Abstract
Introduction
Synthetic opioids, such as morphine, fentanyl, and remifentanil, have been used in anesthesia for decades. These agents are well characterized by their strong analgesic potency and have comparatively little cardiovascular side effects. Their introduction has increased patient safety—of hemodynamically unstable patients, in particular—by diminishing the need for high-dose use of propofol, barbiturates or volatile anesthetics. 1 Nevertheless, opioids have numerous side effects, such as respiratory depression, decreased airway patency, nausea and vomiting, constipation, hyperalgesia, tolerance development and dependence. 2 A large meta-analysis from 2013 showed that opioid-related adverse events of clinical relevance occurred in about 12% of all operated patients, which led to longer hospital stays, a higher rate of re-entries and significantly higher costs. 3 Consequently—especially in times of the prevailing opioid crisis in the USA—there is a growing effort to reduce, or entirely prevent, the exposure of patients to opioids both during the intra- and postoperative periods. 4 Accordingly, opioid-sparing anesthesia (OSA) and opioid-free anesthesia (OFA) are becoming ever more popular. 5 Currently, the most common indication for applying OFA techniques is bariatric surgery.6,7 Other common indications include a history of postoperative nausea and vomiting (PONV), sleep apnoea, opioid dependence or chronic pain syndrome.6,8 In the context of OFA, multiple classes of pharmacological agents are used instead of the opioids to achieve sufficient analgesia and autonomic stability, such as α2-agonists (e.g., clonidine, dexmedetomidine (Dex)), local anesthetics (e.g., lidocaine (Lid)), N-methyl-D-aspartate (NMDA) antagonists (e.g. ketamine (Ket)), or combinations there of.6-8 Magnesium (Mg) is another important component of OFA; it acts as an NMDA antagonist (particularly in combination with Ket) but also has anti-inflammatory effects. 6
At our hospital, OFA is used several times a week for a variety of indications. We generally apply Dex, intravenous Lid, Ket, and Mg, in addition to regular analgesic drugs such as diclofenac, metamizol, and acetaminophene. For shorter procedures, Dex, Lid, Ket, and Mg are administered separately because of the better controllability, but for longer procedures, Dex, Lid, and Ket are mixed in a perfusor syringe and administered continuously. Other hospitals use yet other combinations of the different “OFA ingredients” in their clinical practice. The existing data on the stability of different combinations of these agents in different carrier solutions and under different storage conditions is very limited. However, these stability and compatibility issues are obviously crucial: if it turned out that the mixed solutions are both compatible with each other and stable over time, well-trained pharmaceutical personnel could produce the mixtures for the clinicians’ use ahead of time, both under optimized hygienic conditions and with increased accuracy regarding the concentrations of the individual agents.9-12
Several research groups have previously investigated the stability of single analytes of the aforementioned drugs in aqueous solution stored in polypropylene syringes. For example, ML Storms et al 13 proved stability of a 20 mg/ml Lid solution at ambient temperature and 4°C in polypropylene syringes for up to 90 days. Furthermore, stability of 4 µg/ml Dex was proved by Anderson et al 14 for at least 48 hours at 20°C to 25°C and 14 days at 5°C when stored in polypropylene syringes. Sarver et al 15 showed that there is no consistent decrease in the concentration of a Mg in Lactated Ringer’s solution or 0.9% saline at room temperature for a period of 3 months. Foy et al 16 proved physico chemical stability of a 1 mg/ml Ket solution in portable PCA systems for 28 days at room temperature. Furthermore, some studies have demonstrated the stability and compatibility of dual combinations of the aforementioned drugs in polypropylene syringes. Recently, Beiler et al 17 showed the physico chemical stability of a 20 mg/ml Lid and 2.5 mg/ml Ket in polypropylene syringes at 28°C over a time period of 48 hours. Along these lines, Houlihan et al 18 proved the physical compatibility and chemical stability of Mg and Lid in prefilled polypropylene syringes at 25°C and 40°C over 6 months.
According to our knowledge, however, there are no reports on the stability of ternary or quaternary mixtures of the aforementioned target analytes. The aim of this study was therefore to evaluate the chemical stability and physical compatibility of different combinations of Dex, Ket, Lid, and Mg diluted with 0.9% saline stored in polypropylene syringes under clinically used storage conditions for 56 days. For this purpose, an Ultra Performance Liquid Chromatography (UPLC) method was developed and validated and pH measurements as well as visual checks for color changes and particle formation were carried out.
Materials and Methods
Chemicals and Reagents
Formic acid (FA) and analytical reference standard of Lid and Ket were obtained from Sigma Aldrich (Buchs, Switzerland). Dex was obtained from Tokyo Chemical Industry (Tokyo, Japan). Acetonitrile (ACN) and water were purchased from Merck (Massachusetts, USA) and were of LC-MS grade. HPLC vials, screwcaps and inlets were purchased from BGB (Boeckten, Switzerland).
Hydrochloride acid, EDTA 0.1 M (Titrisol® Triplex III) and calcium carbonate were obtained from Merck (Massachusetts, USA), ammonia solution 25% from VWR Chemicals (Pennsylvania, USA) and ammonium chloride from Sigma Aldrich (Buchs, Switzerland). These substances were used for titration of magnesium sulfate.
For all experiments, Gilson pipettes and Gilson DIAMOND tips were used (Mettmenstetten, Switzerland). pH meter was calibrated using commercially available standards (ROTI® Calipure) from Carl Roth GmbH (Karlsruhe, Germany).
Chromatographic Conditions
All samples were analyzed on a Waters Acquity UPLC system equipped with an autosampler, a binary pump, and a column oven (Waters, Milford (MA), USA). Detection was done on a Thermo LTQ XL linear ion trap mass spectrometer (Thermo Scientific, San Jose (CA), USA). For all separations, water with 0.1% FA (solvent A) and ACN with 0.1% FA (solvent B) were used. Analytes were separated on Acquity BEH C18, 50 × 2.1 mm, 1.7 μm (Waters, Milford (MA), USA). Chromatographic conditions were as follows: flow set 0.5 ml/minute; 0 to 5.5 minute 95% A, 5.5 to 6.0 minute 60% A, 6.0 to 8.5 minute 5% A, 8.5 to 11.0 minute 95% A. Column oven temperature was set to 30°C. Autosampler was kept at 10°C and injection volume was 10 µl. Settings of the mass spectrometry (MS) detector are used as reported previously. 19 Analysis was performed in the positive ionization mode. The LTQ-XL was equipped with a heated ESI II source set to 150°C. Sheath gas 40 arbitrary units (AU); auxiliary gas 20 AU; source voltage 3.00 kV; ion transfer capillary 300°C; capillary voltage 31 V; tube lens voltage 80 V. Automatic gain control was set to 15 000 ions for full scan and 5000 for MSn. Collision induced dissociation (CID)-MSn experiments were performed on precursor ions selected for MS1. Using information dependent acquisition. MS1 was performed in full scan mode (m/z 100-500). MS2 and MS3 were performed in the IDA mode: 4 IDA MS2 experiments were performed on the 4 most intensive signal from MS1 and additionally 8 MS3 scan filters were chosen to record the most and second most ions from MS2.
Method Validation
The assay was validated according to international guidelines in respect of accuracy, precision and linearity.20,21 In short, 8 replicates (on 8 different days) were analyzed according to the procedure described above. Regression lines based a non-weighted or weighted (1/x2) least-squares regression model. Each calibrant (CAL 1-4) was back-calculated using daily regression concentrations and was compared to corresponding theoretical values. QC samples (QC low, QC high) were prepared and analyzed in duplicate on each of 8 days. Bias was defined as the percent deviation of the mean calculated concentration at each QC level from their respective nominal concentration. Bias should not exceed ± 10% of target concentration. Intra-day and inter-day imprecision were calculated as relative standard deviation (RSD) according to Peter et al. 20 During the validation period, the solutions of the quality controls and the calibrations were stored in the freezer at −18°C.
Sample Preparation
Preparation of calibration and quality control samples
Calibration solutions (CAL) and quality control solutions (QC) were prepared from independent stock solutions in solvent A. Concentration of stock solutions was 50 mg/ml Lid, 5 mg/ml Ket and 5 µg/ml Dex, respectively. Stock solutions were diluted with solvent A to obtain final concentrations.
Preparation of therapeutic samples
Three different therapeutic solutions were prepared using commercially available finished medicinal products (Lidocain® 2%, Streuli Pharma AG, Uznach, Switzerland; Ketamin® 10 mg/ml, Sintetica SA, Mendrisio, Switzerland; Dexdor® 200 µg/ml, Orion Pharma AG, Zug, Switzerland; Magnesiumsulfat® 20% and 50%, Bichsel, Interlaken, Switzerland). 0.9% saline (250 ml Ecoflac), 20 ml propylene syringes and corresponding plastic tip caps were obtained from B. Braun (Sempach, Switzerland). Substances were diluted with 0.9% saline to the final concentration. Each solution was distributed to 20 ml polypropylene syringes and closed with plastic tip caps. For each mixture, syringes (n = 5) were stored at ambient temperature (15°C-25°C) with or without light protection or in the refrigerator (2°C-8°C).
Sample preparation for chromatographic analysis
Before analysis, CAL, QC, and test samples were diluted with solvent in 2 different schemes. For determination of Dex, 20 µl of sample were diluted in 980 µl solvent A. Subsequently, 50 µl of this solution were further diluted with 950 µl of solvent A for the determination of Lid and Ket, respectively. The content of test solutions were determined on day 0, 2, 7, 14, 28, and 56. Content of mixture containing Lid-Ket-Dex-Mg was analyzed on day 148 instead of day 56. On the selected days, solutions were also examined for obvious changes in color and precipitation. The mixture was considered stable if the concentration was not less than 90% of the original concentration for all drugs. This is in accordance with the requirements of the Pharmacopoeia Helvetica (Ph. Helv., 11th edition, chapter 17.1.2.1): The content of the active pharmaceutical ingredient (API) in preparations must be within 90% to 110% of the declared content until the expiry date. 22
Titration and pH Determination
The content of magnesium was determined by complexometric titration as described in the European Pharmacopoeia (Ph. Eur., 10th edition, chapter 2.5.11) with minor modifications. 23 Titration was done in triplicate on day 0 and day 56 or day 148 respectively, using an automatic titrator (809 Titrando, Metrohm AG). Standard solutions were adjusted using 100 mg of calcium carbonate as primary standard. Test solutions (2.0 ml, 100 mg/ml) were diluted with water to 300 ml. After addition of 10 ml ammonium chloride buffer solution (pH 10.0) and 50 mg Eriochrome black T trituration, titration was performed with 0.1 M EDTA solution. Titration was monitored using an optrode (Methrom AG) set to λ = 610 nm. On indicated time points, pH was determined in each syringe using a calibrated pH-electrode (Schott Instruments).
Data Analysis
Peak integration was performed in Agilent EZChrom Elite (Version 3.3.2 SP2) software. GraphPad Prism 8.2 (GraphPad Software, San Diego, CA, USA) was used for regression analysis, 95% confidence interval (CI) determination and visualization. Outlier tests were performed according to Dixon’s Q test. 24
Results
Method Validation
In here, a new chromatographic method for the determination of Dex, Lid, and Ket is developed and validated. The described method is useful for the quantification of aforementioned analytes in pharmaceutical preparations using 0.9% saline as excipient. The retention time was 3.3 minute for Lid, 3.6 minute for Ket and about 4.5 minute for Dex. Since therapeutically applied concentrations of Dex, Lid, and Ket are up to 104 fold different (e.g. Dex-Lid), a 2-step dilution scheme was applied prior to analysis. In the selected concentration range, analytes concentrations showed a linear relationship to the detector response. Correlation coefficient (R2) for all analytes is above 0.96. For Lid lowest accuracy was found for QC high (97%), whereas lowest intra-day (RSDR) and inter-day impression (RSDT) at QC low and QC high level were detected (<5%). Ket showed highest accuracies at all levels and imprecision of less than 7%. Highest imprecision was detected for Dex (QC high). Intra-day (RSDR) and inter-day precision (RSDT) was 7.6% and 7.3%, respectively. Metrics of the method validation are shown in Table 1. Blank runs were performed after both the highest calibrator and QC high samples and were evaluated for detectable peaks. No carry-over was observed.
Method Validation Data.
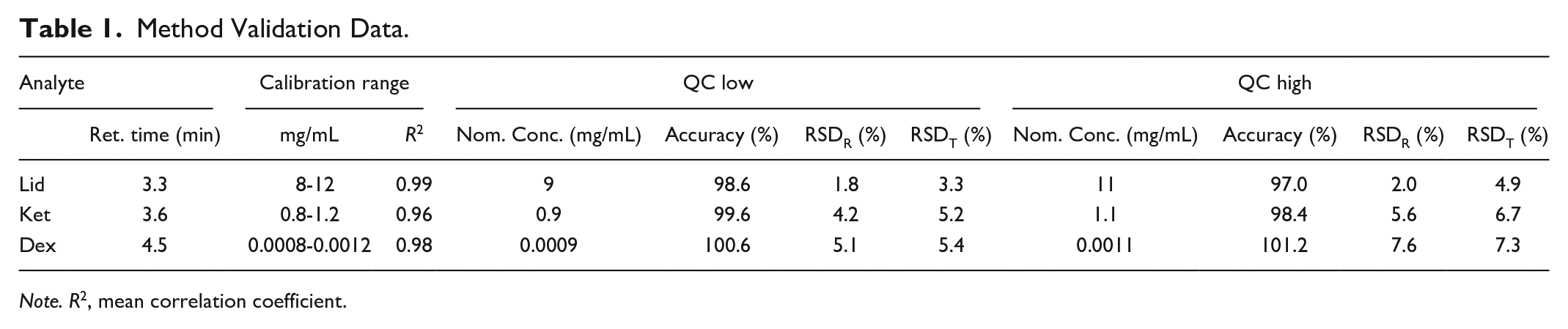
Note. R2, mean correlation coefficient.
Stability
pH measurement and visual examination
All preparations remained clear and colorless at visual inspection for the duration of the study. Additionally, no precipitation was detected. The pH-value remained unchanged over the whole measurement period among all mixtures and all 3 storage conditions (Table 2). Slight deviations were observed for Mix Lid-Mg (5.6 to 5.8). For all other combinations, pH was 5.8.
Stability Data for All Tested Combinations.
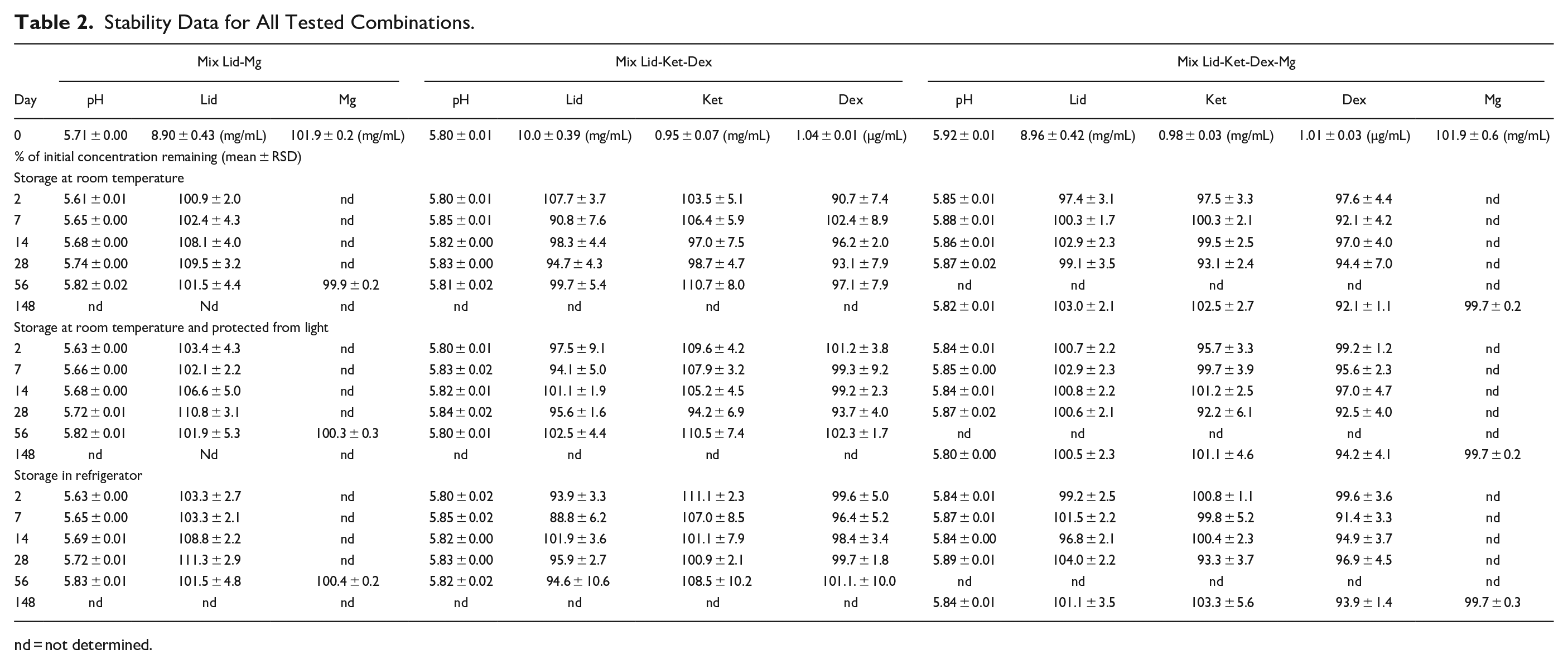
nd = not determined.
Stability of Active Pharmaceutical Ingredients
Detected concentrations of Dex, Ket, Lid, and Mg at indicated time points are summarized in Table 2. Data are given as a percentage of the initial concentration remaining. No continuous decrease in concentration can be observed for all combinations over 56 and 148 days respectively. Content of all analytes was higher than 90% at the final measurement day in all mixtures. Only in mixtures containing Lid, Ket, and Dex and stored in the refrigerator, the concentration of Lid fell below 90% of the initial content at day 2. Figure 1 shows the course of the measured concentrations of Lid in 0.9% saline stored in combination with Mg under different conditions over the whole storage period. 95% CI bands indicate the likely location of the true curve. Statistical analysis revealed that the slope of the regression lines is not significant different from zero. Therefore, no loss of Lid is expected. In Supplemental Figures S1 and S2, the course of the measured concentrations of Lid, Ket, and Dex without Mg and with Mg is visualized with the 95% CI bands. Regression lines indicate no loss of the API in the prepared admixtures. Only for Dex, a slightly decline in the regression line is observed. Obviously, decline is more prominent for mixtures stored at room temperature. However, the slope of the regression line is not significant from zero. Additionally, it needs to be mentioned that last measuring point of this mixture corresponds to day 148. For all storage conditions, final values are always higher than 90% of the initial reported values and pre-set goals are fulfilled.
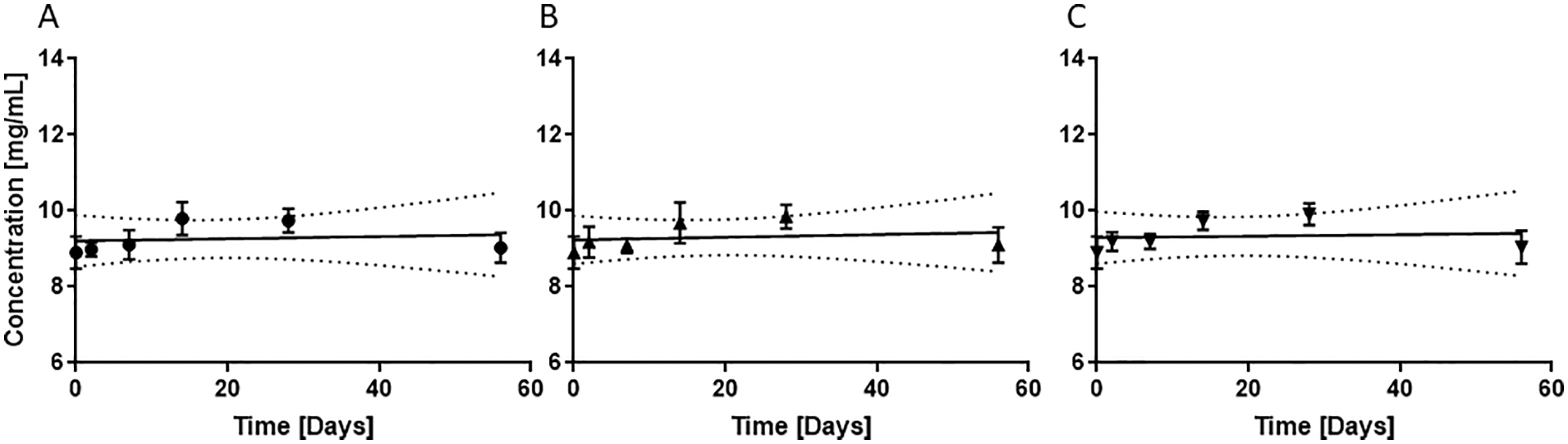
Stability of Lid in 0.9% saline in combination with Mg at different storage conditions: (A) room temperature, (B) room temperature and light protection, and (C) refrigerator (2°C-8°C); Dotted lines represent 95% CI bands.
Discussion
Knowledge about physicochemical stability of drugs in carrier solutions supports preparation of anesthetics in centralized hospital pharmacies. Data on stability of Dex, Ket, and Lid in 0.9% saline as single preparation for parenteral use is available in current literature. However, dual or triple combination with magnesium have not been investigated yet. Chromatographic method development was challenging, since there is a large difference regarding the therapeutic concentrations of the target substances. Ratio of Dex/Lid and Dex/Ket is 1:104 (10 mg/ml vs 1 µg/ml) and 1:103 (1 mg/ml vs 1 µg/ml), respectively. In order to detect all target analytes, a 2-step dilution procedure was developed and applied. For the determination of Dex, therapeutic solutions were diluted 50 times with mobile phase A. Subsequently, a 20-fold dilution with the same solvent was applied and was sufficient for Lid and Ket determination. The linear range of the analytical method was set according to clinical target concentration of selected API’s. Therefore, lower limit of detection and lower limit of quantification was not investigated systematically. The developed analytical procedure was validated in terms of accuracy, linearity and repeatability. QC low was set at the 90% level of starting concentration and defined as acceptance threshold for stability. Accuracy for all analytes were in the acceptable range at QC high and QC low level. However, inter-day imprecision was higher at QC high level for all analytes. One may speculate, if low concentration of Dex and the 2-step dilution process for Lid and Ket can explain these variations. In general, also higher relative standard deviations were recorded with LC/MS methods compared to LC-UV methods in other studies. 25
In general, our results confirm previous findings obtained for single analytes and compatibility with polypropylene syringes is given. The current study extends information on stability of dual, triple and quadruple combinations of selected analytes and the stability of admixtures after 56 or 148 days, respectively. The lower content of Lid at starting conditions for Mix Lid-Mg and Mix Lid-Ket-Dex-Mg is a result of using syringes for preparation of the therapeutic solutions. Physical stability of prepared mixtures was given, since no precipitation and color changes were observed. Also, pH was constant over time and in the acceptable range for parenteral preparations. 26 Additionally, influence of the bivalent cation magnesium was investigated in detail. It is know that magnesium can accelerate the degradation of analytes in tablet formulations. 27 Since titration experiments are time consuming and laborsome, Mg content was only analyzed at day 0 and on the last day of the study. Admixtures containing Mg (Lid-Mg, Lid-Ket-Dex-Mg) showed no lower concentration of any target analyte. Accordingly, storage in the refrigerator and at room temperature (with and without light protection) is appropriate. Nevertheless, storage in refrigerator would be more reasonable according to the microbial stability. It is noteworthy to say, that co-administration of other API’s to the aforementioned admixtures needs further evaluation particularly regarding compatibility and precipitation.
Limitations
In this study, there are also some limitations. Although several combination and conditions were investigated, only a limited number of therapeutic samples was analyzed (n = 5). However, the validated chromatographic methods provides a reliable starting point for further stability and compatibility studies.
Conclusion
In here, we presented a fast and accurate method for analysis of commonly used anesthetics in OSA and OFA settings. It has been shown, that selected admixtures can be prepared as ready to use syringes. The results of our study indicated no significant loss of compounds in different mixtures and storage conditions over a time period of 56 and 148 days respectively. All admixtures showed a constant pH value and the content remained higher than 90% of their initial concentration. None of the composition of the admixture and none of the investigated storage conditions showed a significant influence on the stability of the substances over the test period. These results will support anesthetists in daily practice, especially as the mixtures could be prepared in advance by well-trained pharmaceutical personal. Due to the worldwide use of OFA, the results are not only of local but also of international interest.
Supplemental Material
sj-pdf-1-hpx-10.1177_00185787211016336 – Supplemental material for Opioid-Free Anesthesia: Physico Chemical Stability Studies on Multi-Analyte Mixtures Intended for Use in Clinical Anesthesiology
Supplemental material, sj-pdf-1-hpx-10.1177_00185787211016336 for Opioid-Free Anesthesia: Physico Chemical Stability Studies on Multi-Analyte Mixtures Intended for Use in Clinical Anesthesiology by Larissa Schenkel, Irene Vogel Kahmann and Christian Steuer in Hospital Pharmacy
Footnotes
Acknowledgements
We would like to thank Danielle Lüthi for her assistance with the titration experiments. We are grateful to Dr. Kaspar Meyer for advises in writing the introduction
Authors’ Contributions
All mentioned authors significantly contributed to the current study. LS and CS prepared and analyzed selected mixtures. LS, IV, and CS designed the study protocol. LS, CS, and IV interpreted all obtained data. Writing the manuscript was equally done by LS and CS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
