between patients that were prescribed thromboprophylaxis and those that were not (P = .164). There was also no significant difference in mean platelet level between patients that were prescribed thromboprophylaxis and those that were not (P = .749). Additionally, no patients experienced a platelet reduction greater than 50% or a Hb drop greater than 20 g/L (key thresholds that can be used as markers for potential medication related harm).
Differences in Mean Values of Hemoglobin and Platelet in Gastroenterology Patients by Thromboprophylaxis Prescription (n = 68).

Four patients had missing hemoglobin data and six patients had missing platelet data.
Hemoglobin is a measure for bleeding and a 20 g/L decrease was considered as clinically significant.
Platelets is a measure for HITT and 50% decrease was considered as clinically significant.
Discussion
This study investigated the appropriateness of VTE prophylaxis regimens, and the quality and safety of thromboprophylaxis prescribing according to VTE and bleeding risk, for gastroenterology inpatients at a tertiary Australian hospital. All patients that had thromboprophylaxis prescribed had prescriptions which complied with the dosing, duration, and monitoring recommendations outlined in the Queensland Health State-wide VTE guidelines. However, completion and documentation of a VTE risk assessment was not always done. Thromboprophylaxis prescribing did not change significantly according to VTE risk assessed through Caprini scores, however thromboprophylaxis prescribing significantly decreased as bleeding risk according to HASBLED scores increased. Overall, there were no subsequent thromboprophylaxis-related complications from baseline to discharge.
Local VTE prophylaxis guidelines recommend that a VTE risk assessment is completed for all patients. 8 However, in line with other studies, there was a lack of documentation for the completion of VTE risk assessments for hospital inpatients. 17 Limited documentation can cause inefficiencies in workflow, with multiple clinicians completing the same assessment, which increases patients risk as appropriate thromboprophylaxis prescribing might be missed if it is assumed to have been completed already. 18 With VTEs as a leading cause of preventable patient harm, this highlights the need to improve assessment, and documentation of VTE risk for hospital inpatients. The Australia and New Zealand Working Party on the Management and Prevention of Venous Thromboembolism have strategized that establishing electronic reminder systems may promote optimal use of thromboprophylaxis. 6
One method to improve the screening and prescribing of thromboprophylaxis is using digital tools such as dashboards. Dashboards offer a novel approach for improving patient safety and optimizing high-risk therapies as digital dashboards can stream clinical data from electronic health records.19,20 Dashboards can also be coupled with a VTE stewardship program where a specialist pharmacist can work collaboratively, or as part of a team, to identify, explore, and escalate therapeutic problems to treating clinical teams for timely and corrective action. 21 Pharmacist-led VTE stewardship has demonstrated reductions in mortality, LOS, bleeding complications, blood transfusion requirements, and cost of therapy.22,23 Such a dashboard has recently (March 2021) been implemented at this hospital. An audit of 1024 patients demonstrated that 73 (7%) patients had duplicate therapy (2 anticoagulants) prescribed, and on average 49 patients per day had dosing errors. This equates to 1470 errors each month and 17 885 errors each year. Future research will involve a robust study of the clinical and cost effectiveness of using this dashboard in practice as part of a VTE stewardship program.
Limitations
This study was a retrospective review where the results are reliant on accurate, complete, and consistent documentation in patient clinical records. Undocumented VTE risk factors may result in calculations of lower Caprini scores which could underestimate the VTE risk for patients. Given that VTE risk assessments were often not documented, it was not possible to determine whether this was completed unless this was recorded in the patient’s clinical notes. The HASBLED scoring system was originally designed for patients with atrial fibrillation, therefore, if may not be applicable to this patient cohort. Due to the small sample size, we were unable to adequately assess the incidence of thromboprophylaxis-related complications of thrombotic and haemorrhagic events post discharge. Lastly, patients that presented to a private hospital would not be captured in this study. Despite these limitations, this study provides a basis for future research into the quality and safety of prescribed thromboprophylaxis for gastroenterology inpatients.
Conclusion
The use VTE prophylaxis should be individualized in gastroenterology patients, with each patient assessed for benefits and potential risks, including VTE risk and bleeding risk. Adequate documentation of VTE risk assessment in this specific patient cohort is essential and needs to be improved for hospital inpatients.
Footnotes
Appendices
Acknowledgements
There are no acknowledgements to report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was also approved for ethical exemption by the Princess Alexandra Hospital Drug and Therapeutics Chair and The University of Queensland for the Metro South Human Research Ethics Committee (approval number: 20200705CM).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
