Abstract
Exposure to natural environments has positive psychological effects. These effects have been explained from an evolutionary perspective, emphasizing humans’ innate preference for natural stimuli. We tested whether top-down cognitive processes influence the psychophysiological effects of environments. The source of an ambiguous sound was attributed to either nature (waterfall) or industry (factory). The results suggested that the participants’ subjective experiences were more pleasant and relaxed when the sound was attributed to nature than to industry. The influence of source attribution was also reflected in physiological measures that were free of subjective biases. The power of the brain’s lower alpha band activity was stronger in the nature scenario than in the industry condition. The individuals’ nature connectedness moderated the influence of source attribution on theta band power and electrodermal activity. The results support an evolutionary-constructivist perspective which assumes that the individual’s meanings and associations modulate the innate bottom-up effects of nature exposure.
Exposure to natural environments is linked, as compared with built settings, to several psychological benefits, such as stress reduction, enhancement of positive emotions and subjective well-being, and improved mental health (Berman et al., 2008; Bratman et al., 2015, 2021; Hartig et al., 2014; R. Kaplan & Kaplan, 1989; Ulrich, 1981). Exposure to natural settings can restore human attention and cognitive abilities, as measured using psychological tests (Berto, 2005; Ohly et al., 2016; Stevenson et al., 2018), as well as increase performance in working memory tasks (Bratman et al., 2015) and tasks requiring cognitive control (Berman et al., 2008, 2012; Hartig et al., 2003). The causal evidence for these favorable effects has most often been obtained by using visual stimuli, such as photographs and videos. The effects of auditory stimuli have been studied less, but recent reviews (Franco et al., 2017; Ratcliffe, 2021) suggest that stimuli such as bird song and sounds of water or wind, either alone or on conjunction with visual presentation, may improve mood, relaxation, and cognitive performance in a comparable way to visual stimuli. In the present study, we used an ambiguous auditory stimulus, whose source can be attributed to a natural or non-natural environment, to examine whether top-down cognitive processes contribute to the psychological and physiological effects of exposure to nature.
The psychological effects of exposure to nature have often been explained after considering an evolutionary perspective based on the Biophilia hypothesis (Kellert & Wilson, 1993; Wilson, 1984). Biophilia refers to our “innate tendency to focus on life and lifelike processes” (Wilson, 1984). According to this tendency, humans have an innate propensity to positively respond to unthreatening natural environments because it has proven useful for adaptive purposes during their biological evolution. The Stress Reduction Theory (SRT; Ulrich, 1983; Ulrich et al., 1991) emphasizes the physiological and emotional effects that humans experience when exposed to natural environments. Humans have spent most of their time in nature, which has provided favorable conditions for biological survival; therefore, humans may have developed an inherently positive perception of natural settings. Stress reduction theory proposes that qualitative features in natural settings can support psychophysiological stress recovery by reducing negative affect and stress and improving positive affect. The Attention Restoration Theory (ART; S. Kaplan, 1995; R. Kaplan & Kaplan, 1989) focuses on cognition and emphasizes the restorative effects that natural settings have on human attentional and cognitive fatigue. It proposes that attention to natural environments is relatively effortless and captured by involuntary bottom-up attention. Natural environments place few demands on top-down directed attention and thereby provide an opportunity to restore the cognitive system. These theories may complement one another (Hartig et al., 2003), as cognitive fatigue and stress are interrelated (Cohen & Spacapan, 1978; S. Kaplan, 1995; Ulrich et al., 1991). These leading theories assume that natural environments are characterized by physical features that are responsible for nature’s positive psychological and physiological effects. In the present study, we test an alternative or complementary view (Egner et al., 2020; Haga et al., 2016; Van Hedger et al., 2019) that assumes that the associations and meanings which humans attribute to the physical attributes of nature play a role in nature’s positive psychological and physiological effects.
Researchers have tried to understand the basic mechanisms underlying the restorative effects of nature by studying what makes nature “special” compared to other environments (Berto, 2005; Haga et al., 2016; Joye et al., 2016; Menzel & Reese, 2021). Some of the stimuli in natural settings may be more restorative than others because they have specific features that foster fascination (Berto, 2005; S. Kaplan, 1995). For example, fractal structures of natural stimuli may promote a cognitively less demanding processing of natural stimuli (Joye & Van den Berg, 2011; Joye et al., 2016). In studies on auditory natural stimuli, structural and perceptual properties have been shown to predict the restorative potential of bird sounds (Ratcliffe et al., 2020). In the context of natural sounds, pink noise is a common auditory signal in nature. Pink noise (sometimes also referred to as fractal noise) is comprised of a mixture of sound waves whose intensity decreases proportionally with frequency so that it has approximately equal energy per octave. The sounds of steady rain and ocean waves are examples of pink noise in nature, but pink noise may also resemble the sound of man-made machines (for example, an untuned radio channel), making it an ambiguous stimulus that can be interpreted in multiple ways and a good stimulus for studying top-down cognitive processes.
The empirical research guided by SRT and ART have focused predominantly on stimulus-driven, bottom-up processes when experiencing nature. However, human experience does not arise solely on the basis of bottom-up processes but also according to top-down factors, such as learned associations, memories, meanings, and attitudes that may influence how individuals interpret and experience the world. The constructivist perspective considers these deliberations, assuming that individuals make sense of their experiences and interactions with the world by creating conceptual schemas that organize ideas and experiences (Myers, 2012). The constructivist view stresses the physical features of nature less and instead focuses on the interpretation that the human brain makes of natural signals. According to the constructivist perspective, the restorative value of a given natural environments depends on the extent to which its features can be recognized and associated with the concept of nature (Van Hedger et al., 2019). According to this view, natural stimuli are not restorative per se due to a hardwired biological response. It is important how a person interprets the stimuli and associates them with prior situations and emotions. Van Hedger et al. (2019) found that nature sounds were aesthetically preferred over urban sounds only when they could be recognized and attributed to nature. In their constructivist framework, the features that differentiate between natural and urban stimuli are not inherently preferred, but conceptual knowledge about the environment (e.g., recognizing an environment as being natural) will organize how individuals aesthetically value specific features of the environment. A previously published study (Haga et al., 2016) addressed the contribution of individuals’ beliefs on perceived restoration in a study in which participants who believed they were listening to a sound from nature assessed the sound as being more restorative than participants who believed it originated from an industrial environment. The Conditioned Restoration Theory (CRT; Egner et al., 2020) can also be considered an example of constructivist view. It is based on the idea that natural settings may be restorative due to prior learning experiences. According to this theory, people are conditioned to associate nature with something relaxing because people enjoy relaxing leisure activities in forests, parks, beaches, and mountains (e.g., hiking, family trips, exercise, picnics, and general recreation). When presented with nature, conditioning triggers relaxation and the conditioned response may be generalized to other similar settings. The generalization of conditioned responses explains why simply watching photographs (Grassini et al., 2019; Ulrich, 1981) or videos (Ulrich et al., 1991) of nature may have relaxing psychological and physiological effects. In our view, the evolution-based theories focusing on the bottom-up processing of stimulus features and the constructive perspective are not necessarily mutually exclusive, but an evolutionary-constructivist view is also plausible. In such a view, one of the reasons that stimuli from natural settings are interpreted as restorative and associated with relaxation and pleasure may be because, during evolution, humans have developed an inherent tendency to respond positively to natural stimuli. However, the interpretation of these responses and experiences would be moderated by top-down factors, such as individuals’ learned associations and meanings related to nature. This view explicitly takes into account the possibility that individuals vary in their relationships with nature. The constructivist view is also consistent with major theories of emotions (Moors, 2009), assuming that cognition has an important role in modulating the affective and physiological responses to stimuli.
However, the causal contribution that top-down processes have on the positive psychological effects of nature, assumed in constructivist views, has little empirical support. Haga et al. (2016) conducted a relevant study. They showed that the restorative quality of stimuli may be related to the beliefs that the participants attributed to the stimuli. The participants were exposed to a sound consisting of pink noise with interspersed white noise. A group of participants were told that the sound originated from nature—from a waterfall—while another group was told that the sound originated from industrial machinery. Self-reported questionnaires investigating mental exhaustion showed that those participants who believed that the sound originated from a natural setting were more psychologically restored than those believing that the sound originated from machinery. The authors interpreted these results as supporting the idea that positive meanings and associations to nature may be, in part, responsible for nature-related restoration and not solely related to a hardwired response shaped by evolution and related to low-level qualities of the natural environments. However, the “objective” attention test did not show any effects for attribution to the source. Thus, the supportive evidence for the top-down processes in nature-related restoration effects is only based on subjective reports known to be vulnerable to demand-characteristics and hypotheses that the participants generated. It remains possible that the results are subject to “nature-positive” bias (Corazon et al., 2019).
Individuals vary in their affective connection and feeling of belonging to nature (Tam, 2013). Such nature connectedness (“connection with nature,” “nature in self”) can be considered a trait-like feature that remains stable over time (Mayer & Frantz, 2004), although relatively short-term state-like fluctuations in connectedness can occur after short exposures to nature (e.g., a 15-minute walk; Mayer et al., 2009). Love for nature and a feeling of oneness with nature correlate with past experiences with nature (Kals et al., 1999), and connectedness to nature is positively related with the frequency of time spent in nature and outdoors (Nisbet et al., 2009). The stronger the connectedness to nature, the more likely the individual will be spending time in nature and acquiring positive experiences and associations to nature. The positive effects that exposure to nature has on a person’s psychological well-being are, at least partially, mediated by connectedness to nature (Mayer et al., 2009; Pensini et al., 2016). Thus, the relationship between nature exposure and connectedness seems to be bidirectional: exposure to nature increases connectedness to nature and connectedness to nature creates cognitive structures (i.e., constructs) through which nature is interpreted in a positive way. From the constructivist view’s perspective, we hypothesize that the higher the nature connectedness, the stronger the top-down factors will moderate the positive effects of nature-related exposure.
The present investigation aimed to test whether stimulus-source attribution influences the psychological and physiological effects of nature by manipulating the participant’s belief of whether the soundscape originated from nature or an industrial environment. The stimulus was ambiguous, consisting of pink noise interspersed with white noise. In addition to subjective reports of relaxation and pleasantness, we measured more objective and bias-free physiological responses using electroencephalography (EEG) and by recording electrodermal activity (EDA). In the EEG recordings, we were specifically interested in the effects of the source attribution manipulation on the brain’s alpha band activity (8–13 Hz). The alpha activity increases in calm and relaxed states of mind and reflects the deactivation of cortical areas (Pfurtscheller et al., 1996) and inhibition of cognitive processing (Klimesch et al., 2007). Earlier studies have shown that exposure to natural scenes enhance alpha power (Chang et al., 2008; Elsadek et al., 2019; Grassini et al., 2019; Ulrich, 1981), especially in the lower alpha frequencies in central electrodes (Grassini et al., 2019). These studies suggest that the mind is in a more relaxed and calm state when exposed to natural settings, as compared with urban or built-up settings. On the other hand, the sympathetic nervous system controls EDA and it varies with the state of sweat glands in the skin, reflecting psychological or physiological arousal that is not under conscious control (Critchley, 2002). The EDA does not, however, directly show the valence of arousal. The EDA waveform can be divided into two components: the tonic skin conductance level (SCL) and skin conductance responses (SCRs; Benedek & Kaernbach, 2010). Skin conductive level refers to slowly varying tonic activity, whereas SCR is a quickly varying phasic activity observed as peaks above the SCL. Previous studies indicate that skin conductance varies as a function of whether participants are exposed to natural or non-natural settings (Alvarsson et al., 2010; Elsadek et al., 2019; Ulrich et al., 1991).
We hypothesized, in line with the constructivist view, that the positive psychological and physiological responses to a stimulus are not entirely attributable to the physical characteristics of the stimulus, but also to the meaning assigned to the stimulus and learning factors that shape perception of the stimulus. Specifically, we hypothesized that the same auditory ambiguous stimulus could have different influences depending on whether the source of the stimulus is attributed to nature (waterfall) or to an industrial environment (machinery in factory). We expected that, in the nature condition, the stimulus would be evaluated as being more relaxing and pleasant than in the industrial condition, and the participants would feel relaxed. We hypothesized that the power of the lower alpha band (8–10 Hz), which correlates with a relaxed and positive state of mind (Aftanas & Golocheikine, 2001; Grassini et al., 2019), would be higher in the nature condition as compared with the industrial condition. For EDA, we did not make any predictions concerning the direction of the effect, as skin conductance may reflect either positive or negative affective arousal (Gross & Levenson, 1997) and previous literature on the effects of nature exposure have reported both an increase (Browning et al., 2020) and decrease (Alvarsson et al., 2010; Hedblom et al., 2019) of skin conductance. In addition, because nature connectedness varies between individuals, we hypothesized that it would moderate the effects of the source-attribution manipulation in such a way that, the stronger the nature connection, the stronger the positive effects of nature attribution would be in subjective evaluations and the physiological measures.
Method
Participants
Thirty-three neurologically healthy students (six males; Mage = 24.1 years, SD = 7.2) volunteered from the introductory psychology courses at the University of Turku. The study was advertised on the department’s webpage and the participants were self-selected. They received course credits for their participation and were naïve to the hypotheses. The experiment was conducted in accordance with the Declaration of Helsinki and with each participant’s understanding and written consent. The Ethics Committee for Human Sciences at the University of Turku accepted the study.
The sample size required to produce statistically significant main effects in both the subjective and EEG measures was estimated based on Grassini et al. (2019), who used 32 participants and showed that exposure to images of nature significantly influenced brain activity in lower alpha bands. We also considered Haga et al. (2016), who used 30 participants/conditions in a behavioral source attribution study and found large effect sizes in self-evaluations. However, there was no relevant data for estimating the effect sizes in our EDA measures or in interactions between the physiological measures and nature connectedness. Thus, we aimed for 32 participants, but due to overbooking, we received 33 participants.
Materials
The sound stimulus consisted of continuous pink noise (180 seconds) mixed with 1 second bursts of white noise presented randomly every third, fourth, fifth, sixth, or seventh second (Haga et al., 2016). This stimulus is interpretable in multiple ways. Haga et al. (2016) found that almost half of the participants who were not told anything about the source of the sound interpreted it as coming from nature, whereas others attributed it to a source not related to nature. The sound was presented using Etymotic ER-3A earphones at approximately 55 dBA Leq. The experiment was run using E-prime 2 (Psychology Software Tools, Inc.).
At the end of the experiment, the participants filled in the Extended Inclusion of Nature in Self (EINS) scale (Martin & Czellar, 2016), which measures self-nature connectedness. The EINS consists of four pictorial items (overlap, size, distance, and centrality), each having seven alternatives, and the participants selected the option that best described their relationship with natural environments. Thus, the score could vary between 4 and 28, with the higher scores referring to strong nature connectedness. Cronbach’s alpha for EINS in the present data was 87.3.
Procedure
The experiment used a within-participants design. It consisted of three conditions: a 3 minutes baseline (silence without any stimulation), followed by two 3 minutes conditions during which the sound was presented. The sound was attributed to either a natural environment (waterfall) or to an industrial environment (a machine in a factory). While the experiment always started with the baseline condition, the order of the sound conditions was counterbalanced across the participants. The participants sat in a comfortable armchair in a dimly lit room. During the conditions, the participants were asked to relax and look at the empty screen placed approximately 150 cm in front of them. They were asked to avoid unnecessary eye movement, blinks, and body movement.
After setting up the EEG and EDA electrodes, the experiment began with the baseline (silence) condition. The participants were told that, in this condition, no sound was presented and they should try to relax during the 3 minutes silence. After the silence, they were asked to respond on a scale ranging from 1 (not at all) to 9 (extremely) to three questions that appeared on the screen one at a time: “How relaxed did you feel during the silence?”, “How pleasant was the silence?”, and “How relaxing was the silence?” There was a 2 minutes break after the responses. After the break, the experimenter told the participants that the first soundscape would follow. For half of the participants, the experimenter said that the sound came from a natural environment—from a waterfall—whereas, for the other half, the experimenter said that it came from an industrial environment—from a machine in a factory. The participants were asked to try to relax during the soundscape. Then, the stimulus (i.e., pink noise with short bursts of white noise) was presented for 3 minutes. After the stimulation, the participants were asked to respond (on a scale from 1 to 9) to three questions appearing on the screen: “How relaxed did you feel during the soundscape?”, “How pleasant was the soundscape?”, and “How relaxing was the soundscape?” There was a 2 minutes break after the responses. Then, the experimenter stated, “We are sorry, but we misled you a little. The sound did not come from a waterfall (or a factory). It instead came from an industrial environment, from a machine in a factory (or from a natural environment, from a waterfall). Now, as you know the real source of the soundscape, we are going to play it again. Try to relax during the soundscape.” Then, the stimulus was presented for 3 minutes. At the end of the stimulation, the participants were asked to respond to the same questions as after the first sound condition.
After the last soundscape condition, the participants filled out the EINS scale (Martin & Czellar, 2016). After that, the participants were debriefed about the purpose of the experiment and about the real nature of the sound.
EEG and EDA Recording and Processing
Electroencephalographic activity was measured using NeurOne 1.3.1.26 software and Tesla #MRI 2013011 and #MRI 2013012 amplifiers (Mega Electronics Ltd, Kuopio, Finland) with 500 Hz sampling rate. Twenty-one scalp electrodes (Fp1, Fp2, F3, F4, F7, F8, Fz, FCz, C3, C4, T7, T8, Tp9, Tp10, P3, P4, P7, P8, Pz, O1, and O2) were placed according to the 10–10 electrode system using a cap with sintered Ag/AgCl active electrodes (Easycap GmbH, Herrsching, Germany). The signal was referenced online to Cz and the ground electrode was placed on AFz (Grassini et al., 2019). Eye movements and blinks were recorded using two additional electrodes placed about 2 cm to the left of the left eye and about 2 cm below it.
The EEG data was processed offline using the EEGLAB (v. 2019_1; Delorme & Makeig, 2004), running under MATLAB (v. R2019b; The MathWorks, Inc., Natick, MA). Data was down-sampled to 256 Hz and high pass filtered at 1 Hz. Line noise (50 Hz) was removed using the CleanLine plugin (Mullen, 2012). Bad channels were interpolated using spherical interpolation and the data were then re-referenced to averaged mastoids. Due to the relatively small number of electrodes, we did not use an average reference like Grassini et al. (2019) did with their 64 scalp electrodes. The data was divided into epochs and submitted to extended Infomax independent component analysis (ICA). The IClabel plugin (Pion-Tonachini et al., 2019) was used to identify and eliminate ICs with eye movement and muscular artefacts. Occasionally, low battery charge level in the active electrodes produced artefacts that IClabel did not detect, and they were cleaned on a basis of visual inspection of the ICs. Then, the data was segmented into three 3 minutes conditions (baseline, waterfall, and factory). The power of the lower alpha band (8–10 Hz) in each of the conditions was analyzed with fast Fourier transform (FFT) performed on 2 seconds Hamming windows of 2 seconds with 50% overlap. Darbeliai plugin (Baranauskas, 2008) was used to calculate the absolute power. The data from two participants had to be removed from the analyses (many channels lacked a signal in one of them; the EEG signal was extremely weak for the other). As an exploratory analysis after seeing the results, the power in the higher alpha band (10–13 Hz) and theta band (4–8 Hz) was also analyzed.
The EDA was recorded using Biopac MP150 (Biopac Systems, Inc., Santa Barbara, CA) and AcqKnowledge 5.0 software, with a 2,000 Hz sampling rate. The electrodes were placed on the top surfaces of the left forefinger and middle finger. The data was processed using Ledalab 3.4.9 software (Benedek & Kaernbach, 2010), running under MATLAB (v. R2019b; The MathWorks, Inc., Natick, MA). The data was down-sampled to 10 Hz, then Continuous Decomposition Analysis (CDA) was performed on each 3 minutes condition (baseline, waterfall, and factory) with two sets of initial values considered in optimization. The CDA separated phasic (skin conductance response [SCR]) and continuous tonic (skin conductance level [SCL]) components from the EDA. A significant phasic peak was detected if a local maximum had a difference of ≥0.01 μS from its preceding or following local minimum (Benedek & Kaernbach, 2010). The resulting variables were the number of skin conductance responses (nSCR) and the amplitude (μS) of SCL, computed separately for each of the three 3 minutes conditions.
Statistical Analyses
The analysis scripts and data sets are available at OSF.io (https://osf.io/mfbjv/?view_only=651e0b84bd864568b3771c5f0d7ed0bd). The results were analyzed with R statistical software 3.5.0 (R Core Team, 2018), using packages lme4 (Bates et al., 2015) and Psycho 0.4.0 (Makowski, 2018). Packages ggplot2 (Wickham, 2016), and sjPlot 2.4.1 (Lüdecke, 2019) were used to illustrate the results.
Separate planned analyses were performed on the three subjective variables, the absolute power (µV2) of the lower and higher alpha band (average of the activity in central electrodes C3 and C4), and the EDA variables (nSCR and SCL). The p-values in the planned analyses were Bonferroni-corrected by multiplying the p-values with the number of analyses for each measurement type (subjective, EEG, and EDA). In addition, after inspecting the power spectrum, an exploratory analysis on the theta band was performed. The power in the EEG frequency bands and SCL were log transformed for statistical analyses as the residuals in the corresponding models were not normally distributed; the models with log transformed variables had no problem with the normality of distributions.
The analyses for each dependent variable (DV) were conducted with linear mixed-effect models (Bates et al., 2015). First, we compared the fit of two models, one with Condition (baseline, waterfall, and factory) as the fixed effect (DV − Condition + (1|participant)) and one in which the EINS score and its interactions were additional fixed effects (DV − Condition × EINS+(1|participant)). The models included a separate intercept for each participant as the random-effect. Condition was coded as a factor and the EINS score was a continuous variable centered on the mean. The uncentered mean of the EINS score was 20.2 points (SD = 3.9, minimum = 12, maximum = 27, 95% CI [18.8, 21.6]), and the scores were normally distributed (Shapiro-Wilk test, W = 0.97476, p = .622). We defined planned intervention contrasts for the difference between the waterfall condition and the factory condition and between the waterfall condition and baseline with the hypr package (Rabe et al., 2020). The model with smaller Akaike information criterion (AIC), an estimator of the relative quality of statistical models, was selected for the analysis. For the subjective DVs, the models with only Condition as the fixed effect were the best, whereas for the EEG and EDA variables, the models with Condition and EINS and their interactions as fixed effects were the best.
Finally, to rule out the effects of the order of presenting the waterfall and factory conditions, we compared for each DV the model with the lowest AIC obtained in the previous phase with the model which included the order and its interactions as fixed effects. The model without the order effects had the lowest AIC value for all subjective and EDA variables. The same was true for EEG variables, with an exception that, for the lower alpha band, the model including the order effects had the lowest AIC value. However, because in this model, none of the order effects were statistically significant (p’s > .05), we shall report the results without order effects, consistent with the other models.
For each fixed effect, we report the minimum detectable effect size (MDES) for 80% power with .05 alpha level. The MDES was determined with 100 simulations using the simr package (Green & MacLeod, 2016) in R.
Results
Subjective Ratings
For all subjective variables (Figure 1), the model without EINS score fit the data better than the model with it. Thus, the self-nature connection did not predict subjective ratings in different conditions. The effects of source attribution on the subjective rating variables were tested with the sound condition (baseline, waterfall, and factory) as the only fixed effect. We hypothesized that, if top-down effects play a role in the psychological effects of nature exposure, the participants should rate themselves as more relaxed and the sound as more pleasant and relaxing in the waterfall condition than in the factory condition.
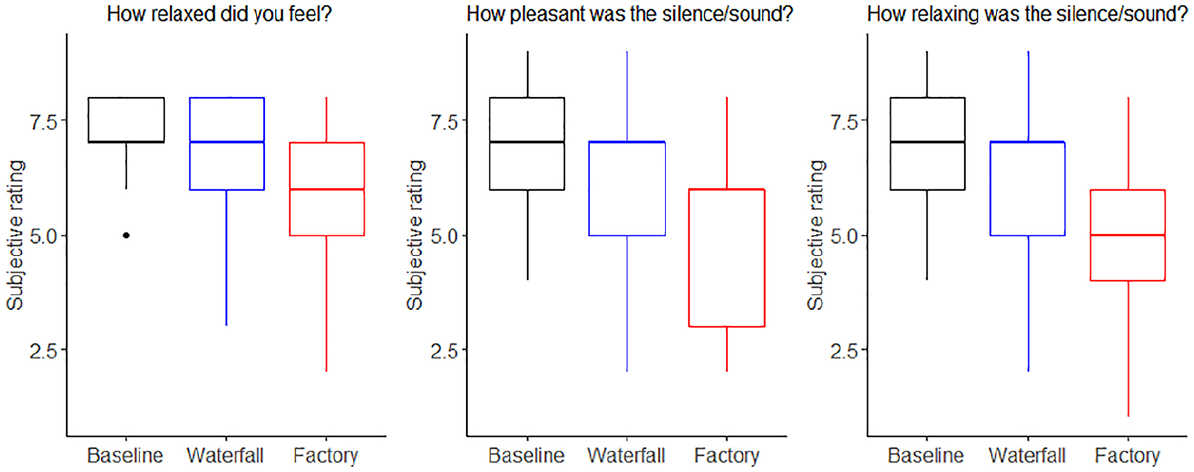
Subjective ratings on scale from 1 (not at all) to 9 (extremely) in baseline, waterfall, and factory conditions.
The model on relaxation did not detect a statistically significant difference between the waterfall and baseline conditions (β = −.45, SE = 0.24, 95% CI [−0.92, 0.0068], t(64) = −1.93, Bonferroni-corrected p = .174, MDEF = −0.65). However, the participants felt more relaxed when the sound was attributed to waterfall than to factory (β = .79, SE = 0.24, 95% CI [0.33, 1.25], t(64) = 3.34, Bonferroni-corrected p = .003, MDEF = 0.68).
The model on pleasantness ratings did not detect a statistically significant difference between the waterfall and baseline conditions (β = −.64, SE = 0.34, 95% CI [−1.31, 0.034], t(64) = −1.86, Bonferroni-corrected p = .204, MDES = −0.92). However, the waterfall condition was rated as more pleasant than the factory sound (β = 1.12, SE = 0.34, 95% CI [0.45, 1.79], t(64) = 3.28, Bonferroni-corrected p = .006, MDES = 0.99).
Finally, the model predicting how relaxing the participants considered the sound suggested that the waterfall condition was considered less relaxing than the silence (i.e., baseline) condition (β = −.79, SE = 0.31, 95% CI [−1.40, −0.18], t(64) = −2.52, Bonferroni-corrected p = .042, MDES = −0.85), but more relaxing than the factory machine condition (β = 1.15, SE = 0.31, 95% CI [0.54, 1.76], t(64) = 3.68, Bonferroni-corrected p = .001; MDES = 0.90).
In summary, the subjective ratings suggest that attributing the source of the sound to a waterfall made the participants feel more relaxed compared to attributing it to a factory. The sound was also considered more pleasant and relaxing in the waterfall condition than in the factory condition.
Alpha Power
In the analysis of EEG, we focused on the alpha band, in which the attribution of the sound to a waterfall was expected to increase the spectral power, specifically in the lower alpha frequencies (8–10 Hz), reflecting a relaxed and calm state of mind. The distribution of power across frequencies in central electrodes and the scalp distributions of alpha power in the lower (8–10 Hz) and higher (10–13 Hz) bands are presented in Figure 2. As we expected, the upper panel of Figure 2 shows that the changes of power due to manipulating the source attribution occurred specifically in the alpha band (around 10 Hz) and most clearly over the central areas of the scalp (lower panel in Figure 2).
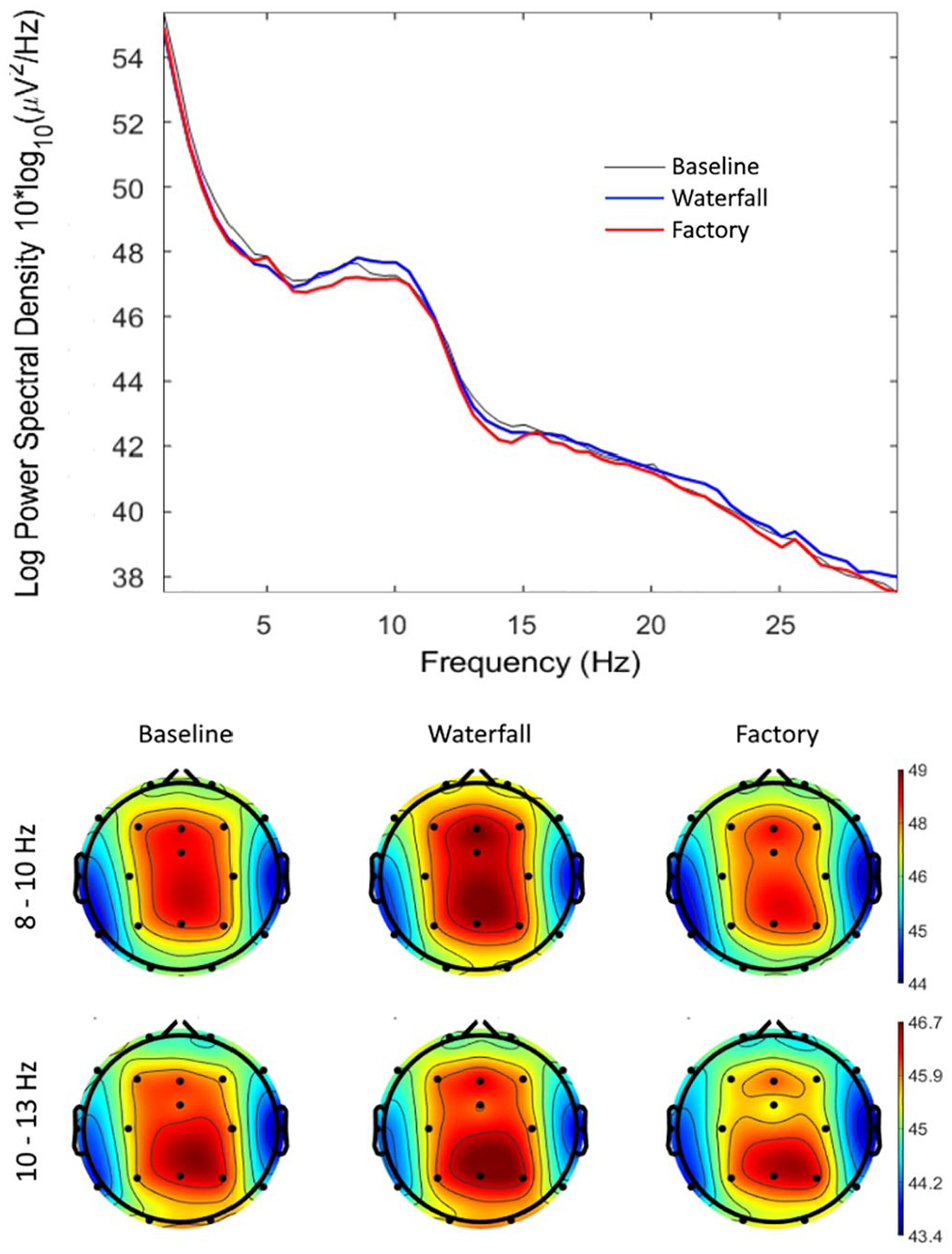
Upper panel: The distribution of power into frequency components in central electrodes (average of c3 and c4). Lower panel: The scalp distribution of power in the lower (8–10 Hz) and higher (10–13 Hz) alpha bands.
Modelling of the spectral power (log(µV2)) in the lower alpha band (8–10 Hz) in central electrodes showed that the model with EINS and its interactions with Condition had a lower AIC value than the model without them. Thus, the mixed-effect model on the lower band was performed with Condition and EINS with its interactions as fixed effects. In the higher alpha band, the model without EINS fit better; therefore, Condition was the only fixed effect in the analysis of the higher alpha band.
The model predicting power in the lower alpha band (8–10 Hz; Figure 3) showed that it was higher in the waterfall condition than in the factory condition (β = 0.14, SE = 0.055, 95% CI [0.033, 0.25], t(58) = 2.53, Bonferroni-corrected p = .028, MDES = 0.14), but the power in the waterfall condition did not differ from that in the baseline (β = .043, SE = 0.055, 95% CI [−0.063, 0.15], t(58) = 0.78, Bonferroni-corrected p = .876, MDES = 0.16). The effect of EINS was not statistically significant (β = −.0042, SE = 0.046, 95% CI [−0.094, 0.086], t(31) = −0.092, Bonferroni-corrected p = 1.00, MDES = 0.14), which means that, in the baseline condition (i.e., silence), the activity did not depend on EINS. However, the interaction between EINS and the waterfall-baseline difference was statistically significant (β = .038, SE = 0.014, 95% CI [0.011, 0.066], t(58) = 2.67, Bonferroni-corrected p = .020, MDES = 0.04), showing that the higher the EINS score, the higher power the waterfall condition elicited as compared with the baseline. The interaction between the EINS and waterfall-factory difference occurred in the same direction, but did not reach a statistical significance (β = .026, SE = 0.014, 95% CI [−0.001, 0.054], t(58) = 1.84, Bonferroni-corrected p = .142, MDES = 0.04). Supplemental Figure S1 presents the relationship between EINS and the different scores between the conditions as scattergrams with a regression line.
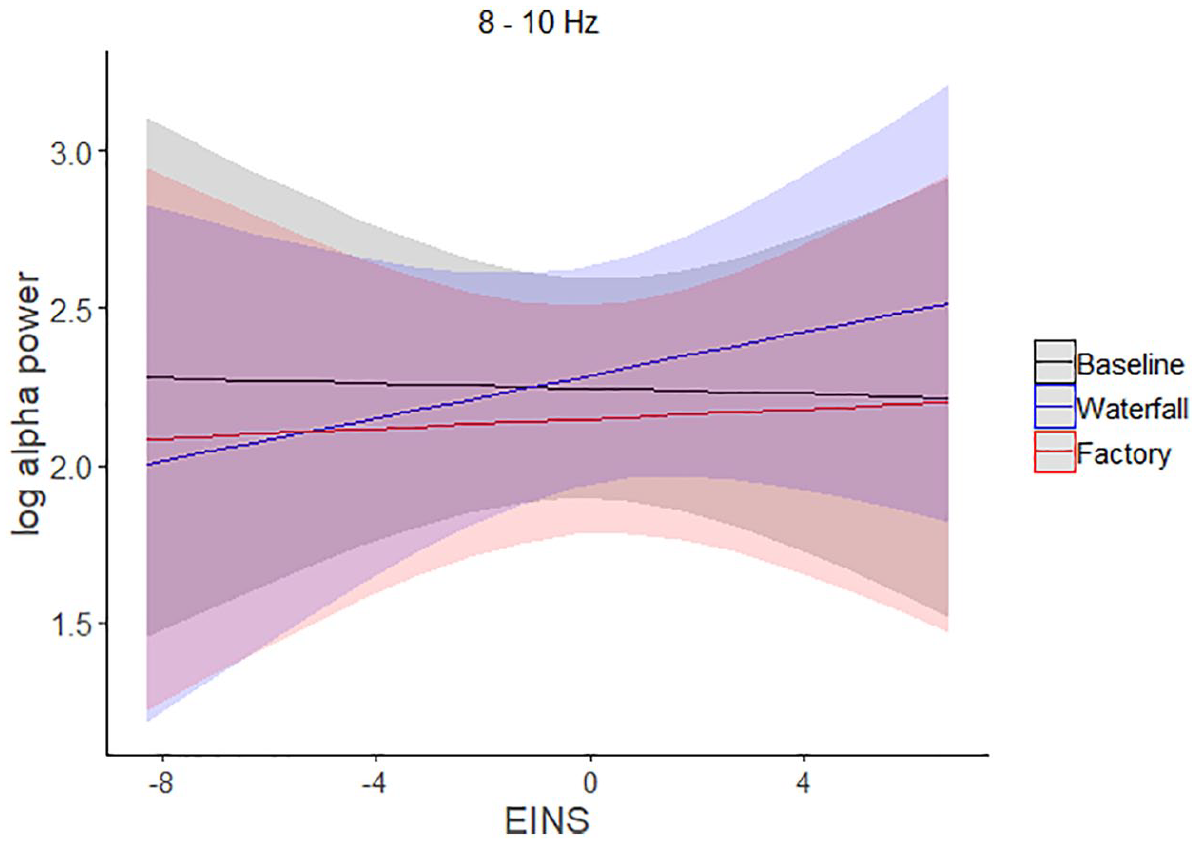
The modelled lower alpha band (8–10 Hz) power (LOG(µV2)) in the baseline, waterfall, and factory conditions, and the interactions between the conditions and the self-nature connection (EINS).
The model predicting power in the higher alpha band (10–13 Hz), with Condition as a fixed effect, did not detect differences between the waterfall and baseline conditions (β = .025, SE = 0.046, 95% CI [−0.065, 0.11], t(60) = 0.54, Bonferroni-corrected p = 1.00, MDES = 0.13) or between waterfall and factory conditions (β = .073, SE = 0.046, 95% CI [−0.017, 0.16], t(60) = 1.60, Bonferroni-corrected p = .232, MDES = 0.13). However, a visual inspection of Figure 2 (lower panel) suggests that the higher alpha band may have maximal activity over the parietal lobes rather than over the central areas of the brain. As a result, we performed an exploratory analysis on the higher alpha power in the parietal electrode Pz, but it did not reveal a significant difference between the waterfall and baseline conditions (β = .070, SE = 0.090, 95% CI [−0.11, 0.25], t(60) = 0.77, p = .442, MDES = 0.25) or between the waterfall and factory conditions (β = .063, SE = 0.090, 95% CI [−0.11, 0.24], t(60) = 0.70, p = .485, MDES = 0.24).
In summary, the power in the lower alpha band depended on the source attribution manipulation. It was higher when the sound was attributed to a waterfall than when attributed to a factory sound. Nature connectedness, measured with EINS, related to brain activity by moderating (i.e., interacting with) the difference between waterfall and baseline conditions in the lower alpha band: the higher the nature connection, the larger the difference between the waterfall and baseline conditions (the relation of EINS to the difference between waterfall condition and factory condition in the lower alpha band was in the same direction but did not reach statistical significance).
Theta Power
The power spectrum (Figure 2, upper panel) suggests that the soundscape condition may also have an effect on the theta band activity (4–8 Hz). We performed an explorative analysis to test whether the effects of Condition and EINS are present in the theta frequency using a statistical model identical to those in the analyses of alpha activity. The model predicting theta power in the central electrodes with both Condition and EINS as fixed effects fitted the data better than the model without EINS. The effect of EINS (β = −.012, SE = 0.027, 95% CI [−0.06, 0.04], t(33) = −0.47, p = .641, MDES = −0.075) and the differences between the waterfall and baseline conditions (β = −.035, SE = 0.047, 95% CI [−0.12, 0.06], t(58) = −0.74, p = .464, MDES = −0.13) and between the waterfall and factory conditions (β = .046, SE = 0.047, 95% CI [−0.04, 0.14], t(58) = 0.98, p = .332, MDES = −0.13) were not statistically significant. However, EINS interacted with the difference between the waterfall and baseline conditions (β = .042, SE = 0.012, 95% CI [0.02, 0.07], t(58) = 3.45, p = .001, MDES = 0.035) and with the difference between the waterfall and factory conditions (β = .033, SE = 0.012, 95% CI [0.01, 0.06], t(58) = 2.73, p = .008, MDES = 0.035). As the scalp distribution of theta power (Figure 4) seems to be more frontal than alpha, we repeated the models on theta power in the frontal electrodes (F3, Fz, and F4). The results were like those reported above: the interactions of EINS with the contrasts between conditions were statistically significant (p = .026 and .028). These interactions suggest that, when compared to those in the baseline and factory conditions, the higher the EINS score, the stronger the theta power in the waterfall condition.
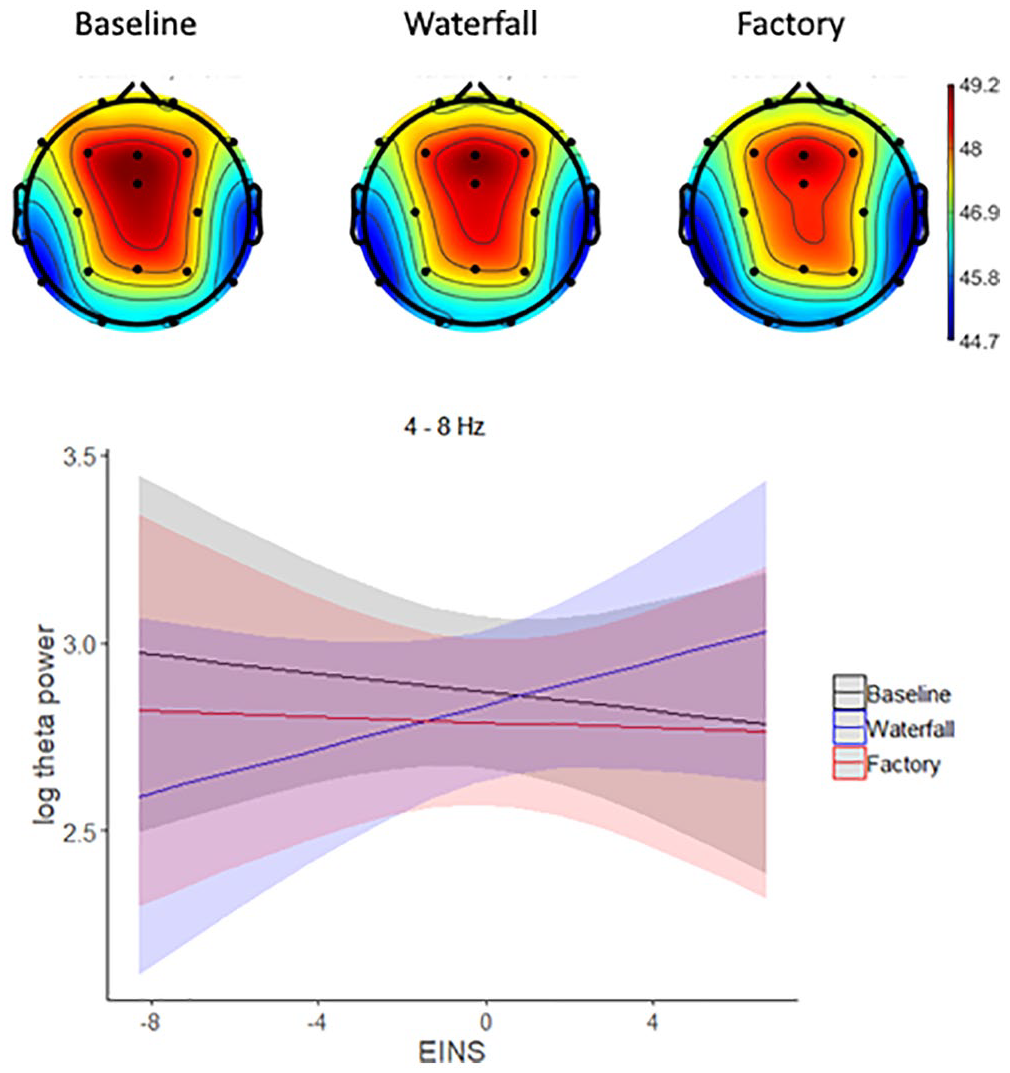
Upper panel: The scalp distribution of power in the theta band (4–8 Hz). Lower panel: The modelled theta band (8–10 Hz) power (log(µV2)) in the baseline, waterfall, and factory conditions, and the interactions between the conditions and the self-nature connection (EINS).
In summary, nature connectedness moderated (EINS) the influence that the source attribution manipulation had on theta activity. The higher the nature connectedness, the more powerful the theta activity in the waterfall condition as compared with that in the other conditions.
Electrodermal Activity (EDA)
In the analysis of EDA, the models with the EINS and its interactions as fixed effects showed the lowest AIC values, suggesting that they fitted the data better than the models without them. The nSCR and SCL in each condition and the modeled results are shown in Figure 5.
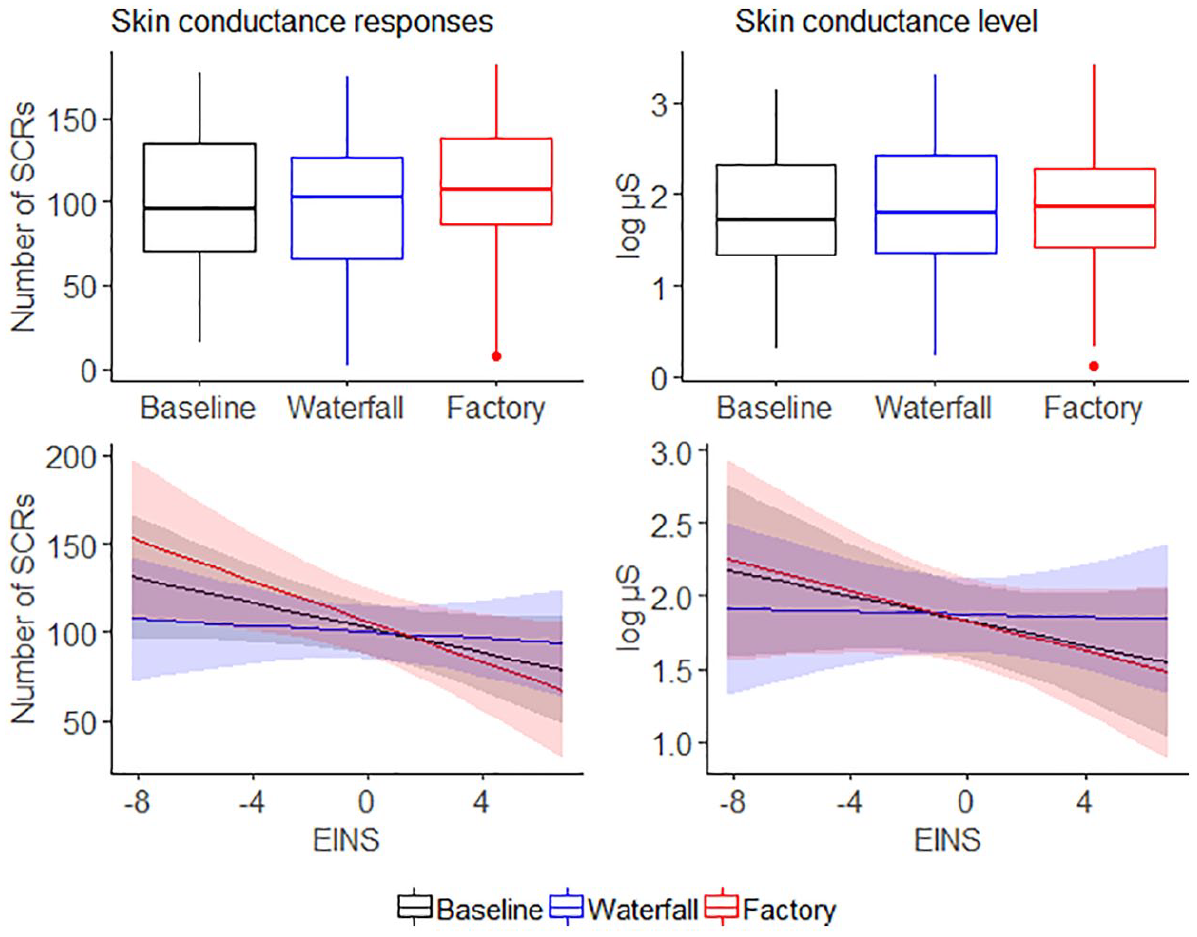
The upper panels show the observed results for skin conductance responses (nSCR) and skin conductance levels (SCL) in the baseline, waterfall, and factory conditions. The lower panels illustrate modeled results with the interactions between the conditions and the self-nature connection (EINS).
The model predicting the number of SCRs did not detect any statistically significant difference between the waterfall and baseline conditions (β = −2.18, SE = 6.05, 95% CI [−13.84, 9.48], t(62) = −0.36, Bonferroni-corrected p = 1.000, MDES = −17) or between the waterfall and factory conditions (β = −5.82, SE = 6.05, 95% CI [−17.48, 5.84], t(62) = −0.96, Bonferroni-corrected p = .680, MDES = −18; Figure 5, upper-left panel). Nature connection did not predict nSCR in the baseline (silence) condition (β = −3.53, SE = 1.97, 95% CI [−7.34, 0.29], t(48) = −1.79, Bonferroni-corrected p = .160, MDES = 5.7). However, the interaction between EINS and the waterfall-factory difference was statistically significant (β = 4.83, SE = 1.57, 95% CI [1.81, 7.85], t(62) = 3.08, Bonferroni-corrected p = .006, MDES = 4.4), suggesting that the higher the EINS score, the more SCRs were elicited in response to the waterfall condition as compared with the factory condition (Figure 5, lower-left panel). Also, the interaction between EINS and the waterfall-baseline difference showed a similar pattern but failed to reach statistical significance (β = 2.64, SE = 1.57, 95% CI [−0.38, 5.66], t(62) = 1.69, Bonferroni-corrected p = .194, MDES = 4.5).
The overall model predicting log-transformed tonic SCL did not detect differences in SCL between waterfall and baseline conditions (β = .046, SE = 0.076, 95% CI [−0.10, 0.19], t(62) = 0.60, Bonferroni-corrected p = 1.00, MDES = 0.20) or between waterfall and factory conditions (β = .047, SE = 0.076, 95% CI [−0.10, 0.19], t(62) = 0.61, Bonferroni-corrected p = 1.00, MDES = 0.21; Figure 4, upper-right panel). The effect of EINS in the baseline condition was not statistically significant (β = −.042, SE = 0.033, 95% CI [−0.11, 0.023], t(39) = −1.27, Bonferroni-corrected p = .424, MDES = −0.09). As in the model on the number of SCRs, the interaction between EINS and the waterfall-factory difference in SCL was statistically significant (β = .047, SE = 0.020, 95% CI [0.01, 0.09], t(62) = 2.38, Bonferroni-corrected p = .042, MDES = 0.05), suggesting that the higher the EINS score, the higher SCL was elicited in response to the waterfall condition as compared with the factory condition. Also, the difference between the waterfall and baseline conditions in SCL showed a similar pattern, but the effect was not statistically significant (β = .038, SE = 0.020, 95% CI [−0.00015, 0.076], t(62) = 1.92, Bonferroni-corrected p = .120, MDES = 0.05).
Skin conductance measures can relate to either positive or negative affective arousal. To explore whether the SCRs and SCL were related to positive or negative arousal, linear mixed effect models with Condition and the centered average of the three subjective variables as fixed effects were used to predict the number of SCRs and SCL. The results showed that subjective ratings interacted with the waterfall-baseline contrast in the analysis of SCRs (β = 12.22, SE = 5.65, 95% CI [1.23, 23.13], t(67) = 2.16, p = .034, MDES = 12) and in the analysis of SCL (β = .16, SE = 0.071, 95% CI [0.03, 0.30], t(64) = 2.28, p = .026, MDES = 0.15). The higher the subjective ratings, the more SCRs and the higher SCL there were in the waterfall condition as compared to the baseline condition. These results imply that a high level of skin conductance was associated with positive subjective evaluations of the waterfall condition.
In summary, EDA was moderated by the nature connection in such way that the higher the nature connection, the greater the number of phasic SCRs and the higher the tonic SCL in the waterfall condition, as compared with the factory condition. High SCL appeared to related to high subjective ratings (the average of the three measures displayed in Figure 1).
The Relationship Between EDA and EEG
Finally, we explored whether the EDA variables (nSCR and SCL) were related to spectral power in those of the frequency bands (lower alpha and theta) which responded to the source attribution manipulation. Since the skin conductance effects depended on nature connectedness, the EINS score was included in the analyses. The linear mixed effects model on the lower alpha band with the number of SCRs, EINS score, and their interaction as fixed effects showed an interaction between the number of SCRs and EINS (β = .0009, SE = 0.0003, t(67.34) = 2.89, 95% CI [0.0003, 0.0014], p = .005, MDES = 0.00083). Also, in the model involving SCL, EINS, and their interaction as fixed effects, the interaction between SCL and EINS was significant (β = .047, SE = 0.021, t(72.16) = 2.29, 95% CI [0.007, 0.088], p = .025, MDES = 0.06). These results suggest that the increase of skin conductance as a function of connectedness to nature was related to an increased lower alpha band power. Similar analyses on theta power did not reveal any statistically significant effects (p > .38), suggesting that theta activity was not related to skin conductance.
Discussion
Recently published scientific literature has proposed that the beneficial psychological effects of nature are affected by top-down factors, such as the meanings and associations associated with the environment (Egner et al., 2020; Haga et al., 2016; Van Hedger et al., 2019). The present study tested such constructivist view and examined whether the contribution of top-down factors would receive support not only from subjective measurements, as in previous research (Haga et al., 2016; Van Hedger et al., 2019), but also from objective and bias-free physiological measurements of the brain’s alpha power and the activation of the sympathetic nervous system, when the source of an ambiguous sound was attributed either to nature (waterwall) or an industrial environment (machinery in a factory). We hypothesized that the stimulus would be considered more relaxing and pleasant in the nature condition than in the industrial condition, and the participants would consider themselves more relaxed in the nature condition than in the industrial condition. This relaxation would be reflected in the brain’s alpha activity. The results in the subjective evaluations and alpha activity were consistent with the hypotheses, showing the expected difference between the conditions, but not in the EDA (nSCR and SCL). In addition, we hypothesized that a connection to nature would strengthen the difference between the nature and industrial conditions in the subjective and physiological measures. The overall pattern of results supported this hypothesis, with the exception that subjective evaluations were not statistically significantly related to nature connectedness. Next, we discuss these findings in more detail.
The subjective ratings given after the 3 minutes exposures to the ambiguous sound suggested that the sound was considered more pleasant and relaxing, and the participants felt more relaxed when the sound was attributed to nature compared to an industrial environment. These findings conceptually replicate the results of Haga et al. (2016) and support the constructivist framework supposing that top-down processing moderates the psychological effects of environmental stimuli. However, nature connectedness did not predict the differences in subjective ratings between nature versus industrial attribution conditions. The persons with a higher than average score in nature connection did not give more nature-positive ratings than other participants. These results can be interpreted in two ways. Either the subjective ratings were bias-free and the participants really experienced the nature-attribution condition more positively than the industrial condition, or the subjective ratings in general were subject to a nature-positive bias that was independent of nature connectedness. The bias-free physiological results from EEG and EDA recordings suggest that the differences between nature and industrial source-attribution conditions cannot be explained solely with a nature-positive bias.
The constructivist (top-down) view predicts that attributing the source of the ambiguous sound to nature would reflect as an increase of spectral power in the lower alpha band because such activity is connected to suppressing sensory and attentional mechanisms (Foxe & Snyder, 2011; Klimesch et al., 2007; Pfurtscheller et al., 1996). Our results confirmed this prediction and showed that, in the same way as observed when watching nature photographs (Grassini et al., 2019), the alpha activity in the lower band increased during the nature-attribution condition. In addition, the increase in the lower alpha activity varied individually. The stronger that the participants were connected to nature, the stronger their alpha band activity was when the source of the stimulus was attributed to nature, as compared with an industrial environment. These results support the hypothesis that top-down factors contribute to the positive effects of nature exposure.
Visual inspection of brainwaves revealed that the effect of environmental attribution was not restricted to the low-alpha band wave. Therefore, we performed an exploratory analysis for the frequency band below (theta band, 4–8 Hz) the lower alpha band and a planned analysis for the band above (higher alpha-band, 10–13 Hz) the lower alpha band in order to verify whether the influence of the source attribution was restricted to the lower alpha band. In the higher alpha band, the effects did not reach statistical significance. However, in the theta band, the effect of the source attribution depended on connectedness to nature. The higher the individual’s connectedness to nature, the higher theta power was observed when the sound was attributed to nature, as compared with the industrial condition or baseline (i.e., silence). Previous studies have shown that mindfulness (Lomas et al., 2015) and meditation (Aftanas & Golocheikine, 2001; Lagopoulos et al., 2009) increase alpha and theta powers, suggesting they are related to relaxed and emotionally positive states. Specifically related to nature exposure, Sahni and Kumar (2020) reported that viewing nature videos increased both alpha and theta powers, suggesting a relaxed yet alert state of mind. Thus, in showing enhancement of theta and alpha activity in nature-related source attribution conditions, especially in persons highly connected to nature, our study suggests that mere attribution of the sound to nature and listening to the sound seems to produce a state of relaxed alertness. Such state may, based on present neurophysiological measures, resemble this alertness during mindfulness or meditation.
Activity of the sympathetic nervous system was operationalized with two variables that measure different, but complementary, aspects of EDA. The amplitude of SCL indicates general, tonic, long-lasting sympathetic nervous system activity, while SCR reflects phasic activity that shows the number of activated nerve impulses regulating sweat secretion (Benedek & Kaernbach, 2010). We did not find any main effects for the source attribution in either measure. However, the EDA depended on the connection with nature. The stronger the nature connectedness, the more phasic activation peaks occurred and the larger the amplitude of tonic activity in response to the nature condition, as compared to the responses in the industrial condition. The finding that attributing the sound to nature was related to an increased difference between the conditions in skin conductance may look strange at first. However, skin conductance measures can be linked to arousal of either positive or negative affect and our exploratory analyses indicated that the increased number of SCRs and amplitude of tonic activity were connected to positive affective arousal and enhanced EEG power in the lower alpha band. The positive affective arousal in response to stimulation interpreted as pleasant is consistent with SRT (Ulrich, 1991), which assumes that unthreatening nature automatically evokes positive affect and correspondingly reduces negative affect and stress. As in the present study, Browning et al. (2020) found that exposure to nature increased skin conductance and was associated with positive self-assessed emotional states. Their study resembled the present study in that it did not include any stressor task prior to exposure, and the task during the exposure was to “try to relax.” Thus, unlike studies that have focused on restoration from stress and showed reduced skin conductance when listening to natural sounds (Alvarsson et al., 2010; Hedblom et al., 2019), the stress level was not particularly high before exposure in our study or in Browning et al. (2020).
The present results, along with those of Haga et al. (2016) and Van Hedger et al. (2019), suggest that physical differences between natural environments and non-natural environments are not sufficient to explain the positive effects that nature exposure has on well-being, but the meanings attributed to the environment contribute to the effects. In addition, there are individual differences in how different stimuli are interpreted and experienced, as shown by the interactions between nature connectedness and physiological measures in the present study. These findings imply that both natural and built environments may have positive psychophysiological impacts depending on the meanings the individuals associate with them. Indeed, people find some of the built environments relaxing, such as historic sites or cafes, and cultural background, age, social components, and individual dispositions may influence these feelings (Weber & Trojan, 2018). In addition, there are differences between what people perceive as relaxing in natural stimuli (Ratcliffe et al., 2013). For example, the singing of any bird species is not perceived as relaxing, but the degree of perceived relaxation is affected by the associations with the bird and the acoustic properties of the sound (Ratcliffe et al., 2013, 2020). Thus, the classification that natural environments promote relaxation and built environments promote anti-relaxation does not make sense. By contrast, the relaxing or restorative value of a specific environment is influenced by the features of the stimulus, as suggested by the evolutionary perspectives stressing the bottom-up processes (Kellert & Wilson, 1993; Ulrich, 1983; Ulrich et al., 1991), and the interaction of bottom-up processes with top-down processes that the constructivist perspective suggested (Haga et al., 2016; Van Hedger et al., 2019). In addition, the dispositions of the individuals, such as nature connectedness, influence how the environments are interpret and experienced. This view can be named the evolutionary-constructivist perspective.
The present study suggests that top-down factors contribute to the physiological (as well as psychological) effects of attributing a stimulus to nature. However, only the constructivist part of the evolutionary-constructivist perspective was tested here. The study’s main limitation was that it examined only one stimulus and two source-attribution conditions. The contrast between natural (waterfall) and industrial (factory) sources was strong, and with different stimulus examples, the results might have been weaker. It also remains possible that the top-down effects are restricted to ambiguous stimuli, as the relative contribution of low-level stimulus features and top-down processes could not be estimated. This might be possible by creating phase-scrambled versions of images of nature and urban settings so that their original environmental category would be difficult to label; a source attribution manipulation could be applied by giving the observers either the label of the original category or that of a different one. It is also clear that the psychological and physiological responses to a single stimulus (e.g., sound or picture) are not comparable with responses elicited during multisensory experiences in real environments. Lived experiences in real settings are likely to generate stronger and richer sensory responses compared to vicarious experiences in laboratory experiments (Mayer et al., 2009). However, the present study’s major contribution is that, by utilizing bias-free physiological measurements, our results suggest that top-down mechanisms related to the beliefs and meanings of individuals contribute to the effects of environmental exposure. Therefore, the influence of people’s beliefs and attitudes should be considered in theories and empirical research on the effects of environmental exposure.
Supplemental Material
sj-docx-1-eab-10.1177_00139165221107535 – Supplemental material for Top-Down Processing and Nature Connectedness Predict Psychological and Physiological Effects of Nature
Supplemental material, sj-docx-1-eab-10.1177_00139165221107535 for Top-Down Processing and Nature Connectedness Predict Psychological and Physiological Effects of Nature by Mika Koivisto, Enni Jalava, Lina Kuusisto, Henry Railo and Simone Grassini in Environment and Behavior
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
